Antibody data
- Antibody Data
- Antigen structure
- References [5]
- Comments [0]
- Validations
- Other assay [8]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA5-13045 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-HIPK2 (Tyr361) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- This antibody is predicted to react with mouse and rat based on sequence homology.
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 400 µL
- Concentration
- 0.5 mg/mL
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
Submitted references Small-molecule probe reveals a kinase cascade that links stress signaling to TCF/LEF and Wnt responsiveness.
Inhibition of invasive pancreatic cancer: restoring cell apoptosis by activating mitochondrial p53.
Functional characterization of DYRK1A missense variants associated with a syndromic form of intellectual deficiency and autism.
Effect of tyrosine autophosphorylation on catalytic activity and subcellular localisation of homeodomain-interacting protein kinases (HIPK).
Transcriptional corepressors HIPK1 and HIPK2 control angiogenesis via TGF-β-TAK1-dependent mechanism.
Cheng J, Tsuda M, Okolotowicz K, Dwyer M, Bushway PJ, Colas AR, Lancman JJ, Schade D, Perea-Gil I, Bruyneel AAN, Lee J, Vadgama N, Quach J, McKeithan WL, Biechele TL, Wu JC, Moon RT, Si Dong PD, Karakikes I, Cashman JR, Mercola M
Cell chemical biology 2021 May 20;28(5):625-635.e5
Cell chemical biology 2021 May 20;28(5):625-635.e5
Inhibition of invasive pancreatic cancer: restoring cell apoptosis by activating mitochondrial p53.
Cheng J, Okolotowicz KJ, Ryan D, Mose E, Lowy AM, Cashman JR
American journal of cancer research 2019;9(2):390-405
American journal of cancer research 2019;9(2):390-405
Functional characterization of DYRK1A missense variants associated with a syndromic form of intellectual deficiency and autism.
Widowati EW, Ernst S, Hausmann R, Müller-Newen G, Becker W
Biology open 2018 Apr 26;7(4)
Biology open 2018 Apr 26;7(4)
Effect of tyrosine autophosphorylation on catalytic activity and subcellular localisation of homeodomain-interacting protein kinases (HIPK).
van der Laden J, Soppa U, Becker W
Cell communication and signaling : CCS 2015 Jan 29;13:3
Cell communication and signaling : CCS 2015 Jan 29;13:3
Transcriptional corepressors HIPK1 and HIPK2 control angiogenesis via TGF-β-TAK1-dependent mechanism.
Shang Y, Doan CN, Arnold TD, Lee S, Tang AA, Reichardt LF, Huang EJ
PLoS biology 2013;11(4):e1001527
PLoS biology 2013;11(4):e1001527
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 5. Reduced tyrosine autophosphorylation of in vitro -translated DYRK1A-L295F. DYRK1A constructs (28-499) were expressed in a cell-free E. coli- derived expression system. Coupled in vitro transcription-translation reactions were incubated for 90 min at 37degC before reaction products were subjected to immunoblot analysis. For quantification, pTyr signals were normalized to the total amounts of recombinant DYRK1A as determined by detection of the N-terminal StrepTag (means and s.d., n =3). The difference in relative tyrosine autophosphorylation was tested for statistical significance by one-sample t -test (* P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Mutation in Y361 of HIPK2 abolishes its ability to suppress Mmp10-Luc activity. (A) Phospho-specific antibody for HIPK2-P-Y361 confirms that TGF-beta-TAK1 can indeed promote the phosphorylation of wild-type HIPK2 protein, but not HIPK2-Y361F mutant, in HEK293T cells. (B) TGF-beta type I receptor (TbetaRI) inhibitor SB431542 blocks TGF-beta-induced phosphorylation, but not basal phosphorylation, on Y361 residue in HIPK2 in HUVEC cells. (C and D) Phosphorylation on the Y361 residue of HIPK2 is required for the transcriptional suppressor effect of HIPK2 on Mmp10 in HEK293T cells (C) and HUVEC cells (D). Data in (C) and (D) are shown as mean +- s.e.m., n = 3. Statistics in (C) and (D) use two-way ANOVA. * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Tyrosine phosphorylation in the activation loop of HIPK1, HIPK2 and HIPK3. HeLa cells were transfected with expression plasmids for wild type GFP-HIPK fusion proteins or the Tyr-Phe mutants thereof (YF). If indicated, cells were treated with sodium orthovanadate (Na 3 VO 4 ) for 1 h or 2 h. Western blots of total cell lysates were detected with antibodies directed against pTyr361 in HIPK2, a general antibody for phosphotyrosine independent of the sequence context (PY99) and a GFP antibody. A, The pTyr361(HIPK2) antibody detects wild type HIPK1-3 but not the Tyr-Phe mutants. B , Effect of vanadate treatment. The column diagrams show the quantitative evaluation of 3 experiments. The background signal in the untransfected control samples was subtracted from the signal intensities obtained with the phosphospecific antibodies. Relative phosphorylation after vanadate treatment was calculated by normalization to the signal measured in untreated cells. Means + SEM, * p < 0.05, analysed by one-sample t test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
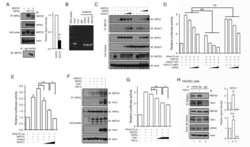
- Figure 3 HIPK2 suppresses Mmp10 expression through interaction with MEF2C and HDAC7. (A) Co-IP assays using protein lysates from HEK293T cells expressing HIPK2 and MEF2C show that HIPK2 can be detected in a protein complex with MEF2C (upper panels). Similar protein complex formation between endogenous HIPK2 and MEF2C can also be detected in wild-type MEF cells (lower panel). The interaction appears to depend on HIPK2 kinase activity as the kinase inactive HIPK2-K221A shows much reduced interaction with MEF2C. (B) ChIP assays using native chromatin from HUVECs show that HIPK1, HIPK2, and MEF2C can be detected in the promoter sequence of Mmp10 . (C) Co-IP assays reveal that HIPK2 can be detected in a protein complex with HDAC7 and MEF2C. (D) HIPK2 and HDAC7 cooperatively suppress MEF2C-dependent activation of Mmp10 -luciferase reporter activity. In the presence of the HDAC7 that lacks MEF2C interaction domain, HIPK2 can still suppress MEF2C in the activation of Mmp10 reporter. (E) HDAC7 continues to suppress MEF2C-dependent activation of Mmp10 in HEK293T cells where the endogenous Hipk2 mRNA is reduced by siRNA. (F) Co-IP assays showing that TGF-beta and TAK1 enhance the interaction between MEF2C and HIPK2. (G) TGF-beta and TAK1 enhance the corepressor effects of HIPK2 on MEF2C-mediated Mmp10 expression. (H) Co-IP assays using HUVEC cell lysates show that TGF-beta promotes the interaction of endogenous HIPK2, TAK1, and MEF2C. Data are shown as mean +- s.e.m. Student's t test, n
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Dot blot analysis of Phospho-HIPK2 pTyr361 polyclonal antibody (Product # PA5-13045) on nitrocellulose membrane. 50 ng of Phospho-peptide or Non Phospho-peptide per dot were adsorbed. Antibody working concentrations are 0.5 µg per mL.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 TGF-beta-TAK1 promotes HIPK2 activity through protein-protein interaction and protects HIPK2 from proteasome-mediated degradation. (A) TGF-beta promotes HIPK2 kinase activity in HEK293T cells, whereas kinase inactive HIPK2-K221A shows no incorporation of gamma- 32 P-ATP upon TGF-beta treatment. (B) The ability of TGF-beta to activate HIPK2 kinase activity can be blocked by TGF-beta type I receptor inhibitor SB431542. (C and D) TGF-beta and wild-type TAK1 activate HIPK2 kinase and maintain the stability of HIPK2 protein. In contrast, dominant negative TAK1 (DN-TAK1) promotes HIPK2 degradation via the proteasome pathway.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 TGF-beta activates HIPK2 by phosphorylating a highly conserved tyrosine residue on position 361. (A) Amino acid sequence alignment of the HIPK protein family from human and mouse reveals a stretch of highly conserved residues from position 346 to 371 in the activation segment of the subdomain VII in HIPK2. (B) Alignment of the similar regions of HIPK2 (346 to 371) from different species confirms that these amino acid residues are highly conserved from nematodes to the vertebrates. Conserved amino acids that can potentially be phosphorylated in MAPK signaling pathway are shown in bold. (C) The combined immunoprecipitation and in vitro kinase (IP-IVK) assays show that TGF-beta treatment promotes the ability of wild-type HIPK2 to incorporate gamma- 32 P-ATP. In contrast, kinase inactive HIPK2-K221A fails to incorporate gamma- 32 P-ATP. While HIPK2-S359A and HIPK2-T360A mutant proteins can still incorporate gamma- 32 P-ATP in response to TGF-beta treatment, the Y361F mutation in HIPK2 completely eliminates its ability to incorporate gamma- 32 P-ATP. (D) TGF-beta and TAK1-induced phosphorylation of HIPK2 occurs primarily on Y361 residue in HIPK2. HIPK2-Y361F mutant completely loses its ability to incorporate gamma- 32 P-ATP upon activation by TGF-beta or TAK1. Data are shown as mean + s.e.m., n = 3. Statistics in (C) and (D) use Student's t test. * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
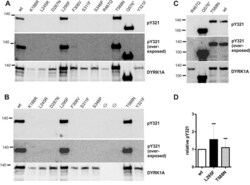
- Fig. 2. Tyrosine autophosphorylation of wild - type and variant DYRK1A constructs. Wild-type (wt) and mutant GFP-DYRK1A fusion proteins were immunoprecipitated with a GFP antibody from cell extracts of transiently transfected HEK293 cells and subjected to immunoblot analysis. Tyrosine phosphorylation of the activation loop (Y321) was analyzed using an antibody against the activation loop tyrosine in HIPK2 that cross-reacts with the corresponding residue in DYRK1A ( Abu Jhaisha et al., 2017 ). Relative tyrosine phosphorylation was calculated ratio of the pY321 signal relative to total amounts DYRK1A constructs as detected with a DYRK1A specific antibody. (A-C) Representative western blots from selected experiments, including overexposed images to demonstrate the absence of pY321 in the weakly expressed variants. (D) Quantitative evaluation of the catalytically active missense variants (means and s.d.; n =5 for wt versus L295F and n =6 for wt versus T588N) . Differences were not statistically significant by one-sample t -test ( P >0.05).
 Explore
Explore Validate
Validate Learn
Learn Other assay
Other assay