Antibody data
- Antibody Data
- Antigen structure
- References [9]
- Comments [0]
- Validations
- Western blot [1]
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 14-5841-80 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- OCT3/4 Monoclonal Antibody (EM92), eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The EM92 monoclonal antibody reacts with mouse and human Oct3/4, encoded by the Pou5F1 gene. Oct3/4 is a POU domain-containing transcription factor that is critical for maintaining embryonic stem (ES) and induced pluripotent stem (iPS) cells in a pluripotent state, and is expressed by ES, embryonic germ cells and embryonic carcinoma cell lines. In cells of the inner cell mass (ICM), reduction of Oct3/4 expression causes dedifferentiation to trophoectoderm, whereas increased expression results in differentiation to mesoderm and primitive endoderm. Oct3/4 regulates the expression of several genes, including FGF-4, UTF1, Sox2, Fbx15, Rex1 and osteopontin through distinct mechanisms. Furthermore, Oct3/4 frequently acts synergistically with Sox2 to regulate target gene expression, as is the case with FGF-4. It has been demonstrated that Oct3/4 expression in ES cells can be negatively regulated by either treatment with retinoic acid, or by removal of leukemia-inhibitory factor (LIF). Applications Reported: This EM92 antibody has been reported for use in immunoblotting (WB). Applications Tested: This EM92 antibody has been tested by western blot analysis of F9 and P19 embryonal carcinoma cell lysates. For western blotting, this antibody can be used at a starting dilution 2 µg/mL. However, the antibody should be titrated for individual experiments. Purity: Greater than 90%, as determined by SDS-PAGE. Aggregation: Less than 10%, as determined by HPLC. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human, Mouse
- Host
- Rat
- Isotype
- IgG
- Antibody clone number
- EM92
- Vial size
- 25 µg
- Concentration
- 0.5 mg/mL
- Storage
- 4° C
Submitted references Comparative analysis of CI- and CIV-containing respiratory supercomplexes at single-cell resolution.
CBP-mediated Wnt3a/β-catenin signaling promotes cervical oncogenesis initiated by Piwil2.
Pressure-Driven Mitochondrial Transfer Pipeline Generates Mammalian Cells of Desired Genetic Combinations and Fates.
TAF5L and TAF6L Maintain Self-Renewal of Embryonic Stem Cells via the MYC Regulatory Network.
Multi-kilobase homozygous targeted gene replacement in human induced pluripotent stem cells.
Highly upregulated Lhx2 in the Foxn1-/- nude mouse phenotype reflects a dysregulated and expanded epidermal stem cell niche.
Induction of pluripotent stem cells from adult human fibroblasts by defined factors.
Retinoic acid represses Oct-3/4 gene expression through several retinoic acid-responsive elements located in the promoter-enhancer region.
A novel octamer binding transcription factor is differentially expressed in mouse embryonic cells.
Bertan F, Wischhof L, Scifo E, Guranda M, Jackson J, Marsal-Cots A, Piazzesi A, Stork M, Peitz M, Prehn JHM, Ehninger D, Nicotera P, Bano D
Cell reports methods 2021 May 24;1(1):100002
Cell reports methods 2021 May 24;1(1):100002
CBP-mediated Wnt3a/β-catenin signaling promotes cervical oncogenesis initiated by Piwil2.
Feng D, Yan K, Liang H, Liang J, Wang W, Yu H, Zhou Y, Zhao W, Dong Z, Ling B
Neoplasia (New York, N.Y.) 2021 Jan;23(1):1-11
Neoplasia (New York, N.Y.) 2021 Jan;23(1):1-11
Pressure-Driven Mitochondrial Transfer Pipeline Generates Mammalian Cells of Desired Genetic Combinations and Fates.
Patananan AN, Sercel AJ, Wu TH, Ahsan FM, Torres A Jr, Kennedy SAL, Vandiver A, Collier AJ, Mehrabi A, Van Lew J, Zakin L, Rodriguez N, Sixto M, Tadros W, Lazar A, Sieling PA, Nguyen TL, Dawson ER, Braas D, Golovato J, Cisneros L, Vaske C, Plath K, Rabizadeh S, Niazi KR, Chiou PY, Teitell MA
Cell reports 2020 Dec 29;33(13):108562
Cell reports 2020 Dec 29;33(13):108562
TAF5L and TAF6L Maintain Self-Renewal of Embryonic Stem Cells via the MYC Regulatory Network.
Seruggia D, Oti M, Tripathi P, Canver MC, LeBlanc L, Di Giammartino DC, Bullen MJ, Nefzger CM, Sun YBY, Farouni R, Polo JM, Pinello L, Apostolou E, Kim J, Orkin SH, Das PP
Molecular cell 2019 Jun 20;74(6):1148-1163.e7
Molecular cell 2019 Jun 20;74(6):1148-1163.e7
Multi-kilobase homozygous targeted gene replacement in human induced pluripotent stem cells.
Byrne SM, Ortiz L, Mali P, Aach J, Church GM
Nucleic acids research 2015 Feb 18;43(3):e21
Nucleic acids research 2015 Feb 18;43(3):e21
Highly upregulated Lhx2 in the Foxn1-/- nude mouse phenotype reflects a dysregulated and expanded epidermal stem cell niche.
Bohr S, Patel SJ, Vasko R, Shen K, Huang G, Yarmush ML, Berthiaume F
PloS one 2013;8(5):e64223
PloS one 2013;8(5):e64223
Induction of pluripotent stem cells from adult human fibroblasts by defined factors.
Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S
Cell 2007 Nov 30;131(5):861-72
Cell 2007 Nov 30;131(5):861-72
Retinoic acid represses Oct-3/4 gene expression through several retinoic acid-responsive elements located in the promoter-enhancer region.
Pikarsky E, Sharir H, Ben-Shushan E, Bergman Y
Molecular and cellular biology 1994 Feb;14(2):1026-38
Molecular and cellular biology 1994 Feb;14(2):1026-38
A novel octamer binding transcription factor is differentially expressed in mouse embryonic cells.
Okamoto K, Okazawa H, Okuda A, Sakai M, Muramatsu M, Hamada H
Cell 1990 Feb 9;60(3):461-72
Cell 1990 Feb 9;60(3):461-72
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- F9 mouse embryonal carcinoma cell line (left) and mouse splenocyte (right) lysates were loaded at 1x10e5 cells/lane, probed with 2 µg/mL of Anti-Mouse OCT3/4 Purified and revealed with Anti-Rat IgG HRP.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
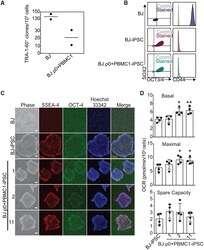
- Figure 3. SIMR Fibroblasts Can Be Reprogrammed (A) Native BJ and SIMR fibroblasts reprogrammed to iPSCs with TRA-1-60 + clones counted by microscopy. Data are the means of biological duplicates. Data for BJ fibroblast control are the same data as in Figure S3A . (B) Flow cytometry of pluripotency biomarkers SOX2 and OCT3/4, and fibroblast biomarker CD44. Immunostained samples are shown in color with isotype negative controls in gray. Representative data for native BJ fibroblasts and BJ-iPSCs, and for BJ rho0+PBMC1-iPSC cells. Data for the native BJ fibroblasts and BJ-iPSCs shown here are the same as in Figure S3B . (C) Representative phase contrast and IF microscopy images of native BJ fibroblast (negative control), BJ-iPSC (positive control), and three BJ rho0+PBMC1-iPSC clones immunostained for pluripotency biomarkers SSEA-4 and OCT4. Scale bars, 100 mum. (D) OCR measurements for ~1.5 x 10 4 native BJ-iPSCs and BJ rho0+PBMC1-iPSC clones 1, 2, and 11. Data for BJ-iPSC control are the same as in Figure S3D . Data are the means +- SD of four technical replicates. Statistical significance by unpaired, two-tailed Student''s t test. *p 0.05; **p 0.01 See also Figure S1 and Table S2 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
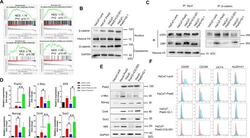
- Figure 4 CBP/beta-catenin promotes the maintenance of stem cell reprogramming by Piwil2. (A) GSEA plot showing significant enrichment of the Wnt/beta-catenin signaling activation modules in HaCaT-Piwil2 cells. (B) Immunoblotting of beta-catenin translocated into the nucleus in HaCaT-Piwil2 cells after treatment with 20 uM ICG-001 or IQ-1 for 24 h. (C) Nuclear lysates from HaCaT-Piwil2 cells treated with 20 uM ICG-001, 20 uM IQ-1, or DMSO were coimmunoprecipitated with antisera to beta-catenin and immunoblotted for CBP and p300. (D and E) The expression of Piwil2 and the ""reprogramming"" factors c-Myc, Nanog, Oct4, Sox2 , and Klf4 was determined by real-time PCR and immunoblotting in HaCaT-Piwil2 cells treated with 20 uM ICG-001, 20 uM IQ-1, or DMSO for 24 h. (F) The proportion of CD49f-, CD338-, OCT4-, and ALDHA1-positive cells, determined by flow cytometry in HaCaT-Piwil2 cells treated with 20 uM ICG-001, 20 uM IQ-1, or DMSO for 24 h. The data are presented as the mean +- SD. * P < 0.05 and ** P < 0.01 by Student's t test. Figure 4
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
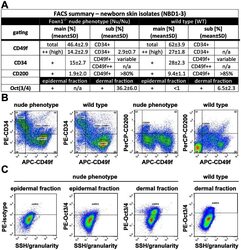
- Figure 4 As Oct3/4+ population is expanded; CD49f+, CD34+ and CD200+ populations are reduced in epithelial isolates from newborn skin of the Foxn1 -/- phenotype vs. wild type. (A) Flow cytometry summary table listing fractions positive for markers of progeny within epithelial isolates from newborns (%+-SD, n>6). Distinct differences between the Foxn1 -/- phenotype and wild type are demonstrated by comparative sample flow cytometry data gated for (B) CD49f, CD34, CD200 and (C) Oct3/4 positive epithelial fractions.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
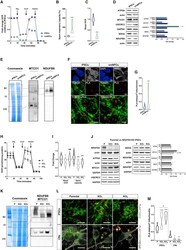
- Experimental details
- Figure 4 CI*CIV-SC remodeling occurs during iPSC differentiation (A) OCR measurement of iPSCs and iPSC-derived smNPCs using a conventional Seahorse protocol (n = 3). (B) Mitochondrial spare respiratory capacity in human iPSCs and smNPCs. Percentage is relative to their respective basal OCRs (Student's t test, ****p < 0.0001). (C) Basal OCR in iPSCs and smNPCs (Student's t test, ****p < 0.0001). (D) Immunoblots using antibodies against OXPHOS system components in iPSCs and smNPCs. Densitometry is relative to iPSCs and reported as mean +- SEM (n = 3, Student's t test, ***p < 0.001, *p < 0.05). (E) BN-PAGE and corresponding immunoblots using antibodies against MTCO1 and NDUFB8. (F) Representative pictures of iPSCs and smNPCs stained with DAPI (nucleus, blue), TOM20 (mitochondria, green), and PLA (CI*CIV-SCs, red). Oct3 and nestin staining (both in gray) was used as markers of pluripotency and differentiation, respectively. Scale bar, 5 mum. (G) Quantification of PLA dots normalized to mitochondrial area in iPSCs and smNPCs (n = 3; iPSCs = 20 cells; smNPCs = 17 cells; Student's t test, ***p < 0.001). (H) OCR measurement of WT (P) and two independent clones of NDUFS4 KO iPSCs (KO 1 and KO 2 ). (I) Basal OCR and mitochondrial spare capacity of WT and NDUFS4 KO iPSCs (fold change, relative to WT; two-way RM ANOVA, ***p < 0.001, *p < 0.05; ns, not significant). (J) Immunoblot analysis of homogenates from parental and NDUFS4 KO iPSCs using antibodies against ETC subunits (CI, NDUFS4,
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunohistochemistry
Immunohistochemistry