Antibody data
- Antibody Data
- Antigen structure
- References [28]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [7]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 14-9989-82 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD279 (PD-1) Monoclonal Antibody (J116), eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The J116 monoclonal antibody reacts with the human PD-1 (programmed death-1), a 55 kDa member of the immunoglobulin superfamily. PD-1 contains the immunoreceptor tyrosine-based inhibitory motif (ITIM) and plays a key role in peripheral tolerance and autoimmune disease. PD-1 is expressed predominantly on activated T and B lymphocytes. Two novel members of the B7 family have been identified as the PD-1 ligands, PD-L1 (B7-H1) and PD-L2 (B7-DC). Evidence reported to date suggests overlapping functions for these two PD-1 ligands and their constitutive expression on some normal tissues and upregulation on activated antigen-presenting cells. Binding of the J116 monoclonal antibody inhibits PD-1 signal transduction, however, it does not block binding of the ligand PD-L1. Applications Reported: This J116 antibody has been reported for use in immunoprecipitation and immunohistology staining of frozen tissue sections. It has also been reported in in vitro functional assays. (Please use Functional Grade purified J116, Product # 16-9989, in functional assays). Applications Tested: The J116 antibody has been tested by flow cytometric analysis of human PD-1 transfected cells. This can be used at less than or equal to 1 µg per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest. Purity: Greater than 90%, as determined by SDS-PAGE. Aggregation: Less than 10%, as determined by HPLC. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- J116
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- 4° C
Submitted references Correlations of the expression of γδ T cells and their co-stimulatory molecules TIGIT, PD-1, ICOS and BTLA with PR and PIBF in the peripheral blood and decidual tissues of women with unexplained recurrent spontaneous abortion.
Idelalisib and caffeine reduce suppression of T cell responses mediated by activated chronic lymphocytic leukemia cells.
PD-L1 expression is associated with advanced non-small cell lung cancer.
Control of signaling-mediated clearance of apoptotic cells by the tumor suppressor p53.
Programmed death-1 blockade enhances the antitumor effects of peptide vaccine-induced peptide-specific cytotoxic T lymphocytes.
Immune-escape markers in relation to clinical outcome of advanced melanoma patients following immunotherapy.
Inhibiting the programmed death 1 pathway rescues Mycobacterium tuberculosis-specific interferon γ-producing T cells from apoptosis in patients with pulmonary tuberculosis.
Foxp3+ regulatory T cells among tuberculosis patients: impact on prognosis and restoration of antigen specific IFN-γ producing T cells.
Cytotoxic activity of dendritic cells as a possible mechanism of negative regulation of T lymphocytes in pulmonary tuberculosis.
IL-17 and IFN-γ expression in lymphocytes from patients with active tuberculosis correlates with the severity of the disease.
T-cell immune function in tumor, skin, and peripheral blood of advanced stage melanoma patients: implications for immunotherapy.
Immunostaining of PD-1/PD-Ls in liver tissues of patients with hepatitis and hepatocellular carcinoma.
Stromal cells' B7-1 is a key stimulatory molecule for interleukin-10 production by HOZOT, a multifunctional regulatory T-cell line.
Role played by the programmed death-1-programmed death ligand pathway during innate immunity against Mycobacterium tuberculosis.
Expression and function of PDCD1 at the human maternal-fetal interface.
Programmed death (PD)-1:PD-ligand 1/PD-ligand 2 pathway inhibits T cell effector functions during human tuberculosis.
Programmed death (PD)-1:PD-ligand 1/PD-ligand 2 pathway inhibits T cell effector functions during human tuberculosis.
FOXP3+ Tregs and B7-H1+/PD-1+ T lymphocytes co-infiltrate the tumor tissues of high-risk breast cancer patients: Implication for immunotherapy.
Expression of B7-H1 in inflammatory renal tubular epithelial cells.
NK cells that are activated by CXCL10 can kill dormant tumor cells that resist CTL-mediated lysis and can express B7-H1 that stimulates T cells.
Cord blood CD4(+)CD25(+)-derived T regulatory cell lines express FoxP3 protein and manifest potent suppressor function.
CD4+ PD-1+ T cells accumulate as unique anergic cells in rheumatoid arthritis synovial fluid.
Microanatomical localization of PD-1 in human tonsils.
Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion.
PD-L2 is a second ligand for PD-1 and inhibits T cell activation.
Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation.
Developmentally regulated expression of the PD-1 protein on the surface of double-negative (CD4-CD8-) thymocytes.
Expression of the PD-1 antigen on the surface of stimulated mouse T and B lymphocytes.
Liang Q, Tong L, Xiang L, Shen S, Pan C, Liu C, Zhang H
Clinical and experimental immunology 2021 Jan;203(1):55-65
Clinical and experimental immunology 2021 Jan;203(1):55-65
Idelalisib and caffeine reduce suppression of T cell responses mediated by activated chronic lymphocytic leukemia cells.
Hock BD, MacPherson SA, McKenzie JL
PloS one 2017;12(3):e0172858
PloS one 2017;12(3):e0172858
PD-L1 expression is associated with advanced non-small cell lung cancer.
Chen Z, Mei J, Liu L, Wang G, Li Z, Hou J, Zhang Q, You Z, Zhang L
Oncology letters 2016 Aug;12(2):921-927
Oncology letters 2016 Aug;12(2):921-927
Control of signaling-mediated clearance of apoptotic cells by the tumor suppressor p53.
Yoon KW, Byun S, Kwon E, Hwang SY, Chu K, Hiraki M, Jo SH, Weins A, Hakroush S, Cebulla A, Sykes DB, Greka A, Mundel P, Fisher DE, Mandinova A, Lee SW
Science (New York, N.Y.) 2015 Jul 31;349(6247):1261669
Science (New York, N.Y.) 2015 Jul 31;349(6247):1261669
Programmed death-1 blockade enhances the antitumor effects of peptide vaccine-induced peptide-specific cytotoxic T lymphocytes.
Sawada Y, Yoshikawa T, Shimomura M, Iwama T, Endo I, Nakatsura T
International journal of oncology 2015 Jan;46(1):28-36
International journal of oncology 2015 Jan;46(1):28-36
Immune-escape markers in relation to clinical outcome of advanced melanoma patients following immunotherapy.
Tjin EP, Krebbers G, Meijlink KJ, van de Kasteele W, Rosenberg EH, Sanders J, Nederlof PM, van de Wiel BA, Haanen JB, Melief CJ, Vyth-Dreese FA, Luiten RM
Cancer immunology research 2014 Jun;2(6):538-46
Cancer immunology research 2014 Jun;2(6):538-46
Inhibiting the programmed death 1 pathway rescues Mycobacterium tuberculosis-specific interferon γ-producing T cells from apoptosis in patients with pulmonary tuberculosis.
Singh A, Mohan A, Dey AB, Mitra DK
The Journal of infectious diseases 2013 Aug 15;208(4):603-15
The Journal of infectious diseases 2013 Aug 15;208(4):603-15
Foxp3+ regulatory T cells among tuberculosis patients: impact on prognosis and restoration of antigen specific IFN-γ producing T cells.
Singh A, Dey AB, Mohan A, Sharma PK, Mitra DK
PloS one 2012;7(9):e44728
PloS one 2012;7(9):e44728
Cytotoxic activity of dendritic cells as a possible mechanism of negative regulation of T lymphocytes in pulmonary tuberculosis.
Sakhno LV, Tikhonova MA, Tyrinova TV, Leplina OY, Shevela EY, Nikonov SD, Zhdanov OA, Ostanin AA, Chernykh ER
Clinical & developmental immunology 2012;2012:628635
Clinical & developmental immunology 2012;2012:628635
IL-17 and IFN-γ expression in lymphocytes from patients with active tuberculosis correlates with the severity of the disease.
Jurado JO, Pasquinelli V, Alvarez IB, Peña D, Rovetta AI, Tateosian NL, Romeo HE, Musella RM, Palmero D, Chuluyán HE, García VE
Journal of leukocyte biology 2012 Jun;91(6):991-1002
Journal of leukocyte biology 2012 Jun;91(6):991-1002
T-cell immune function in tumor, skin, and peripheral blood of advanced stage melanoma patients: implications for immunotherapy.
Tjin EP, Konijnenberg D, Krebbers G, Mallo H, Drijfhout JW, Franken KL, van der Horst CM, Bos JD, Nieweg OE, Kroon BB, Haanen JB, Melief CJ, Vyth-Dreese FA, Luiten RM
Clinical cancer research : an official journal of the American Association for Cancer Research 2011 Sep 1;17(17):5736-47
Clinical cancer research : an official journal of the American Association for Cancer Research 2011 Sep 1;17(17):5736-47
Immunostaining of PD-1/PD-Ls in liver tissues of patients with hepatitis and hepatocellular carcinoma.
Wang BJ, Bao JJ, Wang JZ, Wang Y, Jiang M, Xing MY, Zhang WG, Qi JY, Roggendorf M, Lu MJ, Yang DL
World journal of gastroenterology 2011 Jul 28;17(28):3322-9
World journal of gastroenterology 2011 Jul 28;17(28):3322-9
Stromal cells' B7-1 is a key stimulatory molecule for interleukin-10 production by HOZOT, a multifunctional regulatory T-cell line.
Otani T, Tsuji-Takayama K, Okochi A, Yamamoto M, Takeuchi M, Yamasaki F, Nakamura S, Kibata M
Immunology and cell biology 2011 Feb;89(2):246-54
Immunology and cell biology 2011 Feb;89(2):246-54
Role played by the programmed death-1-programmed death ligand pathway during innate immunity against Mycobacterium tuberculosis.
Alvarez IB, Pasquinelli V, Jurado JO, Abbate E, Musella RM, de la Barrera SS, García VE
The Journal of infectious diseases 2010 Aug 15;202(4):524-32
The Journal of infectious diseases 2010 Aug 15;202(4):524-32
Expression and function of PDCD1 at the human maternal-fetal interface.
Taglauer ES, Trikhacheva AS, Slusser JG, Petroff MG
Biology of reproduction 2008 Sep;79(3):562-9
Biology of reproduction 2008 Sep;79(3):562-9
Programmed death (PD)-1:PD-ligand 1/PD-ligand 2 pathway inhibits T cell effector functions during human tuberculosis.
Jurado JO, Alvarez IB, Pasquinelli V, Martínez GJ, Quiroga MF, Abbate E, Musella RM, Chuluyan HE, García VE
Journal of immunology (Baltimore, Md. : 1950) 2008 Jul 1;181(1):116-25
Journal of immunology (Baltimore, Md. : 1950) 2008 Jul 1;181(1):116-25
Programmed death (PD)-1:PD-ligand 1/PD-ligand 2 pathway inhibits T cell effector functions during human tuberculosis.
Jurado JO, Alvarez IB, Pasquinelli V, Martínez GJ, Quiroga MF, Abbate E, Musella RM, Chuluyan HE, García VE
Journal of immunology (Baltimore, Md. : 1950) 2008 Jul 1;181(1):116-25
Journal of immunology (Baltimore, Md. : 1950) 2008 Jul 1;181(1):116-25
FOXP3+ Tregs and B7-H1+/PD-1+ T lymphocytes co-infiltrate the tumor tissues of high-risk breast cancer patients: Implication for immunotherapy.
Ghebeh H, Barhoush E, Tulbah A, Elkum N, Al-Tweigeri T, Dermime S
BMC cancer 2008 Feb 23;8:57
BMC cancer 2008 Feb 23;8:57
Expression of B7-H1 in inflammatory renal tubular epithelial cells.
Chen Y, Zhang J, Li J, Zou L, Zhao T, Tang Y, Wu Y
Nephron. Experimental nephrology 2006;102(3-4):e81-92
Nephron. Experimental nephrology 2006;102(3-4):e81-92
NK cells that are activated by CXCL10 can kill dormant tumor cells that resist CTL-mediated lysis and can express B7-H1 that stimulates T cells.
Saudemont A, Jouy N, Hetuin D, Quesnel B
Blood 2005 Mar 15;105(6):2428-35
Blood 2005 Mar 15;105(6):2428-35
Cord blood CD4(+)CD25(+)-derived T regulatory cell lines express FoxP3 protein and manifest potent suppressor function.
Godfrey WR, Spoden DJ, Ge YG, Baker SR, Liu B, Levine BL, June CH, Blazar BR, Porter SB
Blood 2005 Jan 15;105(2):750-8
Blood 2005 Jan 15;105(2):750-8
CD4+ PD-1+ T cells accumulate as unique anergic cells in rheumatoid arthritis synovial fluid.
Hatachi S, Iwai Y, Kawano S, Morinobu S, Kobayashi M, Koshiba M, Saura R, Kurosaka M, Honjo T, Kumagai S
The Journal of rheumatology 2003 Jul;30(7):1410-9
The Journal of rheumatology 2003 Jul;30(7):1410-9
Microanatomical localization of PD-1 in human tonsils.
Iwai Y, Okazaki T, Nishimura H, Kawasaki A, Yagita H, Honjo T
Immunology letters 2002 Oct 1;83(3):215-20
Immunology letters 2002 Oct 1;83(3):215-20
Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion.
Dong H, Strome SE, Salomao DR, Tamura H, Hirano F, Flies DB, Roche PC, Lu J, Zhu G, Tamada K, Lennon VA, Celis E, Chen L
Nature medicine 2002 Aug;8(8):793-800
Nature medicine 2002 Aug;8(8):793-800
PD-L2 is a second ligand for PD-1 and inhibits T cell activation.
Latchman Y, Wood CR, Chernova T, Chaudhary D, Borde M, Chernova I, Iwai Y, Long AJ, Brown JA, Nunes R, Greenfield EA, Bourque K, Boussiotis VA, Carter LL, Carreno BM, Malenkovich N, Nishimura H, Okazaki T, Honjo T, Sharpe AH, Freeman GJ
Nature immunology 2001 Mar;2(3):261-8
Nature immunology 2001 Mar;2(3):261-8
Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation.
Freeman GJ, Long AJ, Iwai Y, Bourque K, Chernova T, Nishimura H, Fitz LJ, Malenkovich N, Okazaki T, Byrne MC, Horton HF, Fouser L, Carter L, Ling V, Bowman MR, Carreno BM, Collins M, Wood CR, Honjo T
The Journal of experimental medicine 2000 Oct 2;192(7):1027-34
The Journal of experimental medicine 2000 Oct 2;192(7):1027-34
Developmentally regulated expression of the PD-1 protein on the surface of double-negative (CD4-CD8-) thymocytes.
Nishimura H, Agata Y, Kawasaki A, Sato M, Imamura S, Minato N, Yagita H, Nakano T, Honjo T
International immunology 1996 May;8(5):773-80
International immunology 1996 May;8(5):773-80
Expression of the PD-1 antigen on the surface of stimulated mouse T and B lymphocytes.
Agata Y, Kawasaki A, Nishimura H, Ishida Y, Tsubata T, Yagita H, Honjo T
International immunology 1996 May;8(5):765-72
International immunology 1996 May;8(5):765-72
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
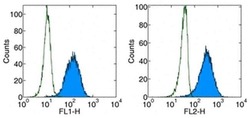
- Experimental details
- Staining of human PD-1 transfected cells with anti-human Anti-Human CD279 (PD-1) FITC (left)or PE (right).Appropriate isotype controls were used (open histogram).Total viable cells were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 2 Fig. The expression of gammadelta T cells and their co-stimulatory molecules T cell immunoglobulin and ITIM domain (TIGIT), programmed cell death 1 (PD-1), B and T lymphocyte attenuator (BTLA) and inducible co-stimulator (ICOS) as measured by flow cytometry and FlowJo software. (a) gammadelta T cell receptor (CD3 + gammadelta TCR + ) gating strategy in peripheral blood. (b) gammadelta T cell (CD45 + CD3 + gammadelta TCR + ) gating strategy in decidual tissue.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
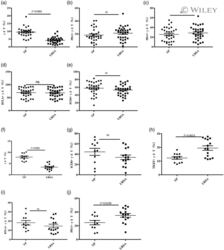
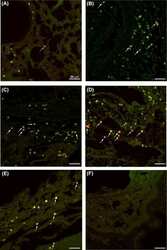
- Experimental details
- 3 Fig. The proportion of gammadelta T cells and the expression of their co-stimulatory molecules T cell immunoglobulin and ITIM domain (TIGIT), inducible co-stimulator (ICOS), B and T lymphocyte attenuator (BTLA) and programmed cell death 1 (PD-1) as determined by flow cytometry. The data are expressed as the mean +- standard error of the mean (s.e.m.). (a-e) Peripheral blood; normal pregnant (NP) group, n = 30; unexplained recurrent spontaneous abortion (URSA) group, n = 32; n.s. = not significant. (f-j) Decidual tissue; NP group, n = 11; URSA group, n = 15.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
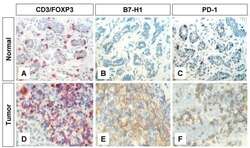
- Experimental details
- Figure 1 Immunohistochemical staining of FOXP3+ T regs B7-H1 +and PD-1+ in T lymphocytes of breast tissues . Representative micrographs at x 530 magnification of (A&D) CD3/FOXP3 double staining (red color, membranous for CD3 and brown nuclear color for FOXP3 expression). (B&E) B7-H1 single staining (brown color, membranous/cytoplasmic expression). (C&F) PD-1 single staining (brown color, membranous expression). Upper panel (A-C) is sections for normal breast duct and lower panel (D-F) is sections for infiltrating ductal carcinoma of the breast.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
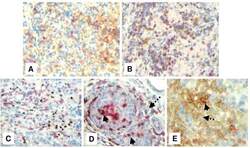
- Experimental details
- Figure 4 Immunohistochemical staining showing the expression of FOXP3, B7-H1 and PD-1 molecules by different subsets of TIL . Representative micrographs at x 530 magnification of (A) double staining of B7-H1 (brown color, membranous/cytoplasmic) and FOXP3 (red color, nuclear) in an area rich in TIL of tumor section. (B) Double staining of PD-1 (brown color, membranous) and FOXP3 (red color, nuclear) in sections from the same tumor as in A. (C) double staining of FOXP3 (brown color, nuclear) and CD8 (red color, membranous). (D) double staining of PD-1 (brown color, membranous) and CD8 (red color, membranous). Solid arrows indicate CD8 + /PD-1 + T lymphocytes and dashed arrow indicates a CD8 - /PD-1 + T lymphocyte. (E) Double staining of B7-H1 (brown color, membranous/cytoplasmic) and CD8 (red color, membranous). Solid arrow indicates a CD8 + /B7-H1 + T lymphocyte and dashed arrow indicates a CD8 - /B7-H1 + T lymphocyte.
 Explore
Explore Validate
Validate Learn
Learn Immunoprecipitation
Immunoprecipitation Flow cytometry
Flow cytometry