Antibody data
- Antibody Data
- Antigen structure
- References [38]
- Comments [0]
- Validations
- Western blot [4]
- Immunocytochemistry [6]
- Flow cytometry [1]
- Other assay [20]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 700062 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Vinculin Recombinant Rabbit Monoclonal Antibody (42H89L44)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Description
- Intact IgG appears on a non-reducing gel as ~150 kDa band and upon reduction generating a ~25 kDa light chain band and a ~50 kDa heavy chain. This antibody reacts with Human Vinculin. Based on sequence similarity, reactivity to chimpanzee, Rhesus monkey, swine, equine, mouse, rat, bovine, and chicken is expected. Immunogen sequence IRGALAEARKIAELCDDPKERDDILRSLGEI SALTSKLADLRRQGKGDSPEARALAKQV ATALQNLQTKTNRAVANSRPAKAAVHLE GKIEQAQRWIDNPTVDDRGVGQAAIRGL VAEGHRLANVMMGPYRQDLLAKCDRVD QLTAQLADLAARGEGESPQARALASQLQ DSLKDLKARMQEAM. Test material for western blotting/immunofluorescence: HeLa cells. Recombinant rabbit monoclonal antibodies are produced using in vitro expression systems. The expression systems are developed by cloning in the specific antibody DNA sequences from immunoreactive rabbits. Then, individual clones are screened to select the best candidates for production. The advantages of using recombinant rabbit monoclonal antibodies include: better specificity and sensitivity, lot-to-lot consistency, animal origin-free formulations, and broader immunoreactivity to diverse targets due to larger rabbit immune repertoire.
- Reactivity
- Human, Mouse
- Host
- Rabbit
- Isotype
- IgG
- Antibody clone number
- 42H89L44
- Vial size
- 100 μg
- Concentration
- 0.5 mg/mL
- Storage
- Store at 4°C short term. For long term storage, store at -20°C, avoiding freeze/thaw cycles.
Submitted references A conserved domain of Drosophila RNA-binding protein Pumilio interacts with multiple CCR4-NOT deadenylase complex subunits to repress target mRNAs.
Increased Stiffness Downregulates Focal Adhesion Kinase Expression in Pancreatic Cancer Cells Cultured in 3D Self-Assembling Peptide Scaffolds.
ATRX loss in glioma results in dysregulation of cell-cycle phase transition and ATM inhibitor radio-sensitization.
Augmin-dependent microtubule self-organization drives kinetochore fiber maturation in mammals.
The Plasma Membrane Ca(2+) Pump PMCA4b Regulates Melanoma Cell Migration through Remodeling of the Actin Cytoskeleton.
Cardiac pericytes function as key vasoactive cells to regulate homeostasis and disease.
Transcriptional Regulation of PIK3CD and PIKFYVE in T-Cell Acute Lymphoblastic Leukemia by IKAROS and Protein Kinase CK2.
Distinct genetic pathways define pre-malignant versus compensatory clonal hematopoiesis in Shwachman-Diamond syndrome.
ARPC1B binds WASP to control actin polymerization and curtail tonic signaling in B cells.
STIM1 Controls the Focal Adhesion Dynamics and Cell Migration by Regulating SOCE in Osteosarcoma.
The Effects of Syndecan on Osteoblastic Cell Adhesion Onto Nano-Zirconia Surface.
Poly(ADP-ribose) Glycohydrolase Inhibition Sequesters NAD(+) to Potentiate the Metabolic Lethality of Alkylating Chemotherapy in IDH-Mutant Tumor Cells.
Succinic Semialdehyde Dehydrogenase Deficiency: In Vitro and In Silico Characterization of a Novel Pathogenic Missense Variant and Analysis of the Mutational Spectrum of ALDH5A1.
A tumor-intrinsic PD-L1/NLRP3 inflammasome signaling pathway drives resistance to anti-PD-1 immunotherapy.
Senescent mesenchymal stem cells remodel extracellular matrix driving breast cancer cells to a more-invasive phenotype.
A fluorescence-activatable reporter of flavivirus NS2B-NS3 protease activity enables live imaging of infection in single cells and viral plaques.
Increased talin-vinculin spatial proximities in livers in response to spotted fever group rickettsial and Ebola virus infections.
IDH-mutant gliomas harbor fewer regulatory T cells in humans and mice.
Culture and Differentiation of Human Hair Follicle Dermal Papilla Cells in a Soft 3D Self-Assembling Peptide Scaffold.
Mechano-signalling, induced by fullerene C(60) nanofilms, arrests the cell cycle in the G2/M phase and decreases proliferation of liver cancer cells.
Minute-scale persistence of a GPCR conformation state triggered by non-cognate G protein interactions primes signaling.
A divalent siRNA chemical scaffold for potent and sustained modulation of gene expression throughout the central nervous system.
16-Hydroxycleroda-3,13-dien-15,16-olide induces anoikis in human renal cell carcinoma cells: involvement of focal adhesion disassembly and signaling.
Transition of responsive mechanosensitive elements from focal adhesions to adherens junctions on epithelial differentiation.
Delineating the role of cooperativity in the design of potent PROTACs for BTK.
Human snoRNA-93 is processed into a microRNA-like RNA that promotes breast cancer cell invasion.
Diamond, graphite, and graphene oxide nanoparticles decrease migration and invasiveness in glioblastoma cell lines by impairing extracellular adhesion.
Biosynthetic PCL-graft-Collagen Bulk Material for Tissue Engineering Applications.
Integrin-mediated traction force enhances paxillin molecular associations and adhesion dynamics that increase the invasiveness of tumor cells into a three-dimensional extracellular matrix.
Decellularized extracellular matrix of human umbilical vein endothelial cells promotes endothelial differentiation of stem cells from exfoliated deciduous teeth.
Human fibroblasts display a differential focal adhesion phenotype relative to chimpanzee.
Angiotensin receptors and β-catenin regulate brain endothelial integrity in malaria.
Effects of decellularized matrices derived from periodontal ligament stem cells and SHED on the adhesion, proliferation and osteogenic differentiation of human dental pulp stem cells in vitro.
A multi-omics approach identifies key hubs associated with cell type-specific responses of airway epithelial cells to staphylococcal alpha-toxin.
Connectivity Map-based discovery of parbendazole reveals targetable human osteogenic pathway.
Susceptibility to tuberculosis is associated with variants in the ASAP1 gene encoding a regulator of dendritic cell migration.
Force engages vinculin and promotes tumor progression by enhancing PI3K activation of phosphatidylinositol (3,4,5)-triphosphate.
Dual-objective STORM reveals three-dimensional filament organization in the actin cytoskeleton.
Haugen RJ, Arvola RM, Connacher RP, Roden RT, Goldstrohm AC
The Journal of biological chemistry 2022 Sep;298(9):102270
The Journal of biological chemistry 2022 Sep;298(9):102270
Increased Stiffness Downregulates Focal Adhesion Kinase Expression in Pancreatic Cancer Cells Cultured in 3D Self-Assembling Peptide Scaffolds.
Betriu N, Andreeva A, Alonso A, Semino CE
Biomedicines 2022 Jul 29;10(8)
Biomedicines 2022 Jul 29;10(8)
ATRX loss in glioma results in dysregulation of cell-cycle phase transition and ATM inhibitor radio-sensitization.
Qin T, Mullan B, Ravindran R, Messinger D, Siada R, Cummings JR, Harris M, Muruganand A, Pyaram K, Miklja Z, Reiber M, Garcia T, Tran D, Danussi C, Brosnan-Cashman J, Pratt D, Zhao X, Rehemtulla A, Sartor MA, Venneti S, Meeker AK, Huse JT, Morgan MA, Lowenstein PR, Castro MG, Yadav VN, Koschmann C
Cell reports 2022 Jan 11;38(2):110216
Cell reports 2022 Jan 11;38(2):110216
Augmin-dependent microtubule self-organization drives kinetochore fiber maturation in mammals.
Almeida AC, Soares-de-Oliveira J, Drpic D, Cheeseman LP, Damas J, Lewin HA, Larkin DM, Aguiar P, Pereira AJ, Maiato H
Cell reports 2022 Apr 5;39(1):110610
Cell reports 2022 Apr 5;39(1):110610
The Plasma Membrane Ca(2+) Pump PMCA4b Regulates Melanoma Cell Migration through Remodeling of the Actin Cytoskeleton.
Naffa R, Padányi R, Ignácz A, Hegyi Z, Jezsó B, Tóth S, Varga K, Homolya L, Hegedűs L, Schlett K, Enyedi A
Cancers 2021 Mar 17;13(6)
Cancers 2021 Mar 17;13(6)
Cardiac pericytes function as key vasoactive cells to regulate homeostasis and disease.
Lee LL, Khakoo AY, Chintalgattu V
FEBS open bio 2021 Jan;11(1):207-225
FEBS open bio 2021 Jan;11(1):207-225
Transcriptional Regulation of PIK3CD and PIKFYVE in T-Cell Acute Lymphoblastic Leukemia by IKAROS and Protein Kinase CK2.
Dovat E, Song C, Hu T, Rahman MA, Dhanyamraju PK, Klink M, Bogush D, Soliman M, Kane S, McGrath M, Ding Y, Desai D, Sharma A, Gowda C
International journal of molecular sciences 2021 Jan 15;22(2)
International journal of molecular sciences 2021 Jan 15;22(2)
Distinct genetic pathways define pre-malignant versus compensatory clonal hematopoiesis in Shwachman-Diamond syndrome.
Kennedy AL, Myers KC, Bowman J, Gibson CJ, Camarda ND, Furutani E, Muscato GM, Klein RH, Ballotti K, Liu S, Harris CE, Galvin A, Malsch M, Dale D, Gansner JM, Nakano TA, Bertuch A, Vlachos A, Lipton JM, Castillo P, Connelly J, Churpek J, Edwards JR, Hijiya N, Ho RH, Hofmann I, Huang JN, Keel S, Lamble A, Lau BW, Norkin M, Stieglitz E, Stock W, Walkovich K, Boettcher S, Brendel C, Fleming MD, Davies SM, Weller EA, Bahl C, Carter SL, Shimamura A, Lindsley RC
Nature communications 2021 Feb 26;12(1):1334
Nature communications 2021 Feb 26;12(1):1334
ARPC1B binds WASP to control actin polymerization and curtail tonic signaling in B cells.
Leung G, Zhou Y, Ostrowski P, Mylvaganam S, Boroumand P, Mulder DJ, Guo C, Muise AM, Freeman SA
JCI insight 2021 Dec 8;6(23)
JCI insight 2021 Dec 8;6(23)
STIM1 Controls the Focal Adhesion Dynamics and Cell Migration by Regulating SOCE in Osteosarcoma.
Lin YS, Lin YH, Nguyen Thi M, Hsiao SC, Chiu WT
International journal of molecular sciences 2021 Dec 23;23(1)
International journal of molecular sciences 2021 Dec 23;23(1)
The Effects of Syndecan on Osteoblastic Cell Adhesion Onto Nano-Zirconia Surface.
Sun L, Hong G, Matsui H, Song YJ, Sasaki K
International journal of nanomedicine 2020;15:5061-5072
International journal of nanomedicine 2020;15:5061-5072
Poly(ADP-ribose) Glycohydrolase Inhibition Sequesters NAD(+) to Potentiate the Metabolic Lethality of Alkylating Chemotherapy in IDH-Mutant Tumor Cells.
Nagashima H, Lee CK, Tateishi K, Higuchi F, Subramanian M, Rafferty S, Melamed L, Miller JJ, Wakimoto H, Cahill DP
Cancer discovery 2020 Nov;10(11):1672-1689
Cancer discovery 2020 Nov;10(11):1672-1689
Succinic Semialdehyde Dehydrogenase Deficiency: In Vitro and In Silico Characterization of a Novel Pathogenic Missense Variant and Analysis of the Mutational Spectrum of ALDH5A1.
Brennenstuhl H, Didiasova M, Assmann B, Bertoldi M, Molla G, Jung-Klawitter S, Kuseyri Hübschmann O, Schröter J, Opladen T, Tikkanen R
International journal of molecular sciences 2020 Nov 13;21(22)
International journal of molecular sciences 2020 Nov 13;21(22)
A tumor-intrinsic PD-L1/NLRP3 inflammasome signaling pathway drives resistance to anti-PD-1 immunotherapy.
Theivanthiran B, Evans KS, DeVito NC, Plebanek M, Sturdivant M, Wachsmuth LP, Salama AK, Kang Y, Hsu D, Balko JM, Johnson DB, Starr M, Nixon AB, Holtzhausen A, Hanks BA
The Journal of clinical investigation 2020 May 1;130(5):2570-2586
The Journal of clinical investigation 2020 May 1;130(5):2570-2586
Senescent mesenchymal stem cells remodel extracellular matrix driving breast cancer cells to a more-invasive phenotype.
Ghosh D, Mejia Pena C, Quach N, Xuan B, Lee AH, Dawson MR
Journal of cell science 2020 Jan 23;133(2)
Journal of cell science 2020 Jan 23;133(2)
A fluorescence-activatable reporter of flavivirus NS2B-NS3 protease activity enables live imaging of infection in single cells and viral plaques.
Arias-Arias JL, MacPherson DJ, Hill ME, Hardy JA, Mora-Rodríguez R
The Journal of biological chemistry 2020 Feb 21;295(8):2212-2226
The Journal of biological chemistry 2020 Feb 21;295(8):2212-2226
Increased talin-vinculin spatial proximities in livers in response to spotted fever group rickettsial and Ebola virus infections.
Liu Y, Xiao J, Zhang B, Shelite TR, Su Z, Chang Q, Judy B, Li X, Drelich A, Bei J, Zhou Y, Zheng J, Jin Y, Rossi SL, Tang SJ, Wakamiya M, Saito T, Ksiazek T, Kaphalia B, Gong B
Laboratory investigation; a journal of technical methods and pathology 2020 Aug;100(8):1030-1041
Laboratory investigation; a journal of technical methods and pathology 2020 Aug;100(8):1030-1041
IDH-mutant gliomas harbor fewer regulatory T cells in humans and mice.
Richardson LG, Nieman LT, Stemmer-Rachamimov AO, Zheng XS, Stafford K, Nagashima H, Miller JJ, Kiyokawa J, Ting DT, Wakimoto H, Cahill DP, Choi BD, Curry WT
Oncoimmunology 2020 Aug 20;9(1):1806662
Oncoimmunology 2020 Aug 20;9(1):1806662
Culture and Differentiation of Human Hair Follicle Dermal Papilla Cells in a Soft 3D Self-Assembling Peptide Scaffold.
Betriu N, Jarrosson-Moral C, Semino CE
Biomolecules 2020 Apr 28;10(5)
Biomolecules 2020 Apr 28;10(5)
Mechano-signalling, induced by fullerene C(60) nanofilms, arrests the cell cycle in the G2/M phase and decreases proliferation of liver cancer cells.
Sosnowska M, Kutwin M, Jaworski S, Strojny B, Wierzbicki M, Szczepaniak J, Łojkowski M, Święszkowski W, Bałaban J, Chwalibog A, Sawosz E
International journal of nanomedicine 2019;14:6197-6215
International journal of nanomedicine 2019;14:6197-6215
Minute-scale persistence of a GPCR conformation state triggered by non-cognate G protein interactions primes signaling.
Gupte TM, Ritt M, Dysthe M, Malik RU, Sivaramakrishnan S
Nature communications 2019 Oct 23;10(1):4836
Nature communications 2019 Oct 23;10(1):4836
A divalent siRNA chemical scaffold for potent and sustained modulation of gene expression throughout the central nervous system.
Alterman JF, Godinho BMDC, Hassler MR, Ferguson CM, Echeverria D, Sapp E, Haraszti RA, Coles AH, Conroy F, Miller R, Roux L, Yan P, Knox EG, Turanov AA, King RM, Gernoux G, Mueller C, Gray-Edwards HL, Moser RP, Bishop NC, Jaber SM, Gounis MJ, Sena-Esteves M, Pai AA, DiFiglia M, Aronin N, Khvorova A
Nature biotechnology 2019 Aug;37(8):884-894
Nature biotechnology 2019 Aug;37(8):884-894
16-Hydroxycleroda-3,13-dien-15,16-olide induces anoikis in human renal cell carcinoma cells: involvement of focal adhesion disassembly and signaling.
Chen YC, Huang BM, Lee WC, Chen YC
OncoTargets and therapy 2018;11:7679-7690
OncoTargets and therapy 2018;11:7679-7690
Transition of responsive mechanosensitive elements from focal adhesions to adherens junctions on epithelial differentiation.
Noethel B, Ramms L, Dreissen G, Hoffmann M, Springer R, Rübsam M, Ziegler WH, Niessen CM, Merkel R, Hoffmann B
Molecular biology of the cell 2018 Sep 15;29(19):2317-2325
Molecular biology of the cell 2018 Sep 15;29(19):2317-2325
Delineating the role of cooperativity in the design of potent PROTACs for BTK.
Zorba A, Nguyen C, Xu Y, Starr J, Borzilleri K, Smith J, Zhu H, Farley KA, Ding W, Schiemer J, Feng X, Chang JS, Uccello DP, Young JA, Garcia-Irrizary CN, Czabaniuk L, Schuff B, Oliver R, Montgomery J, Hayward MM, Coe J, Chen J, Niosi M, Luthra S, Shah JC, El-Kattan A, Qiu X, West GM, Noe MC, Shanmugasundaram V, Gilbert AM, Brown MF, Calabrese MF
Proceedings of the National Academy of Sciences of the United States of America 2018 Jul 31;115(31):E7285-E7292
Proceedings of the National Academy of Sciences of the United States of America 2018 Jul 31;115(31):E7285-E7292
Human snoRNA-93 is processed into a microRNA-like RNA that promotes breast cancer cell invasion.
Patterson DG, Roberts JT, King VM, Houserova D, Barnhill EC, Crucello A, Polska CJ, Brantley LW, Kaufman GC, Nguyen M, Santana MW, Schiller IA, Spicciani JS, Zapata AK, Miller MM, Sherman TD, Ma R, Zhao H, Arora R, Coley AB, Zeidan MM, Tan M, Xi Y, Borchert GM
NPJ breast cancer 2017;3:25
NPJ breast cancer 2017;3:25
Diamond, graphite, and graphene oxide nanoparticles decrease migration and invasiveness in glioblastoma cell lines by impairing extracellular adhesion.
Wierzbicki M, Jaworski S, Kutwin M, Grodzik M, Strojny B, Kurantowicz N, Zdunek K, Chodun R, Chwalibog A, Sawosz E
International journal of nanomedicine 2017;12:7241-7254
International journal of nanomedicine 2017;12:7241-7254
Biosynthetic PCL-graft-Collagen Bulk Material for Tissue Engineering Applications.
Gentile P, McColgan-Bannon K, Gianone NC, Sefat F, Dalgarno K, Ferreira AM
Materials (Basel, Switzerland) 2017 Jun 23;10(7)
Materials (Basel, Switzerland) 2017 Jun 23;10(7)
Integrin-mediated traction force enhances paxillin molecular associations and adhesion dynamics that increase the invasiveness of tumor cells into a three-dimensional extracellular matrix.
Mekhdjian AH, Kai F, Rubashkin MG, Prahl LS, Przybyla LM, McGregor AL, Bell ES, Barnes JM, DuFort CC, Ou G, Chang AC, Cassereau L, Tan SJ, Pickup MW, Lakins JN, Ye X, Davidson MW, Lammerding J, Odde DJ, Dunn AR, Weaver VM
Molecular biology of the cell 2017 Jun 1;28(11):1467-1488
Molecular biology of the cell 2017 Jun 1;28(11):1467-1488
Decellularized extracellular matrix of human umbilical vein endothelial cells promotes endothelial differentiation of stem cells from exfoliated deciduous teeth.
Gong T, Heng BC, Xu J, Zhu S, Yuan C, Lo EC, Zhang C
Journal of biomedical materials research. Part A 2017 Apr;105(4):1083-1093
Journal of biomedical materials research. Part A 2017 Apr;105(4):1083-1093
Human fibroblasts display a differential focal adhesion phenotype relative to chimpanzee.
Advani AS, Chen AY, Babbitt CC
Evolution, medicine, and public health 2016;2016(1):110-6
Evolution, medicine, and public health 2016;2016(1):110-6
Angiotensin receptors and β-catenin regulate brain endothelial integrity in malaria.
Gallego-Delgado J, Basu-Roy U, Ty M, Alique M, Fernandez-Arias C, Movila A, Gomes P, Weinstock A, Xu W, Edagha I, Wassmer SC, Walther T, Ruiz-Ortega M, Rodriguez A
The Journal of clinical investigation 2016 Oct 3;126(10):4016-4029
The Journal of clinical investigation 2016 Oct 3;126(10):4016-4029
Effects of decellularized matrices derived from periodontal ligament stem cells and SHED on the adhesion, proliferation and osteogenic differentiation of human dental pulp stem cells in vitro.
Heng BC, Zhu S, Xu J, Yuan C, Gong T, Zhang C
Tissue & cell 2016 Apr;48(2):133-43
Tissue & cell 2016 Apr;48(2):133-43
A multi-omics approach identifies key hubs associated with cell type-specific responses of airway epithelial cells to staphylococcal alpha-toxin.
Richter E, Harms M, Ventz K, Gierok P, Chilukoti RK, Hildebrandt JP, Mostertz J, Hochgräfe F
PloS one 2015;10(3):e0122089
PloS one 2015;10(3):e0122089
Connectivity Map-based discovery of parbendazole reveals targetable human osteogenic pathway.
Brum AM, van de Peppel J, van der Leije CS, Schreuders-Koedam M, Eijken M, van der Eerden BC, van Leeuwen JP
Proceedings of the National Academy of Sciences of the United States of America 2015 Oct 13;112(41):12711-6
Proceedings of the National Academy of Sciences of the United States of America 2015 Oct 13;112(41):12711-6
Susceptibility to tuberculosis is associated with variants in the ASAP1 gene encoding a regulator of dendritic cell migration.
Curtis J, Luo Y, Zenner HL, Cuchet-Lourenço D, Wu C, Lo K, Maes M, Alisaac A, Stebbings E, Liu JZ, Kopanitsa L, Ignatyeva O, Balabanova Y, Nikolayevskyy V, Baessmann I, Thye T, Meyer CG, Nürnberg P, Horstmann RD, Drobniewski F, Plagnol V, Barrett JC, Nejentsev S
Nature genetics 2015 May;47(5):523-527
Nature genetics 2015 May;47(5):523-527
Force engages vinculin and promotes tumor progression by enhancing PI3K activation of phosphatidylinositol (3,4,5)-triphosphate.
Rubashkin MG, Cassereau L, Bainer R, DuFort CC, Yui Y, Ou G, Paszek MJ, Davidson MW, Chen YY, Weaver VM
Cancer research 2014 Sep 1;74(17):4597-611
Cancer research 2014 Sep 1;74(17):4597-611
Dual-objective STORM reveals three-dimensional filament organization in the actin cytoskeleton.
Xu K, Babcock HP, Zhuang X
Nature methods 2012 Jan 8;9(2):185-8
Nature methods 2012 Jan 8;9(2):185-8
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
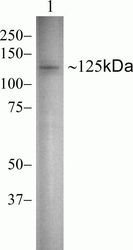
- Experimental details
- Western blot analysis of Vinculin in HeLa cell lysate using a Vinculin recombinant rabbit monoclonal antibody (Product # 700062) at a dilution of 0.5 µg/mL.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of Vinculin was performed by loading 20 µg of HeLa (lane1) and NIH/3T3 (lane2) cell lysates using Novex®NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX), XCell SureLock Electrophoresis System (Product # EI0002), Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Proteins were transferred to a PVDF membrane and blocked with 5 % skim milk for 1 hour at room temperature. Vinculin was detected at ~125 kDa using Vinculin Recombinant Rabbit Monoclonal Antibody (Product # 700062) at 1 µg-3 µg/mL in 2.5 % skim milk at 4°C overnight on a rocking platform. Goat anti-Rabbit IgG-HRP Secondary Antibody (Product # G-21234) at 1:5000 dilution was used and chemiluminescent detection was performed using Pierce™ ECL Western blotting Substrate (Product # 32106).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
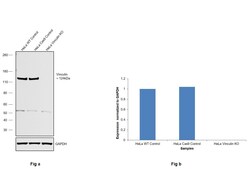
- Experimental details
- Knockout of Vinculin was achieved by CRISPR-Cas9 genome editing using LentiArray™ Lentiviral sgRNA (Product # A32042, Assay ID CRISPR948199_LV) and LentiArray Cas9 Lentivirus (Product # A32064). Western blot analysis of Vinculin was performed by loading 30 µg of HeLa Wild Type (Lane 1), HeLa Cas9 (Lane 2) andHeLa Vinculin KO (Lane 3) membrane enriched extracts. The samples were electrophoresed using NuPAGE™ Novex™ 4-12% Bis-Tris Protein Gel (Product # NP0322BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with Anti-Vinculin Recombinant Rabbit Monoclonal Antibody (42H89L44) (Product # 700062, 1:500 dilution) and Goat anti-Rabbit IgG (Heavy Chain) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036, 1:6,000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005). Loss of signal upon CRISPR mediated knockout (KO) using the LentiArray™ CRISPR product line confirms that antibody is specific to Vinculin. Anuncharacterized band observed at ~55 kDa in all the samples.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-Vinculin Recombinant Rabbit Monoclonal Antibody (42H89L44)(Product # 700062) and a 124 kDa band corresponding to Vinculin was observed across cell lines and tissues tested. Membrane enriched extracts (30 µg lysate) of HeLa (Lane 1), NIH/3T3 (Lane 2), U-87 MG (Lane 3), PC-3 (Lane 4), MCF-7 (Lane 5), MOLT-4 (Lane 6), Jurkat (Lane 7). Tissue extracts of Mouse Lung (Lane 8), Rat Lung (Lane 9), Mouse Brain (Lane 10), Rat Brain (Lane 11) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0321BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1 µg/mL) and detected by chemiluminescence with Goat anti-Rabbit IgG (Heavy Chain) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036,1:4,000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
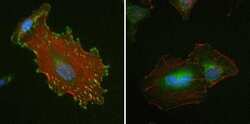
- Experimental details
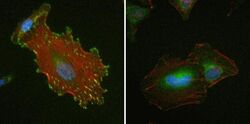
- Immunofluorescent analysis of Vinculin in HeLa cells using a Vinculin recombinant rabbit monoclonal antibody (Product # 700062) at a dilution of 1 µg/mL (left) or preincubated with the recombinant protein used as an immunogen (right), followed by detection using an Alexa Fluor 488-conjugated goat anti-rabbit secondary antibody at a dilution of 1:1000. Nuclei are stained with Hoescht (blue) and actin stained a fluorescent red phalloidin. Note punctate staining at cell-cell and cell-matrix junctions.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
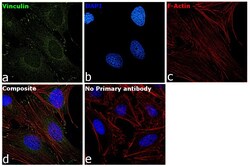
- Experimental details
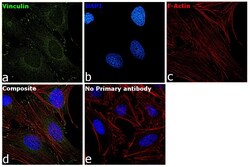
- Immunofluorescence analysis of Vinculin was performed using 70 confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with Vinculin Recombinant Rabbit Monoclonal Antibody (42H89L44) (Product # 700062) at 5 µg/mL in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Donkey anti-Rabbit IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32790), (1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b:Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300 dilution). Panel d represents the merged image showing cytoskeleton along with focal adhesion signal which is also reported localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
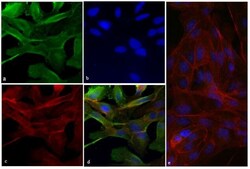
- Immunofluorescent analysis of Vinculin was done on 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 15 minutes; permeabilized with 0.25% Triton X-100 for 10 minutes followed by blocking with 5% BSA for 1 hour at room temperature. The cells were incubated with Vinculin Recombinant Rabbit Monoclonal Antibody (Product # 700062) at 2 µg-4 µg in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor® 488 Goat anti-Rabbit IgG Secondary Antibody (Product # A-11008) at a dilution of 1:400 for 30 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 594 Phalloidin (Product # A12381). Panel d is a merged image showing junction and cytoplasmic localization of Vinculin. Panel e shows no primary antibody control. The images were captured at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
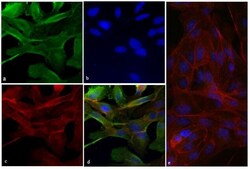
- Immunofluorescent analysis of Vinculin was done on 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 15 minutes; permeabilized with 0.25% Triton X-100 for 10 minutes followed by blocking with 5% BSA for 1 hour at room temperature. The cells were incubated with Vinculin Recombinant Rabbit Monoclonal Antibody (Product # 700062) at 2 µg-4 µg in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor® 488 Goat anti-Rabbit IgG Secondary Antibody (Product # A-11008) at a dilution of 1:400 for 30 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 594 Phalloidin (Product # A12381). Panel d is a merged image showing junction and cytoplasmic localization of Vinculin. Panel e shows no primary antibody control. The images were captured at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Immunofluorescent analysis of Vinculin in HeLa cells using a Vinculin recombinant rabbit monoclonal antibody (Product # 700062) at a dilution of 1 µg/mL (left) or preincubated with the recombinant protein used as an immunogen (right), followed by detection using an Alexa Fluor 488-conjugated goat anti-rabbit secondary antibody at a dilution of 1:1000. Nuclei are stained with Hoescht (blue) and actin stained a fluorescent red phalloidin. Note punctate staining at cell-cell and cell-matrix junctions.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Immunofluorescence analysis of Vinculin was performed using 70 confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with Vinculin Recombinant Rabbit Monoclonal Antibody (42H89L44) (Product # 700062) at 5 µg/mL in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Donkey anti-Rabbit IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32790), (1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b:Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300 dilution). Panel d represents the merged image showing cytoskeleton along with focal adhesion signal which is also reported localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
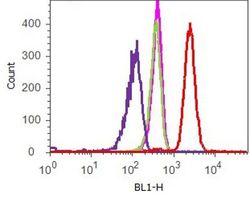
- Flow cytometry analysis of Vinculin was done on HeLa cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Tritonª X-100 for 20 minutes, and blocked with 5% BSA for 1 hour at room temperature. Cells were labeled with ABfinityª Vinculin Recombinant Rabbit Monoclonal Antibody (700062, red histogram) or with rabbit isotype control (pink histogram) at 2 µg-4 µg/million cells in 2.5% BSA. After incubation at room temperature for 2-3 hours, the cells were labeled with Alexa Fluor¨ 488 Goat Anti-Rabbit Secondary Antibody (A11008) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune¨ Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
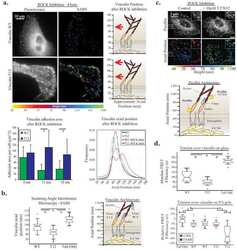
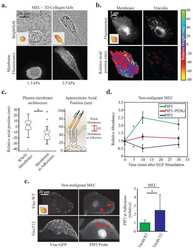
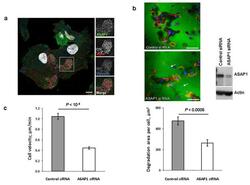
- Figure 3 In DCs, the ASAP1 protein is partly associated with podosomes and its depletion leads to impaired matrix degradation and migration ( a ) Confocal microscopy showing ASAP1 (green), actin (red), vinculin (blue) and nuclei (white) in DCs derived from monocytes of a healthy volunteer. Podosomes are structures characterized by the actin core (visible as red dots) surrounded by other proteins. Boxed area is shown in grayscale enlarged on the right. Scale bar represents 7.5mum. ( b ) Representative confocal microscopy images (left panel) showing monocyte-derived DCs treated with either ASAP1 or control siRNA and seeded on FITC-conjugated gelatine (green) for 4 hours. The cells were stained to visualize actin (phalloidin-red) and nuclei (DAPI-blue). White arrows show examples of the DAPI-stained nuclei of cells that do not degrade matrix. Scale bar represents 50mum. In the same experiment cell lysates were collected for western blotting and probed with anti-ASAP1 and anti-actin antibodies (right panel) to assess knockdown efficiency. Area of degradation of the FITC-conjugated gelatine matrix by DCs (lower panel). In total 394 and 317 DCs treated with either ASAP1 or control siRNA, respectively, were counted in 4 independent experiments. ( c ) Migration velocity of DCs in a Dunn chamber. In total 162 and 186 DCs treated with either ASAP1 or control siRNA, respectively, were tracked in 5 independent experiments. Graphs show mean values +- SEM. P -values were calculated using t
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
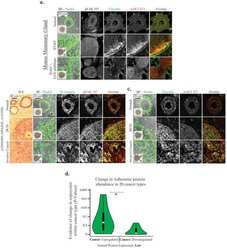
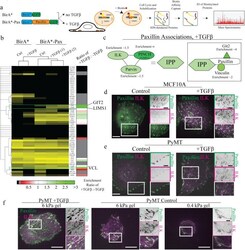
- FIGURE 8: Proteomics identifies unique proteins recruited to paxillin in the integrin adhesions during EMT. (a) BioID technique. A control BirA*-eGFP construct and a BirA*-paxillin-eGFP construct are stably expressed in MCF10A cells, which are then treated with TGFbeta to induce EMT. BirA*-paxillin-GFP localizes to FAs and biotinylates proteins adjacent to paxillin when biotin is added to the medium. Biotinylated proteins are isolated and identified by mass spectrometry. (b) Normalized spectral counts for the consensus adhesome were clustered on the basis of an uncentered Pearson correlation using Cluster 3.0 and visualized using Java TreeView. From a subgroup of known paxillin interactors, we identify GIT2, LIMS1 (PINCH), and vinculin as enriched in TGFbeta-treated cells. Black indicates no protein detected in the +TGFbeta:-TGFbeta scale bar, and gray indicates no enrichment relative to the control. (c) Model for potential paxillin associations altered in response to TGFbeta treatment. PINCH, GIT2, and vinculin localize adjacent to paxillin, and these associations are enriched in TGFbeta-treated cells. (d-f) Immunofluorescence images of paxillin (green) and ILK (magenta). ILK is enriched in paxillin-rich adhesions in (d) MCF10A cells treated with TGFbeta and (e) PyMT cells treated with TGFbeta. Scale bar, 10 um (main image), 5 um (inset). (f) Immunofluorescence images of paxillin and ILK in PyMT cells on stiff (6 kPa) and soft (0.4 kPa) PA gels coated with fibronectin, showi
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
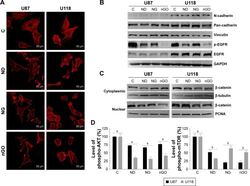
- Figure 5 EGFR/AKT/mTOR and beta-catenin signaling in glioblastoma cells after treated with nanoparticles. Notes: ( A ) Confocal microscope images of U87 and U118 cells actin cytoskeleton. Cells were grown on extracellular matrix for 24 h and treated with diamond nanoparticles, graphite nanoparticles, or graphene oxide nanoparticles at a concentration of 20 mug/mL and incubated for 24 h. F-Actin was stained with phalloidin conjugated with Atto 633. ( B ) Western blot analysis of N-cadherin, pan-cadherin, vinculin, p-EGFR, and EGFR. GAPDH was used as a loading control. ( C ) Western blot analysis of nuclear and cytoplasmic protein fractions used for determination of beta-catenin protein level. PCNA and beta-tubulin were used as loading controls for nuclear and cytoplasmic fractions, respectively. ( D ) ELISA analysis of AKT and mTOR phosphorylation in comparison to control. Treatment with nanoparticles significantly reduced phospho-AKT ( P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
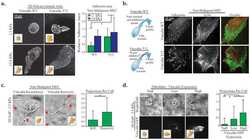
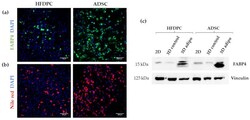
- Figure 9 Characterization of adipogenic phenotype of HFDPC and ADSC cultured in RAD16-I scaffold for 21 days. ( a ) FABP4 immunofluorescence of 3D constructs under adipogenic induction; ( b ) Nile red staining of 3D constructs under adipogenic induction; ( c ) FABP4 western blot of HFDPC and ADSC cultured in 2D cultures and in 3D RAD16-I scaffold under control and adipogenic medium. Vinculin was used as an internal control. The image is representative of three replicates.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
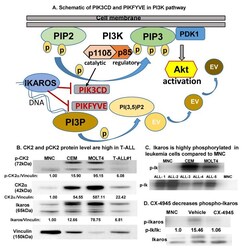
- Figure 1 CK2 is overexpressed in T-ALL compared to normal mononuclear cells . ( A ) Schematic showing PIK3CD and PIKFYVE genes in the PI3K pathway. ( B ) Baseline protein levels of CK2alpha, pCK2, and IKAROS in the T cell leukemia cell panel (CCRF-CEM, MOLT4, and primary T-ALL cells (labeled T-ALL#1)) were measured by Western blot and compared to peripheral blood mononuclear cells (MNCs). The protein level is graphed relative to vinculin as a loading control. ( C ) Radio-immunoblot showing phospho-IKAROS in a leukemia cell panel (CCRF-CEM, MOLT4, and primary T-ALL cells (labeled ALL#1-5)) compared to MNCs. ( D ) Radio-immunoblot showing a decrease in the phospho-IKAROS level following CX-4945 treatment. MOLT4 cells were treated with 10 muM of CX-4945 for 24 h. EV, endosomal vesicle; PIK3CD, phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta; and PIKFYVE, phosphoinositide kinase, FYVE-type zinc finger containing; PI3K, phosphoinositide 3-kinase; P, phospho; PIP2, phosphatidylinositol 4,5-bisphosphate; PIP3, phosphatidylinositol 3,4,5-trisphosphate; PDK1, phosphoinositide-dependent kinase; PI3P, phosphatidylinositol-3-phosphate; PI(3,5)P2, phosphotydile inositol 3,5-biphosphate.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
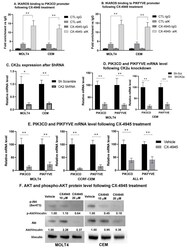
- Figure 4 CK2 inhibition restores IKAROS DNA binding and repression of PIK3CD and PIKFYVE . MOLT4, CEM, and T-ALL#1 cells were treated with 10 muM of CX-4945 for 24 h. IKAROS binding to the ( A ) PIK3CD and ( B ) PIKFYVE promoter region was confirmed using qChIP assay in vehicle- and CX-4945-treated cells. Results are mean +- SD of triplicates representative of one of three independent experiments. Molecular inhibition of CK2alpha in CEM and MOLT4 cells was achieved using shRNA. Two of four shRNAs showed a significant and similar decrease in CK2alpha. qRT-PCR shows the mRNA level of ( C ) CK2alpha and ( D ) PIK3CD and PIKFYVE in CK2-silenced MOLT4 (left panel) and CEM (right panel) cells. CEM and MOLT4 cells were treated with 10 and 20 muM of CX-4945 for 48 h, and T-ALL#1 primary leukemia cells were treated with 10 muM of CX-4945 for 12 h. mRNA and protein were extracted. ( E ) The mRNA level of PIK3CD and PIKFYVE was measured in CX-4945-treated CEM, MOLT4, and T-ALL#1 cells. ( F ) AKT and phosphorylated-AKT (p-AKT) protein levels were measured by Western blot. The protein level is expressed relative to vinculin. The p -value summaries are as follows: p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
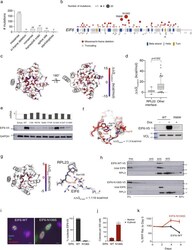
- Fig. 2 EIF6 somatic missense mutations alter EIF6 protein stability or function to improve cell fitness. a Types of somatic EIF6 mutations. b Number and location of EIF6 mutations according to variant type. c Impact on the calculated energy of the folded state (DeltaDelta G mutation ) of EIF6 missense mutations. Mutant residue colored according to DeltaDelta G value. d DeltaDelta G mutation of 12 EIF6 missense mutations located at the RPL23-binding interface versus 85 other EIF6 missense mutations located in the remainder of EIF6 . Boxes center around the median and span the 25th and 75th percentiles, whiskers extend to the 10th and 90th percentiles. p value calculated using unpaired two tailed t -test. e Relative levels of EIF6 mRNA using a V5-specific qPCR primer (top panel) and V5 immunoblot from K562 cells 48 h after doxycycline treatment (bottom panel). Data shown is representative of three independent experiments. f Left panel: In silico modeling of EIF6-R96W. Right panel: V5 and VCL immunoblots of K562 cells with inducible EIF6-R96W versus V5-wild type EIF6 48 h after doxycycline treatment. Data shown is representative of three independent experiments. g Left panel: Change in the energy of binding (DeltaDelta G bind ) of missense mutations at RPL23 interface. Mutant residues are colored according to DeltaDelta G bind . Right panel: In silico modeling of EIF6 N106S mutation. h V5, EIF6, and RPL3 immunoblots of sucrose gradient fractions from polysome profiles of doxycyc
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
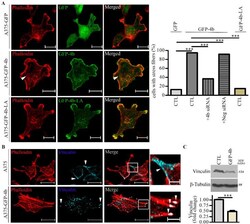
- Figure 3 Lamellipodia and stress fiber formation are increased in A375 cells expressing GFP-PMCA4b but not in those expressing GFP-PMCA4b-LA. ( A ) Confocal microscopy images of A375-GFP, A375-GFP-PMCA4b, and A375-GFP-PMCA4b-LA cells labeled with Phalloidin-TRITC. Arrowheads show stress fibers. Scale bar, 20 um. The fractions of stress fiber-positive cells in A375-GFP ( n = 55), A375-GFP-PMCA4b-LA ( n = 72), A375-GFP-PMCA4b ( n = 60) +- PMCA4b siRNA ( n = 68), and negative siRNA ( n = 49) cultures were determined. siRNA confocal microscopy images are presented in Figure S3A . Relative abundance of cells with stress fibers is indicated by the bar graph. Confocal sections were taken from the bottom of the cells to show the stress fibers. ( B ) Confocal microscopy images of A375 and A375-GFP-PMCA4b cells immunostained with vinculin and labeled with Phalloidin-TRITC. Scale bar, 20 um. Insets show part of the cells with higher magnification. Arrowheads show the differential localization of vinculin. Scale bar, 5 um. ( C ) A375 and A375-GFP-PMCA4b cells were cultured in a 6-well plate for 48 h. Vinculin protein expression from total cell lysate was analyzed by Western blotting. beta-tubulin was used as a loading control. Data represent mean +- S.E.M from three independent experiments (*** p < 0.001).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
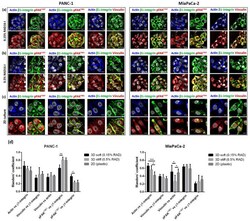
- Experimental details
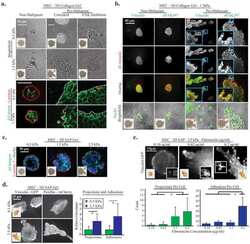
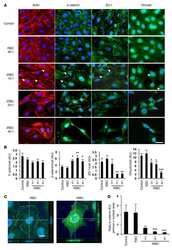
- Immunofluorescence analysis of signal mechanotransduction proteins. Actin (blue) beta1-integrin (green), pFAK Y397 (red), pFAK Y861 (red) and vinculin (red) staining in PANC-1 (left) and MiaPaCa-2 (right) cells in ( a ) 0.15% RAD16-I; ( b ) 0.5% RAD16-I and ( c ) 2D cultures. Merge of green and red channels is also shown. Scale bars represent 10 um; ( d ) Mander's colocalization coefficients of PANC-1 and MiaPaCa-2 cells (n = 5). Statistical differences are indicated as * for p value < 0.05, ** for p value < 0.01 and *** for p value < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
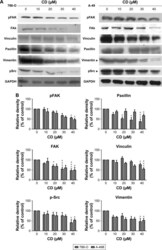
- Experimental details
- Figure 6 ( A ) Immunoblots of FAK-related signaling molecules. The figures shown are representative of one experiment. ( B ) The bar graphs show densitometric data (mean+-SD) from three independent experiments. Note: * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
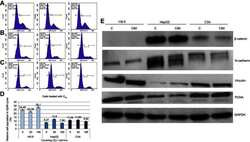
- Experimental details
- Figure 3 Propidium iodide (PI) 488 assay analysis. Notes: Effect of C 60 on the number (percentage) of ( A ) HS-5, ( B ) HepG2 or ( C ) C3A cells in the cycle phases. ( D ) Relative HS-5, HepG2 and C3A cell population in G2/M cycle (%). ( E ) Western blot analysis of beta-catenin, N-cadherin, vinculin and PCNA. GAPDH was used as a loading control. Abbreviations: C 60 , fullerenes; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; PCNA, proliferating cell nuclear antigen.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
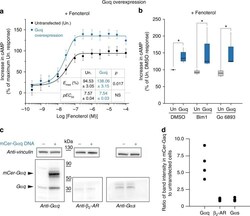
- Experimental details
- Fig. 7 Galphaq overexpression enhances cAMP response of endogenous beta 2 -AR. a cAMP accumulation in HEK293T cells either overexpressing Galphaq (mCer-tag) or an equivalent number of untransfected control cells following stimulation with increasing concentration of fenoterol (0.3 nM to 100 muM) in the presence of 2 muM forskolin (see Methods). The increase in cAMP with fenoterol concentration was fit to a four-parameter logistic function to estimate EC 50 and E max (Methods). Values are mean +- SD from six experiments with at least three repeats per experiment ( n >= 6). Statistically significant differences in the EC 50 and E max values were assessed by Students' t -test. b cAMP accumulation in HEK293T cells either overexpressing Galphaq (mCer-tag) or equivalent number of untransfected control cells, following stimulation with fenoterol (10 muM) and forskolin (2 muM), in the presence of PKC inhibitors Bim1 (1 muM) or Go6983 (1 muM), or DMSO. Cells overexpressing Galphaq had accumulated greater cAMP even in the presence of PKC inhibitors. The drug treatments do not influence the cAMP response across the three untransfected or the three transfected conditions. Box-and-whisker plots: center line is median, box ends are upper and lower quartiles, whisker ends are 1.5 x IQR (interquartile range) from four independent experiments with three technical replicates per experiment ( n >= 4). Statistically significant differences were assessed by two-way ANOVA followed by Tukey's post-
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
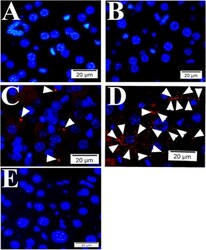
- Experimental details
- Fig. 2 Quality controls for PLA in mouse tissue using primary antibodies raised in mouse. During PLA assay on mouse liver tissue samples from SFGR infection mouse on day 19, there is no specific signals if no primary antibody employed (only normal mouse and rabbit IgGs) ( a ), using rabbit anti-vinculin antibodies and normal mouse IgG employed ( b ), using rabbit anti-vinculin antibodies and mouse anti-talin, but no oligonucleotide probes ( e ). There is specific signal detected when rabbit anti-vinculin and mouse anti-talin antibodies were employed during PLA on mouse liver tissue ( c , d ). Nuclei were counter-stained with DAPI (blue). Scale bars indicate 20 um.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunohistochemistry
Immunohistochemistry