Antibody data
- Antibody Data
- Antigen structure
- References [22]
- Comments [0]
- Validations
- Immunocytochemistry [2]
- Other assay [31]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 34-1500 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Securin Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 μg
- Concentration
- 0.25 mg/mL
- Storage
- -20°C
Submitted references Protein stabilization by RSUME accounts for PTTG pituitary tumor abundance and oncogenicity.
mTOR promotes pituitary tumor development through activation of PTTG1.
Inhibition of PTTG1 expression by microRNA suppresses proliferation and induces apoptosis of malignant glioma cells.
Apratoxin A Shows Novel Pancreas-Targeting Activity through the Binding of Sec 61.
LATS1 and LATS2 phosphorylate CDC26 to modulate assembly of the tetratricopeptide repeat subcomplex of APC/C.
MASTL promotes cyclin B1 destruction by enforcing Cdc20-independent binding of cyclin B1 to the APC/C.
Nek2A destruction marks APC/C activation at the prophase-to-prometaphase transition by spindle-checkpoint-restricted Cdc20.
The role of lipolysis stimulated lipoprotein receptor in breast cancer and directing breast cancer cell behavior.
Aurora kinase inhibitors reveal mechanisms of HURP in nucleation of centrosomal and kinetochore microtubules.
PTTG1 oncogene promotes tumor malignancy via epithelial to mesenchymal transition and expansion of cancer stem cell population.
Human pituitary tumor-transforming gene 1 overexpression reinforces oncogene-induced senescence through CXCR2/p21 signaling in breast cancer cells.
The C-terminus of PARK2 is required for its self-interaction, solubility and role in the spindle assembly checkpoint.
Securin and separase modulate membrane traffic by affecting endosomal acidification.
Substrate degradation by the anaphase promoting complex occurs during mitotic slippage.
Tumor suppressor activity of KLF6 mediated by downregulation of the PTTG1 oncogene.
Thyroid hormone receptors are tumor suppressors in a mouse model of metastatic follicular thyroid carcinoma.
DNA damage triggers p21WAF1-dependent Emi1 down-regulation that maintains G2 arrest.
S100A1 deficiency results in prolonged ventricular repolarization in response to sympathetic activation.
Aberrant accumulation of PTTG1 induced by a mutated thyroid hormone beta receptor inhibits mitotic progression.
p53-regulated transcriptional program associated with genotoxic stress-induced apoptosis.
Overexpressed pituitary tumor-transforming gene causes aneuploidy in live human cells.
DNA damage-induced inhibition of securin expression is mediated by p53.
Fuertes M, Sapochnik M, Tedesco L, Senin S, Attorresi A, Ajler P, Carrizo G, Cervio A, Sevlever G, Bonfiglio JJ, Stalla GK, Arzt E
Endocrine-related cancer 2018 Jun;25(6):665-676
Endocrine-related cancer 2018 Jun;25(6):665-676
mTOR promotes pituitary tumor development through activation of PTTG1.
Chen R, Duan J, Li L, Ma Q, Sun Q, Ma J, Li C, Zhou X, Chen H, Jing Y, Zhao S, Wu X, Zhang H
Oncogene 2017 Feb 16;36(7):979-988
Oncogene 2017 Feb 16;36(7):979-988
Inhibition of PTTG1 expression by microRNA suppresses proliferation and induces apoptosis of malignant glioma cells.
Su X, Chen J, Ni L, Shi W, Shi J, Liu X, Zhang Y, Gong P, Zhu H, Huang Q
Oncology letters 2016 Nov;12(5):3463-3471
Oncology letters 2016 Nov;12(5):3463-3471
Apratoxin A Shows Novel Pancreas-Targeting Activity through the Binding of Sec 61.
Huang KC, Chen Z, Jiang Y, Akare S, Kolber-Simonds D, Condon K, Agoulnik S, Tendyke K, Shen Y, Wu KM, Mathieu S, Choi HW, Zhu X, Shimizu H, Kotake Y, Gerwick WH, Uenaka T, Woodall-Jappe M, Nomoto K
Molecular cancer therapeutics 2016 Jun;15(6):1208-16
Molecular cancer therapeutics 2016 Jun;15(6):1208-16
LATS1 and LATS2 phosphorylate CDC26 to modulate assembly of the tetratricopeptide repeat subcomplex of APC/C.
Masuda K, Chiyoda T, Sugiyama N, Segura-Cabrera A, Kabe Y, Ueki A, Banno K, Suematsu M, Aoki D, Ishihama Y, Saya H, Kuninaka S
PloS one 2015;10(2):e0118662
PloS one 2015;10(2):e0118662
MASTL promotes cyclin B1 destruction by enforcing Cdc20-independent binding of cyclin B1 to the APC/C.
Voets E, Wolthuis R
Biology open 2015 Mar 6;4(4):484-95
Biology open 2015 Mar 6;4(4):484-95
Nek2A destruction marks APC/C activation at the prophase-to-prometaphase transition by spindle-checkpoint-restricted Cdc20.
Boekhout M, Wolthuis R
Journal of cell science 2015 Apr 15;128(8):1639-53
Journal of cell science 2015 Apr 15;128(8):1639-53
The role of lipolysis stimulated lipoprotein receptor in breast cancer and directing breast cancer cell behavior.
Reaves DK, Fagan-Solis KD, Dunphy K, Oliver SD, Scott DW, Fleming JM
PloS one 2014;9(3):e91747
PloS one 2014;9(3):e91747
Aurora kinase inhibitors reveal mechanisms of HURP in nucleation of centrosomal and kinetochore microtubules.
Wu JM, Chen CT, Coumar MS, Lin WH, Chen ZJ, Hsu JT, Peng YH, Shiao HY, Lin WH, Chu CY, Wu JS, Lin CT, Chen CP, Hsueh CC, Chang KY, Kao LP, Huang CY, Chao YS, Wu SY, Hsieh HP, Chi YH
Proceedings of the National Academy of Sciences of the United States of America 2013 May 7;110(19):E1779-87
Proceedings of the National Academy of Sciences of the United States of America 2013 May 7;110(19):E1779-87
PTTG1 oncogene promotes tumor malignancy via epithelial to mesenchymal transition and expansion of cancer stem cell population.
Yoon CH, Kim MJ, Lee H, Kim RK, Lim EJ, Yoo KC, Lee GH, Cui YH, Oh YS, Gye MC, Lee YY, Park IC, An S, Hwang SG, Park MJ, Suh Y, Lee SJ
The Journal of biological chemistry 2012 Jun 1;287(23):19516-27
The Journal of biological chemistry 2012 Jun 1;287(23):19516-27
Human pituitary tumor-transforming gene 1 overexpression reinforces oncogene-induced senescence through CXCR2/p21 signaling in breast cancer cells.
Ruan JW, Liao YC, Lua I, Li MH, Hsu CY, Chen JH
Breast cancer research : BCR 2012 Jul 12;14(4):R106
Breast cancer research : BCR 2012 Jul 12;14(4):R106
The C-terminus of PARK2 is required for its self-interaction, solubility and role in the spindle assembly checkpoint.
Chen Y, Fang ST, Yeh PC, Yang HH, Chen SY, Chang CJ, Zhai WJ, Chen YC, Juang YL
Biochimica et biophysica acta 2012 Apr;1822(4):573-80
Biochimica et biophysica acta 2012 Apr;1822(4):573-80
Securin and separase modulate membrane traffic by affecting endosomal acidification.
Bacac M, Fusco C, Planche A, Santodomingo J, Demaurex N, Leemann-Zakaryan R, Provero P, Stamenkovic I
Traffic (Copenhagen, Denmark) 2011 May;12(5):615-26
Traffic (Copenhagen, Denmark) 2011 May;12(5):615-26
Substrate degradation by the anaphase promoting complex occurs during mitotic slippage.
Lee J, Kim JA, Margolis RL, Fotedar R
Cell cycle (Georgetown, Tex.) 2010 May;9(9):1792-801
Cell cycle (Georgetown, Tex.) 2010 May;9(9):1792-801
Tumor suppressor activity of KLF6 mediated by downregulation of the PTTG1 oncogene.
Lee UE, Ghiassi-Nejad Z, Paris AJ, Yea S, Narla G, Walsh M, Friedman SL
FEBS letters 2010 Mar 5;584(5):1006-10
FEBS letters 2010 Mar 5;584(5):1006-10
Thyroid hormone receptors are tumor suppressors in a mouse model of metastatic follicular thyroid carcinoma.
Zhu XG, Zhao L, Willingham MC, Cheng SY
Oncogene 2010 Apr 1;29(13):1909-19
Oncogene 2010 Apr 1;29(13):1909-19
DNA damage triggers p21WAF1-dependent Emi1 down-regulation that maintains G2 arrest.
Lee J, Kim JA, Barbier V, Fotedar A, Fotedar R
Molecular biology of the cell 2009 Apr;20(7):1891-902
Molecular biology of the cell 2009 Apr;20(7):1891-902
S100A1 deficiency results in prolonged ventricular repolarization in response to sympathetic activation.
Ackermann GE, Domenighetti AA, Deten A, Bonath I, Marenholz I, Pedrazzini T, Erne P, Heizmann CW
General physiology and biophysics 2008 Jun;27(2):127-42
General physiology and biophysics 2008 Jun;27(2):127-42
Aberrant accumulation of PTTG1 induced by a mutated thyroid hormone beta receptor inhibits mitotic progression.
Ying H, Furuya F, Zhao L, Araki O, West BL, Hanover JA, Willingham MC, Cheng SY
The Journal of clinical investigation 2006 Nov;116(11):2972-84
The Journal of clinical investigation 2006 Nov;116(11):2972-84
p53-regulated transcriptional program associated with genotoxic stress-induced apoptosis.
Kho PS, Wang Z, Zhuang L, Li Y, Chew JL, Ng HH, Liu ET, Yu Q
The Journal of biological chemistry 2004 May 14;279(20):21183-92
The Journal of biological chemistry 2004 May 14;279(20):21183-92
Overexpressed pituitary tumor-transforming gene causes aneuploidy in live human cells.
Yu R, Lu W, Chen J, McCabe CJ, Melmed S
Endocrinology 2003 Nov;144(11):4991-8
Endocrinology 2003 Nov;144(11):4991-8
DNA damage-induced inhibition of securin expression is mediated by p53.
Zhou Y, Mehta KR, Choi AP, Scolavino S, Zhang X
The Journal of biological chemistry 2003 Jan 3;278(1):462-70
The Journal of biological chemistry 2003 Jan 3;278(1):462-70
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
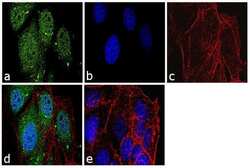
- Experimental details
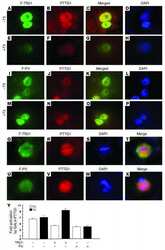
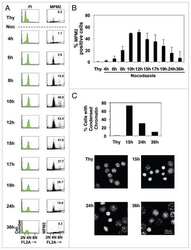
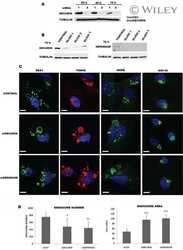
- Immunofluorescence analysis of Securin Polyclonal Antibody was performed using 70% confluent log phase MCF-7 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Securin Rabbit Polyclonal Antibody (Product # 34-1500) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Rabbit IgG (H+L) Superclonal Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing nuclear and cytoplasmic localization. Panel e shows the no primary antibody control. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
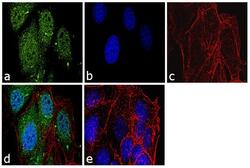
- Experimental details
- Immunofluorescence analysis of Securin Polyclonal Antibody was performed using 70% confluent log phase MCF-7 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Securin Rabbit Polyclonal Antibody (Product # 34-1500) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Rabbit IgG (Heavy Chain) Superclonal Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing nuclear and cytoplasmic localization. Panel e shows the no primary antibody control. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
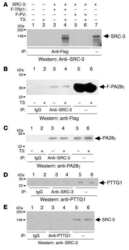
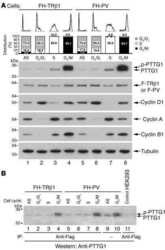
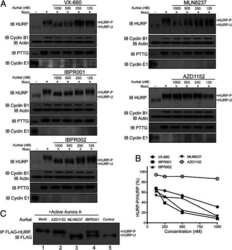
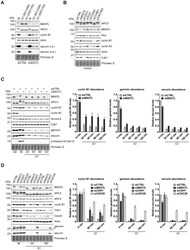
- Fig. 2. MASTL-mediated PP2A inhibition in mitosis is essential to direct cyclin B1 destruction by the APC/C. (A) Forced mitotic exit by either Aurora B or Cdk1 inhibition stabilises cyclin B1 in MASTL RNAi cells. HeLa cells were synchronised in mitosis by thymidine synchronisation and taxol treatment. Mitotic cells were treated with 10 uM of RO-3306 (Cdk1 inhibitor) or 1 uM of AZD1152 (Aurora B inhibitor) for 90 minutes to obtain cells in G1 phase. Extracts were processed for western blot analysis and blotted for indicated proteins. (B) APC/C phosphorylation in mitosis critically depends on cyclin B1-Cdk1 activity. HeLa cells transfected with indicated siRNA pools were synchronised in mitosis by a thymidine block followed by subsequent taxol treatment. Cells arrested in mitosis were collected by mitotic shake-off. Extracts were processed for western blot analysis either 24 hours ( PLK1 RNAi) or 48 hours after the siRNA transfection and blotted for the proteins as indicated. (C) Enforcing mitotic exit in absence of MASTL severely impairs the proteolysis of cyclin B1. HeLa cells transfected with indicated siRNAs were synchronised in mitosis by thymidine synchronisation and taxol treatment. Mitotic cells were treated with 10 uM of RO-3306 for indicated time points to obtain cells in G1 phase. Extracts were processed for western blot analysis and blotted for indicated proteins. The intensity of the cyclin B1, geminin, and securin signals were quantified for each condition and cor
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
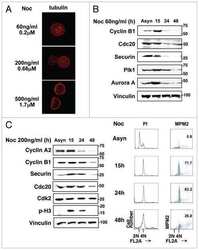
- Fig. 3. MASTL depletion significantly impairs cyclin B1 recruitment to the mitotic APC/C. (A) Cyclin B1 binds to the APC/C specifically in mitosis. HeLa cells were synchronised in G2 and M phase by a thymidine block, followed by nocodazole or taxol treatment. Cells arrested in mitosis were collected by mitotic shake-off, and treated for another 2 hours with 5 uM MG132 where indicated. Cell extracts were subjected to immunoprecipitation using a goat anti-APC4. Aliquots of the immunoprecipitates were analysed by western blotting. (B) MASTL silencing impairs the recruitment of cyclin A2 and cyclin B1 to the mitotic APC/C. HeLa cells transfected with indicated siRNAs were synchronised in G2 phase and mitosis using thymidine and taxol. Cells arrested in mitosis were obtained by mitotic shake-off and lysed directly or subsequently treated with 10 uM of RO-3306 for 90 minutes to obtain G1 phase cells. Cell extracts were subjected to immunoprecipitation using a goat anti-APC4 and aliquots of the immunoprecipitates were analysed by western blotting. Note that equal amounts of APC4 are immunoprecipitated in the mitotic fractions, whereas cyclins A2 and B1 bound to the APC/C are hardly detectable in the MASTL RNAi extracts. The asterisk (*) indicates the remaining securin signal after reprobing the blot with anti-geminin (right-hand panel). (C) Direct binding of cyclin B1 to the APC/C may depend on extensive APC3 phosphorylation in mitosis. HeLa cells treated as in (B), were subjected t
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
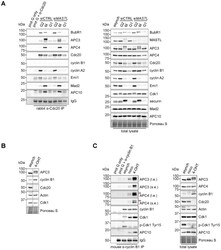
- Fig. 4. Cyclin B1 associates with the mitotic APC/C, independent of Cdc20. (A) Cyclin B1 recruitment to the APC/C does not involve direct Cdc20 binding, different from cyclin A2. HeLa cells transfected with indicated siRNAs were synchronised in G2 phase and mitosis using thymidine and taxol. Cells arrested in mitosis were obtained by mitotic shake-off and lysed directly or subsequently treated with 10 uM of RO-3306 for 90 minutes to obtain G1 phase cells. Cell extracts were subjected to immunoprecipitation using a rabbit anti-Cdc20 and aliquots of the immunoprecipitates were analysed by western blotting. Asynch refers to an asynchronous, non-transfected lysate. (B) Genetic ablation of CDC20 results in a mitotic arrest due to cyclin B1 stabilisation. Cdc20(Delta/Delta); RERT(+/Cre) mouse embryonic fibroblasts (MEFs) were asynchronously grown or thymidine synchronised for 9 hours and treated with 1 uM of 4-hydroxytamoxifen (4-OHT) to induce Cre activity, resulting in the excision of exon 2 of CDC20 (Cdc20(Delta) allele). Cells arrested in mitosis by the 4-OHT treatment were obtained by mitotic shake-off. Extracts of asynchronous or 4-OHT treated cells were subjected to western blot analysis. The dashed line indicates where two lanes have been placed next to each other. (C) Cyclin B1 binds the mitotic APC/C regardless of the presence of Cdc20. Cdc20(Delta/Delta); RERT(+/Cre) MEFs were either asynchronously grown or synchronised in mitosis using thymidine and nocodazole or 4-OHT
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
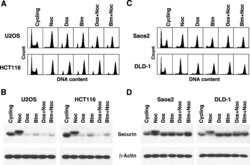
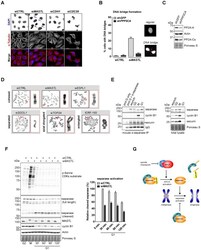
- Fig. 5. MASTL depletion increases the frequency of anaphase bridges that may depend on the activity of separase. (A) DNA bridges formed during anaphase are specific to MASTL silencing. Asynchronously growing HeLa cells were transfected with indicated siRNAs, formaldehyde fixed after 48 hours, and subjected to immunofluorescence staining using anti-alpha-Tubulin and DAPI. Arrows indicate DNA bridges connecting the daughter cells in G1 phase. (B) Combined depletion of MASTL and PPP2CA prevents the formation of DNA bridges. HeLa cells stably infected with an shRNA targeting PPP2CA (sh PPP2CA ) or a control shRNA (sh GFP ) were transfected with indicated siRNAs. Cells were formaldehyde fixed after 48 hours, and subjected to immunofluorescence staining using DAPI. Representative images show a regular cell or two daughter cells that are connected to each other by a DNA bridge (indicated by the red arrow). The amount of cells containing a DNA bridge were scored and quantified for each condition [ n = 3; mean+-standard deviation (s.d.)]. (C) Western blot analysis to show the effects of indicated shRNAs on total protein levels. A reduction in the protein levels of PP2A-A and PP2A-Calpha demonstrates the knockdown efficiency of sh PPP2CA . (D) MASTL silencing does not alter the mitotic chromosome morphology. HeLa cells transfected with indicated siRNAs were synchronised in mitosis by a thymidine block followed by subsequent nocodazole treatment for 5 hours. Cells were processed for chr
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
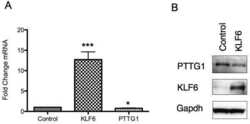
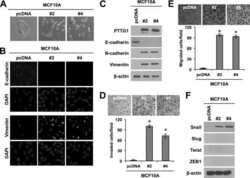
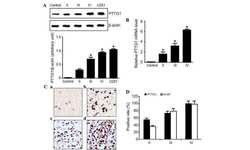
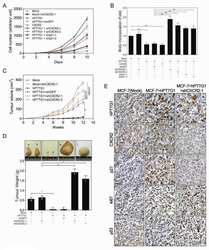
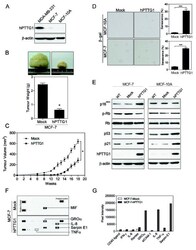
- Figure 1 hPTTG1 overexpression reinforces senescence and senescence-associated secretory phenotype (SASP) . (A) Cell lysates of MDA-MB-231, MCF-7, and MCF-10A cells were collected for immunoblot analysis. (B) MCF-7 cells stably transfected with pcDNA3.1 (Mock; MCF-7 Mock ) or pcDNA3.1- hPTTG1 (hPTTG1; MCF-7 hPTTG1 ) were orthotopically injected into fourth mammary fat pads of SCID mice. The primary tumor weights were measured 18 weeks after implantations. Representative tumors are shown in the upper panel. Quantitative results are shown in the lower panel ( n = 6 per group). The results are presented as the mean +- SEM. * P < 0.05. (C) Tumor growth was measured weekly after orthotopic implantations of MCF-7 cells stably transfected with pcDNA3.1 (Mock) or pcDNA3.1- hPTTG1 (hPTTG1). The experiments were terminated 18 weeks after implantation. Representative tumors are shown in (B). n = 6 per group per time point. Each time point shows the mean +- SEM. * P < 0.05. (D) MCF-7 and MCF-10A cells stably transfected with pcDNA3.1 (Mock) or pcDNA3.1- hPTTG1 (hPTTG1) were plated for the senescence assay. The percentage of positively stained cells is indicated to the right of the respective images. Data represent mean +- SEM ( n = 3). *** P < 0.0001. (E) Cell lysates were collected from MCF-7 and MCF-10A cells stably transfected with pcDNA3.1 (Mock) or pcDNA3.1- hPTTG1 (hPTTG1) for immunoblot analysis. WT, untransfected cells. (F) Conditioned media were collected from MCF-7 cells stably
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
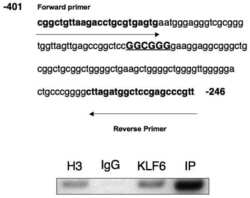
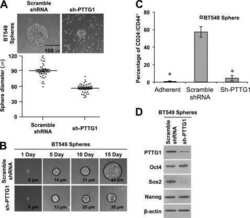
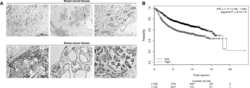
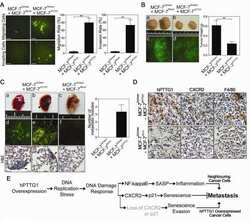
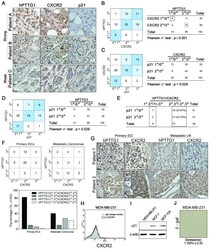
- Figure 7 Positive correlation between hPTTG1, CXCR2, and p21 expression in human breast invasive ductal carcinomas (IDCs) . (A) The expression pattern of hPTTG1, CXCR2, and p21 in representative tumor tissues of patients with breast IDC were immunohistochemically analyzed. Tissues were scored as having strong (3 + ), moderate (2 + ) or weak expression (1 + /0 + ). The criteria are described in the Materials and methods. Original magnification, x400. Scale bars, 25 mum. (B-D) The expression data for hPTTG1, CXCR2, and p21 in 100 IDC specimens. The correlation between the indicated proteins was analyzed by using the Pearson chi 2 test. (E) The expression data for p21 in 84 IDC specimens in which hPTTG1 expression was relevant to CXCR2 expression (the 84 specimens are marked with boxes in (B). (F) The expression data of hPTTG1 and CXCR2 in 50 primary IDC specimens and 50 matched metastatic carcinomas (upper panel). The specimens were classified into four groups: hPTTG1(2 + ,3 + )/CXCR2(2 + ,3 + ), hPTTG1(2 + ,3 + )/CXCR2(1 + ,0 + ), hPTTG1(1 + ,0 + )/CXCR2(2 + ,3 + ), and hPTTG1(1 + ,0 + )/CXCR2(1 + ,0 + ). The percentage of hPTTG1/CXCR2-stained specimens of each group is indicated in the lower panel. (G) The expression pattern of hPTTG1 and CXCR2 in representative breast IDC and matched metastatic lymph nodes (LNs). Original magnification, x400. Scale bars, 25 mum. (H) MDA-MB-231 cells were stained with FITC-conjugated anti-CXCR2 or isotype control IgG for FACS analysis. (I) Ce
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
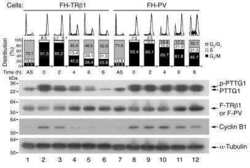
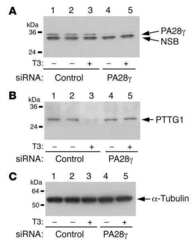
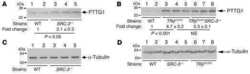
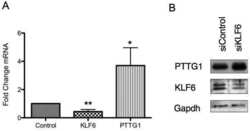
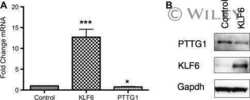
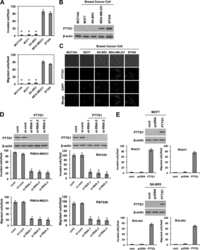
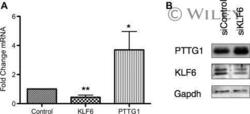
- PTTG1 is upregulated with KLF6 knockdown. HepG2 cells were transfected with siKLF6 or siControl and RNA and protein collected after 24 h. (A) qRT-PCR on cDNA showed 57% decrease in KLF6 mRNA, with a corresponding 3.7-fold increase in PTTG1. (B) Protein extracts were submitted to Western blot analysis and approximately a 50% decrease in KLF6 protein levels resulted in approximately twofold overexpression of PTTG1. The data presented in (B) is representative of at least three independent experiments.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry