PA5-17113
antibody from Invitrogen Antibodies
Targeting: LMNA
CMD1A, HGPS, LGMD1B, LMN1, LMNL1, MADA, PRO1
Antibody data
- Antibody Data
- Antigen structure
- References [4]
- Comments [0]
- Validations
- Western blot [4]
- Immunocytochemistry [1]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA5-17113 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-Lamin A/C (Ser22) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- It is not recommended to aliquot this antibody.
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Concentration
- 0.4 µg/mL
- Storage
- -20°C
Submitted references Depletion of HP1α alters the mechanical properties of MCF7 nuclei.
LAP2alpha maintains a mobile and low assembly state of A-type lamins in the nuclear interior.
Heterochromatin-Driven Nuclear Softening Protects the Genome against Mechanical Stress-Induced Damage.
Proteomics Profiling of KAIMRC1 in Comparison to MDA-MB231 and MCF-7.
Pradhan S, Solomon R, Gangotra A, Yakubov GE, Willmott GR, Whitby CP, Hale TK, Williams MAK
Biophysical journal 2021 Jul 6;120(13):2631-2643
Biophysical journal 2021 Jul 6;120(13):2631-2643
LAP2alpha maintains a mobile and low assembly state of A-type lamins in the nuclear interior.
Naetar N, Georgiou K, Knapp C, Bronshtein I, Zier E, Fichtinger P, Dechat T, Garini Y, Foisner R
eLife 2021 Feb 19;10
eLife 2021 Feb 19;10
Heterochromatin-Driven Nuclear Softening Protects the Genome against Mechanical Stress-Induced Damage.
Nava MM, Miroshnikova YA, Biggs LC, Whitefield DB, Metge F, Boucas J, Vihinen H, Jokitalo E, Li X, García Arcos JM, Hoffmann B, Merkel R, Niessen CM, Dahl KN, Wickström SA
Cell 2020 May 14;181(4):800-817.e22
Cell 2020 May 14;181(4):800-817.e22
Proteomics Profiling of KAIMRC1 in Comparison to MDA-MB231 and MCF-7.
Alghanem B, Ali R, Nehdi A, Al Zahrani H, Altolayyan A, Shaibah H, Baz O, Alhallaj A, Moresco JJ, Diedrich JK, Yates JR 3rd, Boudjelal M
International journal of molecular sciences 2020 Jun 18;21(12)
International journal of molecular sciences 2020 Jun 18;21(12)
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on nuclear enriched cell extracts (30 µg lysate) of HeLa (Lane 1), HeLa treated with Hydroxyurea (4mM for 20 hours) (Lane 2), HeLa treated with Nocodazole (100 ng/mL for 20 hours) (Lane 3), HeLa treated with Paclitaxel (100 nM for 20 hours) (Lane 4), NIH/3T3 (Lane 5), NIH/3T3 treated with Hydroxyurea (4mM for 20 hours) (Lane 6), NIH/3T3 treated with Nocodazole (100 ng/mL for 20 hours) (Lane 7), NIH/3T3 treated with Paclitaxel (100 nM for 20 hours) (Lane 8). The blot was probed with Anti-Phospho-Lamin A/C (Ser22) Polyclonal Antibody (Product # PA5-17113, 1:500 dilution) and detected by chemiluminescence using Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, HRP conjugate (Product # A27036, 0.25 µg/mL, 1:4000 dilution). A 65 and 74 kDa band corresponding to Phospho-Lamin A/C was observed across the cell lines tested and was enhanced upon treatment.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- CRISPR-Cas9 mediated genome editing ofLamin (as confirmed by next generation sequencing) was achieved by using LentiArray™ Lentiviral sgRNA (Product # A32042, AssayID CRISPR817702_LV) and LentiArray Cas9 Lentivirus (Product # A32064). Fig (a) Western blot analysis of Phospho-Lamin AC (Ser22) was performed by loading 30 µg of HeLa wild type (Lane 1), HeLa wild type cells treated with 100 ng/mL Nocodazole for 20hrs (Lane 2), HeLa Cas9 (Lane 3), HeLa Cas9 cells treated with 100 ng/mL Nocodazole for 20hrs (Lane 4), HeLa Cas9 cells transduced with Lamin Lentiviral sgRNA (Lane 5) and HeLa Cas9 cells transduced with Lamin Lentiviral sgRNA and treated with 100 ng/mL Nocodazole for 20hrs (Lane 6) modified whole cell extracts. The samples were electrophoresed using NuPAGE™ Novex™ 4-12% Bis-Tris Protein Gel (Product # NP0321BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with Phospho-Lamin A/C (Ser22) Polyclonal Antibody (Product # PA5-17113, 1:500 dilution) and Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036, 1:5000 dilution). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005). A reduced signal in sgRNA transduced cells using the LentiArray™ CRISPR product line confirms that antibody is specific toLamin.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of Phospho-Lamin A/C pSer22 in extracts from HeLa and NIH/3T3 cells using Phospho-Lamin A/C pSer22 polyclonal antibody (Product # PA5-17113). Lane 1 and 3 are hydroxyurea-treated (4 mM, 20 hours) to induce G1/S phase, lane 2 is paclitaxel-treated (100 nM, 20 hours) to induce G1/S phase, and lane 4 is nocodazole-treated (100 ng/mL, 20 hours) to induce G2/M phase.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of Phospho-Lamin A/C pSer22 in extracts from G2/M phase HeLa cells, untreated or treated with phosphatase, using Phospho-Lamin A/C pSer22 polyclonal antibody (Product # PA5-17113) (upper) or Lamin A/C antibody (lower) as loading control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of Phospho-Lamin A/C pSer22 in HeLa cells using a Phospho-Lamin A/C pSer22 polyclonal antibody (Product # PA5-17113) (green). Actin filaments are labeled with a fluorescent red phalloidin. DNA is labeled using a fluorescent blue dye.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
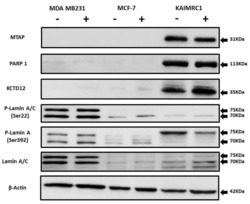
- Experimental details
- Figure 5 Expression of MTAP, PARP1, KCTD12, and Lamin A/C in cancer cell lines: Western blot analysis of protein expression of MTAP, PARP1, KCTD12, Lamin A/C, and phosphorylated Lamin A/C in MDA MB231, MCF-7, and KAIMRC1 cells. Preparation of cell lysates and western blot were performed as described under the Materials and Methods section. In both normal (+) and serum-starved (-) conditions, KAIMRC1 cells showed strong expression of MTAP, PARP1, and KCTD12 compared to MDA-MB231 and MCF-7 cells. Moreover, KAIMRC1 cells showed weak expression of Lamin A/C and phosphorylated Lamin A/C in comparison to MDA MB231 cells, thus validating our proteomics results.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5. LAP2alpha binds to lamin A/C and inhibits their assembly without altering lamin phosphorylation. ( A, B ). Purified recombinant lamin A and LAP2alpha full length (f.l.) ( A ) or lamin A-binding mutant LAP2alpha 1-414 ( B ) were dialyzed either alone or together into assembly buffer with 300 mM NaCl. Samples were separated on a 10% to 30% sucrose gradient, followed by collection of fractions and quantification of protein bands. Exemplary Coomassie stained gels of fractions ,supernatant' (SN) to six are shown on the left. The calculated protein amount per fraction (% of total protein) was plotted as curve chart on the right. ( C ) Lamin A and LAP2alpha were dialyzed either alone or together into assembly buffer as in (D), but with 100 mM NaCl, enabling formation of higher assembly lamin structures. After centrifugation, total (T), supernatant (SN) and pellet (P) fractions were analyzed on a Coomassie gel, protein bands were quantified and normalized to total protein levels. Graph displays lamin A levels in the pellet fraction of mixed samples (lamin A + LAP2alpha) as average fold difference +- S.E.M over samples with lamin A alone, n = 5 (single data points are displayed). ( D ) Lamin A/C was immunoprecipitated from mEos3.2-lamin A/C WT cells and LAP2alpha and lamin A/C knockout controls using the indicated antibodies recognizing different regions of lamin A/C. Immunoprecipitates were analyzed by western blotting using the indicated antibodies (anti lamin A/C 3A6, ant
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot