12-5983-41
antibody from Invitrogen Antibodies
Targeting: CD274
B7-H, B7-H1, B7H1, PD-L1, PDCD1LG1, PDL1
Antibody data
- Antibody Data
- Antigen structure
- References [84]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [39]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 12-5983-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD274 (PD-L1, B7-H1) Monoclonal Antibody (MIH1), PE, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The MIH1 monoclonal antibody reacts with human B7-H1, also known as PD-L1. B7-H1, a member of the B7 family, has a predicted molecular weight of approximately 40 kDa and belongs to the Ig superfamily. B7-H1 is expressed on a majority of leukocytes. B7-H1 is a ligand for PD-1. Interaction of PD-1 with either PD-L1 (B7-H1) or PD-L2 (B7-DC) results in inhibition of T and B cell responses. MIH1 is reported to be a blocking antibody. Applications Reported: The MIH1 antibody has been reported for use in flow cytometric analysis. Applications Tested: This MIH1 antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0.5 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488-561 nm; Emission: 578 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Yellow dye
- Isotype
- IgG
- Antibody clone number
- MIH1
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references CCX559 is a potent, orally-administered small molecule PD-L1 inhibitor that induces anti-tumor immunity.
A Tumor Microenvironment-Driven Network Regulated by STAT3 and p65 Negatively Controls the Enrichment of Cancer Stem Cells in Human HR+/HER2- Breast Cancer.
Cargo and Functional Profile of Saliva-Derived Exosomes Reveal Biomarkers Specific for Head and Neck Cancer.
Chitinase-3 like-protein-1 promotes glioma progression via the NF-κB signaling pathway and tumor microenvironment reprogramming.
Lysosomal acid lipase, CSF1R, and PD-L1 determine functions of CD11c+ myeloid-derived suppressor cells.
WNK3 inhibition elicits antitumor immunity by suppressing PD-L1 expression on tumor cells and activating T-cell function.
PD-L1 CAR effector cells induce self-amplifying cytotoxic effects against target cells.
Cancer genes disfavoring T cell immunity identified via integrated systems approach.
NK cells limit therapeutic vaccine-induced CD8(+)T cell immunity in a PD-L1-dependent manner.
Inhibition of PI3Kδ Differentially Regulates Poly I:C- and Human Metapneumovirus-Induced PD-L1 and PD-L2 Expression in Human Bronchial Epithelial Cells.
Epithelial to Mesenchymal Transition Regulates Surface PD-L1 via CMTM6 and CMTM7 Induction in Breast Cancer.
Interaction between HLA-G and NK cell receptor KIR2DL4 orchestrates HER2-positive breast cancer resistance to trastuzumab.
Protocol for high-throughput compound screening using flow cytometry in THP-1 cells.
Changes in circulating exosome molecular profiles following surgery/(chemo)radiotherapy: early detection of response in head and neck cancer patients.
β-Catenin induces transcriptional expression of PD-L1 to promote glioblastoma immune evasion.
Verteporfin Inhibits PD-L1 through Autophagy and the STAT1-IRF1-TRIM28 Signaling Axis, Exerting Antitumor Efficacy.
The deubiquitinase USP22 regulates PD-L1 degradation in human cancer cells.
PD-L1 Expression in Pediatric Low-Grade Gliomas Is Independent of BRAF V600E Mutational Status.
Combining DNMT and HDAC6 inhibitors increases anti-tumor immune signaling and decreases tumor burden in ovarian cancer.
Altered bioenergetics and mitochondrial dysfunction of monocytes in patients with COVID-19 pneumonia.
Protein kinase D3 regulates the expression of the immunosuppressive protein, PD‑L1, through STAT1/STAT3 signaling.
NPM1 upregulates the transcription of PD-L1 and suppresses T cell activity in triple-negative breast cancer.
Circulating exosomes measure responses to therapy in head and neck cancer patients treated with cetuximab, ipilimumab, and IMRT.
Aberrant upregulation of PDK1 in ovarian cancer cells impairs CD8(+) T cell function and survival through elevation of PD-L1.
HER2 signaling regulates the tumor immune microenvironment and trastuzumab efficacy.
TLR9 Mediated Tumor-Stroma Interactions in Human Papilloma Virus (HPV)-Positive Head and Neck Squamous Cell Carcinoma Up-Regulate PD-L1 and PD-L2.
A kinome-wide screen using a NanoLuc LATS luminescent biosensor identifies ALK as a novel regulator of the Hippo pathway in tumorigenesis and immune evasion.
PD-L1 checkpoint blockade delivered by retroviral replicating vector confers anti-tumor efficacy in murine tumor models.
Phospho‑STAT1 expression as a potential biomarker for anti‑PD‑1/anti‑PD‑L1 immunotherapy for breast cancer.
miRNA-148a-3p Regulates Immunosuppression in DNA Mismatch Repair-Deficient Colorectal Cancer by Targeting PD-L1.
Inhibition of ATM kinase upregulates levels of cell death induced by cannabidiol and γ-irradiation in human glioblastoma cells.
Immunological Properties of Neural Crest Cells Derived from Human Induced Pluripotent Stem Cells.
PD-1 of Sigmodon hispidus: Gene identification, characterization and preliminary evaluation of expression in inactivated RSV vaccine-induced enhanced respiratory disease.
Suppression of Exosomal PD-L1 Induces Systemic Anti-tumor Immunity and Memory.
Disruption of PD-1 Enhanced the Anti-tumor Activity of Chimeric Antigen Receptor T Cells Against Hepatocellular Carcinoma.
Macrophage inducible nitric oxide synthase circulates inflammation and promotes lung carcinogenesis.
Homeobox containing 1 inhibits liver cancer progression by promoting autophagy as well as inhibiting stemness and immune escape.
Targeting the upstream transcriptional activator of PD-L1 as an alternative strategy in melanoma therapy.
Apigenin suppresses PD-L1 expression in melanoma and host dendritic cells to elicit synergistic therapeutic effects.
The PD-L1- and IL6-mediated dampening of the IL27/STAT1 anticancer responses are prevented by α-PD-L1 or α-IL6 antibodies.
Status of programmed death-ligand 1 expression in sarcomas.
Specific expression of PD-L1 in RELA-fusion supratentorial ependymoma: Implications for PD-1-targeted therapy.
Oncofetal gene SALL4 reactivation by hepatitis B virus counteracts miR-200c in PD-L1-induced T cell exhaustion.
PD-L1 expression by tumor cell lines: A predictive marker in melanoma.
Separation of plasma-derived exosomes into CD3((+)) and CD3((-)) fractions allows for association of immune cell and tumour cell markers with disease activity in HNSCC patients.
Antigen-Presenting Cell-Intrinsic PD-1 Neutralizes PD-L1 in cis to Attenuate PD-1 Signaling in T Cells.
PD-1 axis expression in musculoskeletal tumors and antitumor effect of nivolumab in osteosarcoma model of humanized mouse.
PD-L1 expression in medulloblastoma: an evaluation by subgroup.
Mesenchymal Stromal Cell Secretion of Programmed Death-1 Ligands Regulates T Cell Mediated Immunosuppression.
Retroviral Replicating Vector Delivery of miR-PDL1 Inhibits Immune Checkpoint PDL1 and Enhances Immune Responses In Vitro.
Oncogenic RAS Signaling Promotes Tumor Immunoresistance by Stabilizing PD-L1 mRNA.
PD-L1/PD-1 expression and tumor-infiltrating lymphocytes in conjunctival melanoma.
Expression of programmed death 1 ligand 1 on periodontal tissue cells as a possible protective feedback mechanism against periodontal tissue destruction.
CD28 family of receptors on T cells in chronic HBV infection: Expression characteristics, clinical significance and correlations with PD-1 blockade.
Interleukin-25 Mediates Transcriptional Control of PD-L1 via STAT3 in Multipotent Human Mesenchymal Stromal Cells (hMSCs) to Suppress Th17 Responses.
Engineering high-affinity PD-1 variants for optimized immunotherapy and immuno-PET imaging.
Cervical (pre)neoplastic microenvironment promotes the emergence of tolerogenic dendritic cells via RANKL secretion.
PD-L1 expression in melanocytic lesions does not correlate with the BRAF V600E mutation.
Paucity of PD-L1 expression in prostate cancer: innate and adaptive immune resistance.
IL-27 induces the expression of IDO and PD-L1 in human cancer cells.
Interferon beta and vitamin D synergize to induce immunoregulatory receptors on peripheral blood monocytes of multiple sclerosis patients.
Brugia malayi microfilariae induce a regulatory monocyte/macrophage phenotype that suppresses innate and adaptive immune responses.
Inhibition of mitogen-activated protein kinase pathway can induce upregulation of human leukocyte antigen class I without PD-L1-upregulation in contrast to interferon-γ treatment.
Metastasis is regulated via microRNA-200/ZEB1 axis control of tumour cell PD-L1 expression and intratumoral immunosuppression.
Differential potency of regulatory T cell-mediated immunosuppression in kidney tumors compared to subcutaneous tumors.
Th1 cytokines are more effective than Th2 cytokines at licensing anti-tumour functions in CD40-activated human macrophages in vitro.
Alterations of immune response of Non-Small Cell Lung Cancer with Azacytidine.
Gliomas promote immunosuppression through induction of B7-H1 expression in tumor-associated macrophages.
Multiple inhibitory ligands induce impaired T-cell immunologic synapse function in chronic lymphocytic leukemia that can be blocked with lenalidomide: establishing a reversible immune evasion mechanism in human cancer.
Upregulation of programmed death-1 on T cells and programmed death ligand-1 on monocytes in septic shock patients.
Phagocytosis, a potential mechanism for myeloid-derived suppressor cell regulation of CD8+ T cell function mediated through programmed cell death-1 and programmed cell death-1 ligand interaction.
Programmed death ligand 1 is expressed by non-hodgkin lymphomas and inhibits the activity of tumor-associated T cells.
Transfer of regulatory properties from tolerogenic to proinflammatory dendritic cells via induced autoreactive regulatory T cells.
Ex vivo expanded hematopoietic stem cells overcome the MHC barrier in allogeneic transplantation.
The PD-1/PD-L1 axis modulates the natural killer cell versus multiple myeloma effect: a therapeutic target for CT-011, a novel monoclonal anti-PD-1 antibody.
Increase of programmed death-1-expressing intratumoral CD8 T cells predicts a poor prognosis for nasopharyngeal carcinoma.
Novel role of plasmacytoid dendritic cells in humans: induction of interleukin-10-producing Treg cells by plasmacytoid dendritic cells in patients with rheumatoid arthritis responding to therapy.
Critical role for TNF in the induction of human antigen-specific regulatory T cells by tolerogenic dendritic cells.
PD-1 is a regulator of NY-ESO-1-specific CD8+ T cell expansion in melanoma patients.
Neuron-interacting satellite glial cells in human trigeminal ganglia have an APC phenotype.
Expression and function of PDCD1 at the human maternal-fetal interface.
TLR-mediated induction of negative regulatory ligands on dendritic cells.
Expression of B7-H1 in inflammatory renal tubular epithelial cells.
Clinical significance of programmed death-1 ligand-1 and programmed death-1 ligand-2 expression in human esophageal cancer.
Sullivan KMC, Vilalta M, Ertl LS, Wang Y, Dunlap C, Ebsworth K, Zhao BN, Li S, Zeng Y, Miao Z, Fan P, Mali V, Lange C, McMurtrie D, Yang J, Lui R, Scamp R, Chhina V, Kumamoto A, Yau S, Dang T, Easterday A, Liu S, Miao S, Charo I, Schall TJ, Zhang P
PloS one 2023;18(6):e0286724
PloS one 2023;18(6):e0286724
A Tumor Microenvironment-Driven Network Regulated by STAT3 and p65 Negatively Controls the Enrichment of Cancer Stem Cells in Human HR+/HER2- Breast Cancer.
Ben-Yaakov H, Meshel T, Pasmanik-Chor M, Körner C, Ben-Baruch A
Cancers 2023 Apr 12;15(8)
Cancers 2023 Apr 12;15(8)
Cargo and Functional Profile of Saliva-Derived Exosomes Reveal Biomarkers Specific for Head and Neck Cancer.
Hofmann L, Medyany V, Ezić J, Lotfi R, Niesler B, Röth R, Engelhardt D, Laban S, Schuler PJ, Hoffmann TK, Brunner C, Jackson EK, Theodoraki MN
Frontiers in medicine 2022;9:904295
Frontiers in medicine 2022;9:904295
Chitinase-3 like-protein-1 promotes glioma progression via the NF-κB signaling pathway and tumor microenvironment reprogramming.
Zhao T, Zeng J, Xu Y, Su Z, Chong Y, Ling T, Xu H, Shi H, Zhu M, Mo Q, Huang X, Li Y, Zhang X, Ni H, You Q
Theranostics 2022;12(16):6989-7008
Theranostics 2022;12(16):6989-7008
Lysosomal acid lipase, CSF1R, and PD-L1 determine functions of CD11c+ myeloid-derived suppressor cells.
Zhao T, Liu S, Ding X, Johnson EM, Hanna NH, Singh K, Sen CK, Wan J, Du H, Yan C
JCI insight 2022 Sep 8;7(17)
JCI insight 2022 Sep 8;7(17)
WNK3 inhibition elicits antitumor immunity by suppressing PD-L1 expression on tumor cells and activating T-cell function.
Yoon HJ, Kim GC, Oh S, Kim H, Kim YK, Lee Y, Kim MS, Kwon G, Ok YS, Kwon HK, Kim HS
Experimental & molecular medicine 2022 Nov;54(11):1913-1926
Experimental & molecular medicine 2022 Nov;54(11):1913-1926
PD-L1 CAR effector cells induce self-amplifying cytotoxic effects against target cells.
Bajor M, Graczyk-Jarzynka A, Marhelava K, Burdzinska A, Muchowicz A, Goral A, Zhylko A, Soroczynska K, Retecki K, Krawczyk M, Klopotowska M, Pilch Z, Paczek L, Malmberg KJ, Wälchli S, Winiarska M, Zagozdzon R
Journal for immunotherapy of cancer 2022 Jan;10(1)
Journal for immunotherapy of cancer 2022 Jan;10(1)
Cancer genes disfavoring T cell immunity identified via integrated systems approach.
Kishton RJ, Patel SJ, Decker AE, Vodnala SK, Cam M, Yamamoto TN, Patel Y, Sukumar M, Yu Z, Ji M, Henning AN, Gurusamy D, Palmer DC, Stefanescu RA, Girvin AT, Lo W, Pasetto A, Malekzadeh P, Deniger DC, Wood KC, Sanjana NE, Restifo NP
Cell reports 2022 Aug 2;40(5):111153
Cell reports 2022 Aug 2;40(5):111153
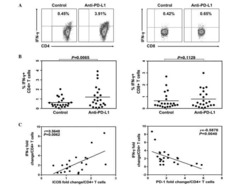
NK cells limit therapeutic vaccine-induced CD8(+)T cell immunity in a PD-L1-dependent manner.
Diniz MO, Schurich A, Chinnakannan SK, Duriez M, Stegmann KA, Davies J, Kucykowicz S, Suveizdyte K, Amin OE, Alcock F, Cargill T, Barnes E, Maini MK
Science translational medicine 2022 Apr 13;14(640):eabi4670
Science translational medicine 2022 Apr 13;14(640):eabi4670
Inhibition of PI3Kδ Differentially Regulates Poly I:C- and Human Metapneumovirus-Induced PD-L1 and PD-L2 Expression in Human Bronchial Epithelial Cells.
Ogawa T, Kan-O K, Shiota A, Fujita A, Ishii Y, Fukuyama S, Matsumoto K
Frontiers in immunology 2021;12:767666
Frontiers in immunology 2021;12:767666
Epithelial to Mesenchymal Transition Regulates Surface PD-L1 via CMTM6 and CMTM7 Induction in Breast Cancer.
Xiao M, Hasmim M, Lequeux A, Moer KV, Tan TZ, Gilles C, Hollier BG, Thiery JP, Berchem G, Janji B, Noman MZ
Cancers 2021 Mar 9;13(5)
Cancers 2021 Mar 9;13(5)
Interaction between HLA-G and NK cell receptor KIR2DL4 orchestrates HER2-positive breast cancer resistance to trastuzumab.
Zheng G, Guo Z, Li W, Xi W, Zuo B, Zhang R, Wen W, Yang AG, Jia L
Signal transduction and targeted therapy 2021 Jun 23;6(1):236
Signal transduction and targeted therapy 2021 Jun 23;6(1):236
Protocol for high-throughput compound screening using flow cytometry in THP-1 cells.
Spangenberg SH, Zavareh RB, Lairson LL
STAR protocols 2021 Jun 18;2(2):100400
STAR protocols 2021 Jun 18;2(2):100400
Changes in circulating exosome molecular profiles following surgery/(chemo)radiotherapy: early detection of response in head and neck cancer patients.
Theodoraki MN, Laban S, Jackson EK, Lotfi R, Schuler PJ, Brunner C, Hoffmann TK, Whiteside TL, Hofmann L
British journal of cancer 2021 Dec;125(12):1677-1686
British journal of cancer 2021 Dec;125(12):1677-1686
β-Catenin induces transcriptional expression of PD-L1 to promote glioblastoma immune evasion.
Du L, Lee JH, Jiang H, Wang C, Wang S, Zheng Z, Shao F, Xu D, Xia Y, Li J, Zheng Y, Qian X, Li X, Kim HR, Xing D, Liu P, Lu Z, Lyu J
The Journal of experimental medicine 2020 Nov 2;217(11)
The Journal of experimental medicine 2020 Nov 2;217(11)
Verteporfin Inhibits PD-L1 through Autophagy and the STAT1-IRF1-TRIM28 Signaling Axis, Exerting Antitumor Efficacy.
Liang J, Wang L, Wang C, Shen J, Su B, Marisetty AL, Fang D, Kassab C, Jeong KJ, Zhao W, Lu Y, Jain AK, Zhou Z, Liang H, Sun SC, Lu C, Xu ZX, Yu Q, Shao S, Chen X, Gao M, Claret FX, Ding Z, Chen J, Chen P, Barton MC, Peng G, Mills GB, Heimberger AB
Cancer immunology research 2020 Jul;8(7):952-965
Cancer immunology research 2020 Jul;8(7):952-965
The deubiquitinase USP22 regulates PD-L1 degradation in human cancer cells.
Wang Y, Sun Q, Mu N, Sun X, Wang Y, Fan S, Su L, Liu X
Cell communication and signaling : CCS 2020 Jul 14;18(1):112
Cell communication and signaling : CCS 2020 Jul 14;18(1):112
PD-L1 Expression in Pediatric Low-Grade Gliomas Is Independent of BRAF V600E Mutational Status.
Martin AM, Bell WR, Yuan M, Harris L, Poore B, Arnold A, Engle EL, Asnaghi L, Lim M, Raabe EH, Eberhart CG
Journal of neuropathology and experimental neurology 2020 Jan 1;79(1):74-85
Journal of neuropathology and experimental neurology 2020 Jan 1;79(1):74-85
Combining DNMT and HDAC6 inhibitors increases anti-tumor immune signaling and decreases tumor burden in ovarian cancer.
Moufarrij S, Srivastava A, Gomez S, Hadley M, Palmer E, Austin PT, Chisholm S, Diab N, Roche K, Yu A, Li J, Zhu W, Lopez-Acevedo M, Villagra A, Chiappinelli KB
Scientific reports 2020 Feb 26;10(1):3470
Scientific reports 2020 Feb 26;10(1):3470
Altered bioenergetics and mitochondrial dysfunction of monocytes in patients with COVID-19 pneumonia.
Gibellini L, De Biasi S, Paolini A, Borella R, Boraldi F, Mattioli M, Lo Tartaro D, Fidanza L, Caro-Maldonado A, Meschiari M, Iadisernia V, Bacca E, Riva G, Cicchetti L, Quaglino D, Guaraldi G, Busani S, Girardis M, Mussini C, Cossarizza A
EMBO molecular medicine 2020 Dec 7;12(12):e13001
EMBO molecular medicine 2020 Dec 7;12(12):e13001
Protein kinase D3 regulates the expression of the immunosuppressive protein, PD‑L1, through STAT1/STAT3 signaling.
Cui B, Chen J, Luo M, Wang L, Chen H, Kang Y, Wang J, Zhou X, Feng Y, Zhang P
International journal of oncology 2020 Apr;56(4):909-920
International journal of oncology 2020 Apr;56(4):909-920
NPM1 upregulates the transcription of PD-L1 and suppresses T cell activity in triple-negative breast cancer.
Qin G, Wang X, Ye S, Li Y, Chen M, Wang S, Qin T, Zhang C, Li Y, Long Q, Hu H, Shi D, Li J, Zhang K, Zhai Q, Tang Y, Kang T, Lan P, Xie F, Lu J, Deng W
Nature communications 2020 Apr 3;11(1):1669
Nature communications 2020 Apr 3;11(1):1669
Circulating exosomes measure responses to therapy in head and neck cancer patients treated with cetuximab, ipilimumab, and IMRT.
Theodoraki MN, Yerneni S, Gooding WE, Ohr J, Clump DA, Bauman JE, Ferris RL, Whiteside TL
Oncoimmunology 2019;8(7):1593805
Oncoimmunology 2019;8(7):1593805
Aberrant upregulation of PDK1 in ovarian cancer cells impairs CD8(+) T cell function and survival through elevation of PD-L1.
Wang JJ, Siu MK, Jiang YX, Leung TH, Chan DW, Cheng RR, Cheung AN, Ngan HY, Chan KK
Oncoimmunology 2019;8(11):e1659092
Oncoimmunology 2019;8(11):e1659092
HER2 signaling regulates the tumor immune microenvironment and trastuzumab efficacy.
Triulzi T, Forte L, Regondi V, Di Modica M, Ghirelli C, Carcangiu ML, Sfondrini L, Balsari A, Tagliabue E
Oncoimmunology 2019;8(1):e1512942
Oncoimmunology 2019;8(1):e1512942
TLR9 Mediated Tumor-Stroma Interactions in Human Papilloma Virus (HPV)-Positive Head and Neck Squamous Cell Carcinoma Up-Regulate PD-L1 and PD-L2.
Baruah P, Bullenkamp J, Wilson POG, Lee M, Kaski JC, Dumitriu IE
Frontiers in immunology 2019;10:1644
Frontiers in immunology 2019;10:1644
A kinome-wide screen using a NanoLuc LATS luminescent biosensor identifies ALK as a novel regulator of the Hippo pathway in tumorigenesis and immune evasion.
Nouri K, Azad T, Lightbody E, Khanal P, Nicol CJ, Yang X
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2019 Nov;33(11):12487-12499
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2019 Nov;33(11):12487-12499
PD-L1 checkpoint blockade delivered by retroviral replicating vector confers anti-tumor efficacy in murine tumor models.
Mitchell LA, Yagiz K, Hofacre A, Viaud S, Munday AW, Espinoza FL, Mendoza D, Rodriguez-Aguirre ME, Bergqvist S, Haghighi A, Miner MV, Accomando WP, Burrascano C, Gammon D, Gruber HE, Jolly DJ, Lin AH
Oncotarget 2019 Mar 19;10(23):2252-2269
Oncotarget 2019 Mar 19;10(23):2252-2269
Phospho‑STAT1 expression as a potential biomarker for anti‑PD‑1/anti‑PD‑L1 immunotherapy for breast cancer.
Nakayama Y, Mimura K, Tamaki T, Shiraishi K, Kua LF, Koh V, Ohmori M, Kimura A, Inoue S, Okayama H, Suzuki Y, Nakazawa T, Ichikawa D, Kono K
International journal of oncology 2019 Jun;54(6):2030-2038
International journal of oncology 2019 Jun;54(6):2030-2038
miRNA-148a-3p Regulates Immunosuppression in DNA Mismatch Repair-Deficient Colorectal Cancer by Targeting PD-L1.
Ashizawa M, Okayama H, Ishigame T, Thar Min AK, Saito K, Ujiie D, Murakami Y, Kikuchi T, Nakayama Y, Noda M, Tada T, Endo H, Fujita S, Sakamoto W, Saito M, Saze Z, Momma T, Ohki S, Mimura K, Kono K
Molecular cancer research : MCR 2019 Jun;17(6):1403-1413
Molecular cancer research : MCR 2019 Jun;17(6):1403-1413
Inhibition of ATM kinase upregulates levels of cell death induced by cannabidiol and γ-irradiation in human glioblastoma cells.
Ivanov VN, Wu J, Wang TJC, Hei TK
Oncotarget 2019 Jan 25;10(8):825-846
Oncotarget 2019 Jan 25;10(8):825-846
Immunological Properties of Neural Crest Cells Derived from Human Induced Pluripotent Stem Cells.
Fujii S, Yoshida S, Inagaki E, Hatou S, Tsubota K, Takahashi M, Shimmura S, Sugita S
Stem cells and development 2019 Jan 1;28(1):28-43
Stem cells and development 2019 Jan 1;28(1):28-43
PD-1 of Sigmodon hispidus: Gene identification, characterization and preliminary evaluation of expression in inactivated RSV vaccine-induced enhanced respiratory disease.
Muralidharan A, Larocque L, Russell M, Creskey M, Li C, Chen W, Van Domselaar G, Cao J, Cyr T, Rosu-Myles M, Wang L, Li X
Scientific reports 2019 Aug 12;9(1):11638
Scientific reports 2019 Aug 12;9(1):11638
Suppression of Exosomal PD-L1 Induces Systemic Anti-tumor Immunity and Memory.
Poggio M, Hu T, Pai CC, Chu B, Belair CD, Chang A, Montabana E, Lang UE, Fu Q, Fong L, Blelloch R
Cell 2019 Apr 4;177(2):414-427.e13
Cell 2019 Apr 4;177(2):414-427.e13
Disruption of PD-1 Enhanced the Anti-tumor Activity of Chimeric Antigen Receptor T Cells Against Hepatocellular Carcinoma.
Guo X, Jiang H, Shi B, Zhou M, Zhang H, Shi Z, Du G, Luo H, Wu X, Wang Y, Sun R, Li Z
Frontiers in pharmacology 2018;9:1118
Frontiers in pharmacology 2018;9:1118
Macrophage inducible nitric oxide synthase circulates inflammation and promotes lung carcinogenesis.
Wang X, Gray Z, Willette-Brown J, Zhu F, Shi G, Jiang Q, Song NY, Dong L, Hu Y
Cell death discovery 2018;4:46
Cell death discovery 2018;4:46
Homeobox containing 1 inhibits liver cancer progression by promoting autophagy as well as inhibiting stemness and immune escape.
Zhao H, Jia H, Han Q, Zhang J
Oncology reports 2018 Sep;40(3):1657-1665
Oncology reports 2018 Sep;40(3):1657-1665
Targeting the upstream transcriptional activator of PD-L1 as an alternative strategy in melanoma therapy.
Zhu B, Tang L, Chen S, Yin C, Peng S, Li X, Liu T, Liu W, Han C, Stawski L, Xu ZX, Zhou G, Chen X, Gao X, Goding CR, Xu N, Cui R, Cao P
Oncogene 2018 Sep;37(36):4941-4954
Oncogene 2018 Sep;37(36):4941-4954
Apigenin suppresses PD-L1 expression in melanoma and host dendritic cells to elicit synergistic therapeutic effects.
Xu L, Zhang Y, Tian K, Chen X, Zhang R, Mu X, Wu Y, Wang D, Wang S, Liu F, Wang T, Zhang J, Liu S, Zhang Y, Tu C, Liu H
Journal of experimental & clinical cancer research : CR 2018 Oct 29;37(1):261
Journal of experimental & clinical cancer research : CR 2018 Oct 29;37(1):261
The PD-L1- and IL6-mediated dampening of the IL27/STAT1 anticancer responses are prevented by α-PD-L1 or α-IL6 antibodies.
Rolvering C, Zimmer AD, Ginolhac A, Margue C, Kirchmeyer M, Servais F, Hermanns HM, Hergovits S, Nazarov PV, Nicot N, Kreis S, Haan S, Behrmann I, Haan C
Journal of leukocyte biology 2018 Nov;104(5):969-985
Journal of leukocyte biology 2018 Nov;104(5):969-985
Status of programmed death-ligand 1 expression in sarcomas.
Park HK, Kim M, Sung M, Lee SE, Kim YJ, Choi YL
Journal of translational medicine 2018 Nov 6;16(1):303
Journal of translational medicine 2018 Nov 6;16(1):303
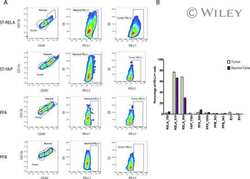
Specific expression of PD-L1 in RELA-fusion supratentorial ependymoma: Implications for PD-1-targeted therapy.
Witt DA, Donson AM, Amani V, Moreira DC, Sanford B, Hoffman LM, Handler MH, Levy JMM, Jones KL, Nellan A, Foreman NK, Griesinger AM
Pediatric blood & cancer 2018 May;65(5):e26960
Pediatric blood & cancer 2018 May;65(5):e26960
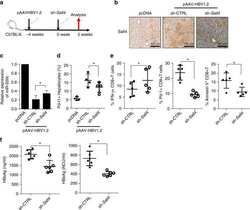
Oncofetal gene SALL4 reactivation by hepatitis B virus counteracts miR-200c in PD-L1-induced T cell exhaustion.
Sun C, Lan P, Han Q, Huang M, Zhang Z, Xu G, Song J, Wang J, Wei H, Zhang J, Sun R, Zhang C, Tian Z
Nature communications 2018 Mar 28;9(1):1241
Nature communications 2018 Mar 28;9(1):1241
PD-L1 expression by tumor cell lines: A predictive marker in melanoma.
Knol AC, Nguyen JM, Pandolfino MC, Denis MG, Khammari A, Dréno B
Experimental dermatology 2018 Jun;27(6):647-655
Experimental dermatology 2018 Jun;27(6):647-655
Separation of plasma-derived exosomes into CD3((+)) and CD3((-)) fractions allows for association of immune cell and tumour cell markers with disease activity in HNSCC patients.
Theodoraki MN, Hoffmann TK, Whiteside TL
Clinical and experimental immunology 2018 Jun;192(3):271-283
Clinical and experimental immunology 2018 Jun;192(3):271-283
Antigen-Presenting Cell-Intrinsic PD-1 Neutralizes PD-L1 in cis to Attenuate PD-1 Signaling in T Cells.
Zhao Y, Harrison DL, Song Y, Ji J, Huang J, Hui E
Cell reports 2018 Jul 10;24(2):379-390.e6
Cell reports 2018 Jul 10;24(2):379-390.e6
PD-1 axis expression in musculoskeletal tumors and antitumor effect of nivolumab in osteosarcoma model of humanized mouse.
Zheng B, Ren T, Huang Y, Sun K, Wang S, Bao X, Liu K, Guo W
Journal of hematology & oncology 2018 Feb 6;11(1):16
Journal of hematology & oncology 2018 Feb 6;11(1):16
PD-L1 expression in medulloblastoma: an evaluation by subgroup.
Martin AM, Nirschl CJ, Polanczyk MJ, Bell WR, Nirschl TR, Harris-Bookman S, Phallen J, Hicks J, Martinez D, Ogurtsova A, Xu H, Sullivan LM, Meeker AK, Raabe EH, Cohen KJ, Eberhart CG, Burger PC, Santi M, Taube JM, Pardoll DM, Drake CG, Lim M
Oncotarget 2018 Apr 10;9(27):19177-19191
Oncotarget 2018 Apr 10;9(27):19177-19191
Mesenchymal Stromal Cell Secretion of Programmed Death-1 Ligands Regulates T Cell Mediated Immunosuppression.
Davies LC, Heldring N, Kadri N, Le Blanc K
Stem cells (Dayton, Ohio) 2017 Mar;35(3):766-776
Stem cells (Dayton, Ohio) 2017 Mar;35(3):766-776
Retroviral Replicating Vector Delivery of miR-PDL1 Inhibits Immune Checkpoint PDL1 and Enhances Immune Responses In Vitro.
Lin AH, Twitty CG, Burnett R, Hofacre A, Mitchell LA, Espinoza FL, Gruber HE, Jolly DJ
Molecular therapy. Nucleic acids 2017 Mar 17;6:221-232
Molecular therapy. Nucleic acids 2017 Mar 17;6:221-232
Oncogenic RAS Signaling Promotes Tumor Immunoresistance by Stabilizing PD-L1 mRNA.
Coelho MA, de Carné Trécesson S, Rana S, Zecchin D, Moore C, Molina-Arcas M, East P, Spencer-Dene B, Nye E, Barnouin K, Snijders AP, Lai WS, Blackshear PJ, Downward J
Immunity 2017 Dec 19;47(6):1083-1099.e6
Immunity 2017 Dec 19;47(6):1083-1099.e6
PD-L1/PD-1 expression and tumor-infiltrating lymphocytes in conjunctival melanoma.
Cao J, Brouwer NJ, Richards KE, Marinkovic M, van Duinen S, Hurkmans D, Verdegaal EME, Jordanova ES, Jager MJ
Oncotarget 2017 Aug 15;8(33):54722-54734
Oncotarget 2017 Aug 15;8(33):54722-54734
Expression of programmed death 1 ligand 1 on periodontal tissue cells as a possible protective feedback mechanism against periodontal tissue destruction.
Zhang J, Wang CM, Zhang P, Wang X, Chen J, Yang J, Lu W, Zhou W, Yuan W, Feng Y
Molecular medicine reports 2016 Mar;13(3):2423-30
Molecular medicine reports 2016 Mar;13(3):2423-30
CD28 family of receptors on T cells in chronic HBV infection: Expression characteristics, clinical significance and correlations with PD-1 blockade.
Tang ZS, Hao YH, Zhang EJ, Xu CL, Zhou Y, Zheng X, Yang DL
Molecular medicine reports 2016 Aug;14(2):1107-16
Molecular medicine reports 2016 Aug;14(2):1107-16
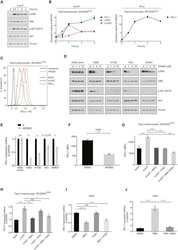
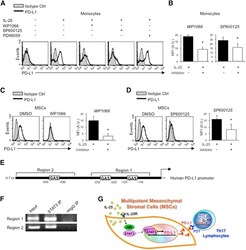
Interleukin-25 Mediates Transcriptional Control of PD-L1 via STAT3 in Multipotent Human Mesenchymal Stromal Cells (hMSCs) to Suppress Th17 Responses.
Wang WB, Yen ML, Liu KJ, Hsu PJ, Lin MH, Chen PM, Sudhir PR, Chen CH, Chen CH, Sytwu HK, Yen BL
Stem cell reports 2015 Sep 8;5(3):392-404
Stem cell reports 2015 Sep 8;5(3):392-404
Engineering high-affinity PD-1 variants for optimized immunotherapy and immuno-PET imaging.
Maute RL, Gordon SR, Mayer AT, McCracken MN, Natarajan A, Ring NG, Kimura R, Tsai JM, Manglik A, Kruse AC, Gambhir SS, Weissman IL, Ring AM
Proceedings of the National Academy of Sciences of the United States of America 2015 Nov 24;112(47):E6506-14
Proceedings of the National Academy of Sciences of the United States of America 2015 Nov 24;112(47):E6506-14
Cervical (pre)neoplastic microenvironment promotes the emergence of tolerogenic dendritic cells via RANKL secretion.
Demoulin SA, Somja J, Duray A, Guénin S, Roncarati P, Delvenne PO, Herfs MF, Hubert PM
Oncoimmunology 2015 Jun;4(6):e1008334
Oncoimmunology 2015 Jun;4(6):e1008334
PD-L1 expression in melanocytic lesions does not correlate with the BRAF V600E mutation.
Rodić N, Anders RA, Eshleman JR, Lin MT, Xu H, Kim JH, Beierl K, Chen S, Luber BS, Wang H, Topalian SL, Pardoll DM, Taube JM
Cancer immunology research 2015 Feb;3(2):110-5
Cancer immunology research 2015 Feb;3(2):110-5
Paucity of PD-L1 expression in prostate cancer: innate and adaptive immune resistance.
Martin AM, Nirschl TR, Nirschl CJ, Francica BJ, Kochel CM, van Bokhoven A, Meeker AK, Lucia MS, Anders RA, DeMarzo AM, Drake CG
Prostate cancer and prostatic diseases 2015 Dec;18(4):325-32
Prostate cancer and prostatic diseases 2015 Dec;18(4):325-32
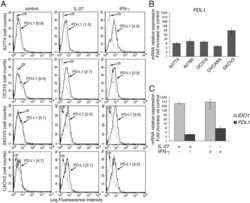
IL-27 induces the expression of IDO and PD-L1 in human cancer cells.
Carbotti G, Barisione G, Airoldi I, Mezzanzanica D, Bagnoli M, Ferrero S, Petretto A, Fabbi M, Ferrini S
Oncotarget 2015 Dec 22;6(41):43267-80
Oncotarget 2015 Dec 22;6(41):43267-80
Interferon beta and vitamin D synergize to induce immunoregulatory receptors on peripheral blood monocytes of multiple sclerosis patients.
Waschbisch A, Sanderson N, Krumbholz M, Vlad G, Theil D, Schwab S, Mäurer M, Derfuss T
PloS one 2014;9(12):e115488
PloS one 2014;9(12):e115488
Brugia malayi microfilariae induce a regulatory monocyte/macrophage phenotype that suppresses innate and adaptive immune responses.
O'Regan NL, Steinfelder S, Venugopal G, Rao GB, Lucius R, Srikantam A, Hartmann S
PLoS neglected tropical diseases 2014 Oct;8(10):e3206
PLoS neglected tropical diseases 2014 Oct;8(10):e3206
Inhibition of mitogen-activated protein kinase pathway can induce upregulation of human leukocyte antigen class I without PD-L1-upregulation in contrast to interferon-γ treatment.
Mimura K, Kua LF, Shiraishi K, Kee Siang L, Shabbir A, Komachi M, Suzuki Y, Nakano T, Yong WP, So J, Kono K
Cancer science 2014 Oct;105(10):1236-44
Cancer science 2014 Oct;105(10):1236-44
Metastasis is regulated via microRNA-200/ZEB1 axis control of tumour cell PD-L1 expression and intratumoral immunosuppression.
Chen L, Gibbons DL, Goswami S, Cortez MA, Ahn YH, Byers LA, Zhang X, Yi X, Dwyer D, Lin W, Diao L, Wang J, Roybal J, Patel M, Ungewiss C, Peng D, Antonia S, Mediavilla-Varela M, Robertson G, Suraokar M, Welsh JW, Erez B, Wistuba II, Chen L, Peng D, Wang S, Ullrich SE, Heymach JV, Kurie JM, Qin FX
Nature communications 2014 Oct 28;5:5241
Nature communications 2014 Oct 28;5:5241
Differential potency of regulatory T cell-mediated immunosuppression in kidney tumors compared to subcutaneous tumors.
Devaud C, Westwood JA, Teng MW, John LB, Yong CS, Duong CP, Smyth MJ, Darcy PK, Kershaw MH
Oncoimmunology 2014 Nov;3(11):e963395
Oncoimmunology 2014 Nov;3(11):e963395
Th1 cytokines are more effective than Th2 cytokines at licensing anti-tumour functions in CD40-activated human macrophages in vitro.
Luheshi N, Davies G, Poon E, Wiggins K, McCourt M, Legg J
European journal of immunology 2014 Jan;44(1):162-72
European journal of immunology 2014 Jan;44(1):162-72
Alterations of immune response of Non-Small Cell Lung Cancer with Azacytidine.
Wrangle J, Wang W, Koch A, Easwaran H, Mohammad HP, Vendetti F, Vancriekinge W, Demeyer T, Du Z, Parsana P, Rodgers K, Yen RW, Zahnow CA, Taube JM, Brahmer JR, Tykodi SS, Easton K, Carvajal RD, Jones PA, Laird PW, Weisenberger DJ, Tsai S, Juergens RA, Topalian SL, Rudin CM, Brock MV, Pardoll D, Baylin SB
Oncotarget 2013 Nov;4(11):2067-79
Oncotarget 2013 Nov;4(11):2067-79
Gliomas promote immunosuppression through induction of B7-H1 expression in tumor-associated macrophages.
Bloch O, Crane CA, Kaur R, Safaee M, Rutkowski MJ, Parsa AT
Clinical cancer research : an official journal of the American Association for Cancer Research 2013 Jun 15;19(12):3165-75
Clinical cancer research : an official journal of the American Association for Cancer Research 2013 Jun 15;19(12):3165-75
Multiple inhibitory ligands induce impaired T-cell immunologic synapse function in chronic lymphocytic leukemia that can be blocked with lenalidomide: establishing a reversible immune evasion mechanism in human cancer.
Ramsay AG, Clear AJ, Fatah R, Gribben JG
Blood 2012 Aug 16;120(7):1412-21
Blood 2012 Aug 16;120(7):1412-21
Upregulation of programmed death-1 on T cells and programmed death ligand-1 on monocytes in septic shock patients.
Zhang Y, Li J, Lou J, Zhou Y, Bo L, Zhu J, Zhu K, Wan X, Cai Z, Deng X
Critical care (London, England) 2011;15(1):R70
Critical care (London, England) 2011;15(1):R70
Phagocytosis, a potential mechanism for myeloid-derived suppressor cell regulation of CD8+ T cell function mediated through programmed cell death-1 and programmed cell death-1 ligand interaction.
Kim YJ, Park SJ, Broxmeyer HE
Journal of immunology (Baltimore, Md. : 1950) 2011 Sep 1;187(5):2291-301
Journal of immunology (Baltimore, Md. : 1950) 2011 Sep 1;187(5):2291-301
Programmed death ligand 1 is expressed by non-hodgkin lymphomas and inhibits the activity of tumor-associated T cells.
Andorsky DJ, Yamada RE, Said J, Pinkus GS, Betting DJ, Timmerman JM
Clinical cancer research : an official journal of the American Association for Cancer Research 2011 Jul 1;17(13):4232-44
Clinical cancer research : an official journal of the American Association for Cancer Research 2011 Jul 1;17(13):4232-44
Transfer of regulatory properties from tolerogenic to proinflammatory dendritic cells via induced autoreactive regulatory T cells.
Kleijwegt FS, Laban S, Duinkerken G, Joosten AM, Koeleman BP, Nikolic T, Roep BO
Journal of immunology (Baltimore, Md. : 1950) 2011 Dec 15;187(12):6357-64
Journal of immunology (Baltimore, Md. : 1950) 2011 Dec 15;187(12):6357-64
Ex vivo expanded hematopoietic stem cells overcome the MHC barrier in allogeneic transplantation.
Zheng J, Umikawa M, Zhang S, Huynh H, Silvany R, Chen BP, Chen L, Zhang CC
Cell stem cell 2011 Aug 5;9(2):119-30
Cell stem cell 2011 Aug 5;9(2):119-30
The PD-1/PD-L1 axis modulates the natural killer cell versus multiple myeloma effect: a therapeutic target for CT-011, a novel monoclonal anti-PD-1 antibody.
Benson DM Jr, Bakan CE, Mishra A, Hofmeister CC, Efebera Y, Becknell B, Baiocchi RA, Zhang J, Yu J, Smith MK, Greenfield CN, Porcu P, Devine SM, Rotem-Yehudar R, Lozanski G, Byrd JC, Caligiuri MA
Blood 2010 Sep 30;116(13):2286-94
Blood 2010 Sep 30;116(13):2286-94
Increase of programmed death-1-expressing intratumoral CD8 T cells predicts a poor prognosis for nasopharyngeal carcinoma.
Hsu MC, Hsiao JR, Chang KC, Wu YH, Su IJ, Jin YT, Chang Y
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2010 Oct;23(10):1393-403
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2010 Oct;23(10):1393-403
Novel role of plasmacytoid dendritic cells in humans: induction of interleukin-10-producing Treg cells by plasmacytoid dendritic cells in patients with rheumatoid arthritis responding to therapy.
Kavousanaki M, Makrigiannakis A, Boumpas D, Verginis P
Arthritis and rheumatism 2010 Jan;62(1):53-63
Arthritis and rheumatism 2010 Jan;62(1):53-63
Critical role for TNF in the induction of human antigen-specific regulatory T cells by tolerogenic dendritic cells.
Kleijwegt FS, Laban S, Duinkerken G, Joosten AM, Zaldumbide A, Nikolic T, Roep BO
Journal of immunology (Baltimore, Md. : 1950) 2010 Aug 1;185(3):1412-8
Journal of immunology (Baltimore, Md. : 1950) 2010 Aug 1;185(3):1412-8
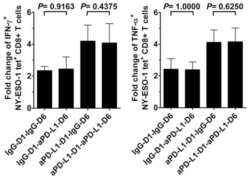
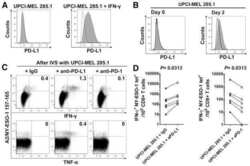
PD-1 is a regulator of NY-ESO-1-specific CD8+ T cell expansion in melanoma patients.
Fourcade J, Kudela P, Sun Z, Shen H, Land SR, Lenzner D, Guillaume P, Luescher IF, Sander C, Ferrone S, Kirkwood JM, Zarour HM
Journal of immunology (Baltimore, Md. : 1950) 2009 May 1;182(9):5240-9
Journal of immunology (Baltimore, Md. : 1950) 2009 May 1;182(9):5240-9
Neuron-interacting satellite glial cells in human trigeminal ganglia have an APC phenotype.
van Velzen M, Laman JD, Kleinjan A, Poot A, Osterhaus AD, Verjans GM
Journal of immunology (Baltimore, Md. : 1950) 2009 Aug 15;183(4):2456-61
Journal of immunology (Baltimore, Md. : 1950) 2009 Aug 15;183(4):2456-61
Expression and function of PDCD1 at the human maternal-fetal interface.
Taglauer ES, Trikhacheva AS, Slusser JG, Petroff MG
Biology of reproduction 2008 Sep;79(3):562-9
Biology of reproduction 2008 Sep;79(3):562-9
TLR-mediated induction of negative regulatory ligands on dendritic cells.
Gröschel S, Piggott KD, Vaglio A, Ma-Krupa W, Singh K, Goronzy JJ, Weyand CM
Journal of molecular medicine (Berlin, Germany) 2008 Apr;86(4):443-55
Journal of molecular medicine (Berlin, Germany) 2008 Apr;86(4):443-55
Expression of B7-H1 in inflammatory renal tubular epithelial cells.
Chen Y, Zhang J, Li J, Zou L, Zhao T, Tang Y, Wu Y
Nephron. Experimental nephrology 2006;102(3-4):e81-92
Nephron. Experimental nephrology 2006;102(3-4):e81-92
Clinical significance of programmed death-1 ligand-1 and programmed death-1 ligand-2 expression in human esophageal cancer.
Ohigashi Y, Sho M, Yamada Y, Tsurui Y, Hamada K, Ikeda N, Mizuno T, Yoriki R, Kashizuka H, Yane K, Tsushima F, Otsuki N, Yagita H, Azuma M, Nakajima Y
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Apr 15;11(8):2947-53
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Apr 15;11(8):2947-53
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
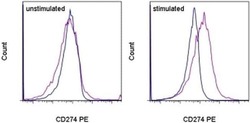
- Experimental details
- Staining of unstimulated (left) or PHA-stimulated (right) normal human peripheral blood cells with Mouse IgG1 K Isotype Control PE (Product # 12-4714-81) (blue histogram) or Anti-Human CD274 (B7-H1) PE (purple histogram). Total viable cells were used for analysis.
- Conjugate
- Yellow dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
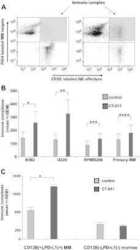
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
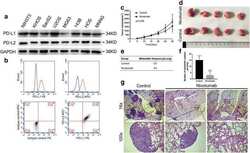
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
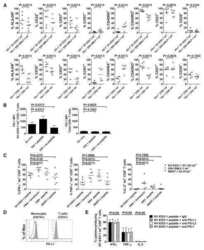
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
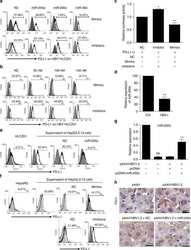
- Figure 6 IL-25-Mediated PD-L1 Expression in Human Monocytes and hMSCs Is Mediated through JNK and STAT3, with STAT3 Involved in Transcriptional Control of PD-L1 (A) Human PBLs were pretreated with inhibitors of STAT3 (WP1066; 2.5 muM), JNK (SP600125; 25 muM), or MEK1 (PD98059; 20 muM) prior to 100 ng/ml rhIL-25 for 18 hr, with subsequent flow cytometric analysis for PD-L1 surface expression on monocytes, gated using FSC and SSC. Filled histograms represent isotype control; unfilled histograms represent PD-L1 antibody staining. (B-D) Pooled data (three donors) are shown (B) with bars representing MFI. hMSCs were treated with inhibitors of STAT3 (C; WP1066, 2.5 muM) and JNK (D; SP600125, 25 muM) for 6 hr, and subsequently assessed by flow cytometric analysis for PD-L1 surface expression. Pooled data (all three donors) for each respective inhibitor are provided (left charts) with bars representing MFI. (E) Putative GAS elements (STAT-binding sites) in the proximal promoter region of human PD-L1 gene (700 bp region upstream from the transcription start site), as determined with TFSearch web-based software. (F) Binding of STAT3 or IgG (negative control) in hMSCs was analyzed by chromatin immunoprecipitation (ChIP) with promoter-specific primers for region 1 and region 2. The input samples (positive control) represent 1% starting chromatin. (G) Schematic shows a model of hMSC-mediated suppression of Th17 responses involving the IL-25/STAT3/PD-L1 axis.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
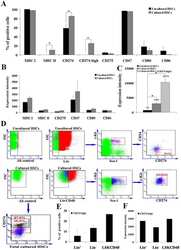
- Figure 2 IL-27 increases PD-L1 surface protein and mRNA expression in EOC cells in vitro A. FACS analysis of surface PD-L1 expression in four EOC cell lines, cultured in the presence of medium (control), IL-27 or IFN-gamma. Dotted lines are isotype-matched unrelated Ig staining controls. Numbers in brackets are Median Fluorescence Intensity (MFI) values calculated as median PD-L1 minus median Ig control. Data are representative of two independent experiments showing similar results. B. QRT-PCR analysis of PDL1 mRNA expression in five IL-27-stimulated EOC cells relative to untreated cells. Data are the mean (+-SD) of three independent experiments. C. Comparative analysis of IDO1 and PDL1 mRNA up-regulation by IL-27 or IFN-gamma in a representative EOC cell line (CAOV3). Data are the mean of two independent replicates and are expressed as DeltaDeltaCT-fold change. Error bars represent the minimum and maximum.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 1 Mesenchymal stromal cell (MSC) cell surface expression and secretion of PD-L1 and PD-L2 are potentiated by pro-inflammatory cytokines, IFNgamma and TNFalpha. MSCs ( n = 4) were exposed to 100 U/ml IFNgamma and 10 ng/ml TNFalpha for 3 days in culture. Cell surface expression (MFI) of (A) PD-L1 and (B) PD-L2 was assessed by flow cytometry. Secretion of (C) soluble (s)PD-L1 and (D) sPD-L2 within the conditioned media of stimulated cells was assessed by ELISA. Bar charts indicate mean +- SEM. Transcriptional regulation of (E) PD-L1 and (F) PD-L2 were assessed by qRT-PCR. mRNA data are expressed as fold change compared to unstimulated, resting MSCs +- SEM. *, p < .05; **, p < .01. Abbreviations: IFNgamma, Interferon gamma; MFI, mean fluorescence intensity; PD-L1 and PD-L2, programmed death 1 ligands 1 and 2; TNFalpha, tumor necrosis factor alpha.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
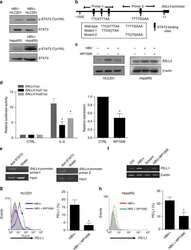
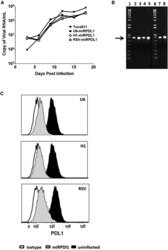
- Figure 4 RRV-RSV-miRPDL1 and RRV-H1-miRPDL1 Exhibit Comparable PDL1 Downregulation Activity as RRV-miRPDL1 (A) Replication kinetics of RRV-RSV-miRPDL1 and RRV-H1-miRPDL1. The viral genome in the supernatants of infected LN-18 cells (MOI of 0.1) at indicated time points were quantified by qRT-PCR using primer set targeted to the env region ( Figure 1 ). RRV-yCD2 and RRV-miRPDL1 (indicated as U6-miRPDL1 in the graph) were included as positive controls. (B) Vector stability of RRV-RSV-miRPDL1 and RRV-H1-miRPDL1 in LN-18 cells was analyzed by endpoint PCR at 14 and 30 days post infection. Lanes 1 and 6: DNA molecular marker (1 Kb Plus marker, Invitrogen); lanes 2, 4, and 7 are positive controls using the corresponding plasmid DNA as the templates; lane 3: RRV-H1-miRPDL1; lane 5: RRV-RSV-miRPDL1; and lane 8: RRV-miRPDL1. The arrow indicates the expected size of the PCR products. (C) LN-18 cells infected with RRV-miRPDL1 (U6), RRV-H1-miRPDL1 (H1), and RRV-RSV-miRPDL1 (RSV) were stained for PDL1 cell surface expression with PDL1 antibody and analyzed by flow cytometry.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
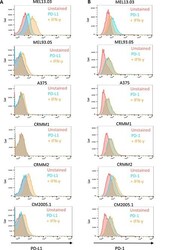
- Figure 6 Cutaneous (MEL13.03, MEL93.05 and A375) and conjunctival melanoma (CRMM1, CRMM2 and CM2005.1) cell lines express various levels of PD-L1 and PD-1 MEL13.03 is the positive control cell line for both PD-L1 and PD-1. Representative histograms show ( A ) PD-L1 and PD-1 ( B ) expression in cell lines with or without IFN-gamma (100 IU/ml) exposure for 48 h. Pink, blue and brown shaded histograms represent unstained, PD-L1 (PD-1) staining, and the effect of IFN-gamma stimulation on PD-L1 and PD-1, respectively.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
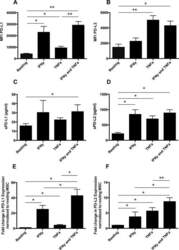
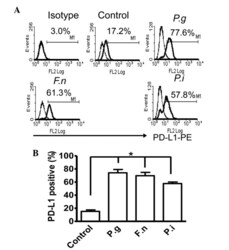
- Figure 2 Common periodontal pathogens induce the expression of PD-L1 on PDLCs. (A) Flow cytometry histogram overlays of PDLCs co-cultured with P.g , F.n and P.i . (B) Comparison of expression levels of PD-L1 induced by P.g, F.n and P.i. Data are expressed as the mean +- standard error of the mean of three independent experiments. All three strains significantly increased the expression of PD-L1 on the PDLCs, * P
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
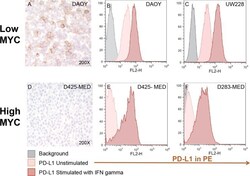
- Figure 4 Medulloblastoma cell lines express PD-L1 in a subgroup dependent manner PD-L1 expression in low and high MYC MB cell lines. Paraffin embedded cell line pellets stained for PD-L1 in DAOY ( A ) and D425-MED ( D ). Histograms of PD-L1 expression by flow cytometry using phycoerythrin (PE) conjugated MIH1 clone of PD-L1 (EBioscience) with and without IFN-gamma stimulation in DAOY ( B ), UW228 ( C ), D425-MED ( E ), and D283-MED ( F ).
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 MYC overexpression in DAOY (YM21) does not alter PD-L1 expression Dot Plots of PD-L1 expression by flow cytometry using PE-conjugated MIH1 clone of PD-L1 (EBioscience) in YM21 construct made by overexpressing MYC in DAOY via stable lentiviral transfection.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
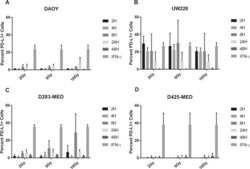
- Figure 6 Radiation induces PD-L1 in most medulloblastoma cell lines Bar graph depicts percent PD-L1 positive cells above baseline as determined by flow cytometry using PE-conjugated MIH1 clone of PD-L1 (EBioscience) in DAOY ( A ), UW228 ( B ), D283-MED ( C ), and D425-MED ( D ) after irradiation with 2, 5, or 10 Gy at 2, 4, 8, 24, and 48 hours. IFN-gamma values were obtained without radiation. Each bar graph depicts findings from 2 different experiments where all data points were repeated. Error bars represent standard deviation of the mean. Levels of PD-L1 that were comparable to those induced by IFN-gamma included UW228 hours 2-24 at 2, 5 and 10 Gy and D283-MED at hour 8, 10 Gy. All other values were significantly less than those induced by IFN-gamma as determined by 2-way ANOVA corrected for multiple comparisons. Full statistical analysis available in Supplementary Table 1-1-1-4 .
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
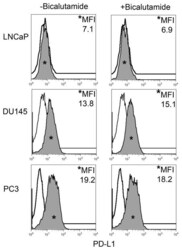
- FIGURE 1 Human Prostate Cancer Cell Lines Display Varying Expression of PD-L1 in Response to IFN-gamma Histograms representing PD-L1 surface expression as detected by flow cytometry in human prostate cancer cell lines with and without exposure to IFN-gamma. Open histograms represent cells stained with an isotype IgG1 antibody tagged with a matching PE fluorochrome. Shaded histograms represent PD-L1. Quantitative measurements reflect mean fluorescence index. LNCaP does not express PD-L1 at rest or in response to IFN-gamma. DU145 expresses PD-L1 at rest and has little response to IFN-gamma indicating an innate immune resistance phenotype. PC3 expresses PD-L1 at rest but robustly up-regulates this expression in response to IFN-gamma displaying a more classic adaptive immune resistance phenotype.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- FIGURE 2 Human Prostate Cancer Cell Lines Display No Change in PD-L1 Expression in Response to Bicalutamide Histograms representing PD-L1 surface expression as detected by flow cytometry in human prostate cancer cell lines with and without exposure to bicalutamide. Open histograms represent cells stained with an isotype IgG1 antibody tagged with a matching PE fluorochrome. Shaded histograms represent PD-L1. Quantitative measurements reflect mean fluorescence index. LNCaP, DU145, and PC3 displayed no changes in PD-L1 expression upon exposure to bicalutamide.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
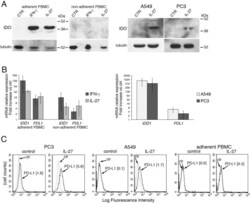
- Figure 7 IL-27 induces PD-L1 and/or IDO expression in human PC3 prostate and A549 lung cancer cells and adherent PBMC A. Western blot analysis of IDO expression in human adherent PBMC, non-adherent PBMC and in PC3 and A549 cells treated with the indicated cytokines or medium only for 48 hours. alpha-tubulin is used as loading control. Similar results were observed in two additional experiments. B. QRT-PCR analysis of IDO1 and PDL1 mRNA expression in cytokine-stimulated adherent or non-adherent PBMC, PC3 and A549 cells relative to untreated cells. Data are expressed as DeltaDeltaCT-fold change. Mean values of three independent experiments. Error bars represent SD. C. FACS analysis of surface PD-L1 in IL-27-treated or untreated PC3 and A549 cell lines and adherent PBMC. Dotted lines are isotype-matched unrelated Ig staining controls. Similar results were obtained in five different experiments (PC3: MFI 5.5 +- 2.7 vs 1.5 +- 0.6, mean +- SD in IL-27-treated vs un-stimulated cells, P = 0.03 by paired Student's t test; A549: MFI 2 +- 1 vs 0.43 +- 0.4, P = 0.01).
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
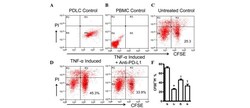
- Figure 4 Expression of PD-L1 on PDLCs improves survival of PDLCs. Flow cytometry histrograms of (A) PDLCs, (B) PHA-activated PBMCs, (C) PDLCs co-cultured with activated PBMCs, (D) PDLCs pretreated with TNF-alpha and co-cultured with activated PBMCs, and (E) PDLCs pretreated with TNF-alpha, and incubated with activated PBMCs and anti-PD-L1 antibodies. (F) Comparison of PDLC survival, according to the percentages of CFSE + /PI - cells. a, c, d and e represent the PDLC control, untreated control, TNF-alpha induced and TNF-alpha induced+anti-PD-L1 groups, respectively. Data are expressed as the mean +- standard error of the mean of three independent experiments. Co-culturing the activated PMBCs with untreated PDLCs resulted in a significant decrease in viable PDLCs ( * P
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
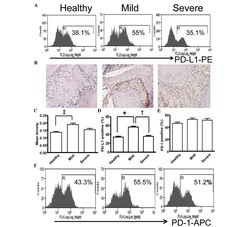
- Figure 5 Expression of PD-L1 is correlated with the severity of periodontitis in the experimental periodontitis model. (A) Flow cytometry histograms of the expression of PD-L1 on the surface of periodontal tissue cells from healthy mice, and mice with mild periodontitis and severe periodontitis. (B) Expression of PD-L1 in periodontal tissues from the three groups, detected by immunohistochemical staining (magnification, x400). (C) Comparison of the expression of PDL-1 in periodontal tissues between the three groups ( ++ P
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
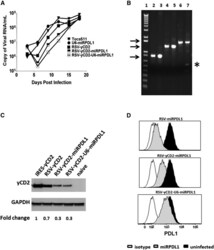
- Figure 5 RRV-RSV-yCD2-miRPDL1 and RRV-RSV-yCD2-U6-miRPDL1 Express yCD2 Protein and Exhibit PDL1 Downregulation Activity (A) Replication kinetics of RRV-RSV-yCD2-miRPDL1 and RRV-RSV-yCD2-U6-miRPDL1. The viral genome in the supernatants of infected LN-18 cells (MOI of 0.1) at indicated time points were quantified by qRT-PCR using primer set targeted to the env region ( Figure 1 ). A paired t test was performed and showed no statistically significant difference in replication kinetics between RRV-RSV-yCD2-miRPDL1 versus RRV-RSV-yCD2 (p = 0.0649) and RRV-RSV-yCD2-U6-miRPDL1 (p = 0.0801). RRV-yCD2, RRV-RSV-yCD2, and RRV-miRPDL1 (indicated as U6-miRPDL1 in the graph) were included as positive controls. (B) Vector stability of RRV-RSV-yCD2-miRPDL1 and RRV-RSV-yCD2-U6-miRPDL1 in LN-18 cells was analyzed by endpoint PCR at 14 days post infection. Lane 1: DNA molecular marker (1 Kb Plus marker, Invitrogen); lanes 2, 4, and 6 are positive controls using the corresponding plasmid DNA as the templates; lane 3: RRV-RSV-miRPDL1; lane 5: RRV-RSV-yCD2-miRPDL1; and lane 7: RRV-RSV-yCD2-U6-miRPDL1. The arrows indicate the expected size of the PCR products (844 bp for RRV-RSV-miRPDL1; 1,326 bp for RRV-RSV-yCD2-miRPDL1; and 1,591 bp for RRV-RSV-yCD2-U6-miRPDL1). (C) yCD2 protein expression in LN-18 cell infected with RRV-yCD2, RRV-RSV-yCD2, RRV-RSV-yCD2-miRPDL1, RRV-RSV-yCD2-U6-miRPDL1, and naive cells. GAPDH is included as loading control. The numbers shown on the bottom of the immunoblot indica
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
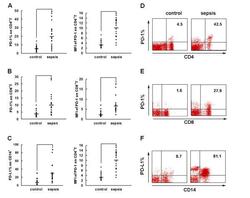
- Figure 2 PD-1 and PD-L1 were upregulated on T cells and monocytes in septic shock patients . Blood samples were obtained from 19 septic shock patients and 22 healthy controls and were stained for programmed death-1 (PD-1) and programmed death ligand-1 (PD-L1) gated on CD4 + T cells, CD8 + T cells, and CD14 + monocytes. (a) to (c) Percentage of PD-1 expression on (a) CD4 + T cells and (b) CD8 + T cells, and (c) percentage of PD-L1 expression on CD14 + monocytes. Each dot represents one individual. Data are mean +- standard error of the mean (SEM) of three independent experiments. ** P < 0.01 compared with healthy controls. (d) to (f) Mean fluorescence intensity (relative fluorescence units) of PD-1 expression on (d) CD4 + T cells, (e) PD-1 expression on CD8 + T cells, and (f) PD-L1 expression on CD14 + monocytes Each dot represents one individual. Data are mean +- SEM of three independent experiments. * P < 0.05 compared with healthy controls. (g) Representative PD-1 expression levels on CD4 + T cells and CD8 + T cells, and PD-L1 expression on CD14 + monocytes. Values in the upper-right quadrant indicate the percentage of cells that express PD-1 or PD-L1.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
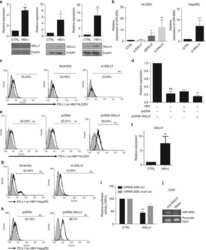
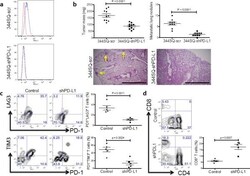
- Figure 5 Genetic targeting of PD-L1 expression on cancer cells reverses the CD8 + TIL dysfunction and suppresses metastasis ( a ) Cell surface expression of PD-L1 on 344SQ PD-L1 knockdown (344SQ-shPD-L1) vs 344SQ scramble control (344SQ-scr) cells by FACS (red line, isotype control staining; blue line, anti-PD-L1 staining). The measurement was independently repeated at least three times. ( b ) Primary tumor mass (top left) and lung metastases (top right) in 129/Sv mice (n = 10) injected subcutaneously with 344SQ-shPD-L1 or 344SQ-scr cancer cells. Micrometastases (bottom) observed in hematoxylin and eosin-stained lung tissue sections are indicated by yellow arrows. Scale bar, 2mm. Samples were obtained 6 weeks post-injection. The data from two independent experiments were pooled. Data are shown as mean +- s.e.m. t -test was used to analyze. P values are shown in the graphs. ( c , d ) FACS analysis of ( c ) surface PD1, LAG3, and TIM3 marker expression levels on CD8 + T cells; ( d ) CD8 + TIL frequency for primary tumors in 129/Sv mice (n = 5) injected subcutaneously with 344SQ-shPD-L1 (shPD-L1) or 344SQ-scr control (Control) cancer cells and necropsied 2 weeks later. The analyses were independently repeated three times. Data are shown as mean +- s.e.m. t -test was used to analyze. P values are shown in the graphs.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 PD-L1 expression on tumor cells is critical to the repression of anti-tumor immunity ( a ) The knockdown (KD) efficiency of PD-L1 in LLC-JSP murine lung cancer cells measured by FACS. Representative histograms (left), and statistical analysis (right). The measurement was independently repeated at least three times. ( b ) Representative FACS histogram of PD-L1 expression on myeloid cells (CD11b + ) in PD-L1 KO or WT mice (n = 3). ( c ) Tumor growth in PD-L1 KO or WT mice (n = 6) of subcutaneously injected LLC-JSP cells (10, 000 cells with 100 mul of PBS per mouse) with differing PD-L1 knockdown (Vector, PD-L1 KD vector control; PD-L1 int , PD-L1 intermediate KD; PD-L1 hi , PD-L1 high level KD). The data is shown from two independent experiments. Data are shown as mean +- s.e.m. ( d ) FACS analysis of CD8 + TIL frequency and T cell exhaustion marker expression levels on CD8 + T cells in subcutaneous primary tumors of LLC-JSP vector control (Vector ctrl) and LLC-JSP intermediate PD-L1 knockdown (Intermediate KD) in PD-L1 WT (WT) and PD-L1 KO (KO) mice (n = 5, from two independent experiments). Analysis was done 3 weeks post-tumor cell injection (20, 000 cells with 100 mul of PBS per mouse). t -test was used to analyze the data. p < 0.0001. ( e ) PD-L1 expression levels on LLC-JSP-shPD-L1 cells (the high knockdown efficiency cells) after reconstitution of PD-L1. The measurement was independently repeated at least three times. Control, LLC-JSP-shPD-L1 + vector control; PD
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
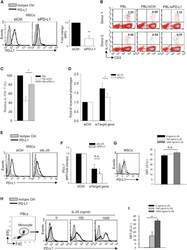
- Figure 5 IL-25 Induces PD-L1 Surface Expression on hMSCs and Human Monocytes (A) PD-L1 in siCtrl MSCs (left) and siPD-L1 MSCs (right) was analyzed by surface staining. (B) Freshly isolated human PBLs were co-cultured without (left) or with siCtrl MSCs (middle) or siPD-L1 MSCs (right) for 3 days, followed by PMA/ionomycin stimulation for 6 hr. IL-17A production in CD3 + T cells was assessed by intracellular staining. Representative data are shown with numbers in the top right quadrants representing the percentages of IL-17A-producing CD3 + T cells. (C) Pooled data from PBLs (n = 4) and two hMSC donors (donors A and B) are shown. (D) Folds of reversed phenotypes of siIL-25 and siPD-L1 are shown. (E) PD-L1 expression on siCtrl hMSCs (left) and siIL-25 hMSCs (right) was assessed by cell surface staining. Filled histograms represent isotype control; unfilled histograms represent PD-L1 antibody staining. (F) Pooled data of PD-L1 expression (indicated by fold change in MFI) on siIL-25 hMSCs and siPD-L1 hMSCs (all three donors) are shown. PD-L1 expression levels were compared between hMSCs silenced for the target gene (IL-25 or PD-L1) and the respective siCtrl. (G) hMSCs were treated with the indicated doses of rhIL-25 for 18 hr and assessed for cell surface PD-L1 expression by cell surface staining. Pooled data (all three donors) are shown in chart to the right with bars representing MFI. (H) Human PBLs were treated with the indicated doses of rhIL-25 for 18 hr and assessed for cell
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
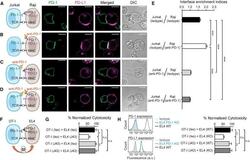
- Figure 7 Blockade of APC-Intrinsic PD-1 Enhances the Synaptic Enrichment of T Cell-Intrinsic PD-1 and Inhibits T Cell-Mediated Cytotoxicity (A) Left: cartoon showing a Jurkat cell expressing PD-1-mGFP (shown as PD-1 in green) conjugated with a Raji cell co-transduced with PD-L1-mCherry and unlabeled PD-1. Right: representative confocal images of the conjugate at the indicated channel acquired 2 min after cell-cell contact. Scale bars, 10 um. (B-D) Same as (A), except that Raji (PD-L1+/PD-1+) cells (B), Jurkat (PD-1-mGFP) cells (C), or both (D) were preincubated with pembrolizumab and washed extensively prior to conjugation. Scale bars, 10 um. (E) Bar graph comparing the interface enrichment indices (calculated as described in STAR Methods) of the four conditions shown in (A)-(D). Data are expressed as mean +- SEM; n = 40 cells from three independent experiments. (F) Cartoon illustrating the cytotoxicity assay, in which EL4 cells were used as the target for OT-I cytotoxic T cells. (G) Bar graph summarizing the OT-I cytotoxicity under indicated conditions, with PD-1 blockade antibody J43 or isotype antibody (Iso) preincubated with neither cell, only EL4, only OT-I, or both cell types. See STAR Methods for details. Cytotoxicity was normalized to the no blockade antibody condition (i.e., EL4 and OT-I preincubated with the isotype control). n.s., not significantly different. Data are presented as mean +- SEM from three independent replicates. (H) Left: FACS histograms showing PD-1
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
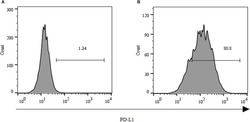
- FIGURE 2 Expression of PD-L1 on human HCC PLC/PRF/5 cells. (A) Human HCC PLC/PRF/5 cells were cultured alone in the absence of GPC3-CAR T cells in RPMI 1640 medium containing 10% FBS. (B) Human HCC PLC/PRF/5 cells were cocultured with GPC3-CAR T cells at an effector:Target ratio of 1:1 for 18 h in RPMI 1640 medium containing 10% FBS. PD-L1 was determined by flow cytometry in the CD3-negative gate, and the fixable, viable stain 780 was used for discriminating live from dead cells.
- Conjugate
- Yellow dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry