Antibody data
- Antibody Data
- Antigen structure
- References [12]
- Comments [0]
- Validations
- Immunocytochemistry [3]
- Flow cytometry [1]
- Other assay [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 14-0499-82 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD49d (Integrin alpha 4) Monoclonal Antibody (9F10), eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 9F10 monoclonal antibody reacts with human CD49d, the 150 kDa integrin alpha4 subunit. The complex of CD49d non-covalently associated with integrin beta1 (CD29), also known as VLA-4, is a receptor for fibronectin and VCAM-1 (CD106). This complex is expressed by thymocytes, peripheral lymphocytes, monocytes and eosinophils. CD49d also associates with integrin beta7 and binds to the Mucosal Addressin Cell-Adhesion Molecule-1 (MadCAM-1). Applications Reported: The 9F10 antibody has been reported for use in flow cytometric analysis, and immunohistochemical staining. 9F10 has also been reported in in vitro functional assays. (Please use Functional Grade purified 9F10, Product # 16-0499, in functional assays). Applications Tested: The 9F10 antibody has been tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at less than or equal to 0.25 µg per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest. Purity: Greater than 90%, as determined by SDS-PAGE. Aggregation: Less than 10%, as determined by HPLC. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 9F10
- Vial size
- 100 μg
- Concentration
- 0.5 mg/mL
- Storage
- 4°C
Submitted references Identification of potential chemical compounds enhancing generation of enucleated cells from immortalized human erythroid cell lines.
A Prime/Boost Vaccine Regimen Alters the Rectal Microbiome and Impacts Immune Responses and Viremia Control Post-Simian Immunodeficiency Virus Infection in Male and Female Rhesus Macaques.
Enhanced control of Mycobacterium tuberculosis extrapulmonary dissemination in mice by an arabinomannan-protein conjugate vaccine.
The APPEESFRS Peptide, Restricted by the HLA-B*35:01 Molecule, and the APPEESFRF Variant Derived from an Autologous HIV-1 Strain Induces Polyfunctional Responses in CD8+ T Cells.
LDL cholesterol modulates human CD34+ HSPCs through effects on proliferation and the IL-17 G-CSF axis.
Incomplete normalization of regulatory t-cell frequency in the gut mucosa of Colombian HIV-infected patients receiving long-term antiretroviral treatment.
Protection against H5N1 highly pathogenic avian and pandemic (H1N1) 2009 influenza virus infection in cynomolgus monkeys by an inactivated H5N1 whole particle vaccine.
Memory immune responses against pandemic (H1N1) 2009 influenza virus induced by a whole particle vaccine in cynomolgus monkeys carrying Mafa-A1*052:02.
Rv2468c, a novel Mycobacterium tuberculosis protein that costimulates human CD4+ T cells through VLA-5.
Characterization of interleukin-17-producing regulatory T cells in inflamed intestinal mucosa from patients with inflammatory bowel diseases.
Expression of endothelia and lymphocyte adhesion molecules in bronchus-associated lymphoid tissue (BALT) in adult human lung.
Interleukin-15 increases effector memory CD8+ t cells and NK Cells in simian immunodeficiency virus-infected macaques.
Soboleva S, Kurita R, Ek F, Åkerstrand H, Silvério-Alves R, Olsson R, Nakamura Y, Miharada K
Communications biology 2021 Jun 3;4(1):677
Communications biology 2021 Jun 3;4(1):677
A Prime/Boost Vaccine Regimen Alters the Rectal Microbiome and Impacts Immune Responses and Viremia Control Post-Simian Immunodeficiency Virus Infection in Male and Female Rhesus Macaques.
Musich T, Thovarai V, Venzon DJ, Mohanram V, Tuero I, Miller-Novak LK, Helmold Hait S, Rahman MA, Hunegnaw R, Huiting E, Yuan W, O'hUigin C, Hoang T, Sui Y, LaBranche C, Montefiori D, Bear J, Rosati M, Bissa M, Berzofsky JA, Pavlakis GN, Felber BK, Franchini G, Robert-Guroff M
Journal of virology 2020 Nov 23;94(24)
Journal of virology 2020 Nov 23;94(24)
Enhanced control of Mycobacterium tuberculosis extrapulmonary dissemination in mice by an arabinomannan-protein conjugate vaccine.
Prados-Rosales R, Carreño L, Cheng T, Blanc C, Weinrick B, Malek A, Lowary TL, Baena A, Joe M, Bai Y, Kalscheuer R, Batista-Gonzalez A, Saavedra NA, Sampedro L, Tomás J, Anguita J, Hung SC, Tripathi A, Xu J, Glatman-Freedman A, Jacobs WR Jr, Chan J, Porcelli SA, Achkar JM, Casadevall A
PLoS pathogens 2017 Mar;13(3):e1006250
PLoS pathogens 2017 Mar;13(3):e1006250
The APPEESFRS Peptide, Restricted by the HLA-B*35:01 Molecule, and the APPEESFRF Variant Derived from an Autologous HIV-1 Strain Induces Polyfunctional Responses in CD8+ T Cells.
Acevedo-Sáenz L, Carmona-Pérez L, Velilla-Hernández PA, Delgado JC, Rugeles L MT
BioResearch open access 2015;4(1):115-20
BioResearch open access 2015;4(1):115-20
LDL cholesterol modulates human CD34+ HSPCs through effects on proliferation and the IL-17 G-CSF axis.
Cimato TR, Palka BA, Lang JK, Young RF
PloS one 2013;8(8):e73861
PloS one 2013;8(8):e73861
Incomplete normalization of regulatory t-cell frequency in the gut mucosa of Colombian HIV-infected patients receiving long-term antiretroviral treatment.
Rueda CM, Velilla PA, Chougnet CA, Rugeles MT
PloS one 2013;8(8):e71062
PloS one 2013;8(8):e71062
Protection against H5N1 highly pathogenic avian and pandemic (H1N1) 2009 influenza virus infection in cynomolgus monkeys by an inactivated H5N1 whole particle vaccine.
Nakayama M, Shichinohe S, Itoh Y, Ishigaki H, Kitano M, Arikata M, Pham VL, Ishida H, Kitagawa N, Okamatsu M, Sakoda Y, Ichikawa T, Tsuchiya H, Nakamura S, Le QM, Ito M, Kawaoka Y, Kida H, Ogasawara K
PloS one 2013;8(12):e82740
PloS one 2013;8(12):e82740
Memory immune responses against pandemic (H1N1) 2009 influenza virus induced by a whole particle vaccine in cynomolgus monkeys carrying Mafa-A1*052:02.
Arikata M, Itoh Y, Okamatsu M, Maeda T, Shiina T, Tanaka K, Suzuki S, Nakayama M, Sakoda Y, Ishigaki H, Takada A, Ishida H, Soda K, Pham VL, Tsuchiya H, Nakamura S, Torii R, Shimizu T, Inoko H, Ohkubo I, Kida H, Ogasawara K
PloS one 2012;7(5):e37220
PloS one 2012;7(5):e37220
Rv2468c, a novel Mycobacterium tuberculosis protein that costimulates human CD4+ T cells through VLA-5.
Li Q, Ding X, Thomas JJ, Harding CV, Pecora ND, Ziady AG, Shank S, Boom WH, Lancioni CL, Rojas RE
Journal of leukocyte biology 2012 Feb;91(2):311-20
Journal of leukocyte biology 2012 Feb;91(2):311-20
Characterization of interleukin-17-producing regulatory T cells in inflamed intestinal mucosa from patients with inflammatory bowel diseases.
Hovhannisyan Z, Treatman J, Littman DR, Mayer L
Gastroenterology 2011 Mar;140(3):957-65
Gastroenterology 2011 Mar;140(3):957-65
Expression of endothelia and lymphocyte adhesion molecules in bronchus-associated lymphoid tissue (BALT) in adult human lung.
Kawamata N, Xu B, Nishijima H, Aoyama K, Kusumoto M, Takeuchi T, Tei C, Michie SA, Matsuyama T
Respiratory research 2009 Oct 22;10(1):97
Respiratory research 2009 Oct 22;10(1):97
Interleukin-15 increases effector memory CD8+ t cells and NK Cells in simian immunodeficiency virus-infected macaques.
Mueller YM, Petrovas C, Bojczuk PM, Dimitriou ID, Beer B, Silvera P, Villinger F, Cairns JS, Gracely EJ, Lewis MG, Katsikis PD
Journal of virology 2005 Apr;79(8):4877-85
Journal of virology 2005 Apr;79(8):4877-85
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
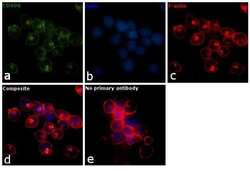
- Experimental details
- Immunofluorescence analysis of CD49d was performed using 70% confluent log phase MOLT-4 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with CD49d Mouse Monoclonal Antibody (Product # 14-0499-82) at 5 µg/mL in 0.1% BSA and incubated overnight at 4 degree and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing membrane localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
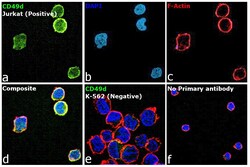
- Experimental details
- Immunofluorescence analysis of CD49d (Integrin alpha 4) was performed using 70% confluent log phase Jurkat cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with CD49d (Integrin alpha 4) Monoclonal Antibody (9F10), eBioscience™ (Product # 14-0499-80) at 5 µg/mL in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766), (1:2000), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b: Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing Plasma Membrane localization. Panel e represents the merged image of K-562 cells having no expression of CD49d. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
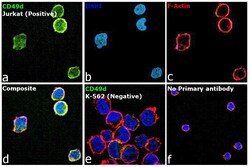
- Experimental details
- Immunofluorescence analysis of CD49d (Integrin alpha 4) was performed using 70% confluent log phase Jurkat cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with CD49d (Integrin alpha 4) Monoclonal Antibody (9F10), eBioscience™ (Product # 14-0499-80) at 5 µg/mL in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766), (1:2000), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b: Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing Plasma Membrane localization. Panel e represents the merged image of K-562 cells having no expression of CD49d. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
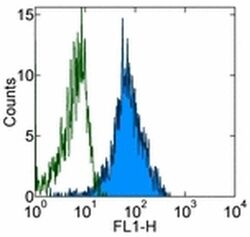
- Experimental details
- Staining of normal human peripheral blood cells with 0.125 µg of Mouse IgG1 kappa Isotype Control Purified (Product # 14-4714-82) (open histogram) or 0.125 µg of Anti-Human CD49d Purified (filled histogram) followed by Anti-Mouse IgG FITC (Product # 11-4011-85). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
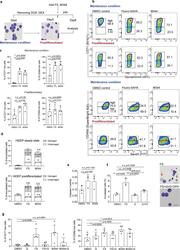
- Experimental details
- Fig. 4 Treatment with HDACi leads to alterations of cell surface phenotype of HiDEP. a Schematic of experimental procedure. HiDEP were treated with FS or M344 for 24 h with or without 5-day pre-differentiation. The cells were then subjected to flow cytometry analyses. b , c Flow cytometry analyses for cell surface expression of representative erythroid cell surface markers. Representative FACS profiles ( b ) and summarized graphs ( c ) are shown. Mean +- SD, n = 4. Significance was determined using one-way ANOVA with Dunnett's multiple comparisons test. d Frequency of enucleated cells after Fluoro-SAHA treatment with or without pre-differentiation treatment. n = 3. Significance was determined using one-way ANOVA with Dunnett's multiple comparisons test. e Measurement of enzymatic activity of caspase-3 in HiDEP treated with DMSO, Fluoro-SAHA (FS), or M344. Mean +- SD, n = 3. Significance was determined using paired t -test. f Effects of caspase inhibition on enucleation of HiDEP upon HDACi treatment. HiDEP were treated with 50 muM of a pan-caspase inhibitor, QVD-OPH, with or without 15 muM of Fluoro-SAHA. Frequency of enucleated cells was analyzed using flow cytometry. FS, Fluoro-SAHA; Q, QVD-OPH. Mean +- SD, n = 3. Significance was determined using one-way ANOVA with Tukey's multiple comparisons test. g Cell surface expression of representative erythroid cell surface markers on HiDEP treated with HDACi with or without QVD-OPH treatment. n = 5. FS, Fluoro-SAHA; Q, QVD-OPH. Mea
 Explore
Explore Validate
Validate Learn
Learn Immunocytochemistry
Immunocytochemistry Immunohistochemistry
Immunohistochemistry