Antibody data
- Antibody Data
- Antigen structure
- References [4]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [3]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 46-0469-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD46 Monoclonal Antibody (8E2), PerCP-eFluor™ 710, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 8E2 monoclonal antibody reacts with human CD46 (membrane cofactor protein). CD46 is a transmembrane glycoprotein consisting of four regularly expressed isoforms, giving an electrophoretic profile of two heterogeneous protein species of 51-58 kDa ("lower band") and 59-68 kDa ("upper band"). CD46 is expressed on the majority of nucleated cells, and functions as a regulator of complement function by binding complement components C3b and C4b, which serves to protect the host from autologous complement attack. CD46 has been called a "pathogen magnet" as it is known to play a role in the entry of several pathogens including strains of measles virus, human herpes virus type 6, group A streptococci and Neisseria. Cross-linking TCR on naive human peripheral blood CD4+ T cells together with CD46, rather than CD28, induces more robust proliferation than achieved with stimulation by the costimulatory molecule CD28, as well as other qualitative differences. There is evidence to suggest that stimulation of TCR and CD46 activates natural CD4+CD25+ T regs, which are non-proliferating, but produce high amounts of granzyme A, and kill activated CD4+, CD8+, CD14+ monocytes, immature dendritic cells and mature dendritic cells. Applications Reported: This 8E2 antibody has been reported for use in flow cytometric analysis. Applications Tested: This 8E2 antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0.25 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. PerCP-eFluor® 710 emits at 710 nm and is excited with the blue laser (488 nm); it can be used in place of PerCP-Cyanine5.5. We recommend using a 710/50 bandpass filter, however, the 695/40 bandpass filter is an acceptable alternative. Please make sure that your instrument is capable of detecting this fluorochrome. Fixation: Samples can be stored in IC Fixation Buffer (Product # 00-8222) (100 µL cell sample + 100 µL IC Fixation Buffer) or 1-step Fix/Lyse Solution (Product # 00-5333) for up to 3 days in the dark at 4°C with minimal impact on brightness and FRET efficiency/compensation. Some generalizations regarding fluorophore performance after fixation can be made, but clone specific performance should be determined empirically. Excitation: 488 nm; Emission: 710 nm; Laser: Blue Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 8E2
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Spectrum-Wide Exploration of Human Adenoviruses for Breast Cancer Therapy.
In vivo antitumor activity by dual stromal and tumor-targeted oncolytic measles viruses.
Complement receptor CD46 co-stimulates optimal human CD8(+) T cell effector function via fatty acid metabolism.
Distinct Contributions of Autophagy Receptors in Measles Virus Replication.
Mach N, Gao J, Schaffarczyk L, Janz S, Ehrke-Schulz E, Dittmar T, Ehrhardt A, Zhang W
Cancers 2020 May 29;12(6)
Cancers 2020 May 29;12(6)
In vivo antitumor activity by dual stromal and tumor-targeted oncolytic measles viruses.
Jing Y, Chavez V, Khatwani N, Ban Y, Espejo AP, Chen X, Merchan JR
Cancer gene therapy 2020 Dec;27(12):910-922
Cancer gene therapy 2020 Dec;27(12):910-922
Complement receptor CD46 co-stimulates optimal human CD8(+) T cell effector function via fatty acid metabolism.
Arbore G, West EE, Rahman J, Le Friec G, Niyonzima N, Pirooznia M, Tunc I, Pavlidis P, Powell N, Li Y, Liu P, Servais A, Couzi L, Fremeaux-Bacchi V, Placais L, Ferraro A, Walsh PR, Kavanagh D, Afzali B, Lavender P, Lachmann HJ, Kemper C
Nature communications 2018 Oct 10;9(1):4186
Nature communications 2018 Oct 10;9(1):4186
Distinct Contributions of Autophagy Receptors in Measles Virus Replication.
Petkova DS, Verlhac P, Rozières A, Baguet J, Claviere M, Kretz-Remy C, Mahieux R, Viret C, Faure M
Viruses 2017 May 22;9(5)
Viruses 2017 May 22;9(5)
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Staining of normal human peripheral blood cells with Mouse IgG1 K Isotype Control PerCP-eFluor® 710 (Product # 46-4714-82) (blue histogram) or Anti-Human CD46 PerCP-eFluor® 710 (purple histogram). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Major adenovirus receptor expression levels on BC cell lines. Cells were stained with antibodies against coxsackievirus and adenovirus receptor (CAR) ( A ), cluster of differentiation 46 (CD46) ( B ), desmoglein-2 receptor (DSG-2) ( C ), and integrins alphavbeta3 and alphavbeta5 ( D,E ). Receptor expression was measured via flow cytometry, expressed as percentage of CAR, CD46, DSG-2, and integrin-receptor-positive cells. Hela cells were used as positive control. Unlabeled cells (negative controls) were used to set the background gate below 1%. A total of 10,000 viable cells were counted.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
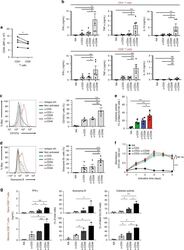
- Experimental details
- Fig. 1 CD46 co-stimulation provides superior support for CTL activity. a CD46 expression on the surface of resting human CD4 + and CD8 + T cells assessed by FACS analysis ( n = 4, gating strategy in Supplementary Fig. 7a ). b Comparison of IFN-gamma, TNF-alpha and IL-10 secretion by CD3 + CD46-activated T cells. Purified CD4 + and CD8 + T cells from healthy donors were left non-activated (NA) or stimulated with immobilized antibodies to CD3, CD3 + CD28 or CD3 + CD46 and cytokines measured 60 h post activation ( n = 5). c, d Degranulation (CD107a staining, ( c )) and granzyme B expression ( d ) by CD8 + T cells upon CD46 co-stimulation. CD8 + T cells were stimulated as in ( a ) and CD107a and granzyme B expression assessed with left panels showing representative cytometry images and right panels corresponding quantifications ( n = 3, gating strategy in Supplementary Fig. 7b ). e Killing activity of CD46-activated CD8 + T cells. T cells were stimulated as depicted for 24 h and cytotoxic activity of differently activated CD8 + T cells towards DU145 target cells assessed 24 h post co-culture of T cells and DU145 cells ( n = 4, gating strategy in Supplementary Fig. 7c ). f Effect of CD46 co-stimulation on CD8 + T-cell proliferation. Purified T cells were activated as indicated for 5 days and cell proliferation measured each day ( n = 4) (black circles, non-activated cells; green, blue, and red circles, CD3, CD3 + CD28 or CD3 + CD46-activated cells, res
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 NF-kappaB-independent role of T6BP and NDP52 in MeV replication. ( A ) p65/RelA-expressing HeLa cells and shControl-expressing HeLa cells were transfected with the indicated siRNAs for 48 h, then lysed, and the expression of relevant proteins was probed by Western blot; ( B ) p65/RelA-expressing HeLa cells and shControl-expressing cells were infected with MeV (MOI 0.1). 48 h post infection, infectious virus particles were titrated by a plaque assay; ( C ) Cells from ( B ) were lysed 48 h post infection. Expression of measles virus N and P proteins were assessed by Western blotting. Representative results from shp65#1 are shown and are accompanied by a graph representing the intensity of MeV-N and MeV-P expression over Actin normalized to shControl-expressing cells condition. Means +- SD of four independent experiments are represented (two with the shp65#1 cell line and two with the shp65#2 cell line); ( D ) p65/RelA-expressing HeLa cells and shControl-expressing HeLa cells were stained for CD46 expression and analyzed by flow cytometry; grey histograms = isotype control, white histograms = CD46 labelling. ( E - G ) p65/RelA-expressing HeLa cells were treated with indicated siRNAs for 48 h; ( E ) Cells were lysed and the expression of relevant proteins was probed by Western blotting. Results regarding cell line shp65 #1 are represented. Similar results were obtained with shp65 #2. Cells were infected with MeV (MOI 0.1) and 48 h post infection, infectious virus part
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry