Antibody data
- Antibody Data
- Antigen structure
- References [7]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [6]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 50-7219-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- IL-21 Monoclonal Antibody (eBio3A3-N2 (3A3-N2)), eFluor™ 660, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The monoclonal antibody eBio3A3-N2 reacts with human IL-21, a potent stimulator of NK and cytolytic T cells. IL-21 is a 131 amino acid protein most closely related to IL-2 and IL-15. Expression is found in CD4+ cells upon activation. In addition to its effect on NK cells, IL-21 stimulates proliferation of B-cell stimulated by crosslinking of the CD40 antigen and stimulates the proliferation of bone marrow progenitor cells and the expression of the NK-cell marker CD56 in the presence of IL-15. Applications Reported: This eBio3A3-N2 (3A3-N2) antibody has been reported for use in intracellular staining followed by flow cytometric analysis. Applications Tested: This eBio3A3-N2 (3A3-N2) antibody has been pre-titrated and tested by intracellular staining and flow cytometric analysis of restimulated, Th17-polarized normal human peripheral blood cells. This can be used at 5 µL (0.125 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Staining for IL-21 has shown donor variability. stimulation times may vary depending on the donor and should be optimized. Increased staining levels have been observed on Th17-polarized normal human peripheral blood cells. eFluor® 660 is a replacement for Alexa Fluor® 647. eFluor® 660 emits at 659 nm and is excited with the red laser (633 nm). Please make sure that your instrument is capable of detecting this fluorochome. Excitation: 633-647 nm; Emission: 668 nm; Laser: Red Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- eBio3A3-N2 (3A3-N2)
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Ageing promotes early T follicular helper cell differentiation by modulating expression of RBPJ.
IL-22 production of effector CD4(+) T-cells is altered in SLE patients.
Effects of Anti-TNFα Treatment on Mucosal Expression of IL-17A, IL-21, and IL-22 and Cytokine-Producing T Cell Subsets in Crohn's Disease.
C5aR1 regulates T follicular helper differentiation and chronic graft-versus-host disease bronchiolitis obliterans.
Recognition of microbial viability via TLR8 drives T(FH) cell differentiation and vaccine responses.
Cutting Edge: Selective Oral ROCK2 Inhibitor Reduces Clinical Scores in Patients with Psoriasis Vulgaris and Normalizes Skin Pathology via Concurrent Regulation of IL-17 and IL-10.
Reprogramming CD19-specific T cells with IL-21 signaling can improve adoptive immunotherapy of B-lineage malignancies.
Webb LMC, Fra-Bido S, Innocentin S, Matheson LS, Attaf N, Bignon A, Novarino J, Fazilleau N, Linterman MA
Aging cell 2021 Jan;20(1):e13295
Aging cell 2021 Jan;20(1):e13295
IL-22 production of effector CD4(+) T-cells is altered in SLE patients.
Dolff S, Scharpenberg C, Specker C, Kribben A, Witzke O, Wilde B
European journal of medical research 2019 Jul 22;24(1):24
European journal of medical research 2019 Jul 22;24(1):24
Effects of Anti-TNFα Treatment on Mucosal Expression of IL-17A, IL-21, and IL-22 and Cytokine-Producing T Cell Subsets in Crohn's Disease.
Dige A, Magnusson MK, Uhrenholt C, Rasmussen TK, Kragstrup T, Öhman L, Dahlerup J, Agnholt J
Mediators of inflammation 2018;2018:3279607
Mediators of inflammation 2018;2018:3279607
C5aR1 regulates T follicular helper differentiation and chronic graft-versus-host disease bronchiolitis obliterans.
Verghese DA, Chun N, Paz K, Fribourg M, Woodruff TM, Flynn R, Hu Y, Xiong H, Zhang W, Yi Z, Du J, Blazar BR, Heeger PS
JCI insight 2018 Dec 20;3(24)
JCI insight 2018 Dec 20;3(24)
Recognition of microbial viability via TLR8 drives T(FH) cell differentiation and vaccine responses.
Ugolini M, Gerhard J, Burkert S, Jensen KJ, Georg P, Ebner F, Volkers SM, Thada S, Dietert K, Bauer L, Schäfer A, Helbig ET, Opitz B, Kurth F, Sur S, Dittrich N, Gaddam S, Conrad ML, Benn CS, Blohm U, Gruber AD, Hutloff A, Hartmann S, Boekschoten MV, Müller M, Jungersen G, Schumann RR, Suttorp N, Sander LE
Nature immunology 2018 Apr;19(4):386-396
Nature immunology 2018 Apr;19(4):386-396
Cutting Edge: Selective Oral ROCK2 Inhibitor Reduces Clinical Scores in Patients with Psoriasis Vulgaris and Normalizes Skin Pathology via Concurrent Regulation of IL-17 and IL-10.
Zanin-Zhorov A, Weiss JM, Trzeciak A, Chen W, Zhang J, Nyuydzefe MS, Arencibia C, Polimera S, Schueller O, Fuentes-Duculan J, Bonifacio KM, Kunjravia N, Cueto I, Soung J, Fleischmann RM, Kivitz A, Lebwohl M, Nunez M, Woodson J, Smith SL, West RF, Berger M, Krueger JG, Ryan JL, Waksal SD
Journal of immunology (Baltimore, Md. : 1950) 2017 May 15;198(10):3809-3814
Journal of immunology (Baltimore, Md. : 1950) 2017 May 15;198(10):3809-3814
Reprogramming CD19-specific T cells with IL-21 signaling can improve adoptive immunotherapy of B-lineage malignancies.
Singh H, Figliola MJ, Dawson MJ, Huls H, Olivares S, Switzer K, Mi T, Maiti S, Kebriaei P, Lee DA, Champlin RE, Cooper LJ
Cancer research 2011 May 15;71(10):3516-27
Cancer research 2011 May 15;71(10):3516-27
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
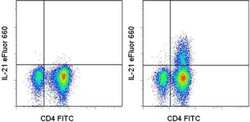
- Experimental details
- Intracellular staining of Th17-polarized normal human peripheral blood cells restimulated with monensin alone (left) or PMA/Ionomycin and monensin for 5 hours (right) with Anti-Human CD4 FITC (Product # 11-0049-42) and Anti-Human IL-21 eFluor® 660. Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Mucosal gene expression and LP T cell producing IL-17A, IL-21, and IL-22 at baseline in inflamed and noninflamed mucosa. Gene expression was determined by rtPCR (a) and frequencies of IL-17A-, IL-21-, and IL-22-producing cells among LP CD3 + T cells were determined by flow cytometry (b). Gene expression data is displayed as the normalized ratios between the relative expression of the gene of interest and the housekeeping gene HPRT-1 . Wilcoxon signed-rank test for comparison was applied on paired samples (rtPCR: IL-17A n = 11; IL-21 n = 9; IL-22 n = 9; flow cytometry: n = 8 for each cytokine).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Comparison of mucosal gene expression and LP T cells expressing IL-17A, IL-21, and IL-22 between baseline and week 4 of adalimumab treatment. Gene expression was determined by rtPCR (a) and frequencies of IL-17A-, IL-21-, and IL-22-producing cells among LP CD3 + T cells were determined by flow cytometry (b). Gene expression data is displayed as the normalized ratios between the relative expression of the gene of interest and the housekeeping gene HPRT-1 . Wilcoxon signed-rank test for comparison was applied on paired samples (rtPCR: IL-17A n = 12; IL-21 n = 11; IL-22 n = 11; flow cytometry: n = 7 for each cytokine).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
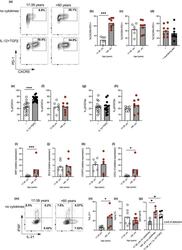
- Experimental details
- FIGURE 2 Age promotes pre-Tfh cell differentiation in humans. Flow plots showing the frequency of CXCR5 + PD-1 + cells amongst CD4 + following 4 days in vitro activation of naive CD4 + T cells taken from younger (17-39 yrs) and older (>60 yrs) donors in the presence/absence IL-12 and TGFbeta (a). Percentage of CXCR5 + PD-1 + following activation of naive CD4 + T cells from younger and older donors in the absence (b) or presence (c) of IL-12 and TGFbeta. Percentage of CXCR5 + PD1 + cells following activation of naive CD4 + T cells from older donors with or without neutralising antibodies to IL-12, Activin A and TGFbeta (d). Bar graphs showing percentage of cells expressing pSTAT3 (e, f), and pSTAT5 (g, h) on day 3 of culture in presence/absence of IL-12 and TGFbeta in donors of the indicated ages. RT-PCR determination MAF (i), BCL6 (j), FOXP3 (k) and CXCL13 (l) following 4 days activation of naive CD4 + T cells from younger and older donors in the absence of polarising cytokines. Flow plots (m-o) and bar graphs (n-o) showing percentage of IFNgamma and IL-21 expressing cells following PMA and ionomycin mediated restimulation after 4 days in vitro culture of naive CD4 + T cells from younger and older donors in the absence of IL-12 and TGFbeta (m-o). Bar graph showing levels of IgG produced by B cells following co-culture with CD4+ T cells from day four cultures from young and older donors, in the presence/absence of Tfh-polarising cytokines (p). Each symbol is representative of
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry