13-9700
antibody from Invitrogen Antibodies
Targeting: CTNNA2
CAP-R, CT114
 Western blot
Western blot ELISA
ELISA Immunocytochemistry
Immunocytochemistry Immunoprecipitation
Immunoprecipitation Immunohistochemistry
Immunohistochemistry Flow cytometry
Flow cytometry Other assay
Other assayAntibody data
- Antibody Data
- Antigen structure
- References [31]
- Comments [0]
- Validations
- Western blot [4]
- Immunocytochemistry [2]
- Other assay [9]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 13-9700 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- alpha Catenin Monoclonal Antibody (alpha-CAT-7A4)
- Antibody type
- Monoclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat, Chicken/Avian, Xenopus
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- alpha-CAT-7A4
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references Loss of NECTIN1 triggers melanoma dissemination upon local IGF1 depletion.
Ontogenesis of the tear drainage system requires Prickle1-driven polarized basement membrane deposition.
In situ molecular characterization of endoneurial microvessels that form the blood-nerve barrier in normal human adult peripheral nerves.
The cell-cell junctions of mammalian testes: II. The lamellar smooth muscle monolayer cells of the peritubular wall are laterally connected by vertical adherens junctions-a novel architectonic cell-cell junction system.
Real-Time Measurement of Melanoma Cell-Mediated Human Brain Endothelial Barrier Disruption Using Electric Cell-Substrate Impedance Sensing Technology.
Vitamin D Receptor Deletion Leads to the Destruction of Tight and Adherens Junctions in Lungs.
α-Catenin-dependent cytoskeletal tension controls Yap activity in the heart.
The Importance of Multifrequency Impedance Sensing of Endothelial Barrier Formation Using ECIS Technology for the Generation of a Strong and Durable Paracellular Barrier.
E-cadherin dynamics is regulated by galectin-7 at epithelial cell surface.
The Human Blood-Nerve Barrier Transcriptome.
Chronic hypoxia down-regulates tight junction protein ZO-2 expression in children with cyanotic congenital heart defect.
Actin-bundling protein plastin 3 is a regulator of ectoplasmic specialization dynamics during spermatogenesis in the rat testis.
Characterization of α-, β- and p120-Catenin Expression in Feline Mammary Tissues and their Relation with E- and P-Cadherin.
Rai14 (retinoic acid induced protein 14) is involved in regulating f-actin dynamics at the ectoplasmic specialization in the rat testis*.
Palladin is a regulator of actin filament bundles at the ectoplasmic specialization in adult rat testes.
α-Catenin contributes to the strength of E-cadherin-p120 interactions.
Melanoma prognostic model using tissue microarrays and genetic algorithms.
Cell discohesion and multifocality of carcinoma in situ of the bladder: new insight from the adhesion molecule profile (e-cadherin, Ep-CAM, and MUC1).
Localization of claudin-5 and ZO-1 in rat spleen sinus endothelial cells.
Expression of the E-cadherin-catenins complex in sentinel node is related to tumor morphology but not to spread to nonsentinel nodes.
Axon behavior in the olfactory nerve reflects the involvement of catenin-cadherin mediated adhesion.
Modulating the strength of cadherin adhesion: evidence for a novel adhesion complex.
Protein expression profiling identifies subclasses of breast cancer and predicts prognosis.
Remodeling of myocyte gap junctions in arrhythmogenic right ventricular cardiomyopathy due to a deletion in plakoglobin (Naxos disease).
Abnormal expression of E-cadherin in early gastric carcinoma: its relationship with macroscopic growth patterns and catenin alpha and beta.
Immunohistochemical patterns for alpha- and beta-catenin, E- and N-cadherin expression in ovarian epithelial tumors.
Distinct subcellular expression of endogenous polycystin-2 in the plasma membrane and Golgi apparatus of MDCK cells.
Progressive changes in adherens junction structure during intestinal adenoma formation in Apc mutant mice.
Monocytes induce reversible focal changes in vascular endothelial cadherin complex during transendothelial migration under flow.
Analysis of gene expression in human bullous keratopathy corneas containing limiting amounts of RNA.
Changing roles of cadherins and catenins during progression of squamous intraepithelial lesions in the uterine cervix.
Ablain J, Al Mahi A, Rothschild H, Prasad M, Aires S, Yang S, Dokukin ME, Xu S, Dang M, Sokolov I, Lian CG, Zon LI
Nature genetics 2022 Dec;54(12):1839-1852
Nature genetics 2022 Dec;54(12):1839-1852
Ontogenesis of the tear drainage system requires Prickle1-driven polarized basement membrane deposition.
Guo D, Ru J, Mao F, Ouyang H, Ju R, Wu K, Liu Y, Liu C
Development (Cambridge, England) 2020 Nov 30;147(22)
Development (Cambridge, England) 2020 Nov 30;147(22)
In situ molecular characterization of endoneurial microvessels that form the blood-nerve barrier in normal human adult peripheral nerves.
Ouyang X, Dong C, Ubogu EE
Journal of the peripheral nervous system : JPNS 2019 Jun;24(2):195-206
Journal of the peripheral nervous system : JPNS 2019 Jun;24(2):195-206
The cell-cell junctions of mammalian testes: II. The lamellar smooth muscle monolayer cells of the peritubular wall are laterally connected by vertical adherens junctions-a novel architectonic cell-cell junction system.
Domke LM, Franke WW
Cell and tissue research 2019 Feb;375(2):451-482
Cell and tissue research 2019 Feb;375(2):451-482
Real-Time Measurement of Melanoma Cell-Mediated Human Brain Endothelial Barrier Disruption Using Electric Cell-Substrate Impedance Sensing Technology.
Anchan A, Kalogirou-Baldwin P, Johnson R, Kho DT, Joseph W, Hucklesby J, Finlay GJ, O'Carroll SJ, Angel CE, Graham ES
Biosensors 2019 Apr 15;9(2)
Biosensors 2019 Apr 15;9(2)
Vitamin D Receptor Deletion Leads to the Destruction of Tight and Adherens Junctions in Lungs.
Chen H, Lu R, Zhang YG, Sun J
Tissue barriers 2018;6(4):1-13
Tissue barriers 2018;6(4):1-13
α-Catenin-dependent cytoskeletal tension controls Yap activity in the heart.
Vite A, Zhang C, Yi R, Emms S, Radice GL
Development (Cambridge, England) 2018 Mar 8;145(5)
Development (Cambridge, England) 2018 Mar 8;145(5)
The Importance of Multifrequency Impedance Sensing of Endothelial Barrier Formation Using ECIS Technology for the Generation of a Strong and Durable Paracellular Barrier.
Robilliard LD, Kho DT, Johnson RH, Anchan A, O'Carroll SJ, Graham ES
Biosensors 2018 Jul 4;8(3)
Biosensors 2018 Jul 4;8(3)
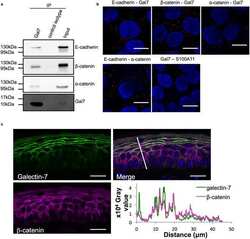
E-cadherin dynamics is regulated by galectin-7 at epithelial cell surface.
Advedissian T, Proux-Gillardeaux V, Nkosi R, Peyret G, Nguyen T, Poirier F, Viguier M, Deshayes F
Scientific reports 2017 Dec 6;7(1):17086
Scientific reports 2017 Dec 6;7(1):17086
The Human Blood-Nerve Barrier Transcriptome.
Palladino SP, Helton ES, Jain P, Dong C, Crowley MR, Crossman DK, Ubogu EE
Scientific reports 2017 Dec 12;7(1):17477
Scientific reports 2017 Dec 12;7(1):17477
Chronic hypoxia down-regulates tight junction protein ZO-2 expression in children with cyanotic congenital heart defect.
Jenkins EL, Caputo M, Angelini GD, Ghorbel MT
ESC heart failure 2016 Jun;3(2):131-137
ESC heart failure 2016 Jun;3(2):131-137
Actin-bundling protein plastin 3 is a regulator of ectoplasmic specialization dynamics during spermatogenesis in the rat testis.
Li N, Mruk DD, Wong CK, Lee WM, Han D, Cheng CY
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2015 Sep;29(9):3788-805
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2015 Sep;29(9):3788-805
Characterization of α-, β- and p120-Catenin Expression in Feline Mammary Tissues and their Relation with E- and P-Cadherin.
Figueira AC, Gomes C, Vilhena H, Miranda S, Carvalheira J, DE Matos AJ, Dias-Pereira P, Gärtner F
Anticancer research 2015 Jun;35(6):3361-9
Anticancer research 2015 Jun;35(6):3361-9
Rai14 (retinoic acid induced protein 14) is involved in regulating f-actin dynamics at the ectoplasmic specialization in the rat testis*.
Qian X, Mruk DD, Cheng CY
PloS one 2013;8(4):e60656
PloS one 2013;8(4):e60656
Palladin is a regulator of actin filament bundles at the ectoplasmic specialization in adult rat testes.
Qian X, Mruk DD, Wong EW, Lie PP, Cheng CY
Endocrinology 2013 May;154(5):1907-20
Endocrinology 2013 May;154(5):1907-20
α-Catenin contributes to the strength of E-cadherin-p120 interactions.
Troyanovsky RB, Klingelhöfer J, Troyanovsky SM
Molecular biology of the cell 2011 Nov;22(22):4247-55
Molecular biology of the cell 2011 Nov;22(22):4247-55
Melanoma prognostic model using tissue microarrays and genetic algorithms.
Gould Rothberg BE, Berger AJ, Molinaro AM, Subtil A, Krauthammer MO, Camp RL, Bradley WR, Ariyan S, Kluger HM, Rimm DL
Journal of clinical oncology : official journal of the American Society of Clinical Oncology 2009 Dec 1;27(34):5772-80
Journal of clinical oncology : official journal of the American Society of Clinical Oncology 2009 Dec 1;27(34):5772-80
Cell discohesion and multifocality of carcinoma in situ of the bladder: new insight from the adhesion molecule profile (e-cadherin, Ep-CAM, and MUC1).
Patriarca C, Colombo P, Pio Taronna A, Wesseling J, Franchi G, Guddo F, Naspro R, Macchi RM, Giunta P, Di Pasquale M, Parente M, Arizzi C, Roncalli M, Campo B
International journal of surgical pathology 2009 Apr;17(2):99-106
International journal of surgical pathology 2009 Apr;17(2):99-106
Localization of claudin-5 and ZO-1 in rat spleen sinus endothelial cells.
Uehara K, Uehara A
Histochemistry and cell biology 2008 Jan;129(1):95-103
Histochemistry and cell biology 2008 Jan;129(1):95-103
Expression of the E-cadherin-catenins complex in sentinel node is related to tumor morphology but not to spread to nonsentinel nodes.
Canavese G, Bernardi A, Candelaresi G, Lovadina P, Amerio S, Rossetti V, Rabagliati C, Berardengo E
Pathology, research and practice 2007;203(7):517-23
Pathology, research and practice 2007;203(7):517-23
Axon behavior in the olfactory nerve reflects the involvement of catenin-cadherin mediated adhesion.
Akins MR, Greer CA
The Journal of comparative neurology 2006 Dec 20;499(6):979-89
The Journal of comparative neurology 2006 Dec 20;499(6):979-89
Modulating the strength of cadherin adhesion: evidence for a novel adhesion complex.
Kim YJ, Sauer C, Testa K, Wahl JK, Svoboda RA, Johnson KR, Wheelock MJ, Knudsen KA
Journal of cell science 2005 Sep 1;118(Pt 17):3883-94
Journal of cell science 2005 Sep 1;118(Pt 17):3883-94
Protein expression profiling identifies subclasses of breast cancer and predicts prognosis.
Jacquemier J, Ginestier C, Rougemont J, Bardou VJ, Charafe-Jauffret E, Geneix J, Adélaïde J, Koki A, Houvenaeghel G, Hassoun J, Maraninchi D, Viens P, Birnbaum D, Bertucci F
Cancer research 2005 Feb 1;65(3):767-79
Cancer research 2005 Feb 1;65(3):767-79
Remodeling of myocyte gap junctions in arrhythmogenic right ventricular cardiomyopathy due to a deletion in plakoglobin (Naxos disease).
Kaplan SR, Gard JJ, Protonotarios N, Tsatsopoulou A, Spiliopoulou C, Anastasakis A, Squarcioni CP, McKenna WJ, Thiene G, Basso C, Brousse N, Fontaine G, Saffitz JE
Heart rhythm 2004 May;1(1):3-11
Heart rhythm 2004 May;1(1):3-11
Abnormal expression of E-cadherin in early gastric carcinoma: its relationship with macroscopic growth patterns and catenin alpha and beta.
Song SY, Kim S, Kim DS, Son HJ, Rhee JC, Kim YI
Journal of clinical gastroenterology 2004 Mar;38(3):252-9
Journal of clinical gastroenterology 2004 Mar;38(3):252-9
Immunohistochemical patterns for alpha- and beta-catenin, E- and N-cadherin expression in ovarian epithelial tumors.
Marques FR, Fonsechi-Carvasan GA, De Angelo Andrade LA, Böttcher-Luiz F
Gynecologic oncology 2004 Jul;94(1):16-24
Gynecologic oncology 2004 Jul;94(1):16-24
Distinct subcellular expression of endogenous polycystin-2 in the plasma membrane and Golgi apparatus of MDCK cells.
Scheffers MS, Le H, van der Bent P, Leonhard W, Prins F, Spruit L, Breuning MH, de Heer E, Peters DJ
Human molecular genetics 2002 Jan 1;11(1):59-67
Human molecular genetics 2002 Jan 1;11(1):59-67
Progressive changes in adherens junction structure during intestinal adenoma formation in Apc mutant mice.
Carothers AM, Melstrom KA Jr, Mueller JD, Weyant MJ, Bertagnolli MM
The Journal of biological chemistry 2001 Oct 19;276(42):39094-102
The Journal of biological chemistry 2001 Oct 19;276(42):39094-102
Monocytes induce reversible focal changes in vascular endothelial cadherin complex during transendothelial migration under flow.
Allport JR, Muller WA, Luscinskas FW
The Journal of cell biology 2000 Jan 10;148(1):203-16
The Journal of cell biology 2000 Jan 10;148(1):203-16
Analysis of gene expression in human bullous keratopathy corneas containing limiting amounts of RNA.
Spirin KS, Ljubimov AV, Castellon R, Wiedoeft O, Marano M, Sheppard D, Kenney MC, Brown DJ
Investigative ophthalmology & visual science 1999 Dec;40(13):3108-15
Investigative ophthalmology & visual science 1999 Dec;40(13):3108-15
Changing roles of cadherins and catenins during progression of squamous intraepithelial lesions in the uterine cervix.
de Boer CJ, van Dorst E, van Krieken H, Jansen-van Rhijn CM, Warnaar SO, Fleuren GJ, Litvinov SV
The American journal of pathology 1999 Aug;155(2):505-15
The American journal of pathology 1999 Aug;155(2):505-15
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of HeLa cell lysates using Ms anti-a-Catenin (Product # 13-9700).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Knockout of Catenin alpha-1 was achieved by CRISPR-Cas9 genome editing using LentiArray™ Lentiviral sgRNA (Product # A32042, Assay ID CRISPR787281_LV) and LentiArray Cas9 Lentivirus (Product # A32064). Western blot analysis of Catenin alpha-1 was performed by loading 30 µg of HeLa Wild Type (Lane 1), HeLa Cas9 (Lane 2) andHeLa Catenin alpha-1 KO (Lane 3) whole cell extracts. The samples were electrophoresed using NuPAGE™ Novex™ 4-12% Bis-Tris Protein Gel (Product # NP0322BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with Anti-alpha Catenin Monoclonal Antibody (alpha-CAT-7A4) (Product # 13-9700, 1:250 dilution) and Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177, 1:5,000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005). Loss of signal upon CRISPR mediated knockout (KO) using the LentiArray™ CRISPR product line confirms that antibody is specific to Catenin alpha-1.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on membrane enriched extracts (30 µg) of A549 (Lane 1), HeLa (Lane 2), MDA-MB-231 (Lane 3), MCF7 (Lane 4), NIH/3T3 (Lane 5) and A-431 (Lane 6).The blots were probed with Anti-alpha Catenin Mouse Monoclonal Antibody (Product # 13-9700, 1:250 dilution) and detected by chemiluminescence using Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, HRP conjugate (Product # A28177, 0.4 µg/mL, 1:2500 dilution). A ~100 kDa band corresponding alpha Catenin was observed across cell lines tested. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane by Pierce™ Power Blotter System (22834). The membrane was probed with the relevant primary and secondary Antibody using iBind™ Flex Western Starter Kit (Product # SLF2000S). Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-alpha Catenin Monoclonal Antibody (alpha-CAT-7A4)(Product # 13-9700) and a 100kDa band corresponding to alpha Catenin was observed across cell lines and tissue extracts tested except PC-3 and Daudi which is reported to be negative. Whole cell extracts (30 µg lysate) of A-431 (Lane 1), HeLa (Lane 2), MCF7 (Lane 3), LNCaP (Lane 4), PC-3 (Lane 5), Daudi (Lane 6), NIH/3T3 (Lane 7) and tissue extracts of Mouse Lung (Lane 8) and Rat Heart (Lane 9) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0322BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:250 dilution) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using SuperSignal™ West Dura Extended Duration Substrate (Product # 34076).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of alpha Catenin was performed using 90% confluent log phase MCF-7 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with alpha Catenin (alpha-CAT-7A4) Mouse Monoclonal Antibody (Product # 13-9700) at 1:250 dilution in0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing cell junctional localization. Panel e shows the no primary antibody control. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Immunocytochemical analysis of Jurkat cell lysates using Mouse anti-Bcl-2 monoclonal antibody (Product # 13-8800)
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 5: Identification of protein domains involved in cross-linking of p120 and alpha-catenin by BM[PEO]3. (A) A cysteine map of p120-F. The protein consists of three major domains--the central arm-repeat domain (individual arm repeats are depicted by open boxes, numbered) flanked by the amino-terminal (N) and carboxy-terminal (C) domains. Note that all 10 cysteines of p120 (indicated by numbered arrows) are located at the arm-repeat domain, one of which, Cys-9, is present in a large loop separating arm repeats 5 and 6. This loop may contain a small insert encoding by the alternative exon C (filled circle). FLAG epitope (F) is indicated by an open circle. (B) Recruitment of the p120-F cysteine mutants into the cadherin-catenin complex. The p120-F mutants were expressed in A-431 cells and immunoprecipitated with anti-FLAG agarose. The immunoprecipitates were probed for the p120 mutants (p120 mutants; the number of the mutant is indicated above the lanes) and for alpha-catenin (alpha-cat) using anti-FLAG or anti-alpha-catenin antibodies. Note that except for Cys-4, all Cys mutants interact with alpha-catenin. (C) Digitonin cell lysates of A-431 cells expressing p120-F (left) and its different cysteine mutants (right; the mutant's numbers are indicated above the lanes) were cross-linked by BM[PEO]3 and analyzed by Western blotting for p120-F or its mutants (p120F, p120F-mutants) and for alpha-catenin (alpha-cat). Note that the addition of the cross-link (+) resulted in ap
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
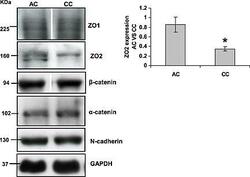
- Figure 3 Down-regulation of zonula occludens protein 2 (ZO-2) protein expression in myocardium of cyanotic compared with acyanotic patients. Myocardium biopsies were lysed to isolate protein content and western blot analysis performed probing for ZO-2, ZO-1, N-cadherin, beta -catenin, and GAPDH. ZO-2 protein expression was significantly down-regulated in cyanotic biopsies compared with acyanotic, whereas no difference in any other adherens junction expressed proteins was detected. All results were normalized to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) levels. Data are mean +- SEM * = P < 0.05 ( n = 5).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
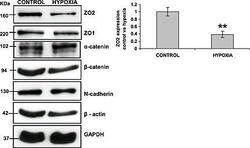
- Figure 4 Down-regulation of zonula occludens protein 2 (ZO-2) levels in rat cardiomyocytes following 24 h hypoxic treatment. Cells were treated with hypoxic conditions by incubating in a chamber with 5% CO 2 / 0.2% O 2 . After 24 h, cells were lysed, and western blot analysis performed probing for ZO-2, ZO-1, N-cadherin, beta -catenin, and beta -actin, normalizing all results to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) levels. ZO-2 protein expression was significantly down-regulated in hypoxic treated cells compared with untreated cells (control), whereas no difference in ZO-1, N-cadherin, beta -catenin, or beta -actin levels was detected between treated and untreated cells. Data are mean +- SEM ** = P < 0.01 ( n = 4).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
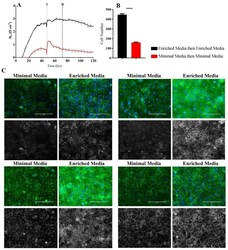
- Figure 6 Immunocytochemistry of brain endothelial junctional space following growth in either Enriched Media or Minimal Media. Cells were seeded at time 0 h at 20,000 cells per 0.3 cm 2 . ( A ) ECIS resistance and R b measurements over 120 h. Cells were grown in either Enriched Media or Minimal Media from T = 0 h, with media changed at T = 48 h (I). Data shown is the mean +- SD (n = 3 wells) of one independent experiment representative of three experimental repeats; ( B ) Cell number count of Hoechst stained nuclei at time point II, obtained through Image J Software analysis. Data shown is the mean +- SEM (n = 18 wells) of 1 independent experiment representative of 3 experimental repeats; ( C ) The junctional space following growth in either Enriched Media or Minimal Media 72 h post seeding. The time point of fixation corresponds to the second vertical dotted line (II) shown on the ECIS traces. Representative GFP/DAPI and GFP monochrome images for the junctional proteins CD144, ZO-1, beta-catenin, and alpha-catenin are shown. Each panel shows cells grown in Minimal Media on the left and cells grown in Enriched Media on the right. The corresponding Alexa Fluor 488 (GFP) monochrome image is shown below each merged image. Green--junctional protein, blue--nuclei. Scale bar is 200 um. Immunocytochemistry data show one representative image from one independent experiment, which is representative of three experimental repeats. Graphical representations of p values are * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
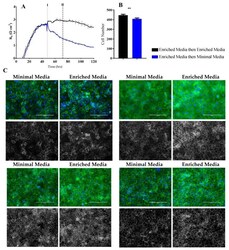
- Figure 7 Immunocytochemistry of brain endothelial junctional space following growth in either Enriched Media or Minimal Media. Cells were seeded at time 0 h at 20,000 cells per 0.3 cm 2 . ( A ) ECIS resistance and R b measurements over 120 h. Cells were grown in either Enriched Media or Minimal Media from T = 0 h, with media changed at T = 48 h (I). Data shown is the mean +- S.D. (n = 3 wells) of 1 independent experiment representative of 3 experimental repeats; ( B ) Cell number count of Hoechst stained nuclei at time point II, obtained through Image J Software analysis. Data shown is the mean +- SEM (n = 18 wells) of 1 independent experiment representative of 3 experimental repeats; ( C ) The junctional space following growth in either Enriched Media or Minimal Media 72 h post seeding. The time point of fixation corresponds to the second vertical dotted line (II) shown on the ECIS traces. Representative GFP/DAPI and GFP monochrome images for the junctional proteins CD144, ZO-1, beta-catenin and alpha-catenin are shown. Each panel shows cells grown in Minimal Media on the left and cells grown in Enriched Media on the right. The corresponding Alexa Fluor 488 (GFP) monochrome image is shown below each merged image. Green--junctional protein, blue--nuclei. Scale bar is 200 um. Immunocytochemistry data shows 1 representative image from 1 independent experiment, which is representative of 3 experimental repeats. Graphical representations of p values are * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
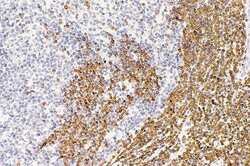
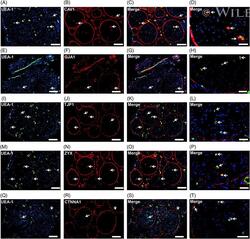
- Endothelial and specialized epithelial-specific proteins. Representative indirect fluorescent digital photomicrographs of cryostat axial sections of normal adult sural nerves show UEA-1-positive endothelial cells (green; A, E, I, M, Q) with expression of CAV1, GJA1, TJP1, ZYX, and CTNNA1 (red; B, F, J, N, R, respectively) restricted to endoneurial microvessels, fenestrated epineurial macrovessels and the perineurium, shown in the merged images at lower and higher magnifications (yellow/orange for endothelial cells). Expression of these proteins by both microvascular and macrovascular endothelial cells and the perineurium suggests non-restrictive barrier, but specialized endothelial and epithelial cell functions in the normal adult human peripheral nerves. White arrows demonstrate positively staining endoneurial microvessels. Blue (4, 6-diamidino-2-phenylindole) staining indicates nuclei. Scale bar 500 mum for A-C, E-G, I-K, M-O, and Q-S and 125 mum for D, H, L, P, and T
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
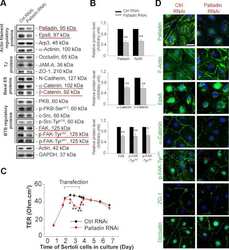
- Figure 5 Knockdown of Rai14 in the Sertoli cell epithelium with an established TJ-permeability barrier in vitro by RNAi disrupts actin filament organization and the TJ barrier. (A) Sertoli cells cultured alone on Matrigel-coated 12-well dishes for 2-day with an established TJ-permeability barrier were transfected with Rai14 siRNA duplexes (Rai14 RNAi) versus non-targeting control duplexes (Ctrl RNAi) at 100 nM using Ribojuice transfection medium for 24 hr, thereafter, cells were washed twice and cultured in F12/DMEM for 12 hr to allow recovery. Thereafter, cells were transfected again under the same conditions for another 24 hr. Thereafter, cells were rinsed with fresh F12/DMEM and cultured for an additional 12 hr before termination, and used to prepare lysates for immunoblotting using antibodies against several BTB-associated constituent or regulatory proteins. A knockdown of Rai14 by ~50% was noted in which the control was arbitrarily set at 1 against which statistical comparison was performed (B) without any apparent off-target effects (A). The findings shown herein are the results of 3 independent experiments excluding pilot experiments which were used to establish optimal experimental conditions, such as different concentrations of siRNA duplexes and Ribojuice. It was noted that we achieved only ~50-60% knockdown of Rai14 in several pilot experiments, unlike other target genes [ e.g ., Scribble, beta1-integrin, and P-glycoprotein] wherein we could silence the target gene
 Explore
Explore Validate
Validate Learn
Learn