Antibody data
- Antibody Data
- Antigen structure
- References [28]
- Comments [0]
- Validations
- Immunocytochemistry [2]
- Immunohistochemistry [3]
- Flow cytometry [1]
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-14029 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- VWF Monoclonal Antibody (F8/86)
- Antibody type
- Monoclonal
- Antigen
- Purifed from natural sources
- Description
- MA5-14029 targets von Willebrand Factor in IHC (P) applications and shows reactivity with Human samples. The MA5-14029 immunogen is partially purified von Willebrand factor from human placenta.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- F8/86
- Vial size
- 500 μL
- Concentration
- 0.2 mg/mL
- Storage
- 4°C
Submitted references Pro-Angiogenic Effects of Natural Antioxidants Extracted from Mango Leaf, Olive Leaf and Red Grape Pomace over Endothelial Colony-Forming Cells.
Inactivation of Cerebral Cavernous Malformation Genes Results in Accumulation of von Willebrand Factor and Redistribution of Weibel-Palade Bodies in Endothelial Cells.
A cellular and spatial map of the choroid plexus across brain ventricles and ages.
Prolyl Carboxypeptidase Mediates the C-Terminal Cleavage of (Pyr)-Apelin-13 in Human Umbilical Vein and Aortic Endothelial Cells.
ABO blood group is a determinant of von Willebrand factor protein levels in human pulmonary endothelial cells.
Primate model of chronic retinal neovascularization and vascular leakage.
Endothelial cells are a source of Nestin expression in Pulmonary Arterial Hypertension.
Human platelet lysate as an alternative to fetal bovine serum for culture and endothelial differentiation of human amniotic fluid mesenchymal stem cells.
VEGFR2-Specific Ligands Based on Affibody Molecules Demonstrate Agonistic Effects when Tetrameric in the Soluble Form or Immobilized via Spider Silk.
Binding of Staphylococcus aureus Protein A to von Willebrand Factor Is Regulated by Mechanical Force.
Establishment and long-term culture of human cystic fibrosis endothelial cells.
Duration of simulated microgravity affects the differentiation of mesenchymal stem cells.
Mechanisms of endothelial cell dysfunction in cystic fibrosis.
Microcirculatory effect of topical vapocoolants.
Tracking fusion of human mesenchymal stem cells after transplantation to the heart.
Endothelium-coated tumor clusters are associated with poor prognosis and micrometastasis of hepatocellular carcinoma after resection.
Prognostic value of bone marrow microvessel density and angiogenic cytokines in patients with multiple myeloma undergoing autologous stem cell transplant.
Lymphatic differentiation in classic Kaposi's sarcoma: patterns of D2-40 immunoexpression in the course of tumor progression.
The urokinase receptor supports tumorigenesis of human malignant pleural mesothelioma cells.
Pulsed acoustic cellular treatment induces expression of proangiogenic factors and chemokines in muscle flaps.
Association of coagulation factor XIII-A with Golgi proteins within monocyte-macrophages: implications for subcellular trafficking and secretion.
Existence of the lymphatic system in the primate corpus luteum.
Fibronectin promotes the phorbol 12-myristate 13-acetate-induced macrophage differentiation in myeloid leukemia cells.
Isolation and characterization of human gingival microvascular endothelial cells.
Epithelioid hemangioendothelioma derived from the spine region: case report and review of the literature.
Heparanase is involved in angiogenesis in esophageal cancer through induction of cyclooxygenase-2.
Regulation of Notch1 and Dll4 by vascular endothelial growth factor in arterial endothelial cells: implications for modulating arteriogenesis and angiogenesis.
Regulation of Notch1 and Dll4 by vascular endothelial growth factor in arterial endothelial cells: implications for modulating arteriogenesis and angiogenesis.
Sánchez-Gomar I, Benítez-Camacho J, Cejudo-Bastante C, Casas L, Moreno-Luna R, Mantell C, Durán-Ruiz MC
Antioxidants (Basel, Switzerland) 2022 Apr 27;11(5)
Antioxidants (Basel, Switzerland) 2022 Apr 27;11(5)
Inactivation of Cerebral Cavernous Malformation Genes Results in Accumulation of von Willebrand Factor and Redistribution of Weibel-Palade Bodies in Endothelial Cells.
Much CD, Sendtner BS, Schwefel K, Freund E, Bekeschus S, Otto O, Pagenstecher A, Felbor U, Rath M, Spiegler S
Frontiers in molecular biosciences 2021;8:622547
Frontiers in molecular biosciences 2021;8:622547
A cellular and spatial map of the choroid plexus across brain ventricles and ages.
Dani N, Herbst RH, McCabe C, Green GS, Kaiser K, Head JP, Cui J, Shipley FB, Jang A, Dionne D, Nguyen L, Rodman C, Riesenfeld SJ, Prochazka J, Prochazkova M, Sedlacek R, Zhang F, Bryja V, Rozenblatt-Rosen O, Habib N, Regev A, Lehtinen MK
Cell 2021 May 27;184(11):3056-3074.e21
Cell 2021 May 27;184(11):3056-3074.e21
Prolyl Carboxypeptidase Mediates the C-Terminal Cleavage of (Pyr)-Apelin-13 in Human Umbilical Vein and Aortic Endothelial Cells.
De Hert E, Bracke A, Pintelon I, Janssens E, Lambeir AM, Van Der Veken P, De Meester I
International journal of molecular sciences 2021 Jun 22;22(13)
International journal of molecular sciences 2021 Jun 22;22(13)
ABO blood group is a determinant of von Willebrand factor protein levels in human pulmonary endothelial cells.
Murray GP, Post SR, Post GR
Journal of clinical pathology 2020 Jun;73(6):347-349
Journal of clinical pathology 2020 Jun;73(6):347-349
Primate model of chronic retinal neovascularization and vascular leakage.
Patel C, Goody R, Hu W, Kurian A, James D, Torres R, Christie LA, Hohman T, Lawrence M
Experimental eye research 2020 Jun;195:108031
Experimental eye research 2020 Jun;195:108031
Endothelial cells are a source of Nestin expression in Pulmonary Arterial Hypertension.
Bhagwani AR, Hultman S, Farkas D, Moncayo R, Dandamudi K, Zadu AK, Cool CD, Farkas L
PloS one 2019;14(3):e0213890
PloS one 2019;14(3):e0213890
Human platelet lysate as an alternative to fetal bovine serum for culture and endothelial differentiation of human amniotic fluid mesenchymal stem cells.
Tancharoen W, Aungsuchawan S, Pothacharoen P, Bumroongkit K, Puaninta C, Pangjaidee N, Narakornsak S, Markmee R, Laowanitwattana T, Thaojamnong C
Molecular medicine reports 2019 Jun;19(6):5123-5132
Molecular medicine reports 2019 Jun;19(6):5123-5132
VEGFR2-Specific Ligands Based on Affibody Molecules Demonstrate Agonistic Effects when Tetrameric in the Soluble Form or Immobilized via Spider Silk.
Güler R, Thatikonda N, Ghani HA, Hedhammar M, Löfblom J
ACS biomaterials science & engineering 2019 Dec 9;5(12):6474-6484
ACS biomaterials science & engineering 2019 Dec 9;5(12):6474-6484
Binding of Staphylococcus aureus Protein A to von Willebrand Factor Is Regulated by Mechanical Force.
Viela F, Prystopiuk V, Leprince A, Mahillon J, Speziale P, Pietrocola G, Dufrêne YF
mBio 2019 Apr 30;10(2)
mBio 2019 Apr 30;10(2)
Establishment and long-term culture of human cystic fibrosis endothelial cells.
Plebani R, Tripaldi R, Lanuti P, Recchiuti A, Patruno S, Di Silvestre S, Simeone P, Anile M, Venuta F, Prioletta M, Mucilli F, Del Porto P, Marchisio M, Pandolfi A, Romano M
Laboratory investigation; a journal of technical methods and pathology 2017 Nov;97(11):1375-1384
Laboratory investigation; a journal of technical methods and pathology 2017 Nov;97(11):1375-1384
Duration of simulated microgravity affects the differentiation of mesenchymal stem cells.
Xue L, Li Y, Chen J
Molecular medicine reports 2017 May;15(5):3011-3018
Molecular medicine reports 2017 May;15(5):3011-3018
Mechanisms of endothelial cell dysfunction in cystic fibrosis.
Totani L, Plebani R, Piccoli A, Di Silvestre S, Lanuti P, Recchiuti A, Cianci E, Dell'Elba G, Sacchetti S, Patruno S, Guarnieri S, Mariggiò MA, Mari VC, Anile M, Venuta F, Del Porto P, Moretti P, Prioletta M, Mucilli F, Marchisio M, Pandolfi A, Evangelista V, Romano M
Biochimica et biophysica acta. Molecular basis of disease 2017 Dec;1863(12):3243-3253
Biochimica et biophysica acta. Molecular basis of disease 2017 Dec;1863(12):3243-3253
Microcirculatory effect of topical vapocoolants.
Galdyn I, Swanson E, Gordon C, Kwiecien G, Bena J, Siemionow M, Zins J
Plastic surgery (Oakville, Ont.) 2015 Summer;23(2):71-6
Plastic surgery (Oakville, Ont.) 2015 Summer;23(2):71-6
Tracking fusion of human mesenchymal stem cells after transplantation to the heart.
Freeman BT, Kouris NA, Ogle BM
Stem cells translational medicine 2015 Jun;4(6):685-94
Stem cells translational medicine 2015 Jun;4(6):685-94
Endothelium-coated tumor clusters are associated with poor prognosis and micrometastasis of hepatocellular carcinoma after resection.
Ding T, Xu J, Zhang Y, Guo RP, Wu WC, Zhang SD, Qian CN, Zheng L
Cancer 2011 Nov 1;117(21):4878-89
Cancer 2011 Nov 1;117(21):4878-89
Prognostic value of bone marrow microvessel density and angiogenic cytokines in patients with multiple myeloma undergoing autologous stem cell transplant.
Sucak GT, Aki SZ, Yüzbaşioğlu B, Akyürek N, Yağci M, Bağriaçik U, Haznedar R
Leukemia & lymphoma 2011 Jul;52(7):1281-9
Leukemia & lymphoma 2011 Jul;52(7):1281-9
Lymphatic differentiation in classic Kaposi's sarcoma: patterns of D2-40 immunoexpression in the course of tumor progression.
Kandemir NO, Barut F, Gun BD, Keser SH, Karadayi N, Gun M, Ozdamar SO
Pathology oncology research : POR 2011 Dec;17(4):843-51
Pathology oncology research : POR 2011 Dec;17(4):843-51
The urokinase receptor supports tumorigenesis of human malignant pleural mesothelioma cells.
Tucker TA, Dean C, Komissarov AA, Koenig K, Mazar AP, Pendurthi U, Allen T, Idell S
American journal of respiratory cell and molecular biology 2010 Jun;42(6):685-96
American journal of respiratory cell and molecular biology 2010 Jun;42(6):685-96
Pulsed acoustic cellular treatment induces expression of proangiogenic factors and chemokines in muscle flaps.
Krokowicz L, Cwykiel J, Klimczak A, Mielniczuk M, Siemionow M
The Journal of trauma 2010 Dec;69(6):1448-56
The Journal of trauma 2010 Dec;69(6):1448-56
Association of coagulation factor XIII-A with Golgi proteins within monocyte-macrophages: implications for subcellular trafficking and secretion.
Cordell PA, Kile BT, Standeven KF, Josefsson EC, Pease RJ, Grant PJ
Blood 2010 Apr 1;115(13):2674-81
Blood 2010 Apr 1;115(13):2674-81
Existence of the lymphatic system in the primate corpus luteum.
Xu F, Stouffer RL
Lymphatic research and biology 2009;7(3):159-68
Lymphatic research and biology 2009;7(3):159-68
Fibronectin promotes the phorbol 12-myristate 13-acetate-induced macrophage differentiation in myeloid leukemia cells.
Esendagli G, Canpinar H, Yilmaz G, Kaymaz FF, Kansu E, Guc D
International journal of hematology 2009 Mar;89(2):167-172
International journal of hematology 2009 Mar;89(2):167-172
Isolation and characterization of human gingival microvascular endothelial cells.
DeCarlo AA, Cohen JA, Aguado A, Glenn B
Journal of periodontal research 2008 Apr;43(2):246-54
Journal of periodontal research 2008 Apr;43(2):246-54
Epithelioid hemangioendothelioma derived from the spine region: case report and review of the literature.
Gokhan GA, Akyuz M, Gurer IE, Tuncer R
Wiener klinische Wochenschrift 2006 Jun;118(11-12):358-61
Wiener klinische Wochenschrift 2006 Jun;118(11-12):358-61
Heparanase is involved in angiogenesis in esophageal cancer through induction of cyclooxygenase-2.
Okawa T, Naomoto Y, Nobuhisa T, Takaoka M, Motoki T, Shirakawa Y, Yamatsuji T, Inoue H, Ouchida M, Gunduz M, Nakajima M, Tanaka N
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Nov 15;11(22):7995-8005
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Nov 15;11(22):7995-8005
Regulation of Notch1 and Dll4 by vascular endothelial growth factor in arterial endothelial cells: implications for modulating arteriogenesis and angiogenesis.
Liu ZJ, Shirakawa T, Li Y, Soma A, Oka M, Dotto GP, Fairman RM, Velazquez OC, Herlyn M
Molecular and cellular biology 2003 Jan;23(1):14-25
Molecular and cellular biology 2003 Jan;23(1):14-25
Regulation of Notch1 and Dll4 by vascular endothelial growth factor in arterial endothelial cells: implications for modulating arteriogenesis and angiogenesis.
Liu ZJ, Shirakawa T, Li Y, Soma A, Oka M, Dotto GP, Fairman RM, Velazquez OC, Herlyn M
Molecular and cellular biology 2003 Jan;23(1):14-25
Molecular and cellular biology 2003 Jan;23(1):14-25
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
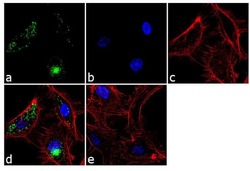
- Experimental details
- Immunofluorescence analysis of von Willebrand Factor was performed using 70% confluent log phase HUVEC cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with von Willebrand Factor Mouse Monoclonal Antibody (Product # MA5-14029) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing punctate cytoplasmic localization. Panel e shows the no primary antibody control. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
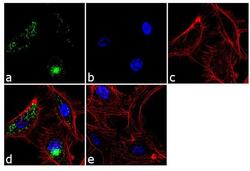
- Experimental details
- Immunofluorescence analysis of von Willebrand Factor was performed using 70% confluent log phase HUVEC cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with von Willebrand Factor Mouse Monoclonal Antibody (Product # MA5-14029) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing punctate cytoplasmic localization. Panel e shows the no primary antibody control. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
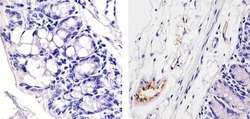
- Experimental details
- Immunohistochemistry analysis of von Willebrand Factor (F8/86) showing staining in the endothelial cells of paraffin-embedded mouse colon tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a von Willebrand Factor Antibody (F8/86) Mouse Monoclonal Antibody (Product # MA5-14029) diluted in 3% BSA-PBS at a dilution of 1:20 for 1 hour at 37°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
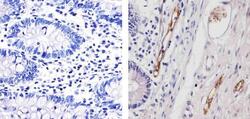
- Experimental details
- Immunohistochemistry analysis of von Willebrand Factor (F8/86) showing staining in the endothelial cells of paraffin-embedded human colon tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a von Willebrand Factor Antibody (F8/86) Mouse Monoclonal Antibody (Product # MA5-14029) diluted in 3% BSA-PBS at a dilution of 1:20 for 1 hour at 37°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
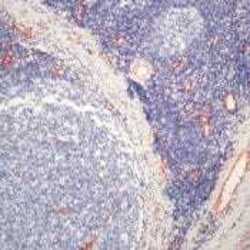
- Experimental details
- Formalin-fixed, paraffin-embedded human tonsil stained with Factor VIII antibody using peroxidase-conjugate and AEC chromogen. Note cytoplasmic staining of endothelial cells.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
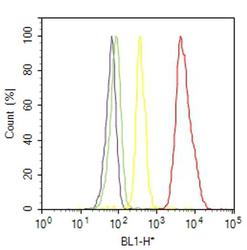
- Experimental details
- Flow cytometry analysis of von Willebrand Factor was done on HUVEC cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with von Willebrand Factor Mouse Monoclonal Antibody (MA514029, red histogram) or with mouse isotype control (yellow histogram) at 3-5 ug/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Rabbit Anti-Mouse Secondary Antibody (A11059) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
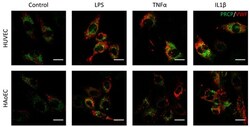
- Experimental details
- Figure 7 PRCP did not show overlap with vWF in stimulated HUVEC and HAoEC. Double staining of PRCP and VWF in control, LPS-, TNFalpha-, and IL-1beta-stimulated HUVEC. Cells were incubated for 16 h with the different stimuli, fixed with 4% PFA, permeabilized with 0.1% Triton X-100 and stained for PRCP (green) and vWF (red). Representative images of two independent experiments. (bar = 20 um).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
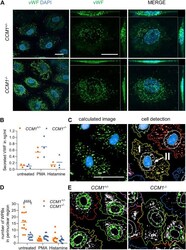
- FIGURE 3 High VWF content and aberrant WPB distribution in CCM1 -/- BOECs. (A) Immunofluorescent staining demonstrated high-level VWF expression (green) in clonally expanded CCM1 -/- BOECs. DAPI (blue) was used as nuclear counterstain. Confocal images were acquired using a 63x (NA 1.4) oil objective (left) and at higher magnification shown as maximum intensity projection of image stacks (0.2 um z planes). Scale bars indicate 20 um. (B) Absolute amount of secreted VWF from CCM1 +/- and CCM1 -/- BOECs as quantified by ELISA. Data are presented as single data points with the mean ( n = 3). (C) Analysis strategy of high-content imaging for untreated and stimulated BOECs. As basis for quantification, a sliding parabola function was applied for contrast enhancement of the VWF signal (green). Cell nuclei were segmented by their DAPI signal (blue) and the surrounding cytosolic area and perinuclear region were detected. VWF-positive granules in the cytosol (I) and the perinuclear region (II) were quantified. Scale bar indicates 30 um. (D) WPBs in the perinuclear region were quantified as shown in untreated CCM1 +/- and CCM1 -/- BOECs. Data are presented as single data points with the mean. Two-way ANOVA with Sidak correction for multiple comparisons was used for statistical analysis. p < 0.05. All experiments were performed in triplicates and four biological replicates. (E) Representative images of WPBs in the perinuclear region in untreated CCM1 +/- (left) and CCM1 -/- BOECs (right)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
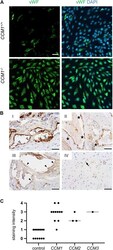
- FIGURE 4 High-level VWF expression is a common feature of CCM disease. (A) Strong immunopositivity for VWF (shown in green) was also found in clonally expanded CCM1 -/- CI-huVECs. DAPI (blue) was used as nuclear counterstain. Confocal images were acquired using a 10x (NA 0.45) objective. Scale bar indicates 50 um. (B) Immunohistochemistry demonstrated medium to strong VWF staining intensities (SI) in CCM tissue samples of hereditary cases (I-III). IV = normal brain. Representative images are shown. Open arrowhead = [SI] 4, black asterisk = [SI] 3, filled arrowhead = [SI] 2, open asterisk = [SI] 1, arrow = [SI] 0. Scale bars indicate 50 um. The graph displays the staining intensity of normal brain vessels in the vicinity of CCMs ( n = 13) and of cavernous vessel endothelia in CCM1 ( n = 11), CCM2 ( n = 3), and CCM3 ( n = 1) probands. Bars indicate the median.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
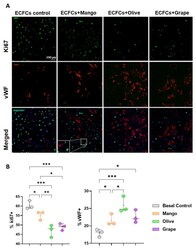
- Effect of the selected extracts on ECFC proliferation and differentiation. ( A ) Representative images of Ki67 and vWF staining of ECFCs treated 48 h with the ethanolic extracts of mango (4.99 mug/mL) and olive (46.67 mug/mL) leaves, and red grape pomace (11.67 mug/mL). ( B ) Box and whisker diagrams of the effect of the selected extracts on the proliferation and differentiation of ECFCs after 48 h of treatment. * p -value: * p < 0.05; ** p < 0.01; *** p < 0.001.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry