MA3-019
antibody from Invitrogen Antibodies
Targeting: P4HB
DSI, ERBA2L, GIT, P4Hbeta, PDI, PDIA1, PO4DB, PO4HB, PROHB
 Western blot
Western blot Immunocytochemistry
Immunocytochemistry Immunoprecipitation
Immunoprecipitation Immunohistochemistry
Immunohistochemistry Flow cytometry
Flow cytometry Immunoelectron microscopy
Immunoelectron microscopy Other assay
Other assayAntibody data
- Antibody Data
- Antigen structure
- References [92]
- Comments [0]
- Validations
- Immunocytochemistry [18]
- Immunohistochemistry [3]
- Flow cytometry [6]
- Other assay [51]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA3-019 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- PDI Monoclonal Antibody (RL90)
- Antibody type
- Monoclonal
- Antigen
- Purifed from natural sources
- Description
- MA3-019 detects protein disulphide-isomerase (PDI) from human, rat, porcine and mouse tissues as well as hamster cells. MA3-019 has been successfully used in Western blot, immunofluorescence, immunohistochemical, flow cytometry, and immunoprecipitation procedures. By Western blot, this antibody detects a protein at 59 kDa representing PDI from rat liver extract or a slightly higher protein at 61 kDa representing PDI from human liver extract. Immunohistochemical staining of PDI in rat intestine with MA3-019 yields a pattern consistent with cytoplasmic staining. In immunoprecipitation procedures MA3-019 has been shown to inhibit the activity of PDI in vitro. MA3-019 has also been found to inhibit disulfide bond reduction of the HIV protein, gp120, at the cell surface of CHO cells and human lymphoid cells. In immunohistochemistry procedures, formalin-fixed paraffin-embedded tissue sections are recommended. The MA3-019 antigen is purified rat PDI.
- Reactivity
- Human, Mouse, Rat, Hamster, Porcine
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- RL90
- Vial size
- 100 μL
- Concentration
- 1 mg/mL
- Storage
- -20°C, Avoid Freeze/Thaw Cycles
Submitted references A discovery-based proteomics approach identifies protein disulphide isomerase (PDIA1) as a biomarker of β cell stress in type 1 diabetes.
von Willebrand factor propeptide variants lead to impaired storage and ER retention in patient-derived endothelial colony-forming cells.
Homodimerized cytoplasmic domain of PD-L1 regulates its complex glycosylation in living cells.
Engrailed 1 coordinates cytoskeletal reorganization to induce myofibroblast differentiation.
Identification and Characterization of Human Norovirus NTPase Regions Required for Lipid Droplet Localization, Cellular Apoptosis, and Interaction with the Viral P22 Protein.
The c.863A>G (p.Glu288Gly) variant of the CTSD gene is not associated with CLN10 disease.
Potential Dual Role of West Nile Virus NS2B in Orchestrating NS3 Enzymatic Activity in Viral Replication.
The Golgi-associated retrograde protein (GARP) complex plays an essential role in the maintenance of the Golgi glycosylation machinery.
Overexpression of FAM46A, a Non-canonical Poly(A) Polymerase, Promotes Hemin-Induced Hemoglobinization in K562 Cells.
Shear stress-exposed pulmonary artery endothelial cells fail to upregulate HSP70 in chronic thromboembolic pulmonary hypertension.
TMEM79/MATTRIN defines a pathway for Frizzled regulation and is required for Xenopus embryogenesis.
TGF-β-induced epigenetic deregulation of SOCS3 facilitates STAT3 signaling to promote fibrosis.
Novel role of dynamin-related-protein 1 in dynamics of ER-lipid droplets in adipose tissue.
Influences of cyclosporin A and non-immunosuppressive derivatives on cellular cyclophilins and viral nucleocapsid protein during human coronavirus 229E replication.
Heterotypic CAF-tumor spheroids promote early peritoneal metastatis of ovarian cancer.
Alzheimer Aβ Assemblies Accumulate in Excitatory Neurons upon Proteasome Inhibition and Kill Nearby NAKα3 Neurons by Secretion.
Caveolin-1 impairs PKA-DRP1-mediated remodelling of ER-mitochondria communication during the early phase of ER stress.
Cell-surface HSP70 associates with thrombomodulin in endothelial cells.
Redox Activation of Nox1 (NADPH Oxidase 1) Involves an Intermolecular Disulfide Bond Between Protein Disulfide Isomerase and p47(phox) in Vascular Smooth Muscle Cells.
Subverted regulation of Nox1 NADPH oxidase-dependent oxidant generation by protein disulfide isomerase A1 in colon carcinoma cells with overactivated KRas.
Influenza A virus ribonucleoproteins form liquid organelles at endoplasmic reticulum exit sites.
Endocytosis of Activated Muscarinic m2 Receptor (m2R) in Live Mouse Hippocampal Neurons Occurs via a Clathrin-Dependent Pathway.
StAR-related lipid transfer domain 11 (STARD11)-mediated ceramide transport mediates extracellular vesicle biogenesis.
Endoplasmic reticulum, Golgi, and lysosomes are disorganized in lung fibroblasts from chronic obstructive pulmonary disease patients.
Mitochondrial fragmentation affects neither the sensitivity to TNFα-induced apoptosis of Brucella-infected cells nor the intracellular replication of the bacteria.
Subcellular Localization and Functional Characterization of GII.4 Norovirus-Encoded NTPase.
Endoplasmic reticulum-resident protein 57 (ERp57) oxidatively inactivates human transglutaminase 2.
Vesicular acetylcholine transporter (VAChT) over-expression induces major modifications of striatal cholinergic interneuron morphology and function.
Evaluation of the selectivity and sensitivity of isoform- and mutation-specific RAS antibodies.
HLA-DP(84Gly) constitutively presents endogenous peptides generated by the class I antigen processing pathway.
Conserved Gene Microsynteny Unveils Functional Interaction Between Protein Disulfide Isomerase and Rho Guanine-Dissociation Inhibitor Families.
Succination of Protein Disulfide Isomerase Links Mitochondrial Stress and Endoplasmic Reticulum Stress in the Adipocyte During Diabetes.
EMC3 coordinates surfactant protein and lipid homeostasis required for respiration.
Embryonic lethal abnormal vision proteins and adenine and uridine-rich element mRNAs after global cerebral ischemia and reperfusion in the rat.
Peri/Epicellular Protein Disulfide Isomerase Sustains Vascular Lumen Caliber Through an Anticonstrictive Remodeling Effect.
An ERp57-mediated disulphide exchange promotes the interaction between Burkholderia cenocepacia and epithelial respiratory cells.
Optogenetic control of organelle transport and positioning.
Optogenetic acidification of synaptic vesicles and lysosomes.
Biogenesis and cytotoxicity of APOL1 renal risk variant proteins in hepatocytes and hepatoma cells.
An analysis of critical factors for quantitative immunoblotting.
Effects of oxidative stress on the solubility of HRD1, a ubiquitin ligase implicated in Alzheimer's disease.
Characterization of N-glycosylation sites on the extracellular domain of NOX1/NADPH oxidase.
Transport of estradiol-17β-glucuronide, estrone-3-sulfate and taurocholate across the endoplasmic reticulum membrane: evidence for different transport systems.
Oxidoreductase activity is necessary for N-glycosylation of cysteine-proximal acceptor sites in glycoproteins.
Depletion of molecular chaperones from the endoplasmic reticulum and fragmentation of the Golgi apparatus associated with pathogenesis in Pelizaeus-Merzbacher disease.
mRNA redistribution during permanent focal cerebral ischemia.
Inhibiting rotavirus infection by membrane-impermeant thiol/disulfide exchange blockers and antibodies against protein disulfide isomerase.
Derlin-1 and UBXD8 are engaged in dislocation and degradation of lipidated ApoB-100 at lipid droplets.
Unraveling the human dendritic cell phagosome proteome by organellar enrichment ranking.
Protective role of Gipie, a Girdin family protein, in endoplasmic reticulum stress responses in endothelial cells.
Elimination of hepatitis C virus from hepatocytes by a selective activation of therapeutic molecules.
Organelles do not colocalize with mRNA granules in post-ischemic neurons.
NLRP7, a nucleotide oligomerization domain-like receptor protein, is required for normal cytokine secretion and co-localizes with Golgi and the microtubule-organizing center.
mRNA escape from stress granule sequestration is dictated by localization to the endoplasmic reticulum.
Ero1alpha is expressed on blood platelets in association with protein-disulfide isomerase and contributes to redox-controlled remodeling of alphaIIbbeta3.
Mobile ER-to-Golgi but not post-Golgi membrane transport carriers disappear during the terminal myogenic differentiation.
Production of anti-carbohydrate antibodies by phage display technologies: potential impairment of cell growth as a result of endogenous expression.
Regulation of ER molecular chaperone prevents bone loss in a murine model for osteoporosis.
Biomechanical stress induces novel arterial intima-enriched genes: implications for vascular adaptation to stress.
Loss of HRD1-mediated protein degradation causes amyloid precursor protein accumulation and amyloid-beta generation.
DC-STAMP interacts with ER-resident transcription factor LUMAN which becomes activated during DC maturation.
Membrane topology of human AGPAT3 (LPAAT3).
Characterization of the expression, localization, and secretion of PANDER in alpha-cells.
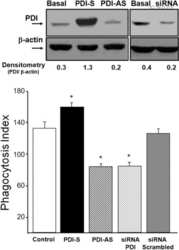
Protein disulfide isomerase (PDI) associates with NADPH oxidase and is required for phagocytosis of Leishmania chagasi promastigotes by macrophages.
Calsyntenins mediate TGN exit of APP in a kinesin-1-dependent manner.
Comparative proteomics profiling of a phospholamban mutant mouse model of dilated cardiomyopathy reveals progressive intracellular stress responses.
Endoplasmic reticulum (ER) chaperone regulation and survival of cells compensating for deficiency in the ER stress response kinase, PERK.
Protein disulphide isomerase in platelet function.
Interaction and functional association of protein disulfide isomerase with alphaVbeta3 integrin on endothelial cells.
Dynamics of somatostatin type 2A receptor cargoes in living hippocampal neurons.
Identification of a novel isoform of microsomal triglyceride transfer protein.
Activated somatostatin type 2 receptors traffic in vivo in central neurons from dendrites to the trans Golgi before recycling.
The dendritic cell-derived protein DC-STAMP is highly conserved and localizes to the endoplasmic reticulum.
Restricted distribution of mRNAs encoding a sarcoplasmic reticulum or transverse tubule protein in skeletal myofibers.
A role for the thiol isomerase protein ERP5 in platelet function.
Regulation of NAD(P)H oxidase by associated protein disulfide isomerase in vascular smooth muscle cells.
A cellular UDP-glucose deficiency causes overexpression of glucose/oxygen-regulated proteins independent of the endoplasmic reticulum stress elements.
Thiol/disulfide exchange is a prerequisite for CXCR4-tropic HIV-1 envelope-mediated T-cell fusion during viral entry.
Intracellularly located misfolded glycoprotein hormone receptors associate with different chaperone proteins than their cognate wild-type receptors.
Enzymatically catalyzed disulfide exchange is required for platelet adhesion to collagen via integrin alpha2beta1.
Protein disulfide isomerase, a component of the estrogen receptor complex, is associated with Chlamydia trachomatis serovar E attached to human endometrial epithelial cells.
Structural requirements for the apical sorting of human multidrug resistance protein 2 (ABCC2).
Structural requirements for the apical sorting of human multidrug resistance protein 2 (ABCC2).
Selective intracellular retention of extracellular matrix proteins and chaperones associated with pseudoachondroplasia.
Selective intracellular retention of extracellular matrix proteins and chaperones associated with pseudoachondroplasia.
Dimerization of P-selectin in platelets and endothelial cells.
Protein-disulfide isomerase (PDI) in FRTL5 cells. pH-dependent thyroglobulin/PDI interactions determine a novel PDI function in the post-endoplasmic reticulum of thyrocytes.
Up-regulation of protein-disulfide isomerase in response to hypoxia/brain ischemia and its protective effect against apoptotic cell death.
Sulfhydryl regulation of L-selectin shedding: phenylarsine oxide promotes activation-independent L-selectin shedding from leukocytes.
Sulfhydryl regulation of L-selectin shedding: phenylarsine oxide promotes activation-independent L-selectin shedding from leukocytes.
Inhibition of human immunodeficiency virus infection by agents that interfere with thiol-disulfide interchange upon virus-receptor interaction.
Protein disulphide-isomerase from human placenta and rat liver. Purification and immunological characterization with monoclonal antibodies.
Syed F, Singhal D, Raedschelders K, Krishnan P, Bone RN, McLaughlin MR, Van Eyk JE, Mirmira RG, Yang ML, Mamula MJ, Wu H, Liu X, Evans-Molina C
EBioMedicine 2023 Jan;87:104379
EBioMedicine 2023 Jan;87:104379
von Willebrand factor propeptide variants lead to impaired storage and ER retention in patient-derived endothelial colony-forming cells.
Bowman M, Casey L, Selvam SN, Lima PDA, Rawley O, Hinds M, Tuttle A, Grabell J, Iorio A, Walker I, Lillicrap D, James P
Journal of thrombosis and haemostasis : JTH 2022 Jul;20(7):1599-1609
Journal of thrombosis and haemostasis : JTH 2022 Jul;20(7):1599-1609
Homodimerized cytoplasmic domain of PD-L1 regulates its complex glycosylation in living cells.
Zhou L, Chai F, He Y, Zhou Z, Guo S, Li P, Sun Q, Zu X, Liu X, Huang Q, Zhong Y, Zhou A, Wang X, Ren H
Communications biology 2022 Aug 30;5(1):887
Communications biology 2022 Aug 30;5(1):887
Engrailed 1 coordinates cytoskeletal reorganization to induce myofibroblast differentiation.
Györfi AH, Matei AE, Fuchs M, Liang C, Rigau AR, Hong X, Zhu H, Luber M, Bergmann C, Dees C, Ludolph I, Horch RE, Distler O, Wang J, Bengsch B, Schett G, Kunz M, Distler JHW
The Journal of experimental medicine 2021 Sep 6;218(9)
The Journal of experimental medicine 2021 Sep 6;218(9)
Identification and Characterization of Human Norovirus NTPase Regions Required for Lipid Droplet Localization, Cellular Apoptosis, and Interaction with the Viral P22 Protein.
Yen JB, Chen LW, Wei LH, Hung CH, Wang SS, Lin CL, Chang PJ
Microbiology spectrum 2021 Sep 3;9(1):e0042221
Microbiology spectrum 2021 Sep 3;9(1):e0042221
The c.863A>G (p.Glu288Gly) variant of the CTSD gene is not associated with CLN10 disease.
Yang J, Ding X, Meng S, Cai J, Zhou W
Molecular genetics & genomic medicine 2021 Oct;9(10):e1777
Molecular genetics & genomic medicine 2021 Oct;9(10):e1777
Potential Dual Role of West Nile Virus NS2B in Orchestrating NS3 Enzymatic Activity in Viral Replication.
Tseng AC, Nerurkar VR, Neupane KR, Kae H, Kaufusi PH
Viruses 2021 Jan 31;13(2)
Viruses 2021 Jan 31;13(2)
The Golgi-associated retrograde protein (GARP) complex plays an essential role in the maintenance of the Golgi glycosylation machinery.
Khakurel A, Kudlyk T, Bonifacino JS, Lupashin VV
Molecular biology of the cell 2021 Aug 15;32(17):1594-1610
Molecular biology of the cell 2021 Aug 15;32(17):1594-1610
Overexpression of FAM46A, a Non-canonical Poly(A) Polymerase, Promotes Hemin-Induced Hemoglobinization in K562 Cells.
Lin HH, Lo YL, Wang WC, Huang KY, I KY, Chang GW
Frontiers in cell and developmental biology 2020;8:414
Frontiers in cell and developmental biology 2020;8:414
Shear stress-exposed pulmonary artery endothelial cells fail to upregulate HSP70 in chronic thromboembolic pulmonary hypertension.
Salibe-Filho W, Araujo TLS, G Melo E, B C T Coimbra L, Lapa MS, Acencio MMP, Freitas-Filho O, Capelozzi VL, Teixeira LR, Fernandes CJCS, Jatene FB, Laurindo FRM, Terra-Filho M
PloS one 2020;15(12):e0242960
PloS one 2020;15(12):e0242960
TMEM79/MATTRIN defines a pathway for Frizzled regulation and is required for Xenopus embryogenesis.
Chen M, Amado N, Tan J, Reis A, Ge M, Abreu JG, He X
eLife 2020 Sep 14;9
eLife 2020 Sep 14;9
TGF-β-induced epigenetic deregulation of SOCS3 facilitates STAT3 signaling to promote fibrosis.
Dees C, Pötter S, Zhang Y, Bergmann C, Zhou X, Luber M, Wohlfahrt T, Karouzakis E, Ramming A, Gelse K, Yoshimura A, Jaenisch R, Distler O, Schett G, Distler JH
The Journal of clinical investigation 2020 May 1;130(5):2347-2363
The Journal of clinical investigation 2020 May 1;130(5):2347-2363
Novel role of dynamin-related-protein 1 in dynamics of ER-lipid droplets in adipose tissue.
Li X, Yang L, Mao Z, Pan X, Zhao Y, Gu X, Eckel-Mahan K, Zuo Z, Tong Q, Hartig SM, Cheng X, Du G, Moore DD, Bellen HJ, Sesaki H, Sun K
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2020 Jun;34(6):8265-8282
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2020 Jun;34(6):8265-8282
Influences of cyclosporin A and non-immunosuppressive derivatives on cellular cyclophilins and viral nucleocapsid protein during human coronavirus 229E replication.
Ma-Lauer Y, Zheng Y, Malešević M, von Brunn B, Fischer G, von Brunn A
Antiviral research 2020 Jan;173:104620
Antiviral research 2020 Jan;173:104620
Heterotypic CAF-tumor spheroids promote early peritoneal metastatis of ovarian cancer.
Gao Q, Yang Z, Xu S, Li X, Yang X, Jin P, Liu Y, Zhou X, Zhang T, Gong C, Wei X, Liu D, Sun C, Chen G, Hu J, Meng L, Zhou J, Sawada K, Fruscio R, Grunt TW, Wischhusen J, Vargas-Hernández VM, Pothuri B, Coleman RL
The Journal of experimental medicine 2019 Mar 4;216(3):688-703
The Journal of experimental medicine 2019 Mar 4;216(3):688-703
Alzheimer Aβ Assemblies Accumulate in Excitatory Neurons upon Proteasome Inhibition and Kill Nearby NAKα3 Neurons by Secretion.
Komura H, Kakio S, Sasahara T, Arai Y, Takino N, Sato M, Satomura K, Ohnishi T, Nabeshima YI, Muramatsu SI, Kii I, Hoshi M
iScience 2019 Mar 29;13:452-477
iScience 2019 Mar 29;13:452-477
Caveolin-1 impairs PKA-DRP1-mediated remodelling of ER-mitochondria communication during the early phase of ER stress.
Bravo-Sagua R, Parra V, Ortiz-Sandoval C, Navarro-Marquez M, Rodríguez AE, Diaz-Valdivia N, Sanhueza C, Lopez-Crisosto C, Tahbaz N, Rothermel BA, Hill JA, Cifuentes M, Simmen T, Quest AFG, Lavandero S
Cell death and differentiation 2019 Jul;26(7):1195-1212
Cell death and differentiation 2019 Jul;26(7):1195-1212
Cell-surface HSP70 associates with thrombomodulin in endothelial cells.
Araujo TLS, Venturini G, Moretti AIS, Tanaka LY, Pereira AC, Laurindo FRM
Cell stress & chaperones 2019 Jan;24(1):273-282
Cell stress & chaperones 2019 Jan;24(1):273-282
Redox Activation of Nox1 (NADPH Oxidase 1) Involves an Intermolecular Disulfide Bond Between Protein Disulfide Isomerase and p47(phox) in Vascular Smooth Muscle Cells.
Gimenez M, Veríssimo-Filho S, Wittig I, Schickling BM, Hahner F, Schürmann C, Netto LES, Rosa JC, Brandes RP, Sartoretto S, De Lucca Camargo L, Abdulkader F, Miller FJ Jr, Lopes LR
Arteriosclerosis, thrombosis, and vascular biology 2019 Feb;39(2):224-236
Arteriosclerosis, thrombosis, and vascular biology 2019 Feb;39(2):224-236
Subverted regulation of Nox1 NADPH oxidase-dependent oxidant generation by protein disulfide isomerase A1 in colon carcinoma cells with overactivated KRas.
De Bessa TC, Pagano A, Moretti AIS, Oliveira PVS, Mendonça SA, Kovacic H, Laurindo FRM
Cell death & disease 2019 Feb 13;10(2):143
Cell death & disease 2019 Feb 13;10(2):143
Influenza A virus ribonucleoproteins form liquid organelles at endoplasmic reticulum exit sites.
Alenquer M, Vale-Costa S, Etibor TA, Ferreira F, Sousa AL, Amorim MJ
Nature communications 2019 Apr 9;10(1):1629
Nature communications 2019 Apr 9;10(1):1629
Endocytosis of Activated Muscarinic m2 Receptor (m2R) in Live Mouse Hippocampal Neurons Occurs via a Clathrin-Dependent Pathway.
Lambert L, Dubayle D, Fafouri A, Herzog E, Csaba Z, Dournaud P, El Mestikawy S, Bernard V
Frontiers in cellular neuroscience 2018;12:450
Frontiers in cellular neuroscience 2018;12:450
StAR-related lipid transfer domain 11 (STARD11)-mediated ceramide transport mediates extracellular vesicle biogenesis.
Fukushima M, Dasgupta D, Mauer AS, Kakazu E, Nakao K, Malhi H
The Journal of biological chemistry 2018 Sep 28;293(39):15277-15289
The Journal of biological chemistry 2018 Sep 28;293(39):15277-15289
Endoplasmic reticulum, Golgi, and lysosomes are disorganized in lung fibroblasts from chronic obstructive pulmonary disease patients.
Weidner J, Jarenbäck L, Åberg I, Westergren-Thorsson G, Ankerst J, Bjermer L, Tufvesson E
Physiological reports 2018 Mar;6(5)
Physiological reports 2018 Mar;6(5)
Mitochondrial fragmentation affects neither the sensitivity to TNFα-induced apoptosis of Brucella-infected cells nor the intracellular replication of the bacteria.
Lobet E, Willemart K, Ninane N, Demazy C, Sedzicki J, Lelubre C, De Bolle X, Renard P, Raes M, Dehio C, Letesson JJ, Arnould T
Scientific reports 2018 Mar 26;8(1):5173
Scientific reports 2018 Mar 26;8(1):5173
Subcellular Localization and Functional Characterization of GII.4 Norovirus-Encoded NTPase.
Yen JB, Wei LH, Chen LW, Chen LY, Hung CH, Wang SS, Chang PJ
Journal of virology 2018 Mar 1;92(5)
Journal of virology 2018 Mar 1;92(5)
Endoplasmic reticulum-resident protein 57 (ERp57) oxidatively inactivates human transglutaminase 2.
Yi MC, Melkonian AV, Ousey JA, Khosla C
The Journal of biological chemistry 2018 Feb 23;293(8):2640-2649
The Journal of biological chemistry 2018 Feb 23;293(8):2640-2649
Vesicular acetylcholine transporter (VAChT) over-expression induces major modifications of striatal cholinergic interneuron morphology and function.
Janickova H, Prado VF, Prado MAM, El Mestikawy S, Bernard V
Journal of neurochemistry 2017 Sep;142(6):857-875
Journal of neurochemistry 2017 Sep;142(6):857-875
Evaluation of the selectivity and sensitivity of isoform- and mutation-specific RAS antibodies.
Waters AM, Ozkan-Dagliyan I, Vaseva AV, Fer N, Strathern LA, Hobbs GA, Tessier-Cloutier B, Gillette WK, Bagni R, Whiteley GR, Hartley JL, McCormick F, Cox AD, Houghton PJ, Huntsman DG, Philips MR, Der CJ
Science signaling 2017 Sep 26;10(498)
Science signaling 2017 Sep 26;10(498)
HLA-DP(84Gly) constitutively presents endogenous peptides generated by the class I antigen processing pathway.
Yamashita Y, Anczurowski M, Nakatsugawa M, Tanaka M, Kagoya Y, Sinha A, Chamoto K, Ochi T, Guo T, Saso K, Butler MO, Minden MD, Kislinger T, Hirano N
Nature communications 2017 May 10;8:15244
Nature communications 2017 May 10;8:15244
Conserved Gene Microsynteny Unveils Functional Interaction Between Protein Disulfide Isomerase and Rho Guanine-Dissociation Inhibitor Families.
Moretti AIS, Pavanelli JC, Nolasco P, Leisegang MS, Tanaka LY, Fernandes CG, Wosniak J Jr, Kajihara D, Dias MH, Fernandes DC, Jo H, Tran NV, Ebersberger I, Brandes RP, Bonatto D, Laurindo FRM
Scientific reports 2017 Dec 8;7(1):17262
Scientific reports 2017 Dec 8;7(1):17262
Succination of Protein Disulfide Isomerase Links Mitochondrial Stress and Endoplasmic Reticulum Stress in the Adipocyte During Diabetes.
Manuel AM, Walla MD, Faccenda A, Martin SL, Tanis RM, Piroli GG, Adam J, Kantor B, Mutus B, Townsend DM, Frizzell N
Antioxidants & redox signaling 2017 Dec 1;27(16):1281-1296
Antioxidants & redox signaling 2017 Dec 1;27(16):1281-1296
EMC3 coordinates surfactant protein and lipid homeostasis required for respiration.
Tang X, Snowball JM, Xu Y, Na CL, Weaver TE, Clair G, Kyle JE, Zink EM, Ansong C, Wei W, Huang M, Lin X, Whitsett JA
The Journal of clinical investigation 2017 Dec 1;127(12):4314-4325
The Journal of clinical investigation 2017 Dec 1;127(12):4314-4325
Embryonic lethal abnormal vision proteins and adenine and uridine-rich element mRNAs after global cerebral ischemia and reperfusion in the rat.
Wang H, Tri Anggraini F, Chen X, DeGracia DJ
Journal of cerebral blood flow and metabolism : official journal of the International Society of Cerebral Blood Flow and Metabolism 2017 Apr;37(4):1494-1507
Journal of cerebral blood flow and metabolism : official journal of the International Society of Cerebral Blood Flow and Metabolism 2017 Apr;37(4):1494-1507
Peri/Epicellular Protein Disulfide Isomerase Sustains Vascular Lumen Caliber Through an Anticonstrictive Remodeling Effect.
Tanaka LY, Araújo HA, Hironaka GK, Araujo TL, Takimura CK, Rodriguez AI, Casagrande AS, Gutierrez PS, Lemos-Neto PA, Laurindo FR
Hypertension (Dallas, Tex. : 1979) 2016 Mar;67(3):613-22
Hypertension (Dallas, Tex. : 1979) 2016 Mar;67(3):613-22
An ERp57-mediated disulphide exchange promotes the interaction between Burkholderia cenocepacia and epithelial respiratory cells.
Pacello F, D'Orazio M, Battistoni A
Scientific reports 2016 Feb 16;6:21140
Scientific reports 2016 Feb 16;6:21140
Optogenetic control of organelle transport and positioning.
van Bergeijk P, Adrian M, Hoogenraad CC, Kapitein LC
Nature 2015 Feb 5;518(7537):111-114
Nature 2015 Feb 5;518(7537):111-114
Optogenetic acidification of synaptic vesicles and lysosomes.
Rost BR, Schneider F, Grauel MK, Wozny C, Bentz C, Blessing A, Rosenmund T, Jentsch TJ, Schmitz D, Hegemann P, Rosenmund C
Nature neuroscience 2015 Dec;18(12):1845-1852
Nature neuroscience 2015 Dec;18(12):1845-1852
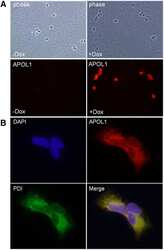
Biogenesis and cytotoxicity of APOL1 renal risk variant proteins in hepatocytes and hepatoma cells.
Cheng D, Weckerle A, Yu Y, Ma L, Zhu X, Murea M, Freedman BI, Parks JS, Shelness GS
Journal of lipid research 2015 Aug;56(8):1583-93
Journal of lipid research 2015 Aug;56(8):1583-93
An analysis of critical factors for quantitative immunoblotting.
Janes KA
Science signaling 2015 Apr 7;8(371):rs2
Science signaling 2015 Apr 7;8(371):rs2
Effects of oxidative stress on the solubility of HRD1, a ubiquitin ligase implicated in Alzheimer's disease.
Saito R, Kaneko M, Kitamura Y, Takata K, Kawada K, Okuma Y, Nomura Y
PloS one 2014;9(5):e94576
PloS one 2014;9(5):e94576
Characterization of N-glycosylation sites on the extracellular domain of NOX1/NADPH oxidase.
Matsumoto M, Katsuyama M, Iwata K, Ibi M, Zhang J, Zhu K, Nauseef WM, Yabe-Nishimura C
Free radical biology & medicine 2014 Mar;68:196-204
Free radical biology & medicine 2014 Mar;68:196-204
Transport of estradiol-17β-glucuronide, estrone-3-sulfate and taurocholate across the endoplasmic reticulum membrane: evidence for different transport systems.
Wlcek K, Hofstetter L, Stieger B
Biochemical pharmacology 2014 Mar 1;88(1):106-18
Biochemical pharmacology 2014 Mar 1;88(1):106-18
Oxidoreductase activity is necessary for N-glycosylation of cysteine-proximal acceptor sites in glycoproteins.
Cherepanova NA, Shrimal S, Gilmore R
The Journal of cell biology 2014 Aug 18;206(4):525-39
The Journal of cell biology 2014 Aug 18;206(4):525-39
Depletion of molecular chaperones from the endoplasmic reticulum and fragmentation of the Golgi apparatus associated with pathogenesis in Pelizaeus-Merzbacher disease.
Numata Y, Morimura T, Nakamura S, Hirano E, Kure S, Goto YI, Inoue K
The Journal of biological chemistry 2013 Mar 15;288(11):7451-7466
The Journal of biological chemistry 2013 Mar 15;288(11):7451-7466
mRNA redistribution during permanent focal cerebral ischemia.
Lewis MK, Jamison JT, Dunbar JC, DeGracia DJ
Translational stroke research 2013 Dec;4(6):604-17
Translational stroke research 2013 Dec;4(6):604-17
Inhibiting rotavirus infection by membrane-impermeant thiol/disulfide exchange blockers and antibodies against protein disulfide isomerase.
Calderon MN, Guerrero CA, Acosta O, Lopez S, Arias CF
Intervirology 2012;55(6):451-64
Intervirology 2012;55(6):451-64
Derlin-1 and UBXD8 are engaged in dislocation and degradation of lipidated ApoB-100 at lipid droplets.
Suzuki M, Otsuka T, Ohsaki Y, Cheng J, Taniguchi T, Hashimoto H, Taniguchi H, Fujimoto T
Molecular biology of the cell 2012 Mar;23(5):800-10
Molecular biology of the cell 2012 Mar;23(5):800-10
Unraveling the human dendritic cell phagosome proteome by organellar enrichment ranking.
Buschow SI, Lasonder E, Szklarczyk R, Oud MM, de Vries IJ, Figdor CG
Journal of proteomics 2012 Feb 16;75(5):1547-62
Journal of proteomics 2012 Feb 16;75(5):1547-62
Protective role of Gipie, a Girdin family protein, in endoplasmic reticulum stress responses in endothelial cells.
Matsushita E, Asai N, Enomoto A, Kawamoto Y, Kato T, Mii S, Maeda K, Shibata R, Hattori S, Hagikura M, Takahashi K, Sokabe M, Murakumo Y, Murohara T, Takahashi M
Molecular biology of the cell 2011 Mar 15;22(6):736-47
Molecular biology of the cell 2011 Mar 15;22(6):736-47
Elimination of hepatitis C virus from hepatocytes by a selective activation of therapeutic molecules.
Wen X, Abe T, Kukihara H, Taguwa S, Mori Y, Tani H, Kato N, Suzuki T, Tatsumi M, Moriishi K, Matsuura Y
PloS one 2011 Jan 6;6(1):e15967
PloS one 2011 Jan 6;6(1):e15967
Organelles do not colocalize with mRNA granules in post-ischemic neurons.
Jamison JT, Szymanski JJ, Degracia DJ
Neuroscience 2011 Dec 29;199:394-400
Neuroscience 2011 Dec 29;199:394-400
NLRP7, a nucleotide oligomerization domain-like receptor protein, is required for normal cytokine secretion and co-localizes with Golgi and the microtubule-organizing center.
Messaed C, Akoury E, Djuric U, Zeng J, Saleh M, Gilbert L, Seoud M, Qureshi S, Slim R
The Journal of biological chemistry 2011 Dec 16;286(50):43313-23
The Journal of biological chemistry 2011 Dec 16;286(50):43313-23
mRNA escape from stress granule sequestration is dictated by localization to the endoplasmic reticulum.
Unsworth H, Raguz S, Edwards HJ, Higgins CF, Yagüe E
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2010 Sep;24(9):3370-80
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2010 Sep;24(9):3370-80
Ero1alpha is expressed on blood platelets in association with protein-disulfide isomerase and contributes to redox-controlled remodeling of alphaIIbbeta3.
Swiatkowska M, Padula G, Michalec L, Stasiak M, Skurzynski S, Cierniewski CS
The Journal of biological chemistry 2010 Sep 24;285(39):29874-83
The Journal of biological chemistry 2010 Sep 24;285(39):29874-83
Mobile ER-to-Golgi but not post-Golgi membrane transport carriers disappear during the terminal myogenic differentiation.
Nevalainen M, Kaisto T, Metsikkö K
Cell and tissue research 2010 Oct;342(1):107-16
Cell and tissue research 2010 Oct;342(1):107-16
Production of anti-carbohydrate antibodies by phage display technologies: potential impairment of cell growth as a result of endogenous expression.
Yuasa N, Zhang W, Goto T, Sakaue H, Matsumoto-Takasaki A, Kimura M, Ohshima H, Tsuchida Y, Koizumi T, Sakai K, Kojima T, Yamamoto K, Nakata M, Fujita-Yamaguchi Y
The Journal of biological chemistry 2010 Oct 1;285(40):30587-97
The Journal of biological chemistry 2010 Oct 1;285(40):30587-97
Regulation of ER molecular chaperone prevents bone loss in a murine model for osteoporosis.
Hino S, Kondo S, Yoshinaga K, Saito A, Murakami T, Kanemoto S, Sekiya H, Chihara K, Aikawa Y, Hara H, Kudo T, Sekimoto T, Funamoto T, Chosa E, Imaizumi K
Journal of bone and mineral metabolism 2010 Mar;28(2):131-8
Journal of bone and mineral metabolism 2010 Mar;28(2):131-8
Biomechanical stress induces novel arterial intima-enriched genes: implications for vascular adaptation to stress.
Pyle AL, Li B, Maupin AB, Guzman RJ, Crimmins DL, Olson S, Atkinson JB, Young PP
Cardiovascular pathology : the official journal of the Society for Cardiovascular Pathology 2010 Mar-Apr;19(2):e13-20
Cardiovascular pathology : the official journal of the Society for Cardiovascular Pathology 2010 Mar-Apr;19(2):e13-20
Loss of HRD1-mediated protein degradation causes amyloid precursor protein accumulation and amyloid-beta generation.
Kaneko M, Koike H, Saito R, Kitamura Y, Okuma Y, Nomura Y
The Journal of neuroscience : the official journal of the Society for Neuroscience 2010 Mar 17;30(11):3924-32
The Journal of neuroscience : the official journal of the Society for Neuroscience 2010 Mar 17;30(11):3924-32
DC-STAMP interacts with ER-resident transcription factor LUMAN which becomes activated during DC maturation.
Eleveld-Trancikova D, Sanecka A, van Hout-Kuijer MA, Looman MW, Hendriks IA, Jansen BJ, Adema GJ
Molecular immunology 2010 Jul;47(11-12):1963-73
Molecular immunology 2010 Jul;47(11-12):1963-73
Membrane topology of human AGPAT3 (LPAAT3).
Schmidt JA, Yvone GM, Brown WJ
Biochemical and biophysical research communications 2010 Jul 9;397(4):661-7
Biochemical and biophysical research communications 2010 Jul 9;397(4):661-7
Characterization of the expression, localization, and secretion of PANDER in alpha-cells.
Carnegie JR, Robert-Cooperman CE, Wu J, Young RA, Wolf BA, Burkhardt BR
Molecular and cellular endocrinology 2010 Aug 30;325(1-2):36-45
Molecular and cellular endocrinology 2010 Aug 30;325(1-2):36-45
Protein disulfide isomerase (PDI) associates with NADPH oxidase and is required for phagocytosis of Leishmania chagasi promastigotes by macrophages.
Santos CX, Stolf BS, Takemoto PV, Amanso AM, Lopes LR, Souza EB, Goto H, Laurindo FR
Journal of leukocyte biology 2009 Oct;86(4):989-98
Journal of leukocyte biology 2009 Oct;86(4):989-98
Calsyntenins mediate TGN exit of APP in a kinesin-1-dependent manner.
Ludwig A, Blume J, Diep TM, Yuan J, Mateos JM, Leuthäuser K, Steuble M, Streit P, Sonderegger P
Traffic (Copenhagen, Denmark) 2009 May;10(5):572-89
Traffic (Copenhagen, Denmark) 2009 May;10(5):572-89
Comparative proteomics profiling of a phospholamban mutant mouse model of dilated cardiomyopathy reveals progressive intracellular stress responses.
Gramolini AO, Kislinger T, Alikhani-Koopaei R, Fong V, Thompson NJ, Isserlin R, Sharma P, Oudit GY, Trivieri MG, Fagan A, Kannan A, Higgins DG, Huedig H, Hess G, Arab S, Seidman JG, Seidman CE, Frey B, Perry M, Backx PH, Liu PP, MacLennan DH, Emili A
Molecular & cellular proteomics : MCP 2008 Mar;7(3):519-33
Molecular & cellular proteomics : MCP 2008 Mar;7(3):519-33
Endoplasmic reticulum (ER) chaperone regulation and survival of cells compensating for deficiency in the ER stress response kinase, PERK.
Yamaguchi Y, Larkin D, Lara-Lemus R, Ramos-Castañeda J, Liu M, Arvan P
The Journal of biological chemistry 2008 Jun 20;283(25):17020-9
The Journal of biological chemistry 2008 Jun 20;283(25):17020-9
Protein disulphide isomerase in platelet function.
Manickam N, Sun X, Li M, Gazitt Y, Essex DW
British journal of haematology 2008 Jan;140(2):223-9
British journal of haematology 2008 Jan;140(2):223-9
Interaction and functional association of protein disulfide isomerase with alphaVbeta3 integrin on endothelial cells.
Swiatkowska M, Szymański J, Padula G, Cierniewski CS
The FEBS journal 2008 Apr;275(8):1813-23
The FEBS journal 2008 Apr;275(8):1813-23
Dynamics of somatostatin type 2A receptor cargoes in living hippocampal neurons.
Lelouvier B, Tamagno G, Kaindl AM, Roland A, Lelievre V, Le Verche V, Loudes C, Gressens P, Faivre-Baumann A, Lenkei Z, Dournaud P
The Journal of neuroscience : the official journal of the Society for Neuroscience 2008 Apr 23;28(17):4336-49
The Journal of neuroscience : the official journal of the Society for Neuroscience 2008 Apr 23;28(17):4336-49
Identification of a novel isoform of microsomal triglyceride transfer protein.
Mohler PJ, Zhu MY, Blade AM, Ham AJ, Shelness GS, Swift LL
The Journal of biological chemistry 2007 Sep 14;282(37):26981-8
The Journal of biological chemistry 2007 Sep 14;282(37):26981-8
Activated somatostatin type 2 receptors traffic in vivo in central neurons from dendrites to the trans Golgi before recycling.
Csaba Z, Lelouvier B, Viollet C, El Ghouzzi V, Toyama K, Videau C, Bernard V, Dournaud P
Traffic (Copenhagen, Denmark) 2007 Jul;8(7):820-34
Traffic (Copenhagen, Denmark) 2007 Jul;8(7):820-34
The dendritic cell-derived protein DC-STAMP is highly conserved and localizes to the endoplasmic reticulum.
Eleveld-Trancikova D, Triantis V, Moulin V, Looman MW, Wijers M, Fransen JA, Lemckert AA, Havenga MJ, Figdor CG, Janssen RA, Adema GJ
Journal of leukocyte biology 2005 Mar;77(3):337-43
Journal of leukocyte biology 2005 Mar;77(3):337-43
Restricted distribution of mRNAs encoding a sarcoplasmic reticulum or transverse tubule protein in skeletal myofibers.
Nissinen M, Kaisto T, Salmela P, Peltonen J, Metsikkö K
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2005 Feb;53(2):217-27
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2005 Feb;53(2):217-27
A role for the thiol isomerase protein ERP5 in platelet function.
Jordan PA, Stevens JM, Hubbard GP, Barrett NE, Sage T, Authi KS, Gibbins JM
Blood 2005 Feb 15;105(4):1500-7
Blood 2005 Feb 15;105(4):1500-7
Regulation of NAD(P)H oxidase by associated protein disulfide isomerase in vascular smooth muscle cells.
Janiszewski M, Lopes LR, Carmo AO, Pedro MA, Brandes RP, Santos CX, Laurindo FR
The Journal of biological chemistry 2005 Dec 9;280(49):40813-9
The Journal of biological chemistry 2005 Dec 9;280(49):40813-9
A cellular UDP-glucose deficiency causes overexpression of glucose/oxygen-regulated proteins independent of the endoplasmic reticulum stress elements.
Flores-Diaz M, Higuita JC, Florin I, Okada T, Pollesello P, Bergman T, Thelestam M, Mori K, Alape-Giron A
The Journal of biological chemistry 2004 May 21;279(21):21724-31
The Journal of biological chemistry 2004 May 21;279(21):21724-31
Thiol/disulfide exchange is a prerequisite for CXCR4-tropic HIV-1 envelope-mediated T-cell fusion during viral entry.
Markovic I, Stantchev TS, Fields KH, Tiffany LJ, Tomiç M, Weiss CD, Broder CC, Strebel K, Clouse KA
Blood 2004 Mar 1;103(5):1586-94
Blood 2004 Mar 1;103(5):1586-94
Intracellularly located misfolded glycoprotein hormone receptors associate with different chaperone proteins than their cognate wild-type receptors.
Mizrachi D, Segaloff DL
Molecular endocrinology (Baltimore, Md.) 2004 Jul;18(7):1768-77
Molecular endocrinology (Baltimore, Md.) 2004 Jul;18(7):1768-77
Enzymatically catalyzed disulfide exchange is required for platelet adhesion to collagen via integrin alpha2beta1.
Lahav J, Wijnen EM, Hess O, Hamaia SW, Griffiths D, Makris M, Knight CG, Essex DW, Farndale RW
Blood 2003 Sep 15;102(6):2085-92
Blood 2003 Sep 15;102(6):2085-92
Protein disulfide isomerase, a component of the estrogen receptor complex, is associated with Chlamydia trachomatis serovar E attached to human endometrial epithelial cells.
Davis CH, Raulston JE, Wyrick PB
Infection and immunity 2002 Jul;70(7):3413-8
Infection and immunity 2002 Jul;70(7):3413-8
Structural requirements for the apical sorting of human multidrug resistance protein 2 (ABCC2).
Nies AT, König J, Cui Y, Brom M, Spring H, Keppler D
European journal of biochemistry 2002 Apr;269(7):1866-76
European journal of biochemistry 2002 Apr;269(7):1866-76
Structural requirements for the apical sorting of human multidrug resistance protein 2 (ABCC2).
Nies AT, König J, Cui Y, Brom M, Spring H, Keppler D
European journal of biochemistry 2002 Apr;269(7):1866-76
European journal of biochemistry 2002 Apr;269(7):1866-76
Selective intracellular retention of extracellular matrix proteins and chaperones associated with pseudoachondroplasia.
Vranka J, Mokashi A, Keene DR, Tufa S, Corson G, Sussman M, Horton WA, Maddox K, Sakai L, Bächinger HP
Matrix biology : journal of the International Society for Matrix Biology 2001 Nov;20(7):439-50
Matrix biology : journal of the International Society for Matrix Biology 2001 Nov;20(7):439-50
Selective intracellular retention of extracellular matrix proteins and chaperones associated with pseudoachondroplasia.
Vranka J, Mokashi A, Keene DR, Tufa S, Corson G, Sussman M, Horton WA, Maddox K, Sakai L, Bächinger HP
Matrix biology : journal of the International Society for Matrix Biology 2001 Nov;20(7):439-50
Matrix biology : journal of the International Society for Matrix Biology 2001 Nov;20(7):439-50
Dimerization of P-selectin in platelets and endothelial cells.
Barkalow FJ, Barkalow KL, Mayadas TN
Blood 2000 Nov 1;96(9):3070-7
Blood 2000 Nov 1;96(9):3070-7
Protein-disulfide isomerase (PDI) in FRTL5 cells. pH-dependent thyroglobulin/PDI interactions determine a novel PDI function in the post-endoplasmic reticulum of thyrocytes.
Mezghrani A, Courageot J, Mani JC, Pugniere M, Bastiani P, Miquelis R
The Journal of biological chemistry 2000 Jan 21;275(3):1920-9
The Journal of biological chemistry 2000 Jan 21;275(3):1920-9
Up-regulation of protein-disulfide isomerase in response to hypoxia/brain ischemia and its protective effect against apoptotic cell death.
Tanaka S, Uehara T, Nomura Y
The Journal of biological chemistry 2000 Apr 7;275(14):10388-93
The Journal of biological chemistry 2000 Apr 7;275(14):10388-93
Sulfhydryl regulation of L-selectin shedding: phenylarsine oxide promotes activation-independent L-selectin shedding from leukocytes.
Bennett TA, Edwards BS, Sklar LA, Rogelj S
Journal of immunology (Baltimore, Md. : 1950) 2000 Apr 15;164(8):4120-9
Journal of immunology (Baltimore, Md. : 1950) 2000 Apr 15;164(8):4120-9
Sulfhydryl regulation of L-selectin shedding: phenylarsine oxide promotes activation-independent L-selectin shedding from leukocytes.
Bennett TA, Edwards BS, Sklar LA, Rogelj S
Journal of immunology (Baltimore, Md. : 1950) 2000 Apr 15;164(8):4120-9
Journal of immunology (Baltimore, Md. : 1950) 2000 Apr 15;164(8):4120-9
Inhibition of human immunodeficiency virus infection by agents that interfere with thiol-disulfide interchange upon virus-receptor interaction.
Ryser HJ, Levy EM, Mandel R, DiSciullo GJ
Proceedings of the National Academy of Sciences of the United States of America 1994 May 10;91(10):4559-63
Proceedings of the National Academy of Sciences of the United States of America 1994 May 10;91(10):4559-63
Protein disulphide-isomerase from human placenta and rat liver. Purification and immunological characterization with monoclonal antibodies.
Kaetzel CS, Rao CK, Lamm ME
The Biochemical journal 1987 Jan 1;241(1):39-47
The Biochemical journal 1987 Jan 1;241(1):39-47
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
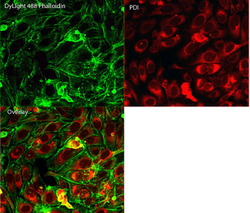
- Immunofluorescent analysis of Phalloidin (green) and PDI (red) in U2OS cells. Formalin fixed cells were permeabilized with 0.1% Triton X-100 in PBS for 10 minutes at room temperature and blocked with 2% BSA (Product # 37525) in PBS + 0.1% Triton X-100 for 30 minutes at room temperature. Cells were probed with a PDI monoclonal antibody (Product # MA3-019) at a dilution of 1:75 for at least 1 hour at room temperature, washed with PBS, and incubated with DyLight 550 goat anti-mouse IgG secondary antibody (Product # 84540) at a dilution of 1:250 for 30 minutes at room temperature. Actin was stained with DyLight 488 Phalloidin (Product # 21833) at a dilution of 1:300 (1 unit/mL final concentration) for 30 minutes. Images were taken on a Thermo Scientific ArrayScan VTI at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of PDI (red) in MDCK cells. The cells were permeabilized with 0.1% Triton X-100 in PBS for 15 minutes, and blocked with 3% BSA in PBS (Product # 37525) for 15 minutes at room temperature. Cells were stained with a PDI mouse monoclonal antibody (Product # MA3-019), at a concentration of 10 µg/mL in blocking buffer for at least 1 hour at room temperature, and then incubated with a Rabbit anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 594 conjugate (Product # A27027) at a dilution of 1:1000 for 30 minutes at room temperature (red). Nuclei (blue) were stained with Hoechst 33342 dye (Product # 62249). Images were taken on a Thermo Scientific ToxInsight Instrument at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
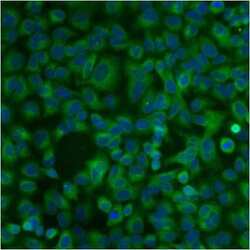
- Immunofluorescent analysis of PDI using anti-PDI monoclonal antibody (Product # MA3-019) shows staining in A549 Cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
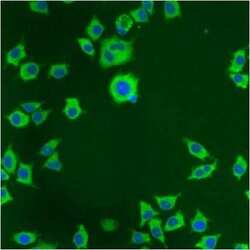
- Immunofluorescent analysis of PDI using anti-PDI monoclonal antibody (Product # MA3-019) shows staining in NS-1 Cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
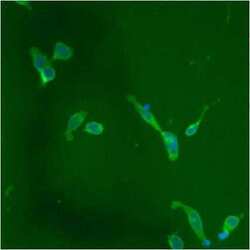
- Immunofluorescent analysis of PDI using anti-PDI monoclonal antibody (Product # MA3-019) shows staining in p19 Cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
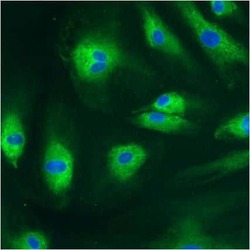
- Experimental details
- Immunofluorescent analysis of PDI using anti-PDI monoclonal antibody (Product # MA3-019) shows staining in HMVEC Cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
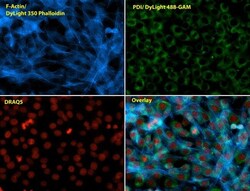
- Experimental details
- Immunofluorescent analysis of Phalloidin (blue) and PDI (green) in NIH 3T3 cells. Formalin fixed cells were permeabilized with 0.1% Triton X-100 in PBS for 10 minutes at room temperature and blocked with 2% BSA in PBS + 0.1% Triton X-100 (Product # 37525) for 30 minutes at room temperature. Cells were probed with a PDI monoclonal antibody (Product # MA3-019) at a dilution of 1:75 for at least 1 hour at room temperature, washed with PBS, and incubated with Dylight 488 goat anti-mouse IgG secondary antibody (Product # 35502) at a dilution of 1:250 for 30 minutes at room temperature. Actin was stained with Dylight 350 Phalloidin (Product # 21830) at a dilution of 1:120 (2.5units/mL final concentration) and nuclei (red) were stained with DRAQ5 (Product # 62254) at a concentration of 1 µg/mL for 30 minutes. Images were taken on a Zeiss Axio Observer Z1 microscope at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
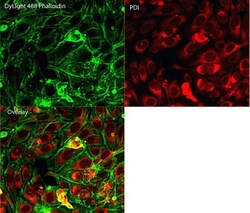
- Experimental details
- Immunofluorescent analysis of Phalloidin (green) and PDI (red) in U2OS cells. Formalin fixed cells were permeabilized with 0.1% Triton X-100 in PBS for 10 minutes at room temperature and blocked with 2% BSA (Product # 37525) in PBS + 0.1% Triton X-100 for 30 minutes at room temperature. Cells were probed with a PDI monoclonal antibody (Product # MA3-019) at a dilution of 1:75 for at least 1 hour at room temperature, washed with PBS, and incubated with DyLight 550 goat anti-mouse IgG secondary antibody (Product # 84540) at a dilution of 1:250 for 30 minutes at room temperature. Actin was stained with DyLight 488 Phalloidin (Product # 21833) at a dilution of 1:300 (1 unit/mL final concentration) for 30 minutes. Images were taken on a Thermo Scientific ArrayScan VTI at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
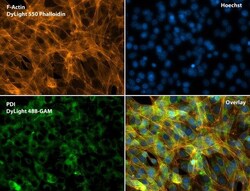
- Experimental details
- Immunofluorescent analysis of Phalloidin (orange) and PDI (green) in NIH 3T3 cells. Formalin fixed cells were permeabilized with 0.1% Triton X-100 in PBS for 10 minutes at room temperature and blocked with 2% BSA (Product # 37525) in PBS + 0.1% Triton X-100 for 30 minutes at room temperature. Cells were probed with a PDI monoclonal antibody (Product # MA3-019) at a dilution of 1:75 for at least 1 hour at room temperature, washed with PBS, and incubated with DyLight 488 goat anti-mouse IgG secondary antibody (Product # 35502) at a dilution of 1:250 for 30 minutes at room temperature. Actin was stained with DyLight 550 Phalloidin (Product # 21835) at a dilution of 1:120 (2.5 units/mL final concentration) and nuclei (blue) were stained with Hoechst (Product # 62249) at a concentration of 1 µg/mL for 30 minutes. Images were taken on a Zeiss Axio Observer Z1 microscope at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
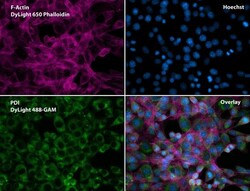
- Experimental details
- Immunofluorescent analysis of Phalloidin (purple) and PDI (green) in NIH 3T3 cells. Formalin fixed cells were permeabilized with 0.1% Triton X-100 in PBS for 10 minutes at room temperature and blocked with 2% BSA (Product # 37525) in PBS + 0.1% Triton X-100 for 30 minutes at room temperature. Cells were probed with a PDI monoclonal antibody (Product # MA3-019) at a dilution of 1:75 for at least 1 hour at room temperature, washed with PBS, and incubated with DyLight 488 goat anti-mouse IgG secondary antibody (Product # 35502) at a dilution of 1:250 for 30 minutes at room temperature. Actin was stained with DyLight 650 Phalloidin (Product # 21838) at a dilution of 1:120 (2.5 units/mL final concentration) and nuclei (blue) were stained with Hoechst (Product # 62249) at a concentration of 1 µg/mL for 30 minutes. Images were taken on a Zeiss Axio Observer Z1 microscope at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
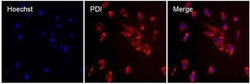
- Experimental details
- Immunofluorescent analysis of PDI (red) in MDCK cells. The cells were permeabilized with 0.1% Triton X-100 in PBS for 15 minutes, and blocked with 3% BSA in PBS (Product # 37525) for 15 minutes at room temperature. Cells were stained with a PDI mouse monoclonal antibody (Product # MA3-019), at a concentration of 10 µg/mL in blocking buffer for at least 1 hour at room temperature, and then incubated with a Rabbit anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 594 conjugate (Product # A27027) at a dilution of 1:1000 for 30 minutes at room temperature (red). Nuclei (blue) were stained with Hoechst 33342 dye (Product # 62249). Images were taken on a Thermo Scientific ToxInsight Instrument at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
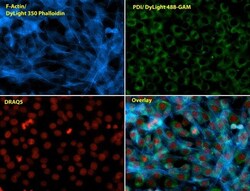
- Experimental details
- Immunofluorescent analysis of Phalloidin (blue) and PDI (green) in NIH 3T3 cells. Formalin fixed cells were permeabilized with 0.1% Triton X-100 in PBS for 10 minutes at room temperature and blocked with 2% BSA in PBS + 0.1% Triton X-100 (Product # 37525) for 30 minutes at room temperature. Cells were probed with a PDI monoclonal antibody (Product # MA3-019) at a dilution of 1:75 for at least 1 hour at room temperature, washed with PBS, and incubated with Dylight 488 goat anti-mouse IgG secondary antibody (Product # 35502) at a dilution of 1:250 for 30 minutes at room temperature. Actin was stained with Dylight 350 Phalloidin (Product # 21830) at a dilution of 1:120 (2.5units/mL final concentration) and nuclei (red) were stained with DRAQ5 (Product # 62254) at a concentration of 1 µg/mL for 30 minutes. Images were taken on a Zeiss Axio Observer Z1 microscope at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
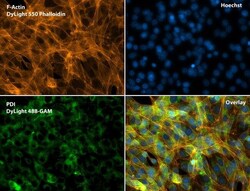
- Experimental details
- Immunofluorescent analysis of Phalloidin (orange) and PDI (green) in NIH 3T3 cells. Formalin fixed cells were permeabilized with 0.1% Triton X-100 in PBS for 10 minutes at room temperature and blocked with 2% BSA (Product # 37525) in PBS + 0.1% Triton X-100 for 30 minutes at room temperature. Cells were probed with a PDI monoclonal antibody (Product # MA3-019) at a dilution of 1:75 for at least 1 hour at room temperature, washed with PBS, and incubated with DyLight 488 goat anti-mouse IgG secondary antibody (Product # 35502) at a dilution of 1:250 for 30 minutes at room temperature. Actin was stained with DyLight 550 Phalloidin (Product # 21835) at a dilution of 1:120 (2.5 units/mL final concentration) and nuclei (blue) were stained with Hoechst (Product # 62249) at a concentration of 1 µg/mL for 30 minutes. Images were taken on a Zeiss Axio Observer Z1 microscope at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
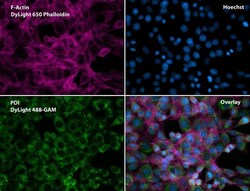
- Experimental details
- Immunofluorescent analysis of Phalloidin (purple) and PDI (green) in NIH 3T3 cells. Formalin fixed cells were permeabilized with 0.1% Triton X-100 in PBS for 10 minutes at room temperature and blocked with 2% BSA (Product # 37525) in PBS + 0.1% Triton X-100 for 30 minutes at room temperature. Cells were probed with a PDI monoclonal antibody (Product # MA3-019) at a dilution of 1:75 for at least 1 hour at room temperature, washed with PBS, and incubated with DyLight 488 goat anti-mouse IgG secondary antibody (Product # 35502) at a dilution of 1:250 for 30 minutes at room temperature. Actin was stained with DyLight 650 Phalloidin (Product # 21838) at a dilution of 1:120 (2.5 units/mL final concentration) and nuclei (blue) were stained with Hoechst (Product # 62249) at a concentration of 1 µg/mL for 30 minutes. Images were taken on a Zeiss Axio Observer Z1 microscope at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
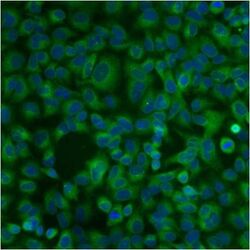
- Experimental details
- Immunofluorescent analysis of PDI using anti-PDI monoclonal antibody (Product # MA3-019) shows staining in A549 Cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
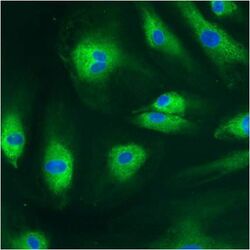
- Experimental details
- Immunofluorescent analysis of PDI using anti-PDI monoclonal antibody (Product # MA3-019) shows staining in HMVEC Cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
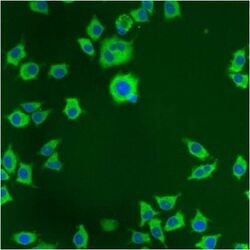
- Experimental details
- Immunofluorescent analysis of PDI using anti-PDI monoclonal antibody (Product # MA3-019) shows staining in NS-1 Cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
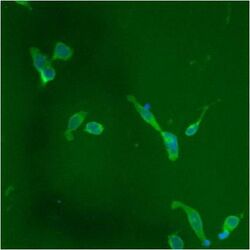
- Experimental details
- Immunofluorescent analysis of PDI using anti-PDI monoclonal antibody (Product # MA3-019) shows staining in p19 Cells.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
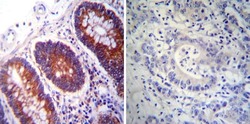
- Experimental details
- Immunohistochemistry was performed on normal deparaffinized Human colon tissue tissues. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:200 with a mouse monoclonal antibody recognizing PDI (Product # MA3-019) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
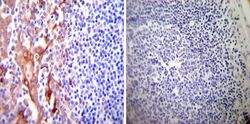
- Experimental details
- Immunohistochemistry was performed on normal deparaffinized Human tonsil tissue tissues. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:200 with a mouse monoclonal antibody recognizing PDI (Product # MA3-019) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
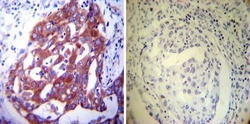
- Experimental details
- Immunohistochemistry was performed on cancer biopsies of deparaffinized Human lung adenocarcinoma tissues. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:200 with a mouse monoclonal antibody recognizing PDI (Product # MA3-019) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
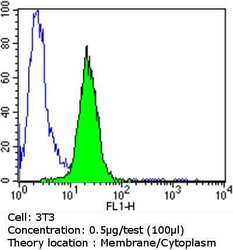
- Experimental details
- Flow cytometry analysis of PDI in NIH/3T3 cells compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a PDI monoclonal antibody (Product # MA3-019) at a dilution of 0.5 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated goat anti-mouse IgG (H+L) secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
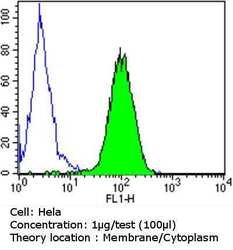
- Experimental details
- Flow cytometry analysis of PDI in Hela cells compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a PDI monoclonal antibody (Product # MA3-019) at a dilution of 1 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated goat anti-mouse IgG (H+L) secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
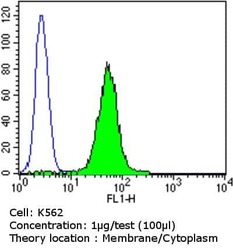
- Experimental details
- Flow cytometry analysis of PDI in K562 cells compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a PDI monoclonal antibody (Product # MA3-019) at a dilution of 1 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated goat anti-mouse IgG (H+L) secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
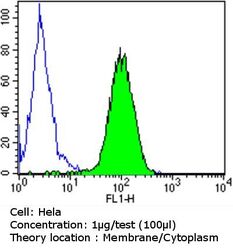
- Experimental details
- Flow cytometry analysis of PDI in Hela cells compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a PDI monoclonal antibody (Product # MA3-019) at a dilution of 1 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated goat anti-mouse IgG (H+L) secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
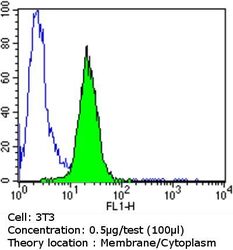
- Experimental details
- Flow cytometry analysis of PDI in NIH/3T3 cells compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a PDI monoclonal antibody (Product # MA3-019) at a dilution of 0.5 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated goat anti-mouse IgG (H+L) secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
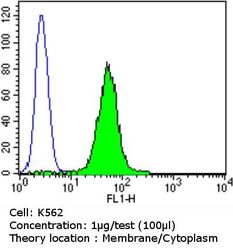
- Experimental details
- Flow cytometry analysis of PDI in K562 cells compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a PDI monoclonal antibody (Product # MA3-019) at a dilution of 1 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated goat anti-mouse IgG (H+L) secondary antibody and re-suspended in PBS for FACS analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
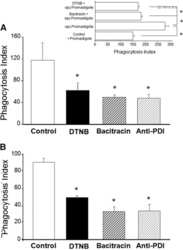
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
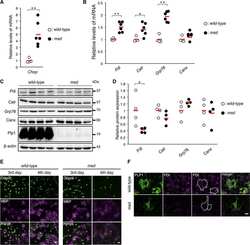
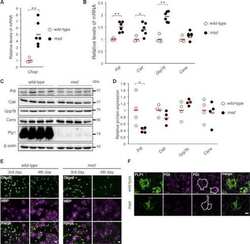
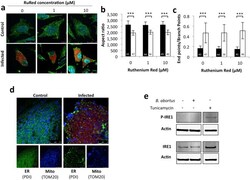
- Figure 4 Search for potential mechanisms involved in Brucella abortus -induced mitochondrial fragmentation ( a ). TOM20 immunostaining in HeLa cells pre-incubated for 30 min with the indicated concentration of ruthenium red and infected with B. abortus 2308 mCherry with or without the inhibitor - 48 h PI (Representative of n = 3). Green: TOM20 (Alexa488)/Red: B. abortus 2308 (mCherry)/Turquoise: Nuclei (Hoechst) ( b , c ). Aspect ratio ( b ) and end point/branch point ratio ( c ) of HeLa cells pre-incubated for 30 min with the indicated concentration of ruthenium red and infected (white) or not (black) with B. abortus 2308 mCherry with or without the inhibitor - 48 h PI. Results represent means +- SD for three independent experiments (n = 3). Statistical analysis: two-way ANOVA on Box Cox transformed data. P value for interaction: 0.446 and 0.051 respectively. The numbers indicated in the columns represent the number of cells analysed for each condition. ( d ) PDI and TOM20 co-immunostaining in HeLa cells infected or not (control) with B. abortus 2308 mCherry - 48 h PI (Representative of n = 3). Green: PDI (Alexa488)/Blue: TOM20 (Alexa633)/Red: B. abortus 2308 (mCherry)/Turquoise: Nuclei (Hoechst). ( e ) Western blot analysis of P-IRE1 and IRE1 abundance in RAW 264.7 macrophages infected or not with B. abortus 2308 mCherry - 48 h PI (Representative of n = 3 in different conditions). RAW 264.7 cells treated for 6 h with 10 uM tunicamycin and then left for 18 h for
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
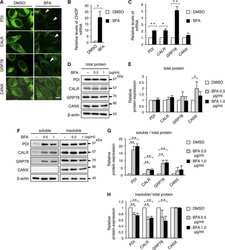
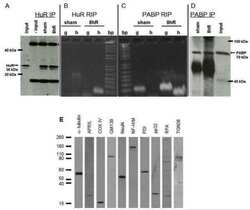
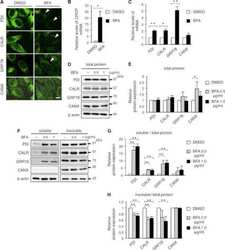
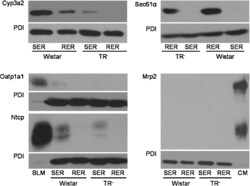
- Fig. 1 Western blot analysis of Cyp3a2, Sec61alpha, Oatp1a1, Ntcp, Mrp2, and PDI in SER and RER isolated from Wistar and TR - rat livers. 5 mug (Cyp3a2, Mrp2), 50 mug (Sec61alpha) or 100 mug (Oatp1a1, Ntcp) protein was loaded per lane and separated by SDS-PAGE as given in section 2.4. Samples were probed against Cyp3a2, Sec61alpha, Oatp1a1, Ntcp, Mrp2, and PDI as described in 2.4. PDI was used as loading control in SER and RER fractions. Basolateral membrane fraction (BLM) isolated from Sprague Dawley rat liver was used as positive control for Oatp1a1 and Ntcp expression. Canalicular (CM) membrane fraction isolated from Sprague Dawley rat liver was used as positive control for Mrp2 expression. The figure shows a representative Western blot of at least two independent SER and RER isolations.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
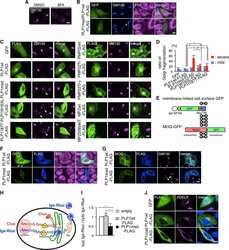
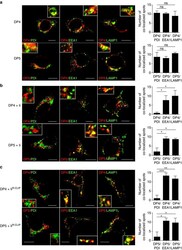
- Figure 4 Subcellular localization of DP4 and DP5 in the presence or absence of Ii. ( a - c ) HEK293 cells were transfected with the indicated combinations of genes. Fixed cells were permeabilized and stained for DP (red) in conjunction with an ER marker, PDI (green, left panels), an early endosomal marker, EEA1 (green, middle panels) or a late endosomal/lysosomal marker, LAMP-1 (green, right panels) and analysed by confocal microscopy. Inset boxes indicate the areas depicted at higher magnification. Scale bar in all images, 10 mum. Quantification of co-localized spots represents means+-s.d. of three counted cells in each condition. ns, not significant; * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
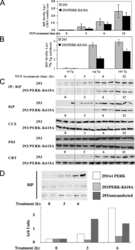
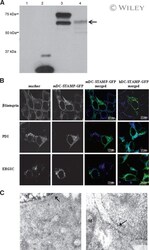
- 3 (A) Western blot analysis of HEK293 cells transfected with mDC-STAMP-GFP constructs. (Lane 1) HEK293 cells; (lane 2) HEK293/GFP; (lane 3) HEK293/mDC-STAMP-GFP; and (lane 4) HEK293/mDC-STAMP-GFP plus tunicamycin. Western blots were stained with anti-GFP mAb. The arrow indicates unglycosylated mDC-STAMP-GFP. (B) HEK293 cells, stably transfected with mDC-STAMP-GFP or hDC-STAMP-GFP constructs, respectively, were stained with antibodies against beta1 integrins, against the protein present in the ER (anti-PDI), and the intermediate compartment (anti-ERGIC-53). Cells adherent to the fibronectin-coated slides were visualized by CLSM for GFP autofluorescence (green) and the markers (blue) as indicated. (C) Immunoelectron microscopy of HEK293 cells stably transfected with hDC-STAMP-GFP. Sections were stained with the antibody against GFP and incubated with the 10-nm gold beads. The arrows point to the gold beads at the nuclear membrane (left) and in the ER (right). The other gold particles in the left panel are located on ER structures, as is more clearly shown in the right panel. No staining was visible at the mitochondria (M) or in the nucleus (N). Original magnification, 20,000x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
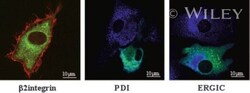
- 4 DC-STAMP-GFP localizes to the ER in DC. Mo-IDC transduced with the adenovirus Ad5fib35 encoding hDC-STAMP-GFP (green) were stained with a beta2-integrin antibody (red), an ER marker (anti-PDI, blue), and an intermediate compartment marker (anti-ERGIC-53, blue), and fluorescence was visualized by CLSM.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
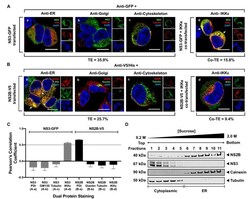
- Experimental details
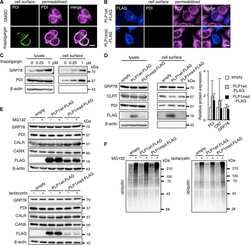
- Figure 2 WNV NS3 is distributed throughout the cytoplasm, while NS2B remains in the ER in transfected cells. ( A ) NS3-GFP transfected ( a - c ) or NS3-GFP and IKKepsilon-FLAG co-transfected ( d ), HEK293T cells were fixed at 24 h post-transfection and immunostained with antibodies against protein disulfide isomerase (PDI), an ER marker, (red; a ), anti-GM130, a Golgi apparatus marker, (red; b ), anti-tubulin, a cytoskeletal marker, (red; c ), or anti-IKKepsilon, a soluble cytoplasmic protein marker, (red; d ). ( B ) NS2B-V5/His transfected ( a - c ) or NS2B-V5/His and IKKepsilon-FLAG co-transfected ( d ) HEK293T cells were fixed 24 h after transfection and immunolabeled with rabbit anti-PDI (green; a ), anti-giantin (green; b ), anti-tubulin (green; c ) or anti-IKKepsilon (green; d ) antibodies to visualize the ER, Golgi apparatus, cytoskeleton or the cytoplasmic protein kinase IKKepsilon protein, respectively. Visualized viral and host proteins are listed on the top right corner of each panel. Nuclear DNA was labeled with DAPI. Confocal microscopy images were of optical slice thickness ~1 mum. Scale bar, 10 mum. The main image depicts the merged image from three channels separately shown on the side. White arrows indicate colocalization between the green and red channels. The transfection efficiency (TE) for cells transfected with one construct or the co-transfection efficiency (Co-TE) for cells co-expressing the viral protein and IKKepsilon are listed at the bottom of each
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
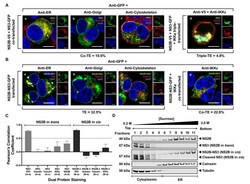
- Experimental details
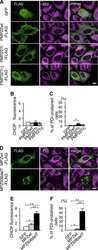
- Figure 3 NS3 localizes to the ER when NS2B is provided in trans and in cis. ( A ) HEK293T cells were co-transfected with NS3-GFP and NS2B-V5 expressing plasmids ( a - c ). At 24 h post-transfection, cells were fixed and co-stained with anti-GFP and antibodies against PDI (red; a ), giantin (red; b ) or tubulin (red; c ). HEK293T cells were also triple-transfected with NS3-GFP, NS2B-V5/His and IKKepsilon-FLAG expressing plasmids ( d ) and triple stained with anti-GFP (green; d ), anti-V5/His (red; d ) and anti-IKKepsilon (blue; d ) antibodies. ( B ) HEK293T cells were transfected with NS2B-NS3-GFP ( a - c ) or co-transfected with NS2B-NS3-GFP and IKKepsilon-FLAG expressing plasmids ( d ) and processed for immunofluorescence at 24 h after transfection using antibodies against PDI (red; a ), giantin (red; b ), tubulin (red; c ) and IKKepsilon (red; d ). The detected viral and host proteins are listed on the top right corner of each panel. White arrows indicate colocalization between the GFP-tagged viral protein and the ER marker. Nuclear DNA was labeled with DAPI. Scale bar represents 10 mum. The main panels depict merged images with the side panels showing the individual channels. The transfection efficiency (TE), the co-transfection efficiency (Co-TE) and the triple-transfection efficiency (Triple-TE) for cells expressing one, two, or three gene constructs, respectively, are listed at the bottom of the panel. Images are representative of two independent transfection experiment
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
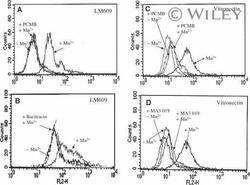
- Experimental details
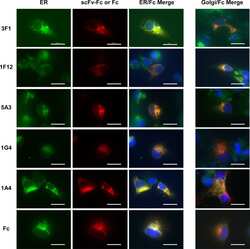
- 3 Increased binding of vitronectin and LM609 to Mn 2+ -stimulated endothelial cells requires free thiols in alpha V beta 3 and active PDI. Endothelial cells were stimulated with Mn 2+ , and their binding of LM609 (A, B) and vitronectin (C, D) was evaluated by flow cytometry in the absence or presence of the membrane-impermeant sulfhydryl group reagent PCMBS, a PDI-specific inhibitor (A, C), bacitracin (B), or blocking antibody to PDI (mAb 019) (D).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
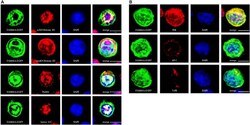
- Experimental details
- FIGURE 3 Confocal analysis of the subcellular localization of FAM46A in transfected K562 cells. Cells were transfected with an expression construct encoding the FAM46A-EGFP protein, which was detected in the nuclear (A) and cytoplasmic (B) compartments. Within the nucleus (A) , FAM46A was predominantly confined in the chromatin domains more accessible to the transcription complexes (as indicated by the acK9-Histone H3 staining) and were mostly excluded from the regions with condensed chromatin structures (as indicated by the meK9-Histone H3 staining). In addition, significant co-localization of FAM46A with NuMA and lamin A/C was observed. DAPI staining defined the morphology of nuclei. Scale bar: 5 mum. Within the cytoplasm (B) , the majority of FAM46A was found to co-localize with the endoplasmic reticulum marker, protein disulfide-isomerase (PDI). By contrast, co-localization of FAM46A with trans-Golgi network marker gamma-adaptin (AP-1) and early endosome marker transferrin receptor (TrfR) is relatively low. DAPI staining defines the morphology of nuclei. Scale bar: 5 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
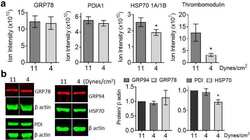
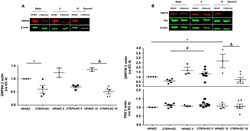
- 10.1371/journal.pone.0242960.g003 Fig 3 Endoplasmic reticulum chaperones are downregulated in CTEPH-EC. (A) Representative blot of the GRP94 expression in static, low, and high magnitude shear stress (0, 5, and 15 dynes/cm 2 ). Data are presented as the mean +- standard error: (*) HPAEC vs CTEPH-EC, (&) HPAEC 15 vs CTEPH-EC 15. (n = 5-7). (B) GRP78 and PDI expression before and after shear stress exposure as described in methods. Data are presented as the mean +- standard error: (*) HPAEC vs HPAEC 15, (#) CTEPH-EC vs CTEPH-EC 5 (&) HPAEC 15 vs CTEPH-EC 15, P < 0.05. (n = 6-7). All of the samples were normalized to ss-actin and run in the same gel.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
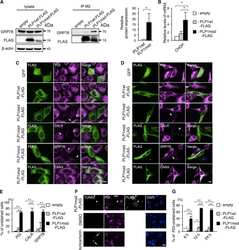
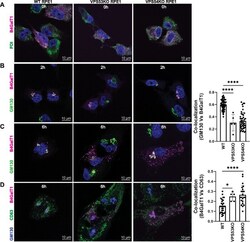
- FIGURE 5: RUSH assay reveals mislocalization of B4GalT1 to endolysosomes in GARP-KO RPE1 cells. RPE1 cells were transfected with plasmids encoding Str-KDEL_flB4GalT1-SBP-mCherry in biotin-free medium. Following overnight incubation, biotin (40 uM) and cycloheximide (50 uM) were added for 0, 2, and 6 h. (A) Colocalization of B4GalT1 with ER marker PDI at time 0. (B, C) Colocalization of B4GalT1 with Golgi protein GM130 at 2 h (B) and 6 h (C). The graph on the right side of C shows the quantification of B4GalT1 and GM130 colocalization at 6 h of biotin/cycloheximide addition. Values in the bar graph represent the mean +- SD of the colocalization between B4GalT1 and GM130 in approximately 40 cells in WT and VPS54-KOs. Colocalization in VPS53-KO cells was measured using each field in approximately 20 cells. Statistical significance was calculated using one-way ANOVA. **** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
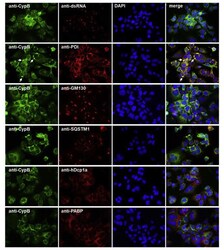
- Fig. 6 Co-immunostaining of CypB and cell organelles in HCoV-229E-infected Huh7 cells . For the identification of the intense cyclophilin B bleb-like structures infected cells (MOI = 1; 48 h p.i.) were co-stained with anti-CypB and antibodies directed against markers of the ER (anti-PDI), cis-GOLGI (anti-GM130), autophagosomes (anti-SQSTM1), anti-P-bodies (anti-hDcp1a) and stress granules (anti-PABP). Cyclophilin B normally distributes within the ER. In the presence of HCoV-229E, it intriguingly concentrates at bleb-like structures of the ER. Fig. 6
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- FIG 6 The LTM-1 and LTM-2 motifs of NTPase have the potential to direct fusion proteins to the ER. (A) Schematic diagram of various NTPase(1-179) deletion mutants. The ability and degree of NTPase(1-179)-GFP and its deletion mutants for LD localization and for ER localization are summarized. (B) Colocalization of NTPase(1-179) deletion constructs with LDs or with the ER in cells. A7 cells were transfected with the indicated expression plasmids for 24 h, and then the transfected cells were dually stained with LipidTOX and anti-PDI antibody; scale bars, 10 mum. The quantitative results are plotted as mean +- SEM ( n = 15 cells obtained from two independent experiments); *, P < 0.05 for results compared to those from the empty vector group (GFP); #, P < 0.05 for results compared to those from the NTPase(1-179)-GFP-transfected group. (C) Confocal microscopic analysis of the localization of NTPase(1-179)-GFP in cells at different time points (6, 18, and 24 h) after transfection. A7 cells were transfected with the NTPase(1-179)-GFP expression plasmid for 6, 18, or 24 h, and then the subcellular localization of this protein to LDs or the ER in transfected cells was analyzed by confocal microscopy; scale bars, 10 mum. The quantitative results are plotted as mean +- SEM ( n = 15 cells obtained from two independent experiments); *, P < 0.05 for results compared to those from the 6 h group.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
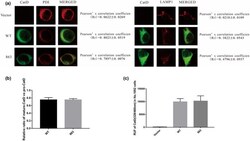
- Experimental details
- FIGURE 4 The intracellular localization of CatD and enzyme activity in No. 1002 cells. (a) The immunofluorescence cell localization of wild-type and mutant CatD expressed in No. 1002 cells. After transfecting vector, wild-type, and mutant plasmids into No. 1002 cells, the co-localization of the CatD protein (green) and lysosome (red), as well as the co-localization of the CatD protein (green) and endoplasmic reticulum (red) were observed. (b) The ratio of mature-CatD and pro-CatD in No. 1002 cells transfected with the wild-type and mutant of the CTSD gene, respectively. (c) The CatD activity of wild-type and mutant No. 1002 cells. CatD activity expressed as relative fluorescence units (RFU) per million cells
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
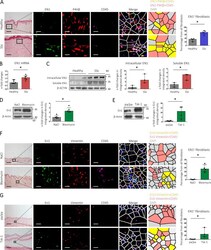
- Experimental details
- Figure 1. EN1 expression is up-regulated in fibrotic skin. (A) Representative immunofluorescence stainings for EN1 (green) and costainings with P4H (red) and CD45 (magenta) in the dermis of healthy donors and SSc patients at 1,000-fold magnification (scale bars = 20 um). Hematoxylin and eosin stainings (200-fold magnification, scale bars = 100 um), Voronoi diagrams, and percentages of EN1-positive fibroblasts from all fibroblasts (defined as P4H + CD45 - cells) are included. Data are shown from one experiment ( n >= 4). (B and C) EN1 mRNA (B) and EN1 intracellular or secreted protein levels (C) in cultured fibroblasts or their supernatants from healthy donors and SSc patients. Representative Western blot images and quantifications are included (C). Data are shown from one experiment ( n = 3). (D and E) En1 protein levels in the skin of mice challenged with bleomycin (D) or Tsk-1 mice (E) and their respective controls. Representative Western blot images and quantifications are included. Data are compiled from two independent experiments ( n >= 4). (F and G) Representative immunofluorescence stainings for EN1 (green) and costainings with Vimentin (red) and CD45 (magenta) in the dermis of mice challenged with bleomycin and their controls at 1,000-fold magnification (F; scale bars = 20 um) and in the hypodermis of Tsk-1 mice and their controls, respectively, at 1,000-fold magnification (G; scale bars = 20 um). Hematoxylin and eosin stainings (100-fold magnification, scale bars =
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
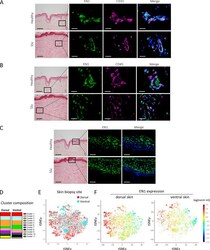
- Experimental details
- Figure S1. EN1 expression in healthy and SSc skin. (A-C) Representative immunofluorescence stainings for EN1 (green) and costainings with CD31 (A) and CD45 (B; both magenta) in the dermis of healthy donors and SSc patients at 1,000-fold magnification (scale bars = 20 um). Hematoxylin and eosin stainings (200-fold magnification, scale bars = 100 um) are included. Data for A-C were obtained from n >= 4 biological samples from two independent experiments. (D) Composition of clusters from the segmented IMC images in ventral and dorsal skin biopsies from SSc patients. (E) tSNE plot showing distribution of cells from ventral and dorsal SSc skin across clusters. (F) tSNE plot showing expression of EN1 across the ventral and dorsal SSc clusters. Data for D-F were obtained from n = 8 biological samples from four SSc patients (paired ventral and dorsal biopsies).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
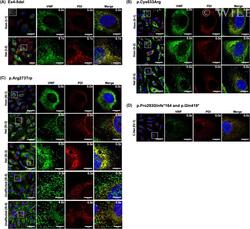
- Experimental details
- 3 FIGURE ER retention of VWF in ECFCs with VWF propeptide variants. A, Ex 4-5del; B, p.Cys633Arg; C, p.Arg273Trp; D, p.Pro293Glnfs*164 and p.Gln419* ECFCs. Co-staining of ECFCs for VWF (green) and the ER marker PDI (red) shows co-localization (yellow) indicating ER retention of the variant (diffuse) VWF in heterozygous and homozygous VWF propeptide ECFCs. In normal ECFCs (Unaffected III-5, III-6), VWF is in WPBs and not associated with the ER marker. Images were obtained using a Leica TCS SP8 microscope at 63x magnification, upgraded with Hyvolution software (scale bars = 50 mum; and 10 mum for zoomed in panels). White boxes show the magnified area. The optical zoom is provided at the top right corner of each zoomed in image. C. Het, compound heterozygous; ECFCs, endothelial colony-forming cells; ER, endoplasmic reticulum; Het, heterozygous; Hom, homozygous; PDI, protein disulfide-isomerase; VWF, von Willebrand factor; WPBs, Weibel-Palade bodies
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
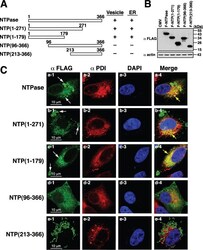
- Experimental details
- FIG 3 N-terminal 179-aa region of GII-NTPase is required for vesicle formation and ER colocalization. (A) Schematic diagram of the full-length and deletion constructs of F-NTPase. The abilities of F-NTPase and its deletions to promote vesicle formation and ER colocalization are summarized in the diagram. (B) Western blot analysis of F-NTPase and its deletions expressed in 293T cells. (C) Representative confocal immunofluorescence images of F-NTPase constructs (green) and PDI (red) in A7 cells. White arrows indicate the positions of vesicle-like structures formed by F-NTPase or its deletions. Yellow arrows represent the colocalization between the indicated F-NTPase constructs and PDI in the merged images. Scale bars, 10 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
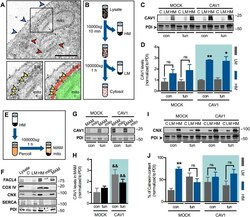
- Experimental details
- Fig. 3 Caveolin-1 accumulates in mitochondria-associated ER membranes, precluding their remodelling upon early-phase ER stress. a Wild-type HeLa cells were labelled by immunogold staining with antibodies against CAV1. Blue arrowheads indicate CAV1 signals at the plasma membrane; red arrowheads indicate CAV1 signals at ER cisternae; yellow arrowheads indicate CAV1 at the ER-mitochondria interface. In the lower right panel, the ER is highlighted in red and mitochondria (mito) in green. Scale bar: 1 um. b Cells lysates were fractionated by differential sedimentation. HM heavy membranes (crude mitochondria), LM light membranes (microsomes). c HeLa mock and CAV1 cells in control condition (con) or under early ER stress with tunicamycin (tun) were processed and fractionated as in b . CAV1 levels were analysed by western blotting using PDI as a loading control. d Quantification of membrane-bound CAV1 analysed in c ( n = 3). e HM fraction obtained as in ( b ) was separated into MAM and pure mitochondria (mito) fractions by Percoll density gradient centrifugation. f Markers of cell fractionation procedures shown in b and e : FACL4 short isoform (mito), long isoform (MAM); COX IV (mito), Calnexin (CNX) (LM and MAM), SERCA2b (LM) and PDI (C, LM and HM). g Experimental groups as in c were processed and fractionated as in e . CAV1 levels were analysed by western blotting using PDI as a loading control. h Quantification of CAV1 in MAM analysed in g ( n = 3). i Experimental groups as in c w
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 6 Caveolin-1 expression precludes DRP1 phosphorylation and redistribution during the early phase of ER stress. a Mock and CAV1 cells in control condition (con) or under early ER stress with tunicamycin (tun) were fractionated and processed for detection of phospho-DRP1 and DRP1 by western blotting using PDI as a loading control. C cytosol, LM light membranes (microsomes), HM heavy membranes (crude mitochondria). b Quantification of DRP1 phosphorylation analysed in ( a ) ( n = 3). c Quantification of membrane-bound DRP1 distribution analysed in a ( n = 3). d Experimental groups as in ( a ) were processed for immunofluorescence analysis. CAV1, mtHSP70 (mitochondrial marker, Mito) and DRP1 were stained with respective antibodies and then imaged using confocal microscopy. Merge between DRP1 and CAV1 images shows colocalized areas in yellow. Merge between DRP1 and Mito images shows colocalized areas in cyan. In merge images of Mito, DRP1 and CAV1, triple colocalization areas appear as white pixels. e CAV1 fluorescence per cell was quantified from images obtained in d ( n = 3). f DRP1-CAV1 colocalization was quantified as Manders' coefficients of images obtained in ( d ) ( n = 3). g DRP1-Mito colocalization was quantified as Manders' coefficients of images obtained in ( d ) ( n = 3). h From images obtained in d , thresholded CAV1 (C) and Mito (M) images were generated. The intersection between M and DRP1 corresponds to mitochondrial DRP1 (DRP1 mito ). Merge between M and C ima
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 1 Role of PDIA1 in oxidant generation by colon carcinoma cells with distinct levels of KRas activation. a KRas expression and activity in Caco2, HKE3, and HCT116 cells. Active KRas was pulled down with GST-RBD beads from lysates of serum-starved cells treated with EGF 25 ng/mL for 10 min. Aliquots of the lysates were blotted for total KRas and GAPDH as loading control. b RNAseq analysis of P4HB (PDIA1) gene in Caco2, HCT116, and HCT15 cells, showing mean expression in FPKM (Fragments Per Kilobase Million), P4HB fold-change expression in HCT15 vs. Caco2 (q < 0.000001) and HCT116 vs. Caco2 (q < 0.000001). c PDIA1 basal protein expression by western analysis, normalized for GAPDH ( n = 3) and quantified using Odyssey software. d Intracellular PDIA1 titration, using Human P4HB ELISA Pair Set (SinoBiological). Test t ** p < 0.01, ( n = 4); e-g ROS production after 72 h of PDIA1 silencing, measured by HPLC analysis of DHE oxidation products. DHE oxidation produces, among many others, two major products: 2-hidroxyethidium (EOH), which is representative of superoxide species and ethidium, representative of other oxidant species. Ctrl negative, si-RNA control; si-PDI, si-RNA against PDIA1 protein. Representative immunoblots of PDI silencing for each cell type Test t * p < 0.05; ** p < 0.01. For Caco2 cells, n = 3; HKE3 cells, n = 4; HCT116 cells, n = 5
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
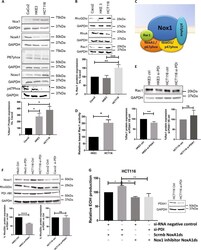
- Experimental details
- Fig. 2 Nox NADPH oxidase expression and superoxide generation by HCT116 cells with overactivated KRas. a Nox1, NoxA1, p67phox, NoxO1, and p47phox basal protein expression by western analysis normalized for GADPH. Nox1 immunoblot densities were quantified using Odyssey software. * P < 0.05, ANOVA plus Tukey's multiple comparisons test ( n = 3). b RhoGDIalpha, RhoA, and Rac1 basal protein expression by western analysis normalized for GAPDH. Rac1 immunoblot densities were quantified using Odyssey software. ** P < 0.01; *** P < 0.005, ANOVA plus Tukey's multiple comparisons test ( n = 3). c Nox1 complex scheme. d Rac1 basal activity, using Rac1 G-LISA activation assay (Cytoskeleton,inc). Test t * p < 0.05, ( n = 2). e Rac1 protein expression by western analysis after 72 h of PDIA1 silencing. Rac1 Immunoblot densities were quantified using Odyssey software ** P < 0.01; ns: not significant, Student's t -test ( n = 3). f Western analysis of NoxO1 and RhoGDIalpha protein expression before and after PDIA1 silencing, normalized for GAPDH. RhoGDIalpha immunoblot densities were quantified using Odyssey software, **** P < 0.001; ns: not significant, Student's t -test ( n = 3). g Superoxide production after 72h of PDIA1 silencing in HCT116 cells treated or not with 10 uM of NoxA1ds Nox1's peptide inhibitor. EOH 2-hidroxyethidiium relative superoxide production, si-PDI, si-RNA against PDIA1 protein; Scrmb NoxA1ds, NoxA1ds negative control peptide; NoxA1ds, Nox1 peptide inhibitor. * P < 0.05
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
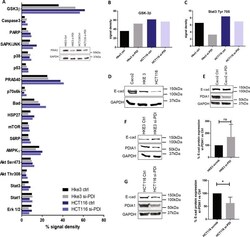
- Experimental details
- Fig. 5 Effects of PDIA1 silencing in signaling targets and EMT marker expression. PathScan array analysis. All array's spots were quantified and analyzed using ImageJ software. PDIA1 silencing was checked by immunoblot analysis. Ctrl, si-RNA negative control; si-PDI, si-RNA against PDIA1; ERK1/2, Thr202/Tyr204 phosphorylation; Stat1, Tyr701 phosphorylation; Stat3, Tyr705 phosphorylation; Akt, Thr308 phosphorylation; Akt, Ser473 phosphorylation; AMPKa, Thr172 phosphorylation, s6 ribosomal protein, Ser235/236 phosphorylation; mTOR, Ser2448 phosphorylation; HSP27, Ser78 phosphorylation; Bad, Ser112 phosphorylation; p70 S6 kinase, Thr389 phosphorylation; PRAS40, Thr246 phosphorylation; p53, Ser15 phosphorylation; p38, Thr180/Tyr182 phosphorylation; SAPK/JNK, Thr183/Tyr185 phosphorylation; PARP, Asp214 cleavage; Caspase-3, Asp175 Cleavage; GSK-3beta, Ser9 phosphorylation. b GSK-3beta, Ser9 phosphorylation plot. c Stat3, Tyr705 phosphorylation. d Basal E-cadherin (E-cad) protein expression by immunoblot normalized for GAPDH ( n = 3). e Immunoblot for E-cadherin before and after PDIA1 silencing in Caco2 cells ( n = 2). f Immunoblot for E-cadherin before and after PDIA1 silencing in HKE3 cells.; ns: not significant, Student's t test ( n = 3). g Immunoblot for E-cadherin before and after PDIA1 silencing in HCT116 cells * P < 0.05, Student's t test ( n = 3). Immunoblot densities were quantified using Odyssey software
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
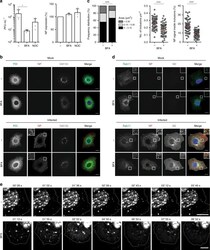
- Experimental details
- Fig. 6 Disruption of ER-Golgi trafficking disassembles vRNP hotspots. A549 cells were infected or mock-infected with PR8 virus at an MOI of 3. a At 90 min p.i., 2 mug mL -1 of brefeldin A (BFA) or 10 mug mL -1 nocodazole (NOC) were added and incubated for 10 h until the supernatant was collected and viral titres determined or cells were harvested in Laemmli's and NP levels detected by western blotting. Statistical analysis of data was performed using a non-parametric one-way ANOVA, followed by Friedman's multiple comparisons test (* p < 0.05). b - d At 8 hpi, cells were also treated or mock-treated with 2 mug mL -1 of BFA for 1 h. b Cells were immunostained for the ER marker PDI (in green), the viral protein NP (in red) and the cis -Golgi marker GM130 (in gray) and imaged by confocal microscopy. Selected areas of the cytoplasm are marked by white boxes and displayed on the top left corner of the images. Bar = 10 mum. c The frequency distribution of NP inclusions within the three size categories (in mum 2 ), the number of inclusions per mum 2 , and the percentage of NP staining that is inside inclusions were plotted for each condition. Statistical analysis of data was performed using a non-parametric Kruskal-Wallis test, followed by Dunn's multiple comparisons test (*** p < 0.001). An average of 60 cells from 2 independent experiments was analyzed per condition. d Infected cells were stained for the host protein Rab11 (in green) and the viral proteins NP (in re
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
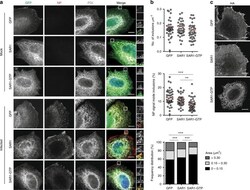
- Experimental details
- Fig. 7 Disruption of ER exit sites dissolves viral inclusions. HeLa cells were transfected with plasmids encoding GFP, SAR1 WT-GFP (SAR1) or SAR1-GTP-GFP (SAR1-GTP) and, 24 h later, infected or mock-infected with PR8 virus, at an MOI of 10. At 16 hpi, cells were fixed and processed for immunofluorescence. a Cells were stained for the viral protein NP (in red) and for the ER protein PDI (in gray). Areas highlighted by the white box are shown on the right of each panel. b The number of inclusions per mum 2 , the percentage of NP staining that is inside inclusions and the frequency distribution of NP inclusions within the three area categories (in mum 2 ) were plotted for each condition. Statistical analysis of data was performed using a non-parametric Kruskal-Wallis test, followed by Dunn's multiple comparisons test (*** p < 0.001). More than 40 cells from 2 independent experiments were analyzed per condition. c Infected cells were stained for the viral protein HA and imaged by confocal microscopy. Bar = 10 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
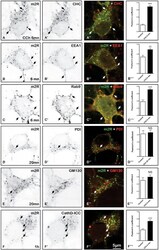
- Experimental details
- FIGURE 9 Immunohistochemical localization of m2R in neuronal compartments involved in endocytosis, synthesis, maturation, and degradation in fixed hippocampal neurons. Hippocampal neurons were transfected with m2R-WT. Neurons were stimulated with CCh at 30 muM for 6, 20 min and 1 h fixed, and processed for visualization of m2R together with markers of intraneuronal compartments and observed by confocal microscopy. ( A-C) 6 min after CCh stimulation (30 muM), some m2R immunopositive punta colocalize with CHC in clathrin-coated pits, EEA1 in early endosomes and Rab9 in late endosomes (arrow heads). (D-E) Twenty minutes after CCh stimulation (30 muM), we failed to detect no colocalization of m2R with PDI, a marker of endoplasmic reticulum and GM130, a marker of Golgi apparatus. (F-F) One hour after CCh stimulation (30 muM), some m2R immunopositive puncta colocalize with CathD, a marker of lysosomes (arrow heads). The quantitative analysis of the colocalization of m2R and markers of subcellular compartment in neurons was performed using the Jacop Plugin of ImageJ and statistical data are reported from the Costes's randomization-based colocalization module (see Materials and Methods). Data are expressed as a Pearson's coefficient (pc) and pc were compared using the Mann-Whitney U -test. Our analysis shows that the colocalization of the immunofluorescent signals for m2R with CHC, EEA1, Rab9, and CathD is higher after treatment with CCh compared to untreated neurons (CHC, Rab9
 Explore
Explore Validate
Validate Learn
Learn