Antibody data
- Antibody Data
- Antigen structure
- References [19]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [20]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 67-0739-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD73 Monoclonal Antibody (AD2), Super Bright™ 702, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: This AD2 monoclonal antibody reacts with human CD73, a 5'-ectonucleotidase that converts 5'-adenosine monophosphate to adenosine. CD73 is expressed on the surface of endothelial cells, as well as B and T cells, including some CD4+Foxp3+ regulatory T cells. Adenosine production by these cells has been linked to the inhibition of CD4 T cell effector functions such as proliferation and cytokine secretion. Applications Reported: This AD2 antibody has been reported for use in flow cytometric analysis. Applications Tested: This AD2 antibody has been pre-diluted and tested by flow cytometric analysis of normal human peripheral blood cells. This may be used at 5 µL (0.25 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Super Bright 702 is a tandem dye that can be excited with the violet laser line (405 nm) and emits at 702 nm. We recommend using a 710/50 bandpass filter. Please make sure that your instrument is capable of detecting this fluorochrome. When using two or more Super Bright dye-conjugated antibodies in a staining panel, it is recommended to use Super Bright Complete Staining Buffer (Product # SB-4401) to minimize any non-specific polymer interactions. Please refer to the datasheet for Super Bright Staining Buffer for more information. Light sensitivity: This tandem dye is sensitive to photo-induced oxidation. Please protect this vial and stained samples from light. Fixation: Samples can be stored in IC Fixation Buffer (Product # 00-8222) (100 µL of cell sample + 100 µL of IC Fixation Buffer) or 1-step Fix/Lyse Solution (Product # 00-5333) for up to 3 days in the dark at 4°C with minimal impact on brightness and FRET efficiency/compensation. Some generalizations regarding fluorophore performance after fixation can be made, but clone specific performance should be determined empirically. Excitation: 405 nm; Emission: 702 nm; Laser: Violet Laser Super Bright Polymer Dyes are sold under license from Becton, Dickinson and Company.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- AD2
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Exosomal CD73 from serum of patients with melanoma suppresses lymphocyte functions and is associated with therapy resistance to anti-PD-1 agents.
Induced Neurodifferentiation of hBM-MSCs through Activation of the ERK/CREB Pathway via Pulsed Electromagnetic Fields and Physical Stimulation Promotes Neurogenesis in Cerebral Ischemic Models.
Reprogramming bone progenitor identity and potency through control of collagen density and oxygen tension.
In vitro differentiation of human embryonic stem cells to hemogenic endothelium and blood progenitors via embryoid body formation.
Bcl-xL mutant promotes cartilage differentiation of BMSCs by upregulating TGF-β/BMP expression levels.
Therapeutic potential of human umbilical cord mesenchymal stem cells on aortic atherosclerotic plaque in a high-fat diet rabbit model.
In Vitro Anti-cancer Activity of Adipose-Derived Mesenchymal Stem Cells Increased after Infection with Oncolytic Reovirus.
VAP-PLGA microspheres (VAP-PLGA) promote adipose-derived stem cells (ADSCs)-induced wound healing in chronic skin ulcers in mice via PI3K/Akt/HIF-1α pathway.
Peptide 11R‑VIVIT promotes fracture healing in osteoporotic rats.
TRPA1 triggers hyperalgesia and inflammation after tooth bleaching.
Down-Regulated Exosomal MicroRNA-221 - 3p Derived From Senescent Mesenchymal Stem Cells Impairs Heart Repair.
Differentiation Potential of Early- and Late-Passage Adipose-Derived Mesenchymal Stem Cells Cultured under Hypoxia and Normoxia.
Vasculogenesis from Human Dental Pulp Stem Cells Grown in Matrigel with Fully Defined Serum-Free Culture Media.
Chondrogenic Differentiation from Induced Pluripotent Stem Cells Using Non-Viral Minicircle Vectors.
Prospective isolation of human fibroadipogenic progenitors with CD73.
Circulating Exosomes Inhibit B Cell Proliferation and Activity.
PF-127 hydrogel plus sodium ascorbyl phosphate improves Wharton's jelly mesenchymal stem cell-mediated skin wound healing in mice.
A high-yield isolation and enrichment strategy for human lung microvascular endothelial cells.
Myoblasts derived from normal hESCs and dystrophic hiPSCs efficiently fuse with existing muscle fibers following transplantation.
Turiello R, Capone M, Morretta E, Monti MC, Madonna G, Azzaro R, Del Gaudio P, Simeone E, Sorrentino A, Ascierto PA, Morello S
Journal for immunotherapy of cancer 2022 Mar;10(3)
Journal for immunotherapy of cancer 2022 Mar;10(3)
Induced Neurodifferentiation of hBM-MSCs through Activation of the ERK/CREB Pathway via Pulsed Electromagnetic Fields and Physical Stimulation Promotes Neurogenesis in Cerebral Ischemic Models.
Park HJ, Choi JH, Nam MH, Seo YK
International journal of molecular sciences 2022 Jan 21;23(3)
International journal of molecular sciences 2022 Jan 21;23(3)
Reprogramming bone progenitor identity and potency through control of collagen density and oxygen tension.
Al Hosni R, Bozec L, Roberts SJ, Cheema U
iScience 2022 Apr 15;25(4):104059
iScience 2022 Apr 15;25(4):104059
In vitro differentiation of human embryonic stem cells to hemogenic endothelium and blood progenitors via embryoid body formation.
Garcia-Alegria E, Potts B, Menegatti S, Kouskoff V
STAR protocols 2021 Mar 19;2(1):100367
STAR protocols 2021 Mar 19;2(1):100367
Bcl-xL mutant promotes cartilage differentiation of BMSCs by upregulating TGF-β/BMP expression levels.
Xiao K, Yang L, Xie W, Gao X, Huang R, Xie M
Experimental and therapeutic medicine 2021 Jul;22(1):736
Experimental and therapeutic medicine 2021 Jul;22(1):736
Therapeutic potential of human umbilical cord mesenchymal stem cells on aortic atherosclerotic plaque in a high-fat diet rabbit model.
Li Y, Shi G, Han Y, Shang H, Li H, Liang W, Zhao W, Bai L, Qin C
Stem cell research & therapy 2021 Jul 15;12(1):407
Stem cell research & therapy 2021 Jul 15;12(1):407
In Vitro Anti-cancer Activity of Adipose-Derived Mesenchymal Stem Cells Increased after Infection with Oncolytic Reovirus.
Babaei A, Bannazadeh Baghi H, Nezhadi A, Jamalpoor Z
Advanced pharmaceutical bulletin 2021 Feb;11(2):361-370
Advanced pharmaceutical bulletin 2021 Feb;11(2):361-370
VAP-PLGA microspheres (VAP-PLGA) promote adipose-derived stem cells (ADSCs)-induced wound healing in chronic skin ulcers in mice via PI3K/Akt/HIF-1α pathway.
Jiang W, Zhang J, Zhang X, Fan C, Huang J
Bioengineered 2021 Dec;12(2):10264-10284
Bioengineered 2021 Dec;12(2):10264-10284
Peptide 11R‑VIVIT promotes fracture healing in osteoporotic rats.
Hou C, Wang X, Jiang W, Bian Z, Zhu L, Li M
International journal of molecular medicine 2021 Aug;48(2)
International journal of molecular medicine 2021 Aug;48(2)
TRPA1 triggers hyperalgesia and inflammation after tooth bleaching.
Chen C, Huang X, Zhu W, Ding C, Huang P, Li R
Scientific reports 2021 Aug 31;11(1):17418
Scientific reports 2021 Aug 31;11(1):17418
Down-Regulated Exosomal MicroRNA-221 - 3p Derived From Senescent Mesenchymal Stem Cells Impairs Heart Repair.
Sun L, Zhu W, Zhao P, Zhang J, Lu Y, Zhu Y, Zhao W, Liu Y, Chen Q, Zhang F
Frontiers in cell and developmental biology 2020;8:263
Frontiers in cell and developmental biology 2020;8:263
Differentiation Potential of Early- and Late-Passage Adipose-Derived Mesenchymal Stem Cells Cultured under Hypoxia and Normoxia.
Zhao AG, Shah K, Freitag J, Cromer B, Sumer H
Stem cells international 2020;2020:8898221
Stem cells international 2020;2020:8898221
Vasculogenesis from Human Dental Pulp Stem Cells Grown in Matrigel with Fully Defined Serum-Free Culture Media.
Luzuriaga J, Irurzun J, Irastorza I, Unda F, Ibarretxe G, Pineda JR
Biomedicines 2020 Nov 9;8(11)
Biomedicines 2020 Nov 9;8(11)
Chondrogenic Differentiation from Induced Pluripotent Stem Cells Using Non-Viral Minicircle Vectors.
Rim YA, Nam Y, Park N, Jung H, Lee K, Lee J, Ju JH
Cells 2020 Mar 1;9(3)
Cells 2020 Mar 1;9(3)
Prospective isolation of human fibroadipogenic progenitors with CD73.
Goloviznina NA, Xie N, Dandapat A, Iaizzo PA, Kyba M
Heliyon 2020 Jul;6(7):e04503
Heliyon 2020 Jul;6(7):e04503
Circulating Exosomes Inhibit B Cell Proliferation and Activity.
Schroeder JC, Puntigam L, Hofmann L, Jeske SS, Beccard IJ, Doescher J, Laban S, Hoffmann TK, Brunner C, Theodoraki MN, Schuler PJ
Cancers 2020 Jul 29;12(8)
Cancers 2020 Jul 29;12(8)
PF-127 hydrogel plus sodium ascorbyl phosphate improves Wharton's jelly mesenchymal stem cell-mediated skin wound healing in mice.
Deng Q, Huang S, Wen J, Jiao Y, Su X, Shi G, Huang J
Stem cell research & therapy 2020 Apr 3;11(1):143
Stem cell research & therapy 2020 Apr 3;11(1):143
A high-yield isolation and enrichment strategy for human lung microvascular endothelial cells.
Gaskill C, Majka SM
Pulmonary circulation 2017 Mar;7(1):108-116
Pulmonary circulation 2017 Mar;7(1):108-116
Myoblasts derived from normal hESCs and dystrophic hiPSCs efficiently fuse with existing muscle fibers following transplantation.
Goudenege S, Lebel C, Huot NB, Dufour C, Fujii I, Gekas J, Rousseau J, Tremblay JP
Molecular therapy : the journal of the American Society of Gene Therapy 2012 Nov;20(11):2153-67
Molecular therapy : the journal of the American Society of Gene Therapy 2012 Nov;20(11):2153-67
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
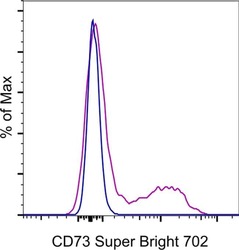
- Experimental details
- Normal human peripheral blood cells were stained with Mouse IgG1 kappa Isotype Control, Super Bright 702 (Product # 67-4714-82) (blue histogram) or CD73 Monoclonal Antibody, Super Bright 702 (purple histogram). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
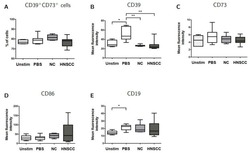
- Experimental details
- Figure 5 B cells were harvested after 2 days of co-culture with either NC or HNSCC exosomes or PBS and stained for FACS analysis. ( A ) Frequency of CD39 + CD73 + regulatory B cells. ( B ) The expression of CD39 on B cells was reduced after co-culture with NC or HNSCC exosomes. ( C ) Expression of CD73 on B cells. ( D ) Expression of CD86 on B cells. ( E ) The expression of CD19 on B cells was increased by stimulation with CD40L and IL-4. **: p < 0.01; *: p < 0.05, n = 8 (HNSCC), n = 6 (NC), n = 5 (Unstim). Unstim = Unstimulated B cells, NC = no cancer (exosomes from blood plasma of healthy volunteers), HNSCC, exosomes from blood plasma of HNSCC patients.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
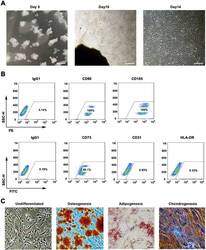
- Experimental details
- Fig. 1 WJMSCs isolation and characterization. a Primary cell isolation procedure from Wharton''s jelly tissue. The migrated cells exhibited typical fibroblast-like morphology. Scale bar, 500 mum. b Flow cytometry analysis of P4 cells using mesenchymal stem cell markers (CD90, CD105, CD73), endothelial cell marker (CD31), and MHC class II protein HLA-DR. Isotypic antibodies (IgG1-PE and IgG1-FITC) were used as negative controls. c Representative stained images show that the fourth passage WJMSCs could differentiate into osteocytes (Alizarin Red S), adipocytes (Oil Red O), and chondrocytes (Alcian blue). Scale bar, 100 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
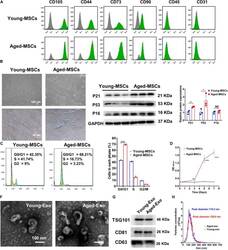
- Experimental details
- FIGURE 1 Characterization of young and aged MSCs and exosomes. (A) Surface marker profiling of young-MSCs and aged-MSCs. (B) SA-beta-Gal staining showed that senescence increased significantly in aged MSCs. (C) Representative immunoblot images and quantitative analysis of p21, p53, and p16 protein level in young and aged-MSCs. ( n = 3). (D) Quantitation of cell cycle phases by propidium iodide staining. ( n = 3). (E) The CCK-8 assay showed that aged MSCs grew more slowly than young MSCs. ( n = 6). (F) Young and aged exosomes were observed using TEM. (G) The exosome surface markers were analyzed by Western blot. (H) Nanoparticle tracking analysis was used to analyze the particle size and concentration of Young-Exo and Aged-Exo. * p < 0.05; ** p < 0.01; *** p < 0.001; **** p < 0.0001; NS, not significant.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
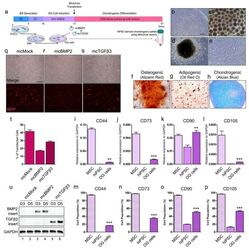
- Experimental details
- Figure 2 Chondrogenesis using minicircle-transfected hiPSC-derived OG cells. ( a ) Scheme of chondrogenic differentiation process from hiPSCs. Minicircles were transfected after OG cells were induced. ( b ) Morphology of the hiPSC colony. ( c ) Morphology of the generated EBs. ( d ) Image of outgrowth cells derived from EBs attached to a gelatin-coated culture dish. ( e ) Morphology of OG cells before transfection. ( f ) Alizarin red-stained osteogenic cells differentiated from OG cells. ( g ) Oil red O staining image of adipogenic cells differentiated from OG cells. ( h ) Chondrogenic pellet generated from OG cells stained with alcian blue. Relative gene expression of ( i ) CD44, ( j ) CD73, ( k ) CD90, and ( l ) CD105 in OG cells. Percentage of ( m ) CD44, ( n ) CD73, ( o ) CD90, and ( p ) CD105 positive cells. ( q ) Fluorescence microscopy of mcMock-transfected OG cells. ( r ) Fluorescence microscopy of mcBMP2-transfected OG cells. ( s ) Fluorescence microscopy of mcTGFbeta3-transfected OG cells. ( t ) Percentage of OG cells transfected with each minicircle vectors. ( u ) Gel image of the PCR results against the insert of mcBMP2 and mcTGFbeta3 in transfected OG cells. Data are presented as mean +- standard deviation from three independent sets of experiments. Scale bars represents 200 mum. ** p < 0.01 and *** p < 0.001 indicate statistical significance. EB: embryonic body; OG: outgrowth; CDM: chondrogenic differentiation media; RFP: red fluorescence protein; MSC: mesenchym
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
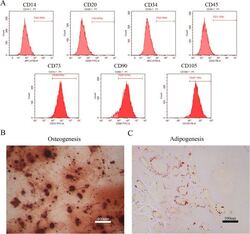
- Experimental details
- Figure 1 Identification of dental pulp stem cells (DPSCs). Human DPSCs were positive for the cell surface antigens CD73, CD90, and CD105, as well as negative for CD14, CD20, CD34, and CD45 demonstrated by flow cytometry ( A ). DPSCs were cultured under osteogenic ( B , 14 days) or adipogenic ( C , 21 days) conditions, and showed mineralized nodules and lipid clusters as revealed by alizarin red and oil red staining, respectively. Scale bar = 400 ( B ) or 100 ( C ) mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3 Expression of endothelial cell surface determinants by primary lung MVEC. Lung cells were incubated with primary antibodies directly conjugated to fluorophores and staining intensity analyzed by flow cytometry. Representative dot plots are presented. Human lung FB and MSC were used as known negative controls, and PAEC as a known positive control. DAPI was used to exclude dead cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
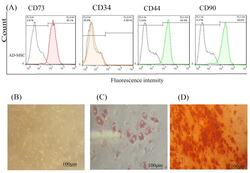
- Experimental details
- Figure 1 AD-MSCs characterization; (A) Flow cytometry assay to assess the CD markers in the surface of extracted AD-MSCs. Target cells were positive for CD73 (95.1%), and CD90 (90.6%) surface markers and were negative for CD44 and CD34 markers. (B) Morphology of AD-MSCs at passage 3. (C) Oil red staining to prove the adipogenic potential differentiation of AD-MSCs. (D) Alizarin-red staining to confirm the osteogenic potential differentiation of AD-MSCs. Scale bar = 100 mum. Abbreviations: AD-MSCs: adipose-derived mesenchymal stem cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
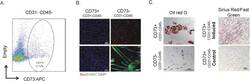
- Experimental details
- Figure 1 CD73 positive cells are fibrogenic and adipogenic but not myogenic in culture. A. FACS analysis showing CD73 staining of single cells from human muscle biopsy. Average fraction of CD73+ CD45- CD31- cells in wild type muscle is 4.5% (n = 4). B. CD73+ and CD73- cells were cultured in myogenic growth medium and stained with MyoD; or cultured in myogenic differentiation medium, and stained with MHC. C. CD73+ and CD73- cells were cultured in adipogenic differentiation medium, then stained with Oil red O. CD73+ cells were also cultured in fibrogenic differentiation medium, then stained with Sirius Red/Fast Green. Figure 1
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
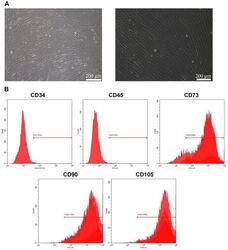
- Figure 1 Culture and identification of bone marrow mesenchymal stem cells. (A) Light microscopy of BMSCs (scale bar=100 um for the left image and 200 um for the right image). (B) Percentage of CD34-, CD45-, CD73-, CD90- and CD105-positive BMSCs were detected by flow cytometry. BMSC, bone marrow mesenchymal stem cell.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
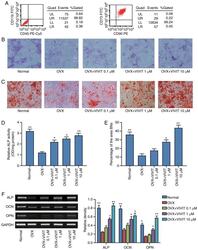
- Figure 2 11R-VIVIT increases the osteogenic potential of osteoporotic BMSCs. (A) Flow cytometric analysis of BMSC surface antigens. (B) ALP staining results. (C) Alizarin Red S staining results. (D) Quantitative results of ALP staining. (E) Quantitative results of Alizarin Red S staining. (F) Semi-quantitative PCR detection of osteogenesis-related gens. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 1 Characterization of UCSCs. A UCSCs display a spindle shaped and fibroblast-like morphology. B High UCSCs expression of CD90, CD29, CD73, and CD105, and low expression of HLA using flow cytometry
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
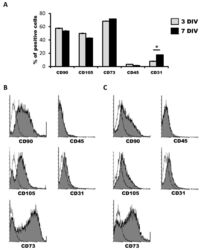
- Figure 1 Characterization of human dental pulp stem cells (hDPSCs) cultured in Neurocult(tm) proliferation medium by flow cytometry. ( A ) Data quantification as representative histograms at 3 days in vitro (DIV) (gray) and 7DIV (black) for CD90, CD105 and CD73 mesenchymal stromal cell (MSC) markers, CD45 hematopoietic and CD31 endothelial markers ( n = 3). Data are represented as the average percentage and standard error of the mean (SEM+-), with respect to the total cell population. ( B ) Representative cytometry of CD90, CD105 and CD73 mesenchymal, CD45 hematopoietic and CD31 endothelial markers in hDPSCs cultured in Neurocult proliferation medium at 3DIV (gray filling), with respect to negative controls (no filling). ( C ) Representative cytometry of CD90, CD105 and CD73 mesenchymal, CD45 hematopoietic and CD31 endothelial markers in hDPSCs cultured in Neurocult proliferation medium at 7DIV (gray filling), with respect to negative controls (no filling). *: p < 0.05. Kruskal-Wallis with Dunn's post hoc test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
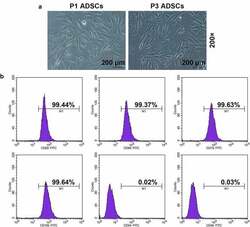
- Figure 1. Morphology and immune phenotype of adipose-derived stem cells (ADSCs) were identified by morphological observation and flow cytometry. (a) Morphology of the primary (P1) and third passage (P3) of ADSCs. Images were acquired at 200x magnification. (b) Immune phenotype of ADSCs. The average data from three independent experiments were shown as mean +- standard deviation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
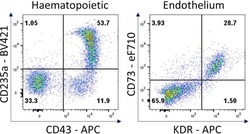
- Experimental details
- Figure 7 Hematopoietic and endothelial cell characterization Flow cytometry analysis of day 4 hemogenic culture for hematopoietic (CD43, CD235a) and endothelial (KDR, CD73) cell surface markers.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
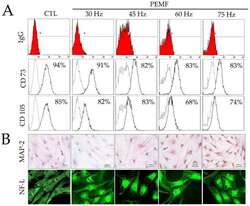
- Experimental details
- Figure 2 ( A ) Results of fluorescence-activated cell sorting (FACS) analysis on hBM-MSCs surface markers (CD73 and CD105) and IgG control after PEMF for three days. ( B ) Immunohistochemical analysis of MAP-2 antibodies on hBM-MSCs cultured after PEMF for three days (original magnification: 100x); and immunofluorescence staining of NF-L antibodies on hBM-MSCs after PEMF for three days (original magnification: 400x).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
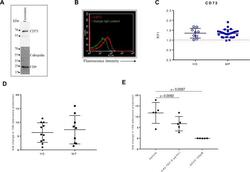
- Experimental details
- Figure 2 CD73 expression and activity in serum-derived exosomes. (A) A representative western blot image for CD73 in purified exosomes (10 ug loaded protein). CD9 was used as specific exosomal marker and calregulin as negative marker. (B) A representative flow cytometry dot plot showing the expression of CD73 on isolated exosomes captured for CD63 and bound to magnetic beads, labeled with a specific antibody anti-CD73 (red line) or the isotype IgG control (green line). (C) Levels of CD73 expression as relative fluorescence intensity (RFI) values on exosomes isolated from healthy donors (HD) (n=9) and patients with melanoma (MP) at baseline (n=27). Comparison of the fold change in the 15 N adenosine production, measured by mass spectrometry, after incubating for 2 hours 5 ug of exosomes isolated from serum of HD (n=6) or MP (n=9) with 15 N AMP (10 uM) (D), with or without the anti-human CD73 mAb (5 ug/mL) or APCP (100 uM) (E), (n=5). Data are mean+-SD (C, D, E). P values are obtained from a repeated measures (RM) one-way analysis of variance, followed by Dunnet's multiple comparison test (E).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
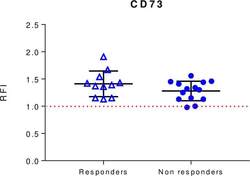
- Experimental details
- Baseline levels of exosomal CD73 expression in responders and non-responders to anti-PD-1 blockers nivolumab or pembrolizumab. CD73 expression was evaluated by beads-assisted flow cytometry on exosomes isolated from patients with melanoma prior to initiation of treatment. Responders n=11. Non-responders n=14. Data are mean+-SD. PD-1, programmed cell-death protein 1; RFI, relative fluorescence intensity.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Cell surface analysis of AMSCs within the transitional culture AMSCs were cultured in a 10% collagen type I gel for 7 days and embedded in a 0.2% collagen type I gel, making up the transitional culture for an additional 14 days. Flow cytometric analysis was conducted for PDPN, CD73, and CD146 (representative data presented).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Flow cytometry of CD cell surface markers for cells cultured under hypoxia and normoxia. The positive CD markers for MSCs as detected by the fluorescent antibodies anti-CD73 FITC, anti-CD105 PE, and anti-CD90 PE Cy7. The negative markers of MSCs were detected using anti-CD14 FITC, anti-CD45 PerCP, anti-CD34-R-PE, and anti-CD19 PE-Cy7 antibodies. Unstained cell for each condition was used as negative controls.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
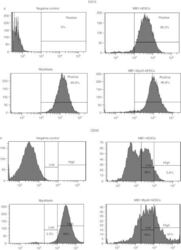
- Experimental details
- Figure 5 FACS analysis of hESC-derived mesenchymal-like precursors generated by culture in MB1 . ( a ) The culture of hESCs in the MB1 culture medium (MB1-hESCs) induced their differentiation in mesenchymal-like stem cells expressing CD73. ( b ) However, in the MB-1 medium,
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry