12-6644-42
antibody from Invitrogen Antibodies
Targeting: TNFRSF10A
Apo2, CD261, DR4, TRAILR-1, TRAILR1
Antibody data
- Antibody Data
- Antigen structure
- References [25]
- Comments [0]
- Validations
- Flow cytometry [2]
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 12-6644-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD261 (DR4) Monoclonal Antibody (DJR1), PE, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The DJR1 monoclonal antibody reacts with human DR4, also known as TRAIL-R1. DR4 binds to TRAIL, activates NF-kappaB, and induces TRAIL-mediated apoptosis. DR4 is expressed by a variety of human tumor cells and at very low level on human leukocytes. Expression has also been found inside the cell on some cell lines (Zhang et al). This has been confirmed with intracellular staining of Jurkat cell line. Please contact tech@ebioscience.com for more information. Applications Reported: The DJR1 antibody has been reported for use in flow cytometric analysis. Applications Tested: This DJR1 antibody has been pre-titrated and tested by flow cytometric analysis of human DR4-transfected cells. This can be used at 5 µL (0.5 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488-561 nm; Emission: 578 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Yellow dye
- Isotype
- IgG
- Antibody clone number
- DJR1
- Vial size
- 100 Tests
- Concentration
- 5 μL/Test
- Storage
- 4°C, store in dark, DO NOT FREEZE!
Submitted references Doxorubicin sensitizes breast cancer cells to natural killer cells in connection with increased Fas receptors.
Genistein Sensitizes Human Cholangiocarcinoma Cell Lines to Be Susceptible to Natural Killer Cells.
Systematic genetic mapping of necroptosis identifies SLC39A7 as modulator of death receptor trafficking.
Regulation of human glioblastoma cell death by combined treatment of cannabidiol, γ-radiation and small molecule inhibitors of cell signaling pathways.
Dual Agonist Surrobody Simultaneously Activates Death Receptors DR4 and DR5 to Induce Cancer Cell Death.
Potential Therapeutic Effect of Natural Killer Cells on Doxorubicin-Resistant Breast Cancer Cells In Vitro.
Combination of systemic chemotherapy with local stem cell delivered S-TRAIL in resected brain tumors.
Increased growth-inhibitory and cytotoxic activity of arsenic trioxide in head and neck carcinoma cells with functional p53 deficiency and resistance to EGFR blockade.
TRAIL-mediated apoptosis in breast cancer cells cultured as 3D spheroids.
MADD knock-down enhances doxorubicin and TRAIL induced apoptosis in breast cancer cells.
The oncogene metadherin modulates the apoptotic pathway based on the tumor necrosis factor superfamily member TRAIL (Tumor Necrosis Factor-related Apoptosis-inducing Ligand) in breast cancer.
Real-time imaging of the dynamics of death receptors and therapeutics that overcome TRAIL resistance in tumors.
A whole-genome RNAi screen identifies an 8q22 gene cluster that inhibits death receptor-mediated apoptosis.
Molecular identification of an MHC-independent ligand recognized by a human {alpha}/{beta} T-cell receptor.
Predominant antitumor effects by fully human anti-TRAIL-receptor 2 (DR5) monoclonal antibodies in human glioma cells in vitro and in vivo.
c-Jun NH2-terminal kinase-dependent upregulation of DR5 mediates cooperative induction of apoptosis by perifosine and TRAIL.
TRAIL death receptor-4, decoy receptor-1 and decoy receptor-2 expression on CD8+ T cells correlate with the disease severity in patients with rheumatoid arthritis.
CAAT/enhancer binding protein homologous protein-dependent death receptor 5 induction is a major component of SHetA2-induced apoptosis in lung cancer cells.
Apoptotic activity and mechanism of 2-cyano-3,12-dioxoolean-1,9-dien-28-oic-acid and related synthetic triterpenoids in prostate cancer.
The farnesyltransferase inhibitor lonafarnib induces CCAAT/enhancer-binding protein homologous protein-dependent expression of death receptor 5, leading to induction of apoptosis in human cancer cells.
Human astrocytes are resistant to Fas ligand and tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis.
Enhancement of Ad5-TRAIL cytotoxicity against renal cell carcinoma with histone deacetylase inhibitors.
Difference in cytotoxicity against hepatocellular carcinoma between liver and periphery natural killer cells in humans.
Synthetic triterpenoids cooperate with tumor necrosis factor-related apoptosis-inducing ligand to induce apoptosis of breast cancer cells.
c-Jun NH2-terminal kinase-mediated up-regulation of death receptor 5 contributes to induction of apoptosis by the novel synthetic triterpenoid methyl-2-cyano-3,12-dioxooleana-1, 9-dien-28-oate in human lung cancer cells.
Sawasdee N, Wattanapanitch M, Thongsin N, Phanthaphol N, Chiawpanit C, Thuwajit C, Yenchitsomanus PT, Panya A
International journal of molecular medicine 2022 Mar;49(3)
International journal of molecular medicine 2022 Mar;49(3)
Genistein Sensitizes Human Cholangiocarcinoma Cell Lines to Be Susceptible to Natural Killer Cells.
Chiawpanit C, Panwong S, Sawasdee N, Yenchitsomanus PT, Panya A
Biology 2022 Jul 23;11(8)
Biology 2022 Jul 23;11(8)
Systematic genetic mapping of necroptosis identifies SLC39A7 as modulator of death receptor trafficking.
Fauster A, Rebsamen M, Willmann KL, César-Razquin A, Girardi E, Bigenzahn JW, Schischlik F, Scorzoni S, Bruckner M, Konecka J, Hörmann K, Heinz LX, Boztug K, Superti-Furga G
Cell death and differentiation 2019 Jun;26(6):1138-1155
Cell death and differentiation 2019 Jun;26(6):1138-1155
Regulation of human glioblastoma cell death by combined treatment of cannabidiol, γ-radiation and small molecule inhibitors of cell signaling pathways.
Ivanov VN, Wu J, Hei TK
Oncotarget 2017 Sep 26;8(43):74068-74095
Oncotarget 2017 Sep 26;8(43):74068-74095
Dual Agonist Surrobody Simultaneously Activates Death Receptors DR4 and DR5 to Induce Cancer Cell Death.
Milutinovic S, Kashyap AK, Yanagi T, Wimer C, Zhou S, O'Neil R, Kurtzman AL, Faynboym A, Xu L, Hannum CH, Diaz PW, Matsuzawa S, Horowitz M, Horowitz L, Bhatt RR, Reed JC
Molecular cancer therapeutics 2016 Jan;15(1):114-24
Molecular cancer therapeutics 2016 Jan;15(1):114-24
Potential Therapeutic Effect of Natural Killer Cells on Doxorubicin-Resistant Breast Cancer Cells In Vitro.
Hwang MH, Li XJ, Kim JE, Jeong SY, Lee SW, Lee J, Ahn BC
PloS one 2015;10(8):e0136209
PloS one 2015;10(8):e0136209
Combination of systemic chemotherapy with local stem cell delivered S-TRAIL in resected brain tumors.
Redjal N, Zhu Y, Shah K
Stem cells (Dayton, Ohio) 2015 Jan;33(1):101-10
Stem cells (Dayton, Ohio) 2015 Jan;33(1):101-10
Increased growth-inhibitory and cytotoxic activity of arsenic trioxide in head and neck carcinoma cells with functional p53 deficiency and resistance to EGFR blockade.
Boyko-Fabian M, Niehr F, Distel L, Budach V, Tinhofer I
PloS one 2014;9(6):e98867
PloS one 2014;9(6):e98867
TRAIL-mediated apoptosis in breast cancer cells cultured as 3D spheroids.
Chandrasekaran S, Marshall JR, Messing JA, Hsu JW, King MR
PloS one 2014;9(10):e111487
PloS one 2014;9(10):e111487
MADD knock-down enhances doxorubicin and TRAIL induced apoptosis in breast cancer cells.
Turner A, Li LC, Pilli T, Qian L, Wiley EL, Setty S, Christov K, Ganesh L, Maker AV, Li P, Kanteti P, Das Gupta TK, Prabhakar BS
PloS one 2013;8(2):e56817
PloS one 2013;8(2):e56817
The oncogene metadherin modulates the apoptotic pathway based on the tumor necrosis factor superfamily member TRAIL (Tumor Necrosis Factor-related Apoptosis-inducing Ligand) in breast cancer.
Zhang N, Wang X, Huo Q, Li X, Wang H, Schneider P, Hu G, Yang Q
The Journal of biological chemistry 2013 Mar 29;288(13):9396-407
The Journal of biological chemistry 2013 Mar 29;288(13):9396-407
Real-time imaging of the dynamics of death receptors and therapeutics that overcome TRAIL resistance in tumors.
Bagci-Onder T, Agarwal A, Flusberg D, Wanningen S, Sorger P, Shah K
Oncogene 2013 Jun 6;32(23):2818-27
Oncogene 2013 Jun 6;32(23):2818-27
A whole-genome RNAi screen identifies an 8q22 gene cluster that inhibits death receptor-mediated apoptosis.
Dompe N, Rivers CS, Li L, Cordes S, Schwickart M, Punnoose EA, Amler L, Seshagiri S, Tang J, Modrusan Z, Davis DP
Proceedings of the National Academy of Sciences of the United States of America 2011 Oct 25;108(43):E943-51
Proceedings of the National Academy of Sciences of the United States of America 2011 Oct 25;108(43):E943-51
Molecular identification of an MHC-independent ligand recognized by a human {alpha}/{beta} T-cell receptor.
Hanada K, Wang QJ, Inozume T, Yang JC
Blood 2011 May 5;117(18):4816-25
Blood 2011 May 5;117(18):4816-25
Predominant antitumor effects by fully human anti-TRAIL-receptor 2 (DR5) monoclonal antibodies in human glioma cells in vitro and in vivo.
Nagane M, Shimizu S, Mori E, Kataoka S, Shiokawa Y
Neuro-oncology 2010 Jul;12(7):687-700
Neuro-oncology 2010 Jul;12(7):687-700
c-Jun NH2-terminal kinase-dependent upregulation of DR5 mediates cooperative induction of apoptosis by perifosine and TRAIL.
Fu L, Lin YD, Elrod HA, Yue P, Oh Y, Li B, Tao H, Chen GZ, Shin DM, Khuri FR, Sun SY
Molecular cancer 2010 Dec 20;9:315
Molecular cancer 2010 Dec 20;9:315
TRAIL death receptor-4, decoy receptor-1 and decoy receptor-2 expression on CD8+ T cells correlate with the disease severity in patients with rheumatoid arthritis.
Bisgin A, Terzioglu E, Aydin C, Yoldas B, Yazisiz V, Balci N, Bagci H, Gorczynski RM, Akdis CA, Sanlioglu S
BMC musculoskeletal disorders 2010 Aug 27;11:192
BMC musculoskeletal disorders 2010 Aug 27;11:192
CAAT/enhancer binding protein homologous protein-dependent death receptor 5 induction is a major component of SHetA2-induced apoptosis in lung cancer cells.
Lin YD, Chen S, Yue P, Zou W, Benbrook DM, Liu S, Le TC, Berlin KD, Khuri FR, Sun SY
Cancer research 2008 Jul 1;68(13):5335-44
Cancer research 2008 Jul 1;68(13):5335-44
Apoptotic activity and mechanism of 2-cyano-3,12-dioxoolean-1,9-dien-28-oic-acid and related synthetic triterpenoids in prostate cancer.
Hyer ML, Shi R, Krajewska M, Meyer C, Lebedeva IV, Fisher PB, Reed JC
Cancer research 2008 Apr 15;68(8):2927-33
Cancer research 2008 Apr 15;68(8):2927-33
The farnesyltransferase inhibitor lonafarnib induces CCAAT/enhancer-binding protein homologous protein-dependent expression of death receptor 5, leading to induction of apoptosis in human cancer cells.
Sun SY, Liu X, Zou W, Yue P, Marcus AI, Khuri FR
The Journal of biological chemistry 2007 Jun 29;282(26):18800-9
The Journal of biological chemistry 2007 Jun 29;282(26):18800-9
Human astrocytes are resistant to Fas ligand and tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis.
Song JH, Bellail A, Tse MC, Yong VW, Hao C
The Journal of neuroscience : the official journal of the Society for Neuroscience 2006 Mar 22;26(12):3299-308
The Journal of neuroscience : the official journal of the Society for Neuroscience 2006 Mar 22;26(12):3299-308
Enhancement of Ad5-TRAIL cytotoxicity against renal cell carcinoma with histone deacetylase inhibitors.
VanOosten RL, Earel JK Jr, Griffith TS
Cancer gene therapy 2006 Jun;13(6):628-32
Cancer gene therapy 2006 Jun;13(6):628-32
Difference in cytotoxicity against hepatocellular carcinoma between liver and periphery natural killer cells in humans.
Ishiyama K, Ohdan H, Ohira M, Mitsuta H, Arihiro K, Asahara T
Hepatology (Baltimore, Md.) 2006 Feb;43(2):362-72
Hepatology (Baltimore, Md.) 2006 Feb;43(2):362-72
Synthetic triterpenoids cooperate with tumor necrosis factor-related apoptosis-inducing ligand to induce apoptosis of breast cancer cells.
Hyer ML, Croxton R, Krajewska M, Krajewski S, Kress CL, Lu M, Suh N, Sporn MB, Cryns VL, Zapata JM, Reed JC
Cancer research 2005 Jun 1;65(11):4799-808
Cancer research 2005 Jun 1;65(11):4799-808
c-Jun NH2-terminal kinase-mediated up-regulation of death receptor 5 contributes to induction of apoptosis by the novel synthetic triterpenoid methyl-2-cyano-3,12-dioxooleana-1, 9-dien-28-oate in human lung cancer cells.
Zou W, Liu X, Yue P, Zhou Z, Sporn MB, Lotan R, Khuri FR, Sun SY
Cancer research 2004 Oct 15;64(20):7570-8
Cancer research 2004 Oct 15;64(20):7570-8
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
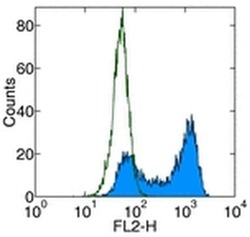
- Experimental details
- Staining of human CD261-transfected cells with Anti-Human CD261 (DR4) Biotin followed by Streptavidin PE (Product # 12-4317-87). Appropriate isotype controls were used (open histogram). Total viable cells were used for analysis.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
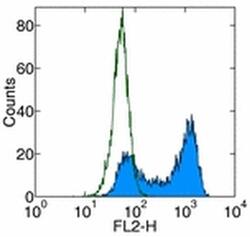
- Experimental details
- Staining of human CD261-transfected cells with Anti-Human CD261 (DR4) Biotin followed by Streptavidin PE (Product # 12-4317-87). Appropriate isotype controls were used (open histogram). Total viable cells were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
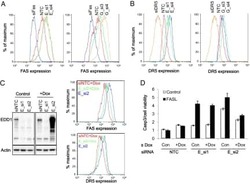
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
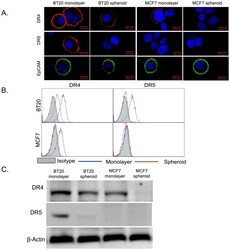
- Experimental details
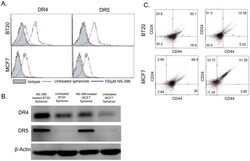
- Figure 1 BT20 and MCF7 spheroids have a lower expression of death receptors. (A) Confocal micrographs of BT20 and MCF7 cells cultured as 3D spheroids and 2D monolayer stained with antibodies against DR4 (red), DR5 (red) and EpCAM (green). (Scale bar = 20 um) (B) Flow cytometry histograms comparing DR4 and DR5 expression in BT20 and MCF7 spheroids and monolayer. (C) Western blot analysis of total DR4 and DR5 expression in cell lysates from BT20 and MCF7 spheroids and monolayer.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
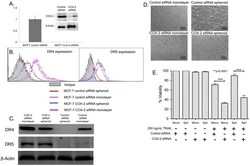
- Figure 6 COX-2 inhibitor treatment upregulates the expression of DR4 and DR5 in BT20 and MCF7 spheroids. (A) Flow cytometry histograms showing the effect of 100 uM NS-398 treatment on death receptor expression in BT20 and MCF7 cells cultured as spheroids. (B) Western blot analysis of whole cell lysates from untreated and NS-398 treated BT20 and MCF7 spheroids for DR4 and DR5 expression. (C) Flow cytometry scatter plots showing the effect of NS-398 treatment on the prevalence of CD44 hi CD24 lo population in BT20 and MCF7 cells cultured as spheroids.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 8 COX-2 knockdown reverses TRAIL-resistance in MCF7 spheroid and monolayer cells. (A) qPCR and western blot data showing the relative expression of COX-2 gene (n = 3) and COX-2 protein in control siRNA and COX-2 siRNA transfected MCF7 cells. (B) Flow cytometry histograms and (C) western blot analysis comparing the expression of DR4 in MCF7 cells transfected with control siRNA and COX-2 siRNA cultured as monolayer and spheroids. (D) Bright field images and (E) MTT assay results (n = 5) quantifying the effect of 200 ng/mL of TRAIL in control siRNA and COX-2 siRNA transfected cells cultured as monolayer and spheroids.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 Orthogonal genetic screens and surface marker analysis specify SLC39A7 loss-of-function phenotype on receptor trafficking. a Spider plot summarizing the results of 8 independent CRISPR/ Cas9 screens in KBM7 wildtype, KBM7 FADD - , or HAP1 cells using an SLC-specific gRNA library. Each plot section represents one screen with the indicated stimuli. Screen analysis was performed by identifying differentially enriched sgRNAs using DESeq2 and then aggregating sgRNAs to genes using Gene Set Enrichment Analysis. Identified hits are ranked according to the adjusted p -value of enrichment (-log10(p adj )), bubble size indicates the number of significantly enriched sgRNAs and color the average log2 fold-change (aLFC) of the enriched sgRNAs. Screens were performed in duplicate. SLC39A7 is highlighted in orange. No gene was identified in KBM7 wildtype cells upon TRAIL treatment. b MCA of Jurkat E6.1 SpCas9 cells transduced with a GFP marker (GFP + ) and sgRNAs targeting either SLC39A7 or Renilla luciferase (sg Ren ) as control, against cells transduced with sg Ren and an mCherry marker (mCherry + ). The cell populations were mixed at 1:1 ratio, treated with TRAIL (20 ng/ml) or FASL (1 ng/ml), and analyzed after 14 days by flow cytometry. Data represent mean value +- s.d. of two independent experiments performed in duplicates, n.d. (not determined) indicates wells with no outgrowth. c Flow cytometry analysis for TRAILR1 (left) and TRAILR2 (right) surface expression in KBM7 FADD - ,
- Conjugate
- Yellow dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry