Antibody data
- Antibody Data
- Antigen structure
- References [9]
- Comments [0]
- Validations
- Immunohistochemistry [1]
- Flow cytometry [2]
- Other assay [7]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA5-14264 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- TMPRSS2 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 400 μL
- Concentration
- 2.0 mg/mL
- Storage
- Store at 4°C short term. For long term storage, store at -20°C, avoiding freeze/thaw cycles.
Submitted references Highly restricted SARS-CoV-2 receptor expression and resistance to infection by primary human monocytes and monocyte-derived macrophages.
Peptidomimetic inhibitors of TMPRSS2 block SARS-CoV-2 infection in cell culture.
Antiviral Potential of the Antimicrobial Drug Atovaquone against SARS-CoV-2 and Emerging Variants of Concern.
Human Erythroid Progenitors Are Directly Infected by SARS-CoV-2: Implications for Emerging Erythropoiesis in Severe COVID-19 Patients.
Alpha-1 antitrypsin inhibits TMPRSS2 protease activity and SARS-CoV-2 infection.
A high-throughput screen for TMPRSS2 expression identifies FDA-approved compounds that can limit SARS-CoV-2 entry.
Expression of SARS-CoV-2-related receptors in cells of the neurovascular unit: implications for HIV-1 infection.
Development and validation of IMMUNO-COV™: a high-throughput clinical assay for detecting antibodies that neutralize SARS-CoV-2.
Infection of bat and human intestinal organoids by SARS-CoV-2.
Zankharia U, Yadav A, Yi Y, Hahn BH, Collman RG
Journal of leukocyte biology 2022 Sep;112(3):569-576
Journal of leukocyte biology 2022 Sep;112(3):569-576
Peptidomimetic inhibitors of TMPRSS2 block SARS-CoV-2 infection in cell culture.
Wettstein L, Knaff PM, Kersten C, Müller P, Weil T, Conzelmann C, Müller JA, Brückner M, Hoffmann M, Pöhlmann S, Schirmeister T, Landfester K, Münch J, Mailänder V
Communications biology 2022 Jul 8;5(1):681
Communications biology 2022 Jul 8;5(1):681
Antiviral Potential of the Antimicrobial Drug Atovaquone against SARS-CoV-2 and Emerging Variants of Concern.
Carter-Timofte ME, Arulanandam R, Kurmasheva N, Fu K, Laroche G, Taha Z, van der Horst D, Cassin L, van der Sluis RM, Palermo E, Di Carlo D, Jacobs D, Maznyi G, Azad T, Singaravelu R, Ren F, Hansen AL, Idorn M, Holm CK, Jakobsen MR, van Grevenynghe J, Hiscott J, Paludan SR, Bell JC, Seguin J, Sabourin LA, Côté M, Diallo JS, Alain T, Olagnier D
ACS infectious diseases 2021 Nov 12;7(11):3034-3051
ACS infectious diseases 2021 Nov 12;7(11):3034-3051
Human Erythroid Progenitors Are Directly Infected by SARS-CoV-2: Implications for Emerging Erythropoiesis in Severe COVID-19 Patients.
Huerga Encabo H, Grey W, Garcia-Albornoz M, Wood H, Ulferts R, Aramburu IV, Kulasekararaj AG, Mufti G, Papayannopoulos V, Beale R, Bonnet D
Stem cell reports 2021 Mar 9;16(3):428-436
Stem cell reports 2021 Mar 9;16(3):428-436
Alpha-1 antitrypsin inhibits TMPRSS2 protease activity and SARS-CoV-2 infection.
Wettstein L, Weil T, Conzelmann C, Müller JA, Groß R, Hirschenberger M, Seidel A, Klute S, Zech F, Prelli Bozzo C, Preising N, Fois G, Lochbaum R, Knaff PM, Mailänder V, Ständker L, Thal DR, Schumann C, Stenger S, Kleger A, Lochnit G, Mayer B, Ruiz-Blanco YB, Hoffmann M, Sparrer KMJ, Pöhlmann S, Sanchez-Garcia E, Kirchhoff F, Frick M, Münch J
Nature communications 2021 Mar 19;12(1):1726
Nature communications 2021 Mar 19;12(1):1726
A high-throughput screen for TMPRSS2 expression identifies FDA-approved compounds that can limit SARS-CoV-2 entry.
Chen Y, Lear TB, Evankovich JW, Larsen MB, Lin B, Alfaras I, Kennerdell JR, Salminen L, Camarco DP, Lockwood KC, Tuncer F, Liu J, Myerburg MM, McDyer JF, Liu Y, Finkel T, Chen BB
Nature communications 2021 Jun 23;12(1):3907
Nature communications 2021 Jun 23;12(1):3907
Expression of SARS-CoV-2-related receptors in cells of the neurovascular unit: implications for HIV-1 infection.
Torices S, Cabrera R, Stangis M, Naranjo O, Fattakhov N, Teglas T, Adesse D, Toborek M
Journal of neuroinflammation 2021 Jul 29;18(1):167
Journal of neuroinflammation 2021 Jul 29;18(1):167
Development and validation of IMMUNO-COV™: a high-throughput clinical assay for detecting antibodies that neutralize SARS-CoV-2.
Vandergaast R, Carey T, Reiter S, Lech P, Gnanadurai C, Tesfay M, Buehler J, Suksanpaisan L, Naik S, Brunton B, Recker J, Haselton M, Ziegler C, Roesler A, Mills JR, Theel E, Weaver SC, Rafael G, Roforth MM, Jerde C, Tran S, Diaz RM, Bexon A, Baum A, Kyratsous CA, Peng KW, Russell SJ
bioRxiv : the preprint server for biology 2020 May 27;
bioRxiv : the preprint server for biology 2020 May 27;
Infection of bat and human intestinal organoids by SARS-CoV-2.
Zhou J, Li C, Liu X, Chiu MC, Zhao X, Wang D, Wei Y, Lee A, Zhang AJ, Chu H, Cai JP, Yip CC, Chan IH, Wong KK, Tsang OT, Chan KH, Chan JF, To KK, Chen H, Yuen KY
Nature medicine 2020 Jul;26(7):1077-1083
Nature medicine 2020 Jul;26(7):1077-1083
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
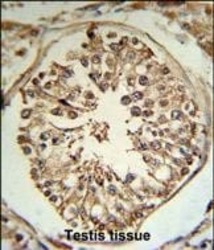
- Experimental details
- Immunohistochemistry analysis of TMPRSS2 in formalin fixed and paraffin embedded human testis tissue. Samples were incubated with TMPRSS2 polyclonal antibody (Product # PA5-14264) followed by peroxidase conjugation of the secondary antibody and DAB staining. This data demonstrates the use of this antibody for immunohistochemistry. Clinical relevance has not been evaluated.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
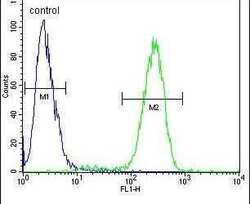
- Experimental details
- Flow cytometry analysis of 293 cells using a TMPRSS2 polyclonal antibody (Product # PA5-14264) (right) compared to a negative control cell (left) at a dilution of 1:10-50, followed by a FITC-conjugated goat anti-rabbit antibody
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
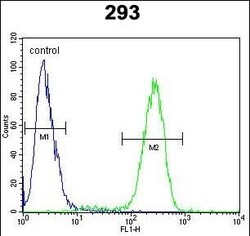
- Experimental details
- Flow cytometry of TMPRSS2 in 293 cells (right histogram). Samples were incubated with TMPRSS2 polyclonal antibody (Product # PA5-14264) followed by FITC-conjugated goat-anti-rabbit secondary antibody. Negative control cell (left histogram).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
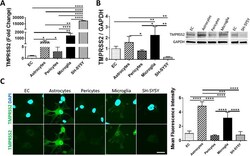
- Experimental details
- Fig. 2 Expression of TMPRSS2 in cells of the NVU. Expression levels of TMPRSS2 were measured by q-PCR ( A ), immunoblotting ( B ), and immunostaining ( C ). GAPDH was used as a housekeeping gene and loading control. Graphs indicate the mean +- SD from three independent experiments. **** p < 0.0001, *** p = 0.0002, ** p = 0.003, * p < 0.0449, n = 3-6 per group; scale bars, 30 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
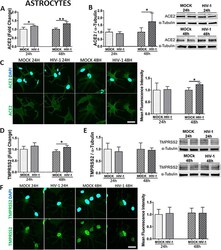
- Experimental details
- Fig. 5 Impact of HIV-1 infection on ACE2 and TMPRSS2 expression in astrocytes. Human primary astrocytes were either mock-infected or infected with HIV-1 with 60 ng/mL HIV-1 p24 for 24 h or 48 h and the expression levels of ACE2 and TMPRSS2 were measured by q-PCR ( A and D , respectively), immunoblotting ( B and E , respectively), and immunostaining ( C and F , respectively). GAPDH was used as a housekeeping gene and alpha-tubulin as a loading control. Graphs indicate the mean +- SD from three independent experiments. ** p = 0.003, * p < 0.0449, n = 4-5 per group; scale bars, 30 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
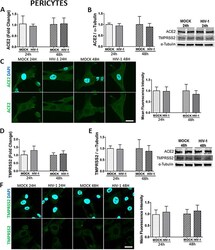
- Experimental details
- Fig. 6 Impact of HIV-1 infection on ACE2 and TMPRSS2 expression in pericytes. Human primary pericytes were either mock-infected or infected with HIV-1 as in Fig. 5 and the expression levels of ACE2 and TMPRSS2 were measured by q-PCR ( A and D , respectively), immunoblotting (B and E , respectively), and immunostaining ( C and F , respectively). GAPDH was used as a housekeeping gene and alpha-tubulin as a loading control. Graphs indicate the mean +- SD from three independent experiments; n = 4 per group; scale bars, 30 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
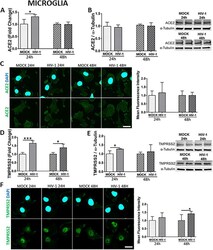
- Experimental details
- Fig. 7 Impact of HIV-1 infection on ACE2 and TMPRSS2 expression in microglial cells. Microglia were either mock-infected or infected with HIV-1 as in Fig. 5 and the expression levels of ACE2 and TMPRSS2 were measured by q-PCR ( A and D , respectively), immunoblotting ( B and E , respectively), and immunostaining ( C and F , respectively). GAPDH was used as a housekeeping gene and alpha-tubulin as a loading control. Graphs indicate the mean +- SD from three independent experiments. *** p = 0.0002, * p < 0.0449, n = 4 per group; scale bars, 30 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
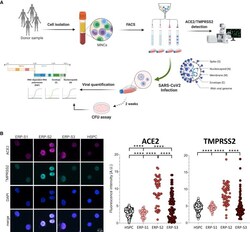
- Experimental details
- Figure 2 Isolation and Analysis of Erythroid Progenitors co-expressing ACE2 and TMPRSS2 (A) Schematic of sample processing: human bone marrow or peripheral blood were processed to isolate by cell sorting ERP populations. ACE2 and TMPRSS2 at the cell membrane was analyzed by confocal microscopy. SARS-CoV-2 infectivity was quantified by the detection of three viral genes (N, Nucleocapsid; E, Envelope; and RdRP, RNA-dependent Polymerase) by real-time qPCR. Created with BioRender. (B) Immunostaining and fluorescence intensity quantification of ACE2 and TMPRSS2 in HSPCs and erythroid progenitors (ERPs) (scale bar, 10 mum). Each dot represents one cell; HSPCs (351 cells), ERP-S1 (50 cells), ERP-S2 (48 cells), ERP-S3 (106 cells). This is representative of one of the two independent immunostainings performed. Two-way ANOVA test was used for the comparison among the different cell populations; **** p < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
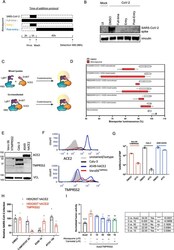
- Figure 6 Atovaquone partially requires TMPRSS2 to drive its antiviral action against SARS-CoV-2 and reduces the interaction between the spike protein and its surface receptor ACE2. (A) Schematic of atovaquone administration. (B) VeroE6 hTMPRSS2 cells were treated with atovaquone (10 muM) for 2 h before infection (full time), at the time of infection (entry), or 1 h after infection (postentry), before challenging with original SARS-CoV-2 at an MOI of 0.1. Infection was carried out for 48 h in the presence of the drug and for all conditions. Infection was assessed by immunoblotting of the spike protein within cell lysates. (C) Structure of constructs for a split NanoLuc-based bioreporter. RBD or S1 from either SARS-CoV1 or SARS-CoV2 was linked to Large BiT (LgBiT) on its N-terminus to form LgBiT-RBD or LgBiT-S1; similarly, the Small BiT (SmBiT) peptide was linked to human ACE2 to form SmBiT-ACE2. ACE2 and RBD or S1 constructs were transfected separately or cotransfected into HEK293 cells for 48 h and lysed with a passive lysis buffer. Mixed lysates or lysates from cotransfected cells were incubated with coelenterazine, and the luminescence measured using a plate reader. (D) Following plasmid transfection into HEK293 cells, the cells were lysed in a NanoLuc-compatible passive lysis buffer and lysates were dispensed into a 96-well plate, to which atovaquone was added at a final concentration of 4 muM. The impact of atovaquone on SARS receptor binding was assessed in two ways: (1)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
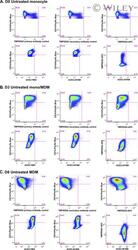
- 1 FIGURE Expression of ACE2 and TMPRSS2 by monocytes and macrophages. Representative FACS stain of ACE2 and TMPRSS2 on (A) freshly isolated monocytes (day 0), and after 3 (B) and 8 days (C) in culture under standard conditions. Top panels show TMPRSS2 negative controls with specific staining. Bottom panels show ACE2 FMO negative control, ACE2 staining, and dual TMPRSS2/ACE2 staining. Data are representative of 5-9 replicate experiments using cells from different donors
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunohistochemistry
Immunohistochemistry