Antibody data
- Antibody Data
- Antigen structure
- References [14]
- Comments [0]
- Validations
- Western blot [2]
- Immunohistochemistry [2]
- Other assay [9]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 40-9800 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- ANGPTL4 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µg
- Concentration
- 0.25 mg/mL
- Storage
- -20°C
Submitted references PPARα and PPARγ activation is associated with pleural mesothelioma invasion but therapeutic inhibition is ineffective.
A TAZ-ANGPTL4-NOX2 Axis Regulates Ferroptotic Cell Death and Chemoresistance in Epithelial Ovarian Cancer.
ANGPTL4 exacerbates pancreatitis by augmenting acinar cell injury through upregulation of C5a.
Circulating progenitor cells and the expression of Cxcl12, Cxcr4 and angiopoietin-like 4 during wound healing in the murine ear.
Hydrogen peroxide promotes the expression of angiopoietin like 4 in RAW264.7 macrophages via MAPK pathways.
ANGPTL4 deficiency in haematopoietic cells promotes monocyte expansion and atherosclerosis progression.
GATA3 suppresses metastasis and modulates the tumour microenvironment by regulating microRNA-29b expression.
Mechanism of hypertriglyceridemia in CTP:phosphoethanolamine cytidylyltransferase-deficient mice.
Angiopoietin like protein 4 expression is decreased in activated macrophages.
Decreased fat storage by Lactobacillus paracasei is associated with increased levels of angiopoietin-like 4 protein (ANGPTL4).
The acute phase response stimulates the expression of angiopoietin like protein 4.
Activating peroxisome proliferator-activated receptor gamma mutant promotes tumor growth in vivo by enhancing angiogenesis.
Enhanced angiogenesis in obesity and in response to PPARgamma activators through adipocyte VEGF and ANGPTL4 production.
Peroxisome proliferator-activated receptor-beta/delta (PPARbeta/delta) ligands do not potentiate growth of human cancer cell lines.
Orozco Morales ML, Rinaldi CA, de Jong E, Lansley SM, Gummer JPA, Olasz B, Nambiar S, Hope DE, Casey TH, Lee YCG, Leslie C, Nealon G, Shackleford DM, Powell AK, Grimaldi M, Balaguer P, Zemek RM, Bosco A, Piggott MJ, Vrielink A, Lake RA, Lesterhuis WJ
iScience 2022 Jan 21;25(1):103571
iScience 2022 Jan 21;25(1):103571
A TAZ-ANGPTL4-NOX2 Axis Regulates Ferroptotic Cell Death and Chemoresistance in Epithelial Ovarian Cancer.
Yang WH, Huang Z, Wu J, Ding CC, Murphy SK, Chi JT
Molecular cancer research : MCR 2020 Jan;18(1):79-90
Molecular cancer research : MCR 2020 Jan;18(1):79-90
ANGPTL4 exacerbates pancreatitis by augmenting acinar cell injury through upregulation of C5a.
Jung KH, Son MK, Yan HH, Fang Z, Kim J, Kim SJ, Park JH, Lee JE, Yoon YC, Seo MS, Han BS, Ko S, Suh YJ, Lim JH, Lee DH, Teo Z, Wee JWK, Tan NS, Hong SS
EMBO molecular medicine 2020 Aug 7;12(8):e11222
EMBO molecular medicine 2020 Aug 7;12(8):e11222
Circulating progenitor cells and the expression of Cxcl12, Cxcr4 and angiopoietin-like 4 during wound healing in the murine ear.
Yellowley CE, Toupadakis CA, Vapniarsky N, Wong A
PloS one 2019;14(9):e0222462
PloS one 2019;14(9):e0222462
Hydrogen peroxide promotes the expression of angiopoietin like 4 in RAW264.7 macrophages via MAPK pathways.
Liu N, Cui C, Sun Y, Zhang F, Wang S, Su G, Cai X
Molecular medicine reports 2017 Nov;16(5):6128-6133
Molecular medicine reports 2017 Nov;16(5):6128-6133
ANGPTL4 deficiency in haematopoietic cells promotes monocyte expansion and atherosclerosis progression.
Aryal B, Rotllan N, Araldi E, Ramírez CM, He S, Chousterman BG, Fenn AM, Wanschel A, Madrigal-Matute J, Warrier N, Martín-Ventura JL, Swirski FK, Suárez Y, Fernández-Hernando C
Nature communications 2016 Jul 27;7:12313
Nature communications 2016 Jul 27;7:12313
GATA3 suppresses metastasis and modulates the tumour microenvironment by regulating microRNA-29b expression.
Chou J, Lin JH, Brenot A, Kim JW, Provot S, Werb Z
Nature cell biology 2013 Feb;15(2):201-13
Nature cell biology 2013 Feb;15(2):201-13
Mechanism of hypertriglyceridemia in CTP:phosphoethanolamine cytidylyltransferase-deficient mice.
Singh RK, Fullerton MD, Vine D, Bakovic M
Journal of lipid research 2012 Sep;53(9):1811-22
Journal of lipid research 2012 Sep;53(9):1811-22
Angiopoietin like protein 4 expression is decreased in activated macrophages.
Feingold KR, Shigenaga JK, Cross AS, Moser A, Grunfeld C
Biochemical and biophysical research communications 2012 May 11;421(3):612-5
Biochemical and biophysical research communications 2012 May 11;421(3):612-5
Decreased fat storage by Lactobacillus paracasei is associated with increased levels of angiopoietin-like 4 protein (ANGPTL4).
Aronsson L, Huang Y, Parini P, Korach-André M, Håkansson J, Gustafsson JÅ, Pettersson S, Arulampalam V, Rafter J
PloS one 2010 Sep 30;5(9)
PloS one 2010 Sep 30;5(9)
The acute phase response stimulates the expression of angiopoietin like protein 4.
Lu B, Moser A, Shigenaga JK, Grunfeld C, Feingold KR
Biochemical and biophysical research communications 2010 Jan 22;391(4):1737-41
Biochemical and biophysical research communications 2010 Jan 22;391(4):1737-41
Activating peroxisome proliferator-activated receptor gamma mutant promotes tumor growth in vivo by enhancing angiogenesis.
Tian L, Zhou J, Casimiro MC, Liang B, Ojeifo JO, Wang M, Hyslop T, Wang C, Pestell RG
Cancer research 2009 Dec 15;69(24):9236-44
Cancer research 2009 Dec 15;69(24):9236-44
Enhanced angiogenesis in obesity and in response to PPARgamma activators through adipocyte VEGF and ANGPTL4 production.
Gealekman O, Burkart A, Chouinard M, Nicoloro SM, Straubhaar J, Corvera S
American journal of physiology. Endocrinology and metabolism 2008 Nov;295(5):E1056-64
American journal of physiology. Endocrinology and metabolism 2008 Nov;295(5):E1056-64
Peroxisome proliferator-activated receptor-beta/delta (PPARbeta/delta) ligands do not potentiate growth of human cancer cell lines.
Hollingshead HE, Killins RL, Borland MG, Girroir EE, Billin AN, Willson TM, Sharma AK, Amin S, Gonzalez FJ, Peters JM
Carcinogenesis 2007 Dec;28(12):2641-9
Carcinogenesis 2007 Dec;28(12):2641-9
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of (A) HepG2 and (B) NGP96 cell lysates using Zymed Rb anti-ANGPTL4 (Mid) (Product # 40-9800).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of (A) HepG2 and (B) NGP96 cell lysates using Zymed Rb anti-ANGPTL4 (Mid) (Product # 40-9800).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of ANGPTL4 showing staining in the cytoplasm and nucleus of paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Anti- ANGPTL4 Polyclonal Antibody (Product # 40-9800) diluted in 3% BSA-PBS at a dilution of 1:50 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of ANGPTL4 showing staining in the cytoplasm and nucleus of paraffin-embedded mouse liver tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Anti- ANGPTL4 Polyclonal Antibody (Product # 40-9800) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 F19 supplementation decreases fat storage in vivo . A) The two groups (5 mice/group) of high-fat (HF) and high-fat supplemented with F19 (HF+F19) were pair-fed, referenced here by food consumption in grams/week. B) Free fatty acid content in the serum of the two groups. C) Lipoprotein profiles of both cholesterol and triglyceride contents of very low density lipoproteins (VLDL), low density lipoproteins (LDL), and high density lipoproteins (HDL). D) Western blot for full length ANGPTL4 in pooled serum (10 ug protein loaded) from HF and HF+F19 mice along with numerical representation of the same. E) Representative images from MRI visualizing fat depots (in white) in both the abdominal and visceral regions. F) Body fat percentages for the two groups. Stars represent P = 0.045 (*), P = 0.002 (**) using Student's t-test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Probiotics upregulate ANGPTL4 expression in colonic cell lines. A) Real-time PCR of ANGPTL4 mRNA in HCT116 cells co-cultured with Lactobacillus F19 (F19; 10 7 /ml), Bifidobacterium lactis (BB12; 10 7 /ml) and Bacteroides thetaiotaomicron (B.theta; 10 7 /ml) respectively for 6 h were compared to non-treated (NT) control. B) Analysis of ANGPTL4 mRNA after 6 h stimulation with F19 at different concentrations in HCT116 cells. C) Time-course of F19 (10 8 ) on ANGPTL4 mRNA expression in HCT116 cells. D) Western (50 ug) of full length ANGPTL4 in HCT116 cells treated with F19 for 6 h and collected after 24 h. Actin is shown as loading control. E) ANGPTL4 mRNA in the colon carcinoma cell lines LoVo, HT29 and SW480. Real-time PCR data are presented as means with standard errors. All data are representative of at least 3 independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 F19 monocolonization in germ-free mice increases ANGPTL4 protein levels in serum. A) Western blot for full length ANGPTL4 levels in a serially diluted (50, 25, 12.5, 6.25 ug protein) serum pool of control (PBS) and mono-infected mice (F19), along with a collated numerical representation of the same western corrected for loading. B) Cholesterol and triglyceride profiles of very low density lipoprotein (VLDL), low density lipoprotein (LDL), and high density lipoprotein (HDL) in control (PBS) and mono-infected serum (F19). Indicated bars represent the average value of each data set, n = 6, while stars represent P = 0.045 (*), P = 0.002 (**) (Student's t-test).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
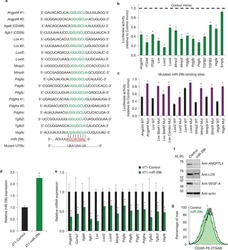
- Figure 1 ANGPTL4 is expressed in macrophages accumulated in atherosclerotic plaques. ( a ) Heat map representation of gene expression from microarray data comparing mouse peritoneal macrophages incubated with or without Ac-LDL (120 mug ml -1 ) for 24 h. Left panel shows upregulated genes ( P =1.4) and right panel shows downregulated genes ( P =2.08) upon Ac-LDL treatment compared with non-treated cells. Samples were analysed in triplicate. ( b ) qRT-PCR validation of selected genes upregulated in the microarray in mouse peritoneal macrophages treated with or without Ac-LDL for 24 h. qRT-PCR analysis of Angptl4 expression levels in macrophages ( c , left) and whole aorta ( c , right) from WT and Ldlr -/- mice fed a WD (left) ( n =4 per group), and in human peripheral blood mononuclear cells ( d ) treated with or without Ac-LDL (120 mug ml -1 ) for 24 h. Abca1 and Hmgcr genes were used as control genes for cholesterol loading and Cd68 was used as a marker for macrophages. All data represent the mean+-s.e.m. and are representative of three experiments in duplicate; * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
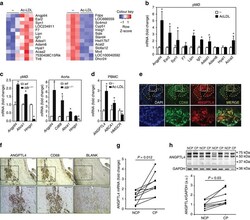
- Figure 3 Alleviation of pancreatitis in ANGPTL 4 -/- ( KO ) mice A AP and SAP were induced in wild-type (WT) and ANGPTL4 -/- mice ( n = 15, each group), as described in the material and methods. H&E staining, TUNEL staining for apoptosis, and immunofluorescence staining for ANGPTL4 (red) and amylase (green) were performed in the pancreatic tissues. ANGPTL4 and IL-6 protein expressions were also determined in pancreatic tissues from ANGPTL4 WT and -/- mice by Western blotting. Scale bar represents 100 mum. B Levels of ANGPTL4, amylase, lipase, and MPO in serum ( n = 15, each group). C Immunofluorescence staining of TNF-alpha and IL-6, and the levels of the inflammatory cytokines TGF-beta, IFN-gamma, and IL-1beta in the sera from ANGPTL4 WT and -/- mice ( n = 10, each group). Scale bar represents 150 mum. Data information: Each value represents the mean +- SEM. (*** P < 0.001). Values of P were calculated using one-way ANOVA with Tukey's post hoc analysis. Exact P values are shown in Appendix Table S2 . Source data are available online for this figure.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 Therapeutic effect of the ANGPTL 4-neutralizing antibody in AP and SAP A Concentrations of C5a in the sera from pancreatitis patients, and correlation between ANGPTL4 and C5a in the sera from pancreatitis patients ( n = 90). The box plots show a typical display consisting of a median value depicted by the line in the center of the box; an interquartile range (IQR; 25 th to the 75 th percentile) depicted by the box; and the maximum (Q3 + 1.5*IQR) and minimum (Q1-1.5*IQR) values depicted by the whisker. Statistical significance of Mann-Whitney U -tests is indicated (*** P < 0.001). Exact P values are shown in Appendix Table S2 . The correlation between C5a and ANGPTL4 is depicted with Pearson correlation coefficient (R). Each dot represents the levels of C5a and ANGPTL4 from an individual patient ( n = 90). The immunofluorescent expression of C5a and amylase was observed in pancreatitis and adjacent normal regions in patient tissues. Scale bar represents 50 mum. B Immunofluorescence staining of C5a in pancreas tissues from ANGPTL4 -/- and WT animals and macrophage depletion/WT models with pancreatitis. Scale bar represents 150 mum. Levels of C5a were measured in serum ( n = 7). C After macrophages were activated with LPS (100 ng/ml), the cells were treated with either neutralizing ANGPTL4 or C5a antibody (100 ng/ml) or an isotype control (IgG). ANGPTL4, C5a, TNF-alpha, and IL-6 levels were measured in culture media ( n = 5). D The neutralizing ANGPTL4 antibody (10 mg/k
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 5 Immunohistochemical staining of ANGPTL4. ANGPTL4 staining (brown) at the wound site immediately (day0) after wounding, and at days 1, 2, 3, 7 and 14 after wounding. Images are representative of n = 3.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot