Antibody data
- Antibody Data
- Antigen structure
- References [62]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [35]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 14-0038-80 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Anti-CD3 Monoclonal Antibody (UCHT1), eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The UCHT1 monoclonal antibody reacts with human CD3e, a 20 kDa subunit of the TCR complex. Along with the other CD3 subunits gamma and delta, the epsilon chain is required for proper assembly, trafficking and surface expression of the TCR complex. CD3 is expressed by thymocytes in a developmentally regulated manner and by all mature T cells. Crosslinking of TCR via immobilized UCHT1 initiates an intracellular biochemical pathway resulting in cellular activation and proliferation. Applications Reported: The UCHT1 antibody has been reported for use in flow cytometric analysis, and immunohistochemical staining. UCHT1 has also been reported in activation of T cells. (Please use Functional Grade purified UCHT1, cat. 16-0038, in functional assays.). Applications Tested: The UCHT1 antibody has been tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at less than or equal to 1 µg per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest. Purity: Greater than 90%, as determined by SDS-PAGE. Aggregation: Less than 10%, as determined by HPLC. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- UCHT1
- Vial size
- 25 µg
- Concentration
- 0.5 mg/mL
- Storage
- 4° C
Submitted references Dendritic Cell Maturation Regulates TSPAN7 Function in HIV-1 Transfer to CD4+ T Lymphocytes.
High-affinity mutant Interleukin-13 targeted CAR T cells enhance delivery of clickable biodegradable fluorescent nanoparticles to glioblastoma.
DNA hypomethylation promotes transposable element expression and activation of immune signaling in renal cell cancer.
Defective glycosylation and multisystem abnormalities characterize the primary immunodeficiency XMEN disease.
Skewed T cell responses to Epstein-Barr virus in long-term asymptomatic kidney transplant recipients.
Inflammation induced by incomplete radiofrequency ablation accelerates tumor progression and hinders PD-1 immunotherapy.
Gpr174-deficient regulatory T cells decrease cytokine storm in septic mice.
Loss of ARPC1B impairs cytotoxic T lymphocyte maintenance and cytolytic activity.
Regulatory T cells use arginase 2 to enhance their metabolic fitness in tissues.
T Cells on Engineered Substrates: The Impact of TCR Clustering Is Enhanced by LFA-1 Engagement.
FUNDAMANT: an interventional 72-week phase 1 follow-up study of AADvac1, an active immunotherapy against tau protein pathology in Alzheimer's disease.
Dysfunction of CD19+CD24hiCD27+ B regulatory cells in patients with bullous pemphigoid.
Biallelic mutations in DNA ligase 1 underlie a spectrum of immune deficiencies.
Antigen receptor-redirected T cells derived from hematopoietic precursor cells lack expression of the endogenous TCR/CD3 receptor and exhibit specific antitumor capacities.
Follicular Regulatory T Cells Are Highly Permissive to R5-Tropic HIV-1.
NK-Cell Recruitment Is Necessary for Eradication of Peritoneal Carcinomatosis with an IL12-Expressing Maraba Virus Cellular Vaccine.
Frequency of CD4+CD161+ T Cell and Interleukin-10 Expression in Inflammatory Bowel Diseases.
Dual role of tumour-infiltrating T helper 17 cells in human colorectal cancer.
Blocking the recruitment of naive CD4+ T cells reverses immunosuppression in breast cancer.
The Effect of Chronic Hepatitis B Virus Infection on BDCA3+ Dendritic Cell Frequency and Function.
Distinct Transcriptional Changes and Epithelial-Stromal Interactions Are Altered in Early-Stage Colon Cancer Development.
MAIT cells are activated during human viral infections.
Interaction among activated lymphocytes and mesenchymal cells through podoplanin is critical for a high IL-17 secretion.
Tissue factor expression by myeloid cells contributes to protective immune response against Mycobacterium tuberculosis infection.
Atypical natural killer T-cell receptor recognition of CD1d-lipid antigens.
Distinct Metabolic Requirements of Exhausted and Functional Virus-Specific CD8 T Cells in the Same Host.
Higher Frequency of NK and CD4+ T-Cells in Mucosa and Potent Cytotoxic Response in HIV Controllers.
Transfer of mRNA Encoding Invariant NKT Cell Receptors Imparts Glycolipid Specific Responses to T Cells and γδT Cells.
Follicular regulatory T cells impair follicular T helper cells in HIV and SIV infection.
A polymorphism in a phosphotyrosine signalling motif of CD229 (Ly9, SLAMF3) alters SH2 domain binding and T-cell activation.
Jejunal T Cell Inflammation in Human Obesity Correlates with Decreased Enterocyte Insulin Signaling.
Thymic HIV-2 infection uncovers posttranscriptional control of viral replication in human thymocytes.
The Nrf2 activator tBHQ inhibits T cell activation of primary human CD4 T cells.
Induction of type I and type III interferons by Borrelia burgdorferi correlates with pathogenesis and requires linear plasmid 36.
Novel lentiviral vectors with mutated reverse transcriptase for mRNA delivery of TALE nucleases.
A comparison of DNA methylation specific droplet digital PCR (ddPCR) and real time qPCR with flow cytometry in characterizing human T cells in peripheral blood.
Safety and immunogenicity of a candidate tuberculosis vaccine MVA85A delivered by aerosol in BCG-vaccinated healthy adults: a phase 1, double-blind, randomised controlled trial.
Signal transduction by different forms of the γδ T cell-specific pattern recognition receptor WC1.
Human mesenchymal stromal cells modulate T-cell responses through TNF-α-mediated activation of NF-κB.
Trypan blue exclusion assay by flow cytometry.
The antiangiogenic insulin receptor substrate-1 antisense oligonucleotide aganirsen impairs AU-rich mRNA stability by reducing 14-3-3β-tristetraprolin protein complex, reducing inflammation and psoriatic lesion size in patients.
MHC multimer-guided and cell culture-independent isolation of functional T cell receptors from single cells facilitates TCR identification for immunotherapy.
Antibody-mediated targeting of the Orai1 calcium channel inhibits T cell function.
Mycobacterial phosphatidylinositol mannoside 6 (PIM6) up-regulates TCR-triggered HIV-1 replication in CD4+ T cells.
Intercellular adhesion molecule 1 mediates migration of Th1 and Th17 cells across human retinal vascular endothelium.
Recombinant influenza virus carrying the respiratory syncytial virus (RSV) F85-93 CTL epitope reduces RSV replication in mice.
Ulcerative colitis impairs the acylethanolamide-based anti-inflammatory system reversal by 5-aminosalicylic acid and glucocorticoids.
TNF-α and TGF-β counter-regulate PD-L1 expression on monocytes in systemic lupus erythematosus.
Epigenetic biomarkers of T-cells in human glioma.
HLA-G level on monocytoid dendritic cells correlates with regulatory T-cell Foxp3 expression in liver transplant tolerance.
Defective response of CD4(+) T cells to retinoic acid and TGFβ in systemic lupus erythematosus.
Immunostaining of PD-1/PD-Ls in liver tissues of patients with hepatitis and hepatocellular carcinoma.
Human Th1 cells that express CD300a are polyfunctional and after stimulation up-regulate the T-box transcription factor eomesodermin.
Accumulation of natural killer T cells in progressive nonalcoholic fatty liver disease.
Construction and preclinical evaluation of an anti-CD19 chimeric antigen receptor.
Loss of the Brm-type SWI/SNF chromatin remodeling complex is a strong barrier to the Tat-independent transcriptional elongation of human immunodeficiency virus type 1 transcripts.
Hepatitis B virus surface antigen impairs myeloid dendritic cell function: a possible immune escape mechanism of hepatitis B virus.
Lung dendritic cell expression of maturation molecules increases with worsening chronic obstructive pulmonary disease.
A role for human skin-resident T cells in wound healing.
Expansion of regulatory T cells in patients with Langerhans cell histiocytosis.
Bacterial superantigens bypass Lck-dependent T cell receptor signaling by activating a Galpha11-dependent, PLC-beta-mediated pathway.
Expression of the novel co-stimulatory molecule B7-H4 by renal tubular epithelial cells.
Perot BP, García-Paredes V, Luka M, Ménager MM
Frontiers in cellular and infection microbiology 2020;10:70
Frontiers in cellular and infection microbiology 2020;10:70
High-affinity mutant Interleukin-13 targeted CAR T cells enhance delivery of clickable biodegradable fluorescent nanoparticles to glioblastoma.
Kim GB, Aragon-Sanabria V, Randolph L, Jiang H, Reynolds JA, Webb BS, Madhankumar A, Lian X, Connor JR, Yang J, Dong C
Bioactive materials 2020 Sep;5(3):624-635
Bioactive materials 2020 Sep;5(3):624-635
DNA hypomethylation promotes transposable element expression and activation of immune signaling in renal cell cancer.
de Cubas AA, Dunker W, Zaninovich A, Hongo RA, Bhatia A, Panda A, Beckermann KE, Bhanot G, Ganesan S, Karijolich J, Rathmell WK
JCI insight 2020 Jun 4;5(11)
JCI insight 2020 Jun 4;5(11)
Defective glycosylation and multisystem abnormalities characterize the primary immunodeficiency XMEN disease.
Ravell JC, Matsuda-Lennikov M, Chauvin SD, Zou J, Biancalana M, Deeb SJ, Price S, Su HC, Notarangelo G, Jiang P, Morawski A, Kanellopoulou C, Binder K, Mukherjee R, Anibal JT, Sellers B, Zheng L, He T, George AB, Pittaluga S, Powers A, Kleiner DE, Kapuria D, Ghany M, Hunsberger S, Cohen JI, Uzel G, Bergerson J, Wolfe L, Toro C, Gahl W, Folio LR, Matthews H, Angelus P, Chinn IK, Orange JS, Trujillo-Vargas CM, Franco JL, Orrego-Arango J, Gutiérrez-Hincapié S, Patel NC, Raymond K, Patiroglu T, Unal E, Karakukcu M, Day AG, Mehta P, Masutani E, De Ravin SS, Malech HL, Altan-Bonnet G, Rao VK, Mann M, Lenardo MJ
The Journal of clinical investigation 2020 Jan 2;130(1):507-522
The Journal of clinical investigation 2020 Jan 2;130(1):507-522
Skewed T cell responses to Epstein-Barr virus in long-term asymptomatic kidney transplant recipients.
Nakid-Cordero C, Arzouk N, Gauthier N, Tarantino N, Larsen M, Choquet S, Burrel S, Autran B, Vieillard V, Guihot A
PloS one 2019;14(10):e0224211
PloS one 2019;14(10):e0224211
Inflammation induced by incomplete radiofrequency ablation accelerates tumor progression and hinders PD-1 immunotherapy.
Shi L, Wang J, Ding N, Zhang Y, Zhu Y, Dong S, Wang X, Peng C, Zhou C, Zhou L, Li X, Shi H, Wu W, Long X, Wu C, Liao W
Nature communications 2019 Nov 28;10(1):5421
Nature communications 2019 Nov 28;10(1):5421
Gpr174-deficient regulatory T cells decrease cytokine storm in septic mice.
Qiu D, Chu X, Hua L, Yang Y, Li K, Han Y, Yin J, Zhu M, Mu S, Sun Z, Tong C, Song Z
Cell death & disease 2019 Mar 8;10(3):233
Cell death & disease 2019 Mar 8;10(3):233
Loss of ARPC1B impairs cytotoxic T lymphocyte maintenance and cytolytic activity.
Randzavola LO, Strege K, Juzans M, Asano Y, Stinchcombe JC, Gawden-Bone CM, Seaman MN, Kuijpers TW, Griffiths GM
The Journal of clinical investigation 2019 Dec 2;129(12):5600-5614
The Journal of clinical investigation 2019 Dec 2;129(12):5600-5614
Regulatory T cells use arginase 2 to enhance their metabolic fitness in tissues.
Lowe MM, Boothby I, Clancy S, Ahn RS, Liao W, Nguyen DN, Schumann K, Marson A, Mahuron KM, Kingsbury GA, Liu Z, Munoz Sandoval P, Rodriguez RS, Pauli ML, Taravati K, Arron ST, Neuhaus IM, Harris HW, Kim EA, Shin US, Krummel MF, Daud A, Scharschmidt TC, Rosenblum MD
JCI insight 2019 Dec 19;4(24)
JCI insight 2019 Dec 19;4(24)
T Cells on Engineered Substrates: The Impact of TCR Clustering Is Enhanced by LFA-1 Engagement.
Benard E, Nunès JA, Limozin L, Sengupta K
Frontiers in immunology 2018;9:2085
Frontiers in immunology 2018;9:2085
FUNDAMANT: an interventional 72-week phase 1 follow-up study of AADvac1, an active immunotherapy against tau protein pathology in Alzheimer's disease.
Novak P, Schmidt R, Kontsekova E, Kovacech B, Smolek T, Katina S, Fialova L, Prcina M, Parrak V, Dal-Bianco P, Brunner M, Staffen W, Rainer M, Ondrus M, Ropele S, Smisek M, Sivak R, Zilka N, Winblad B, Novak M
Alzheimer's research & therapy 2018 Oct 24;10(1):108
Alzheimer's research & therapy 2018 Oct 24;10(1):108
Dysfunction of CD19+CD24hiCD27+ B regulatory cells in patients with bullous pemphigoid.
Liu Z, Dang E, Li B, Qiao H, Jin L, Zhang J, Wang G
Scientific reports 2018 Jan 15;8(1):703
Scientific reports 2018 Jan 15;8(1):703
Biallelic mutations in DNA ligase 1 underlie a spectrum of immune deficiencies.
Maffucci P, Chavez J, Jurkiw TJ, O'Brien PJ, Abbott JK, Reynolds PR, Worth A, Notarangelo LD, Felgentreff K, Cortes P, Boisson B, Radigan L, Cobat A, Dinakar C, Ehlayel M, Ben-Omran T, Gelfand EW, Casanova JL, Cunningham-Rundles C
The Journal of clinical investigation 2018 Dec 3;128(12):5489-5504
The Journal of clinical investigation 2018 Dec 3;128(12):5489-5504
Antigen receptor-redirected T cells derived from hematopoietic precursor cells lack expression of the endogenous TCR/CD3 receptor and exhibit specific antitumor capacities.
Van Caeneghem Y, De Munter S, Tieppo P, Goetgeluk G, Weening K, Verstichel G, Bonte S, Taghon T, Leclercq G, Kerre T, Debets R, Vermijlen D, Abken H, Vandekerckhove B
Oncoimmunology 2017;6(3):e1283460
Oncoimmunology 2017;6(3):e1283460
Follicular Regulatory T Cells Are Highly Permissive to R5-Tropic HIV-1.
Miller SM, Miles B, Guo K, Folkvord J, Meditz AL, McCarter MD, Levy DN, MaWhinney S, Santiago ML, Connick E
Journal of virology 2017 Sep 1;91(17)
Journal of virology 2017 Sep 1;91(17)
NK-Cell Recruitment Is Necessary for Eradication of Peritoneal Carcinomatosis with an IL12-Expressing Maraba Virus Cellular Vaccine.
Alkayyal AA, Tai LH, Kennedy MA, de Souza CT, Zhang J, Lefebvre C, Sahi S, Ananth AA, Mahmoud AB, Makrigiannis AP, Cron GO, Macdonald B, Marginean EC, Stojdl DF, Bell JC, Auer RC
Cancer immunology research 2017 Mar;5(3):211-221
Cancer immunology research 2017 Mar;5(3):211-221
Frequency of CD4+CD161+ T Cell and Interleukin-10 Expression in Inflammatory Bowel Diseases.
Tsuchiya K, Ikeda T, Batmunkh B, Choijookhuu N, Ishizaki H, Hotokezaka M, Hishikawa Y, Nanashima A
Acta histochemica et cytochemica 2017 Feb 28;50(1):21-28
Acta histochemica et cytochemica 2017 Feb 28;50(1):21-28
Dual role of tumour-infiltrating T helper 17 cells in human colorectal cancer.
Amicarella F, Muraro MG, Hirt C, Cremonesi E, Padovan E, Mele V, Governa V, Han J, Huber X, Droeser RA, Zuber M, Adamina M, Bolli M, Rosso R, Lugli A, Zlobec I, Terracciano L, Tornillo L, Zajac P, Eppenberger-Castori S, Trapani F, Oertli D, Iezzi G
Gut 2017 Apr;66(4):692-704
Gut 2017 Apr;66(4):692-704
Blocking the recruitment of naive CD4+ T cells reverses immunosuppression in breast cancer.
Su S, Liao J, Liu J, Huang D, He C, Chen F, Yang L, Wu W, Chen J, Lin L, Zeng Y, Ouyang N, Cui X, Yao H, Su F, Huang JD, Lieberman J, Liu Q, Song E
Cell research 2017 Apr;27(4):461-482
Cell research 2017 Apr;27(4):461-482
The Effect of Chronic Hepatitis B Virus Infection on BDCA3+ Dendritic Cell Frequency and Function.
van der Aa E, Buschow SI, Biesta PJ, Janssen HL, Woltman AM
PloS one 2016;11(8):e0161235
PloS one 2016;11(8):e0161235
Distinct Transcriptional Changes and Epithelial-Stromal Interactions Are Altered in Early-Stage Colon Cancer Development.
Mo A, Jackson S, Varma K, Carpino A, Giardina C, Devers TJ, Rosenberg DW
Molecular cancer research : MCR 2016 Sep;14(9):795-804
Molecular cancer research : MCR 2016 Sep;14(9):795-804
MAIT cells are activated during human viral infections.
van Wilgenburg B, Scherwitzl I, Hutchinson EC, Leng T, Kurioka A, Kulicke C, de Lara C, Cole S, Vasanawathana S, Limpitikul W, Malasit P, Young D, Denney L, STOP-HCV consortium., Moore MD, Fabris P, Giordani MT, Oo YH, Laidlaw SM, Dustin LB, Ho LP, Thompson FM, Ramamurthy N, Mongkolsapaya J, Willberg CB, Screaton GR, Klenerman P
Nature communications 2016 Jun 23;7:11653
Nature communications 2016 Jun 23;7:11653
Interaction among activated lymphocytes and mesenchymal cells through podoplanin is critical for a high IL-17 secretion.
Noack M, Ndongo-Thiam N, Miossec P
Arthritis research & therapy 2016 Jun 23;18:148
Arthritis research & therapy 2016 Jun 23;18:148
Tissue factor expression by myeloid cells contributes to protective immune response against Mycobacterium tuberculosis infection.
Venkatasubramanian S, Tripathi D, Tucker T, Paidipally P, Cheekatla S, Welch E, Raghunath A, Jeffers A, Tvinnereim AR, Schechter ME, Andrade BB, Mackman N, Idell S, Vankayalapati R
European journal of immunology 2016 Feb;46(2):464-79
European journal of immunology 2016 Feb;46(2):464-79
Atypical natural killer T-cell receptor recognition of CD1d-lipid antigens.
Le Nours J, Praveena T, Pellicci DG, Gherardin NA, Ross FJ, Lim RT, Besra GS, Keshipeddy S, Richardson SK, Howell AR, Gras S, Godfrey DI, Rossjohn J, Uldrich AP
Nature communications 2016 Feb 15;7:10570
Nature communications 2016 Feb 15;7:10570
Distinct Metabolic Requirements of Exhausted and Functional Virus-Specific CD8 T Cells in the Same Host.
Schurich A, Pallett LJ, Jajbhay D, Wijngaarden J, Otano I, Gill US, Hansi N, Kennedy PT, Nastouli E, Gilson R, Frezza C, Henson SM, Maini MK
Cell reports 2016 Aug 2;16(5):1243-1252
Cell reports 2016 Aug 2;16(5):1243-1252
Higher Frequency of NK and CD4+ T-Cells in Mucosa and Potent Cytotoxic Response in HIV Controllers.
Taborda NA, González SM, Alvarez CM, Correa LA, Montoya CJ, Rugeles MT
PloS one 2015;10(8):e0136292
PloS one 2015;10(8):e0136292
Transfer of mRNA Encoding Invariant NKT Cell Receptors Imparts Glycolipid Specific Responses to T Cells and γδT Cells.
Shimizu K, Shinga J, Yamasaki S, Kawamura M, Dörrie J, Schaft N, Sato Y, Iyoda T, Fujii S
PloS one 2015;10(6):e0131477
PloS one 2015;10(6):e0131477
Follicular regulatory T cells impair follicular T helper cells in HIV and SIV infection.
Miles B, Miller SM, Folkvord JM, Kimball A, Chamanian M, Meditz AL, Arends T, McCarter MD, Levy DN, Rakasz EG, Skinner PJ, Connick E
Nature communications 2015 Oct 20;6:8608
Nature communications 2015 Oct 20;6:8608
A polymorphism in a phosphotyrosine signalling motif of CD229 (Ly9, SLAMF3) alters SH2 domain binding and T-cell activation.
Margraf S, Garner LI, Wilson TJ, Brown MH
Immunology 2015 Nov;146(3):392-400
Immunology 2015 Nov;146(3):392-400
Jejunal T Cell Inflammation in Human Obesity Correlates with Decreased Enterocyte Insulin Signaling.
Monteiro-Sepulveda M, Touch S, Mendes-Sá C, André S, Poitou C, Allatif O, Cotillard A, Fohrer-Ting H, Hubert EL, Remark R, Genser L, Tordjman J, Garbin K, Osinski C, Sautès-Fridman C, Leturque A, Clément K, Brot-Laroche E
Cell metabolism 2015 Jul 7;22(1):113-24
Cell metabolism 2015 Jul 7;22(1):113-24
Thymic HIV-2 infection uncovers posttranscriptional control of viral replication in human thymocytes.
Nunes-Cabaço H, Matoso P, Foxall RB, Tendeiro R, Pires AR, Carvalho T, Pinheiro AI, Soares RS, Sousa AE
Journal of virology 2015 Feb;89(4):2201-8
Journal of virology 2015 Feb;89(4):2201-8
The Nrf2 activator tBHQ inhibits T cell activation of primary human CD4 T cells.
Turley AE, Zagorski JW, Rockwell CE
Cytokine 2015 Feb;71(2):289-95
Cytokine 2015 Feb;71(2):289-95
Induction of type I and type III interferons by Borrelia burgdorferi correlates with pathogenesis and requires linear plasmid 36.
Krupna-Gaylord MA, Liveris D, Love AC, Wormser GP, Schwartz I, Petzke MM
PloS one 2014;9(6):e100174
PloS one 2014;9(6):e100174
Novel lentiviral vectors with mutated reverse transcriptase for mRNA delivery of TALE nucleases.
Mock U, Riecken K, Berdien B, Qasim W, Chan E, Cathomen T, Fehse B
Scientific reports 2014 Sep 18;4:6409
Scientific reports 2014 Sep 18;4:6409
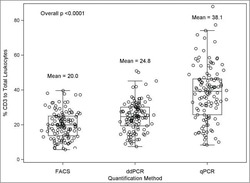
A comparison of DNA methylation specific droplet digital PCR (ddPCR) and real time qPCR with flow cytometry in characterizing human T cells in peripheral blood.
Wiencke JK, Bracci PM, Hsuang G, Zheng S, Hansen H, Wrensch MR, Rice T, Eliot M, Kelsey KT
Epigenetics 2014 Oct;9(10):1360-5
Epigenetics 2014 Oct;9(10):1360-5
Safety and immunogenicity of a candidate tuberculosis vaccine MVA85A delivered by aerosol in BCG-vaccinated healthy adults: a phase 1, double-blind, randomised controlled trial.
Satti I, Meyer J, Harris SA, Manjaly Thomas ZR, Griffiths K, Antrobus RD, Rowland R, Ramon RL, Smith M, Sheehan S, Bettinson H, McShane H
The Lancet. Infectious diseases 2014 Oct;14(10):939-46
The Lancet. Infectious diseases 2014 Oct;14(10):939-46
Signal transduction by different forms of the γδ T cell-specific pattern recognition receptor WC1.
Chen C, Hsu H, Hudgens E, Telfer JC, Baldwin CL
Journal of immunology (Baltimore, Md. : 1950) 2014 Jul 1;193(1):379-90
Journal of immunology (Baltimore, Md. : 1950) 2014 Jul 1;193(1):379-90
Human mesenchymal stromal cells modulate T-cell responses through TNF-α-mediated activation of NF-κB.
Dorronsoro A, Ferrin I, Salcedo JM, Jakobsson E, Fernández-Rueda J, Lang V, Sepulveda P, Fechter K, Pennington D, Trigueros C
European journal of immunology 2014 Feb;44(2):480-8
European journal of immunology 2014 Feb;44(2):480-8
Trypan blue exclusion assay by flow cytometry.
Avelar-Freitas BA, Almeida VG, Pinto MC, Mourão FA, Massensini AR, Martins-Filho OA, Rocha-Vieira E, Brito-Melo GE
Brazilian journal of medical and biological research = Revista brasileira de pesquisas medicas e biologicas 2014 Apr;47(4):307-15
Brazilian journal of medical and biological research = Revista brasileira de pesquisas medicas e biologicas 2014 Apr;47(4):307-15
The antiangiogenic insulin receptor substrate-1 antisense oligonucleotide aganirsen impairs AU-rich mRNA stability by reducing 14-3-3β-tristetraprolin protein complex, reducing inflammation and psoriatic lesion size in patients.
Colin S, Darné B, Kadi A, Ferry A, Favier M, Lesaffre C, Conduzorgues JP, Al-Mahmood S, Doss N
The Journal of pharmacology and experimental therapeutics 2014 Apr;349(1):107-17
The Journal of pharmacology and experimental therapeutics 2014 Apr;349(1):107-17
MHC multimer-guided and cell culture-independent isolation of functional T cell receptors from single cells facilitates TCR identification for immunotherapy.
Dössinger G, Bunse M, Bet J, Albrecht J, Paszkiewicz PJ, Weißbrich B, Schiedewitz I, Henkel L, Schiemann M, Neuenhahn M, Uckert W, Busch DH
PloS one 2013;8(4):e61384
PloS one 2013;8(4):e61384
Antibody-mediated targeting of the Orai1 calcium channel inhibits T cell function.
Cox JH, Hussell S, Søndergaard H, Roepstorff K, Bui JV, Deer JR, Zhang J, Li ZG, Lamberth K, Kvist PH, Padkjær S, Haase C, Zahn S, Odegard VH
PloS one 2013;8(12):e82944
PloS one 2013;8(12):e82944
Mycobacterial phosphatidylinositol mannoside 6 (PIM6) up-regulates TCR-triggered HIV-1 replication in CD4+ T cells.
Rodriguez ME, Loyd CM, Ding X, Karim AF, McDonald DJ, Canaday DH, Rojas RE
PloS one 2013;8(11):e80938
PloS one 2013;8(11):e80938
Intercellular adhesion molecule 1 mediates migration of Th1 and Th17 cells across human retinal vascular endothelium.
Bharadwaj AS, Schewitz-Bowers LP, Wei L, Lee RW, Smith JR
Investigative ophthalmology & visual science 2013 Oct 23;54(10):6917-25
Investigative ophthalmology & visual science 2013 Oct 23;54(10):6917-25
Recombinant influenza virus carrying the respiratory syncytial virus (RSV) F85-93 CTL epitope reduces RSV replication in mice.
De Baets S, Schepens B, Sedeyn K, Schotsaert M, Roose K, Bogaert P, Fiers W, Saelens X
Journal of virology 2013 Mar;87(6):3314-23
Journal of virology 2013 Mar;87(6):3314-23
Ulcerative colitis impairs the acylethanolamide-based anti-inflammatory system reversal by 5-aminosalicylic acid and glucocorticoids.
Suárez J, Romero-Zerbo Y, Márquez L, Rivera P, Iglesias M, Bermúdez-Silva FJ, Andreu M, Rodríguez de Fonseca F
PloS one 2012;7(5):e37729
PloS one 2012;7(5):e37729
TNF-α and TGF-β counter-regulate PD-L1 expression on monocytes in systemic lupus erythematosus.
Ou JN, Wiedeman AE, Stevens AM
Scientific reports 2012;2:295
Scientific reports 2012;2:295
Epigenetic biomarkers of T-cells in human glioma.
Wiencke JK, Accomando WP, Zheng S, Patoka J, Dou X, Phillips JJ, Hsuang G, Christensen BC, Houseman EA, Koestler DC, Bracci P, Wiemels JL, Wrensch M, Nelson HH, Kelsey KT
Epigenetics 2012 Dec 1;7(12):1391-402
Epigenetics 2012 Dec 1;7(12):1391-402
HLA-G level on monocytoid dendritic cells correlates with regulatory T-cell Foxp3 expression in liver transplant tolerance.
Castellaneta A, Mazariegos GV, Nayyar N, Zeevi A, Thomson AW
Transplantation 2011 May 27;91(10):1132-40
Transplantation 2011 May 27;91(10):1132-40
Defective response of CD4(+) T cells to retinoic acid and TGFβ in systemic lupus erythematosus.
Sobel ES, Brusko TM, Butfiloski EJ, Hou W, Li S, Cuda CM, Abid AN, Reeves WH, Morel L
Arthritis research & therapy 2011 Jun 27;13(3):R106
Arthritis research & therapy 2011 Jun 27;13(3):R106
Immunostaining of PD-1/PD-Ls in liver tissues of patients with hepatitis and hepatocellular carcinoma.
Wang BJ, Bao JJ, Wang JZ, Wang Y, Jiang M, Xing MY, Zhang WG, Qi JY, Roggendorf M, Lu MJ, Yang DL
World journal of gastroenterology 2011 Jul 28;17(28):3322-9
World journal of gastroenterology 2011 Jul 28;17(28):3322-9
Human Th1 cells that express CD300a are polyfunctional and after stimulation up-regulate the T-box transcription factor eomesodermin.
Narayanan S, Silva R, Peruzzi G, Alvarez Y, Simhadri VR, Debell K, Coligan JE, Borrego F
PloS one 2010 May 13;5(5):e10636
PloS one 2010 May 13;5(5):e10636
Accumulation of natural killer T cells in progressive nonalcoholic fatty liver disease.
Syn WK, Oo YH, Pereira TA, Karaca GF, Jung Y, Omenetti A, Witek RP, Choi SS, Guy CD, Fearing CM, Teaberry V, Pereira FE, Adams DH, Diehl AM
Hepatology (Baltimore, Md.) 2010 Jun;51(6):1998-2007
Hepatology (Baltimore, Md.) 2010 Jun;51(6):1998-2007
Construction and preclinical evaluation of an anti-CD19 chimeric antigen receptor.
Kochenderfer JN, Feldman SA, Zhao Y, Xu H, Black MA, Morgan RA, Wilson WH, Rosenberg SA
Journal of immunotherapy (Hagerstown, Md. : 1997) 2009 Sep;32(7):689-702
Journal of immunotherapy (Hagerstown, Md. : 1997) 2009 Sep;32(7):689-702
Loss of the Brm-type SWI/SNF chromatin remodeling complex is a strong barrier to the Tat-independent transcriptional elongation of human immunodeficiency virus type 1 transcripts.
Mizutani T, Ishizaka A, Tomizawa M, Okazaki T, Yamamichi N, Kawana-Tachikawa A, Iwamoto A, Iba H
Journal of virology 2009 Nov;83(22):11569-80
Journal of virology 2009 Nov;83(22):11569-80
Hepatitis B virus surface antigen impairs myeloid dendritic cell function: a possible immune escape mechanism of hepatitis B virus.
Op den Brouw ML, Binda RS, van Roosmalen MH, Protzer U, Janssen HL, van der Molen RG, Woltman AM
Immunology 2009 Feb;126(2):280-9
Immunology 2009 Feb;126(2):280-9
Lung dendritic cell expression of maturation molecules increases with worsening chronic obstructive pulmonary disease.
Freeman CM, Martinez FJ, Han MK, Ames TM, Chensue SW, Todt JC, Arenberg DA, Meldrum CA, Getty C, McCloskey L, Curtis JL
American journal of respiratory and critical care medicine 2009 Dec 15;180(12):1179-88
American journal of respiratory and critical care medicine 2009 Dec 15;180(12):1179-88
A role for human skin-resident T cells in wound healing.
Toulon A, Breton L, Taylor KR, Tenenhaus M, Bhavsar D, Lanigan C, Rudolph R, Jameson J, Havran WL
The Journal of experimental medicine 2009 Apr 13;206(4):743-50
The Journal of experimental medicine 2009 Apr 13;206(4):743-50
Expansion of regulatory T cells in patients with Langerhans cell histiocytosis.
Senechal B, Elain G, Jeziorski E, Grondin V, Patey-Mariaud de Serre N, Jaubert F, Beldjord K, Lellouch A, Glorion C, Zerah M, Mary P, Barkaoui M, Emile JF, Boccon-Gibod L, Josset P, Debré M, Fischer A, Donadieu J, Geissmann F
PLoS medicine 2007 Aug;4(8):e253
PLoS medicine 2007 Aug;4(8):e253
Bacterial superantigens bypass Lck-dependent T cell receptor signaling by activating a Galpha11-dependent, PLC-beta-mediated pathway.
Bueno C, Lemke CD, Criado G, Baroja ML, Ferguson SS, Rahman AK, Tsoukas CD, McCormick JK, Madrenas J
Immunity 2006 Jul;25(1):67-78
Immunity 2006 Jul;25(1):67-78
Expression of the novel co-stimulatory molecule B7-H4 by renal tubular epithelial cells.
Chen Y, Yang C, Xie Z, Zou L, Ruan Z, Zhang X, Tang Y, Fei L, Jia Z, Wu Y
Kidney international 2006 Dec;70(12):2092-9
Kidney international 2006 Dec;70(12):2092-9
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
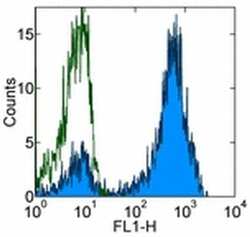
- Experimental details
- Staining of normal human peripheral blood cells with 0.5 µg of Mouse IgG1 K Isotype Control Purified (Product # 14-4714-82) (open histogram) or 0.5 µg of Anti-Human CD3 Purified (filled histogram) followed by Anti-Mouse IgG FITC (Product # 11-4011-85). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Representative high-magnification photomicrographs showing double immunofluorescence for NAAA, CD19, CD3 and CD14 in order to characterize the immune cells in the mucosa infiltrate of UC patients. NAAA immunofluorescence was observed in CD19+ B lymphocytes (A-C), CD3+ T lymphocytes (D-F) and CD14+ macrophages (G-I).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 9 Representative high-magnification photomicrographs showing double immunofluorescence for FAAH, NAPE-PLD, CD38 and CD3 in order to characterize the immune cells in the mucosa infiltrate of UC patients. Nearly all FAAH immunofluorescent cells are plasma cell-specific CD38 (A-C). NAPE-PLD immunofluorescence was observed in both CD38+ plasma cells (D-F) and CD3+ T lymphocytes (G-I).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Profile of T-lymphocytes stained with monoclonal antibody anti-CD3-FITCfollowed by treatment with propidium iodide (PI) and trypan blue (TB) at 0.002and 0.4% (w/v) or PBS (untreated control).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 A , Percentage of dead human T-lymphocytes(CD3-FITC + cells) submitted to cell culture at temperatures (T)of 37degC (physiologic temperature) or 50degC (high-stress temperature) followed bystaining with trypan blue (TB) or propidium iodide (PI). B ,Dot-plot graph profile between human lymphocytes submitted to pretreatment withhigh-stress temperature (50degC) followed by staining with TB and PI andmonoclonal antibody anti-CD3-FITC + . C , Pearson'scorrelation test between dead CD3 + lymphocytes using PI and TB flowcytometry assays.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Functional analysis of NRTLV-delivered TALEN constructs. (a) Schematic vector design of I: LeGO-iTALEN-iG2 (wPRE), II: LeGO-iTALEN-iG2-wPRE-BGH-p(A) and III: LeGO-iTALEN-iG2-wPRE-SV40-p(A) 3 rd generation LVV derived from LeGO-system 11 . CMV = CMV-ie promoter; Delta (DeltaU3), R, U5 = elements of SIN-LTR, self-inactivating long terminal repeat; Psi = Psi , packaging signal; RRE = Rev response element; SFFV = promoter of spleen focus-forming virus; wPRE = Woodchuck hepatitis virus posttranscriptional regulatory element; IRES = internal ribosome entry site; eGFP = enhanced green fluorescent protein; p(A) = polyadenylation signal; BGH = bovine growth hormone; SV40 = simian virus 40. (b) Knockout of CCR5 in reporter cell line CCR5+/293T-cell clones were co-transduced with non-concentrated NRTLVs delivering different iTALEN-constructs with either no internal polyadenylation (p(A)) signal (iTALEN-wPRE), or internal BGH-p(A) (iTALEN-wPRE-BGH-p(A)) or SV40-p(A) (iTALEN-wPRE-SV40-p(A)) signals downstream of the wPRE-element, respectively. Mock-transduction and transduction of left or right TALEN-arms, only, served as negative controls (homodimers only). Measured for 3 independently produced vector preparations, each time in duplicates, *p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
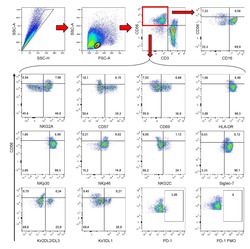
- Figure 2 Single-cell PCR delivers sequences of functional CMV-specific TCRs. (A) PBMCs from Donor 1 were recovered and stained with HLA-B7/pp65 417-426 multimers. Dot plot shows the further analyzed CD8 and MHC multimer double-positive cell population. Cells were pre-gated on live lymphocytes (propidium iodide-negative, and CD3-positive). (B) TCR SCAN as described in Figure 1 and agarose gel electrophoresis of the resulting PCR products was performed. The photography shows the agarose gel. Upper row shows alpha-chain products matched with the respective beta-chains in the lower row. White boxes indicate alpha- and matched beta-chain-products derived from identical single cell samples. (C) The table summarizes the V- D- J- segment type and amino acid sequences of TCRs identified from CMV-multimer positive T cells in Figure 2A . TCR1A was identified 11 times and TCR 1B was detected once. (D) MHC multimer-positive T cells from the same donor were in vitro expanded and six T cell clones were successfully maintained. All clones contained TCR1A as confirmed by PCR and sequencing. The left FACS plot shows HLA-B7/pp65 417-426 staining and the right FACS plot shows staining with an irrelevant MHC multimer. (E) PBMCs from donor 2 were recovered and stained with HLA-B8/IE-1 199-207 multimers. The dot plot shows the further analyzed CD8 and MHC multimer double-positive cell population. Cells were pre-gated on living lymphocytes (propidium iodide negative and CD3 positive). (F) TCR SCAN a
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Characterization of a small diverse T cell repertoire and transgenic expression of detected TCRs. (A) PBMCs from donor 3 were stained with HLA-B8/IE-1 88-96 multimers. Dot plot shows the further analyzed CD8 and MHC multimer double-positive cell population. Cells were pre-gated on living lymphocytes (propidium iodide-negative and CD3-positive). (B) A PCR slide with single antigen-specific T cells from Figure 3A were FACS-isolated. TCR SCAN as described in Figure 1 and agarose gel electrophoresis of the resulting PCR products was performed. Upper row shows alpha-chain products matched with the respective beta-chains in the lower row. White boxes indicate samples alpha- and matched beta-chain-products derived from identical single cell samples. (C) The table summarizes the V- D- J- segment type and amino acid sequences of TCRs identified from CMV-multimer positive T cells in Figure 3A . In three independent experiments we identified nine different TCRs (TCR 3A-I) (D) Pie chart indicates the prevalence of identified TCRs from donor 3. Percentages represent incidence of respective TCR divided by total number of positive samples. (E) Sequences from TCR 3D, 3E and 3G were expressed in Jurkat76 T cells by retroviral gene transfer. Non-transduced (left FACS plot) and transduced Jurkat76 T cells were analyzed for expression of CD3 and MHC multimer binding.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 lp36 contributes to the association of B. burgdorferi with specific populations of dendritic cells. Human PBMCs (4x10 6 ) were co-incubated with 4x10 7 GFP-tagged B31 (black bars), A3-M9 lp36- (white bars) or A3-M9 lp36-/lp36+ (cross-hatched bars) B. burgdorferi for 6 hours at 4degC or 37degC. The percentages of GFP + mDC1s (CD19 - CD3 - BDCA2 - BDCA1 + ) ( A ), pDCs (CD19 - CD3 - BDCA2 + BDCA1 - ) or ( B ) mDC2s (CD19 - CD3 - BDCA3 + BDCA2 - ) ( C ) were determined by multiparameter flow cytometry. Dot plots representing 500,000 collected events are provided to illustrate gating strategies (left). Column graphs represent the mean and standard deviation of three biological replicates (right). Statistical analysis was performed using a one-way ANOVA with a Tukey's post-test for multiple comparisons.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 T FR expansion in lymphoid tissues during chronic SIV infection. ( a ) Disaggregated lymph node and spleen cells from SIV uninfected ( n =9) or chronically SIV-infected rhesus macaques ( n =11) were analysed by flow cytometry. Representative examples of flow cytometry gating are shown. Of viable CD3 + CD8 - cells, follicular subsets were defined as CXCR5 + cells (F) and germinal centre subsets were defined as CXCR5 hi PD-1 hi cells (GC). Of these subsets, regulatory cells were defined as CD25 hi CD127 - . T FR (CXCR5 + CD25 hi CD127 - ) were Foxp3 + , whereas T FH (CXCR5 + CD25 lo/- ) were Foxp3 - . ( b ) The percentages of each rhesus macaque regulatory subset, as analysed in a are shown. ( c ) The ratios of each regulatory cell population to its non-regulatory cell counterpart are shown. ( d ) The percentage of total CTLA-4 expression is shown in SIV-uninfected ( n =9) and chronically SIV-infected ( n =8) rhesus macaques. The horizontal bars of each graph indicate the median value and are listed where appropriate for clarity. Statistical analyses were performed by Mann-Whitney (Wilcoxon) tests to compare unpaired, nonparametric values and significance is denoted by asterisks where * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 HIV entry and replication promote T FR expansion. Disaggregated tonsil cells were spinoculated with X4 or R5 HIV and T FR populations were analysed by flow cytometry ( n =15). ( a ) A representative example of tonsil cell flow gating. From viable CD3 + CD8 - cells, T FR are defined as CXCR5 + and CD25 hi CD127 - . T FR cells contain Foxp3 + cells, whereas remaining T FH (CXCR5 + CD25 lo/- ) cells are Foxp3 - . ( b ) Percentages of T FR determined by gating strategies in a are shown. Experimental conditions include mock-spinoculated cells cultured with PMA (50 ng ml -1 ) and ionomycin (1 mug ml -1 ) or exogenous TGF-beta (100 ng ml -1 ) for 24 h and cells pretreated to block CXCR4 (AMD, 200 muM) and CCR5 (MVC, 2 muM). ( c ) Using flow cytometry counting beads, the number of cells per mul were determined for total (CD3 + CD8 - ), T FH (CXCR5 + CD25 lo/- ) and T FR (CXCR5 + CD25 hi CD127 - ) subsets in mock- and X4-spinoculated samples ( n =3). ( d ) Bcl-6 expression is shown in CXCR5- (grey), T FH (blue) and T FR (red) populations after mock-, X4- or R5-spinoculation ( n =5). ( e ) Blimp-1 expression was also determined as in d . The horizontal bars of each graph indicate the median value and are listed where appropriate for clarity. Statistical analyses were performed by Friedman nonparametric tests ( b , d , e ) and significance is denoted by asterisks where * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
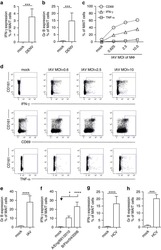
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
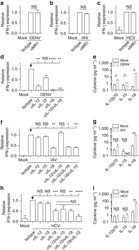
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
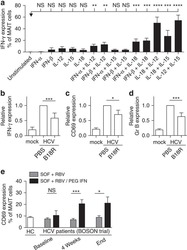
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
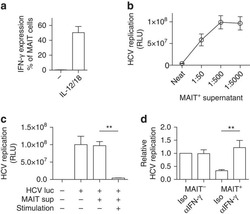
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3. Phenotype and endogenous TCR expression of CD34 + HPC-derived transgenic AR + T cells. Flow cytometric analysis of the AR-transgenic T cells. (A) CAR-transgenic GFP + cells of cultures transduced to express either the CAR:zeta or the CAR:28zeta were analyzed on day 26 of OP9-DL1 culture for CD3 and TCRalphabeta expression. As a control, GFP - cells are shown from the OP9-DL1 culture transduced to express the CAR:zeta ( N = 5). (B) Dot plots show CD3 expression of cells from the OP9-DL1 cultures transgenic for the wtTCR, TCR:zeta and TCR:28zeta. Vbeta14 staining is used to mark transgene expression, as no GFP is expressed by the transgenic cells ( N = 5). (C) Surface and cytoplasmic staining for CD3 of in vitro generated mature T cells that were expanded for one cycle on feeder cells in the presence of cytokines. (D) Expression of various membrane markers by the CD27 + CD1a - mature T cells at the end of OP9-DL1 culture (46 d) ( N = 2). (E) Day 0: fresh cord blood after MACS CD34 enrichment sorted using the sorting window shown. Day 13: cord blood cells cultured on OP9-DL1 were sorted for CD5 CD7 double positive cells, using the indicated sorting window. The cells were then transduced to express CAR:28zeta and further differentiated on OP9-DL1 feeder layer. Day 21: analysis of the transgenic GFP + cultured cells for DP cells and CD27 + CD1a - mature cells. (F) Flow cytometric analysis of GFP + CAR:28zeta-transgenic cultures, gated on GFP + CD27 + CD1a - mature AR + c
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
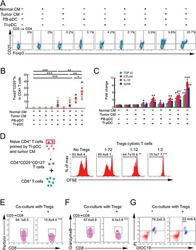
- Experimental details
- Figure 3 Naive CD4 + T cells are converted to functional Tregs by tumor-infiltrating DCs and tumor conditioned medium (CM). (A-C) Naive CD4 + T cells from peripheral blood of patients with invasive breast carcinoma were co-cultured with or without autologous pDCs isolated from tumor (TI) or peripheral blood (PB) for 9 days in the presence or absence of 30% CM from autologous tumor slices or adjacent normal tissue slices. (A , B) Non-adherent cells from co-cultures were stained for CD3, CD4, CD25 and intracellular Foxp3, and analyzed by flow cytometry. Representative plots of gated CD3 + CD4 + cells (A) and quantification of percentage of Foxp3 + CD25 + cells among CD3 + CD4 + cells (B) are shown (mean +- SEM, n = 19; * P < 0.05, ** P < 0.01, *** P < 0.001 by Student's t -test). (C) Expression of Treg-associated genes, assessed by qRT-PCR normalized to GAPDH , in sorted CD4 + T cells, relative to expression in cultures without DCs or CM (mean +- SEM, n = 19; * P < 0.05, ** P < 0.01, *** P < 0.001 compared with naive CD4 + T cells cultured alone by Student's t -test). (D-G) Effect of naive CD4 + T cell-derived Tregs, obtained by co-culture with TI pDCs and tumor CM as above, on function of autologous tumor-specific CD8 + T cells. Tumor-specific CD8 + T cells were generated for each subject by stimulating autologous PB CD8 + T cells with autologous tumor lysate-pulsed autologous DCs. Tregs were recovered from co-cultures by magnetic sorting. (D) CFSE-labeled CD8 + T ce
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 In vivo knockdown of PITPNM3 in CD4 + T cells reverses immunosuppression and inhibits tumor progression in humanized mice. (A) Humanized mice bearing palpable MDA-MB-231 orthotopic xenografts were intraperitoneally injected daily for 14 days with PBS, 1 nmol CD4-aptamer-control siRNA (AsiC-con) or CD4-aptamer-siRNA targeting PITPNM3 (sequence in A , AsiC-PI) to assess the role of PITPNM3 in TI Tregs, and other T cells and tumor control. Experimental schematic is provided in Supplementary information, Figure S9A . (B) Representative immunoblots showing selective knockdown of PITPNM3 protein in PB CD4 + T cells, but not tumor xenografts ( n = 3). (C) PITPNM3 knockdown did not affect the distribution of human CD45 + hematopoietic cells, CD4 + and CD8 + T cells, and CD14 + monocytes in the peripheral blood of humanized mice. Representative flow plots are shown ( n = 3). (D , E) Effect of PITPNM3 knockdown on TI naive CD4 + , Tregs and CD8 + T cell numbers, and apoptosis by TUNEL assay in xenografts. D shows representative immunofluorescence microscopy images. Top row indicates CD4 + naive T cells by arrows; the second row indicates CD4 + CD45RO + Foxp3 - CD4 + memory T cells (yellow arrows) and Foxp3 + Tregs (white arrows). Scale bar, 50 mum. E shows number of cells of each subtype/high power field in eight mice ( ** P < 0.01, *** P < 0.001 compared to PBS group by Student's t -test). (F) Flow cytometry analysis of gated human CD3 + CD4 + cells isolated from xenogra
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
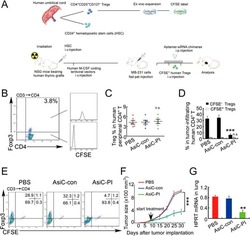
- Experimental details
- Figure 7 CD4-aptamer-siRNA targeting PITPNM3 reduces TI Tregs and inhibits tumor progression in humanized mice with circulating human Tregs. Humanized mice, implanted with MDA-MB-231 tumors and concurrently injected intravenously with autologous Tregs, were intraperitoneally injected daily for 14 days after tumors became palpable with PBS, 1 nmol CD4-aptamer-control siRNA (AsiC-con) or CD4-aptamer-siRNA targeting PITPNM3 to assess the role of PITPNM3 in TI Tregs, and other T cells and tumor control. Tregs were administered every 10 days after the initial injection and mice were sacrificed 30 days after tumor cell inoculation. (A) Experimental schematic. (B , C) Peripheral blood cells of humanized mice were stained for human CD3, CD4 and Foxp3, and analyzed by flow cytometry. A representative flow plot (B) and the percentage (mean +- SEM) of PB CD4 + cells that are CFSE + Tregs in six mice per group (C) are shown. (D , E) Isolated cells from xenografts were stained for human CD3, CD4 and Foxp3. The percentage (mean +- SEM) of six mice per group (D) and representative flow plot (E) of FoxP3 + Tregs are shown. Most Tregs were CFSE - (i.e., did not come from infused Tregs) and the number of TI Tregs was reduced by knocking down PITPNM3 in CD4 + T cells ( *** P < 0.001 compared to the PBS group by Student's t -test). (F) Tumor size (mean +- SEM, n = 6 per group; *** P < 0.001 by two-way ANOVA with Bonferroni multiple comparison tests). (G) Lung metastases assessed by qRT-PCR
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 Absence of ARPC1B alters surface expression of CD8 and GLUT1 in hCTLs. (A-D) HD and ARPC1B-deficient patient hCTLs were fixed in PFA for 20 minutes, permeabilized, and stained with an antibody against CD8 alone (green) ( A and B ) or in combination with anti-GLUT1 (red) and anti-EEA1 (white) antibodies ( C and D ). Images are 3D reconstructions of Z -stack. Scale bars: 4 mum. ( E ) Measurement of the mean intensity of GLUT1 expressed in AU and the degree of colocalization with EEA1 expressed as PCC (see Methods) in HD and ARPC1B-deficient patient hCTLs based on images as sampled in C and D . HD, n = 41 cells; ARPC1B-deficient patient, n = 38 cells. P < 0.005 (unpaired t test). ** P < 0.0013; *** P < 0.0002. ( F ) Flow cytometry analysis of the proliferation capacity of HD and ARPC1B-deficient patient hCTLs (gated on live CD8 + cells) in the absence (blue) or presence (red) of plate-bound anti-CD3 stimulation (1 mug/mL). All data are representative of 3 independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
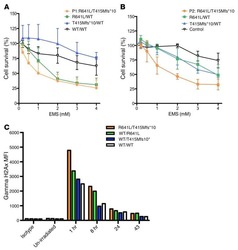
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
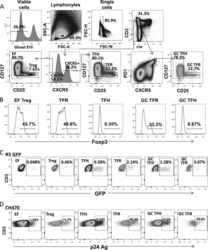
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
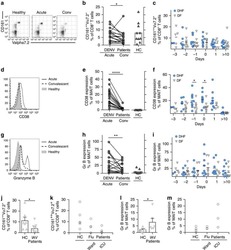
- Figure 5 CD169, as an HIV-1 receptor, mostly impacts transfer from mature MDDCs rather than immature MDDCs. (A) Flow cytometry plots showing CD86, DC-SIGN, and CD169 expression levels on MDDCs (pre-gated on SSC FSC, living cells, CD3 - cells and singlets). Panels show the expression of these proteins in iMDDCs (left panel) and MDDCs with LPS pretreatment at 100 ng/ml for 48 or 24 h before co-culture (middle and right panels, respectively). (B) Percentage of variation of HIV-1 transfer when using iMDDCs or LPS-treated MDDCs (100 ng/ml LPS for different lengths of time) incubated with a blocking antibody against CD169 as compared to an isotype control for each condition. Results are displayed for 4 different blood donors with the mean +- SD of technical triplicates. (C) Percent of variation in HIV-1 transfer to assess the impact of blocking CD169 and TSPAN7 knockdown as compared to scramble shRNA on MDDCs matured with LPS for 48 h treated by an isotype control. Mean +- SD of seven different blood donors in 4 experiments. (B,C) NS, not significant. ** p < 0.01; *** p < 0.001. (D) Confocal microscopy images of iMDDCs (left panel) and mature MDDCs (mMDDCs) right panel, to assess the degree of colocalization between CD169 (magenta) and incoming X4-HIV-1-Gag-iGFP (green). Actin filaments and nuclei were stained with phalloidin (red) and DAPI (blue). Four hundred nanometers of Z-stacks were taken 40 h after the start of the co-culture with CD4 + T cells and X4-HIV-1-Gag-iGFP. The pic
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
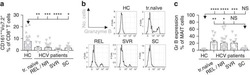
- Fig. 5 Infiltrating myeloid cells inhibit T cell functionality in residual tumors. a Flow cytometric analysis of CD11b + , CD3 + and the ratio of CD3 + /CD11b + in the untreated and iRFA-treated CT26 and MC38 tumors on day 3 and day 9 ( n = 5). b Flow cytometric analysis of CD8 + and CD4 + FoxP3 + cells and the ratio of CD8 + /CD4 + FoxP3 + in the untreated and iRFA-treated CT26 and MC38 tumors on day 3 and day 9 ( n = 5). c Flow cytometric analysis and quantification of granzyme B, IFN-gammaand PD-1 expression in CD8 + cells in the untreated and iRFA-treated CT26 and MC38 tumors on day 3 and day 9 ( n = 5). d In vitro suppressive activity of tumor-infiltrating CD11b + cells purified from spleen or CT26 residual tumors on day 3 after iRFA. Representative histograms of CD8 + T cell proliferation at a ratio of 1:1 CD8 + to CD11b + T cells and percent CD8 + T cell proliferation ( n = 5). e - g CD11b + cells isolated from the residual tumor were mixed with CT26 or MC38 tumor cells and transferred into recipient mice. e Adoptive transfer method. f Growth curve of tumor (one-sided ANOVA test, *** P < 0.001, n = 5). g Flow cytometric analysis and quantification of CD3 + CD8 + cells (gate on single live cells) and Granzyme B expression and IFN-gamma on CD8 + cells ( n = 5). Data represent cumulative results from 1/2 independent experiments with 5 mice/group. The data are represented as mean +- SEM. Statistical differences between pairs of groups
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 8 CCL2/CCR2 blockade inhibits tumor progression and overcomes resistance to anti-PD-1 therapy. a - f iRFA treatment was performed in CT26 and MC38 colon cancer models as shown in Fig. 2a . Anti-PD-1 mAb (200 mug, clone: J43) was administered through intraperitoneal injection to mice every 3 days for a total of four times. The CCR2 antagonist (CCR2a) (RS504393, Tocris) was given subcutaneously at a dose of 5 mg/kg twice per day for 9 days. a Growth curve of the CT26 and MC38 residual tumor (one-sided ANOVA test, n = 8). b The weight of the residual CT26 and MC38 tumor examined on day 14 after iRFA by dissection of the mice ( n = 6). c The number of metastases examined on day 14 after iRFA by dissection the mice ( n = 6). d Kaplan-Meier survival curves are shown, and the log-rank test was performed ( n = 8). e Flow cytometric analysis and quantification of CD3 + and CD8 + infiltration (gate on single live cells) in residual CT26 tumors. f Granzyme B and IFNgamma expression on CD8 + cells in residual CT26 tumors. (gate on CD8 + cells) ( n = 5). g , h iRFA treatment was performed in mice bearing wild type and CCL2 -/- CT26 or MC38 tumor. g Growth curve of the CT26 and MC38 residual tumor (one-sided ANOVA test, n = 5). h Kaplan-Meier survival curves are shown, and the log-rank test was performed ( n = 8). Data represent results from 1/2 independent experiments. The data are represented as mean +- SEM. Statistical differences between pairs o
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Proliferating Cells in LCH Granuloma are Mostly Endothelial Cells, Fibroblasts, and T Cells Paraffin-embedded and frozen sections were stained with antibodies against Ki-67 (which label proliferating cells), CD1a (LCs), CD3 (T cells), CD20 (B cells), CD68, CD31, and CD34 (endothelial cells). (A) Double immunostaining of paraffin-embedded section from LCH eosinophilic granulomas with anti-Ki-67 Ab, (brown nuclear staining) and with anti-CD1a Ab (upper images, blue staining) or anti-CD3 Ab (lower images, blue staining). Open arrowheads indicate double-stained cells, black arrowheads indicate Ki-67 + cells with an endothelial morphology. (B) Histogram represents percentage of CD1a + cells and of CD3 + cells labeled with Ki-67 ( n = 15). (C) Histogram represents percentage of proliferating cells (Ki-67 + ) that express CD1a, CD3, CD20, or CD68 ( n = 15). (D) Histogram represents percentage of proliferating cells (Ki-67 + ) that are endothelial cells, interstitial cells (fibroblasts), and other types based on morphological examination. (E) Immunolabeling of blood vessels on paraffin-embedded section from LCH eosinophilic granulomas with CD34 (left) and CD31 (right) antibodies. (F) Proliferating Ki-67 + cells (brown nuclear staining) with a fibroblast-cell morphology in an eosinophilic granuloma.
 Explore
Explore Validate
Validate Learn
Learn Immunohistochemistry
Immunohistochemistry Flow cytometry
Flow cytometry