36-1700
antibody from Invitrogen Antibodies
Targeting: F11R
CD321, JAM-1, JAM-A, JAM1, JAMA, JCAM, PAM-1
Antibody data
- Antibody Data
- Antigen structure
- References [77]
- Comments [0]
- Validations
- Western blot [1]
- Immunohistochemistry [1]
- Other assay [39]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 36-1700 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- JAM-A (CD321) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µg
- Concentration
- 0.25 mg/mL
- Storage
- -20°C
Submitted references Altered Mucus Barrier Integrity and Increased Susceptibility to Colitis in Mice upon Loss of Telocyte Bone Morphogenetic Protein Signalling.
Soluble JAM-C Ectodomain Serves as the Niche for Adipose-Derived Stromal/Stem Cells.
Helicobacter pylori PqqE is a new virulence factor that cleaves junctional adhesion molecule A and disrupts gastric epithelial integrity.
Melatonin maintains inner blood-retinal barrier via inhibition of p38/TXNIP/NF-κB pathway in diabetic retinopathy.
Impaired Airway Epithelial Barrier Integrity in Response to Stenotrophomonas maltophilia Proteases, Novel Insights Using Cystic Fibrosis Bronchial Epithelial Cell Secretomics.
Restore natural fertility of Kit(w)/Kit(wv) mouse with nonobstructive azoospermia through gene editing on SSCs mediated by CRISPR-Cas9.
Dynein 1 supports spermatid transport and spermiation during spermatogenesis in the rat testis.
Adherens junctions influence tight junction formation via changes in membrane lipid composition.
Secreted Giardia intestinalis cysteine proteases disrupt intestinal epithelial cell junctional complexes and degrade chemokines.
Inhibition of mTOR protects the blood-brain barrier in models of Alzheimer's disease and vascular cognitive impairment.
Elevated expression of JAM-A promotes neoplastic properties of lung adenocarcinoma.
Aroclor1254 disrupts the blood-testis barrier by promoting endocytosis and degradation of junction proteins via p38 MAPK pathway.
Regulation of the blood-testis barrier by a local axis in the testis: role of laminin α2 in the basement membrane.
The serine protease-mediated increase in intestinal epithelial barrier function is dependent on occludin and requires an intact tight junction.
Planar Cell Polarity (PCP) Protein Vangl2 Regulates Ectoplasmic Specialization Dynamics via Its Effects on Actin Microfilaments in the Testes of Male Rats.
Junctional Adhesion Molecule-A Is Highly Expressed on Human Hematopoietic Repopulating Cells and Associates with the Key Hematopoietic Chemokine Receptor CXCR4.
Polarity protein Crumbs homolog-3 (CRB3) regulates ectoplasmic specialization dynamics through its action on F-actin organization in Sertoli cells.
Overexpression of plastin 3 in Sertoli cells disrupts actin microfilament bundle homeostasis and perturbs the tight junction barrier.
NF-κB inhibitors impair lung epithelial tight junctions in the absence of inflammation.
Actin-bundling protein plastin 3 is a regulator of ectoplasmic specialization dynamics during spermatogenesis in the rat testis.
12/15-Lipoxygenase-dependent ROS production is required for diet-induced endothelial barrier dysfunction.
Homoharringtonine increases intestinal epithelial permeability by modulating specific claudin isoforms in Caco-2 cell monolayers.
Deletion of the tyrosine phosphatase Shp2 in Sertoli cells causes infertility in mice.
Cytokines, polarity proteins, and endosomal protein trafficking and signaling-the sertoli cell blood-testis barrier system in vitro as a study model.
Peanut allergens alter intestinal barrier permeability and tight junction localisation in Caco-2 cell cultures.
Differential effects of c-Src and c-Yes on the endocytic vesicle-mediated trafficking events at the Sertoli cell blood-testis barrier: an in vitro study.
rpS6 regulates blood-testis barrier dynamics through Akt-mediated effects on MMP-9.
Perfluorooctanesulfonate (PFOS) perturbs male rat Sertoli cell blood-testis barrier function by affecting F-actin organization via p-FAK-Tyr(407): an in vitro study.
Rai14 (retinoic acid induced protein 14) is involved in regulating f-actin dynamics at the ectoplasmic specialization in the rat testis*.
Build them up and break them down: Tight junctions of cell lines expressing typical hepatocyte polarity with a varied repertoire of claudins.
Palladin is a regulator of actin filament bundles at the ectoplasmic specialization in adult rat testes.
Rictor/mTORC2 regulates blood-testis barrier dynamics via its effects on gap junction communications and actin filament network.
Intercellular adhesion molecule-2 is involved in apical ectoplasmic specialization dynamics during spermatogenesis in the rat.
c-Yes regulates cell adhesion at the apical ectoplasmic specialization-blood-testis barrier axis via its effects on protein recruitment and distribution.
A survey of Sertoli cell differentiation in men after gonadotropin suppression and in testicular cancer.
miR-199a-5p regulates urothelial permeability and may play a role in bladder pain syndrome.
Low doses of celecoxib attenuate gut barrier failure during experimental peritonitis.
Breast cancer resistance protein regulates apical ectoplasmic specialization dynamics stage specifically in the rat testis.
NC1 domain of collagen α3(IV) derived from the basement membrane regulates Sertoli cell blood-testis barrier dynamics.
Unique cell type-specific junctional complexes in vascular endothelium of human and rat liver sinusoids.
rpS6 Regulates blood-testis barrier dynamics by affecting F-actin organization and protein recruitment.
Filamin A is a regulator of blood-testis barrier assembly during postnatal development in the rat testis.
Regulation of the blood-testis barrier by coxsackievirus and adenovirus receptor.
The characterization of arachnoid cell transport II: paracellular transport and blood-cerebrospinal fluid barrier formation.
Differential effects of flavonoids on barrier integrity in human intestinal Caco-2 cells.
Androgen initiates Sertoli cell tight junction formation in the hypogonadal (hpg) mouse.
A marketed fermented dairy product containing Bifidobacterium lactis CNCM I-2494 suppresses gut hypersensitivity and colonic barrier disruption induced by acute stress in rats.
Matrigel improves functional properties of primary human salivary gland cells.
Breast cancer cell migration is regulated through junctional adhesion molecule-A-mediated activation of Rap1 GTPase.
c-Yes regulates cell adhesion at the blood-testis barrier and the apical ectoplasmic specialization in the seminiferous epithelium of rat testes.
Interleukin-1alpha is a regulator of the blood-testis barrier.
Evaluation of soluble junctional adhesion molecule-A as a biomarker of human brain endothelial barrier breakdown.
Adjudin-mediated Sertoli-germ cell junction disassembly affects Sertoli cell barrier function in vitro and in vivo.
Gonadotropins regulate rat testicular tight junctions in vivo.
Maternal glucocorticoid exposure alters tight junction protein expression in the brain of fetal sheep.
Allogeneic human mesenchymal stem cells restore epithelial protein permeability in cultured human alveolar type II cells by secretion of angiopoietin-1.
Disruption of the blood-testis barrier integrity by bisphenol A in vitro: is this a suitable model for studying blood-testis barrier dynamics?
TGF-beta3 and TNFalpha perturb blood-testis barrier (BTB) dynamics by accelerating the clathrin-mediated endocytosis of integral membrane proteins: a new concept of BTB regulation during spermatogenesis.
Conditional forebrain inactivation of nicastrin causes progressive memory impairment and age-related neurodegeneration.
Drug transporter, P-glycoprotein (MDR1), is an integrated component of the mammalian blood-testis barrier.
Interleukin 1 alpha (IL1A) is a novel regulator of the blood-testis barrier in the rat.
Blood-testis barrier dynamics are regulated by testosterone and cytokines via their differential effects on the kinetics of protein endocytosis and recycling in Sertoli cells.
Regulation of testicular tight junctions by gonadotrophins in the adult Djungarian hamster in vivo.
JAM-A is both essential and inhibitory to development of hepatic polarity in WIF-B cells.
Distribution of tight junction proteins in adult human salivary glands.
Coxsackie and adenovirus receptor (CAR) is a product of Sertoli and germ cells in rat testes which is localized at the Sertoli-Sertoli and Sertoli-germ cell interface.
Laminin alpha 3 forms a complex with beta3 and gamma3 chains that serves as the ligand for alpha 6beta1-integrin at the apical ectoplasmic specialization in adult rat testes.
Adjudin-mediated junction restructuring in the seminiferous epithelium leads to displacement of soluble guanylate cyclase from adherens junctions.
Dynamin II interacts with the cadherin- and occludin-based protein complexes at the blood-testis barrier in adult rat testes.
Tumor necrosis factor {alpha} reversibly disrupts the blood-testis barrier and impairs Sertoli-germ cell adhesion in the seminiferous epithelium of adult rat testes.
The Rac activator Tiam1 controls tight junction biogenesis in keratinocytes through binding to and activation of the Par polarity complex.
Development of tight junction molecules in blood vessels of germinal matrix, cerebral cortex, and white matter.
Disruption of Sertoli-germ cell adhesion function in the seminiferous epithelium of the rat testis can be limited to adherens junctions without affecting the blood-testis barrier integrity: an in vivo study using an androgen suppression model.
Junctional adhesion molecule 1 regulates epithelial cell morphology through effects on beta1 integrins and Rap1 activity.
Knockdown of occludin expression leads to diverse phenotypic alterations in epithelial cells.
Endocytosis of epithelial apical junctional proteins by a clathrin-mediated pathway into a unique storage compartment.
Expression and function of tight junctions in the crypt epithelium of human palatine tonsils.
Reyes Nicolás V, Allaire JM, Alfonso AB, Pupo Gómez D, Pomerleau V, Giroux V, Boudreau F, Perreault N
Cells 2021 Oct 29;10(11)
Cells 2021 Oct 29;10(11)
Soluble JAM-C Ectodomain Serves as the Niche for Adipose-Derived Stromal/Stem Cells.
Yamazaki M, Sugimoto K, Mabuchi Y, Yamashita R, Ichikawa-Tomikawa N, Kaneko T, Akazawa C, Hasegawa H, Imura T, Chiba H
Biomedicines 2021 Mar 10;9(3)
Biomedicines 2021 Mar 10;9(3)
Helicobacter pylori PqqE is a new virulence factor that cleaves junctional adhesion molecule A and disrupts gastric epithelial integrity.
Marques MS, Costa AC, Osório H, Pinto ML, Relvas S, Dinis-Ribeiro M, Carneiro F, Leite M, Figueiredo C
Gut microbes 2021 Jan-Dec;13(1):1-21
Gut microbes 2021 Jan-Dec;13(1):1-21
Melatonin maintains inner blood-retinal barrier via inhibition of p38/TXNIP/NF-κB pathway in diabetic retinopathy.
Tang L, Zhang C, Yang Q, Xie H, Liu D, Tian H, Lu L, Xu JY, Li W, Xu G, Qiu Q, Liu K, Luo D, Xu GT, Zhang J
Journal of cellular physiology 2021 Aug;236(8):5848-5864
Journal of cellular physiology 2021 Aug;236(8):5848-5864
Impaired Airway Epithelial Barrier Integrity in Response to Stenotrophomonas maltophilia Proteases, Novel Insights Using Cystic Fibrosis Bronchial Epithelial Cell Secretomics.
Molloy K, Cagney G, Dillon ET, Wynne K, Greene CM, McElvaney NG
Frontiers in immunology 2020;11:198
Frontiers in immunology 2020;11:198
Restore natural fertility of Kit(w)/Kit(wv) mouse with nonobstructive azoospermia through gene editing on SSCs mediated by CRISPR-Cas9.
Li X, Sun T, Wang X, Tang J, Liu Y
Stem cell research & therapy 2019 Aug 24;10(1):271
Stem cell research & therapy 2019 Aug 24;10(1):271
Dynein 1 supports spermatid transport and spermiation during spermatogenesis in the rat testis.
Wen Q, Tang EI, Lui WY, Lee WM, Wong CKC, Silvestrini B, Cheng CY
American journal of physiology. Endocrinology and metabolism 2018 Nov 1;315(5):E924-E948
American journal of physiology. Endocrinology and metabolism 2018 Nov 1;315(5):E924-E948
Adherens junctions influence tight junction formation via changes in membrane lipid composition.
Shigetomi K, Ono Y, Inai T, Ikenouchi J
The Journal of cell biology 2018 Jul 2;217(7):2373-2381
The Journal of cell biology 2018 Jul 2;217(7):2373-2381
Secreted Giardia intestinalis cysteine proteases disrupt intestinal epithelial cell junctional complexes and degrade chemokines.
Liu J, Ma'ayeh S, Peirasmaki D, Lundström-Stadelmann B, Hellman L, Svärd SG
Virulence 2018 Dec 31;9(1):879-894
Virulence 2018 Dec 31;9(1):879-894
Inhibition of mTOR protects the blood-brain barrier in models of Alzheimer's disease and vascular cognitive impairment.
Van Skike CE, Jahrling JB, Olson AB, Sayre NL, Hussong SA, Ungvari Z, Lechleiter JD, Galvan V
American journal of physiology. Heart and circulatory physiology 2018 Apr 1;314(4):H693-H703
American journal of physiology. Heart and circulatory physiology 2018 Apr 1;314(4):H693-H703
Elevated expression of JAM-A promotes neoplastic properties of lung adenocarcinoma.
Magara K, Takasawa A, Osanai M, Ota M, Tagami Y, Ono Y, Takasawa K, Murata M, Hirohashi Y, Miyajima M, Yamada G, Hasegawa T, Sawada N
Cancer science 2017 Nov;108(11):2306-2314
Cancer science 2017 Nov;108(11):2306-2314
Aroclor1254 disrupts the blood-testis barrier by promoting endocytosis and degradation of junction proteins via p38 MAPK pathway.
Jia X, Xu Y, Wu W, Fan Y, Wang G, Zhang T, Su W
Cell death & disease 2017 May 25;8(5):e2823
Cell death & disease 2017 May 25;8(5):e2823
Regulation of the blood-testis barrier by a local axis in the testis: role of laminin α2 in the basement membrane.
Gao Y, Mruk D, Chen H, Lui WY, Lee WM, Cheng CY
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2017 Feb;31(2):584-597
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2017 Feb;31(2):584-597
The serine protease-mediated increase in intestinal epithelial barrier function is dependent on occludin and requires an intact tight junction.
Ronaghan NJ, Shang J, Iablokov V, Zaheer R, Colarusso P, Dion S, Désilets A, Leduc R, Turner JR, MacNaughton WK
American journal of physiology. Gastrointestinal and liver physiology 2016 Sep 1;311(3):G466-79
American journal of physiology. Gastrointestinal and liver physiology 2016 Sep 1;311(3):G466-79
Planar Cell Polarity (PCP) Protein Vangl2 Regulates Ectoplasmic Specialization Dynamics via Its Effects on Actin Microfilaments in the Testes of Male Rats.
Chen H, Mruk DD, Lee WM, Cheng CY
Endocrinology 2016 May;157(5):2140-59
Endocrinology 2016 May;157(5):2140-59
Junctional Adhesion Molecule-A Is Highly Expressed on Human Hematopoietic Repopulating Cells and Associates with the Key Hematopoietic Chemokine Receptor CXCR4.
Chang CH, Hale SJ, Cox CV, Blair A, Kronsteiner B, Grabowska R, Zhang Y, Cook D, Khoo CP, Schrader JB, Kabuga SB, Martin-Rendon E, Watt SM
Stem cells (Dayton, Ohio) 2016 Jun;34(6):1664-78
Stem cells (Dayton, Ohio) 2016 Jun;34(6):1664-78
Polarity protein Crumbs homolog-3 (CRB3) regulates ectoplasmic specialization dynamics through its action on F-actin organization in Sertoli cells.
Gao Y, Lui WY, Lee WM, Cheng CY
Scientific reports 2016 Jun 30;6:28589
Scientific reports 2016 Jun 30;6:28589
Overexpression of plastin 3 in Sertoli cells disrupts actin microfilament bundle homeostasis and perturbs the tight junction barrier.
Li N, Lee WM, Cheng CY
Spermatogenesis 2016 Apr;6(1):e1206353
Spermatogenesis 2016 Apr;6(1):e1206353
NF-κB inhibitors impair lung epithelial tight junctions in the absence of inflammation.
Ward C, Schlingmann B, Stecenko AA, Guidot DM, Koval M
Tissue barriers 2015;3(1-2):e982424
Tissue barriers 2015;3(1-2):e982424
Actin-bundling protein plastin 3 is a regulator of ectoplasmic specialization dynamics during spermatogenesis in the rat testis.
Li N, Mruk DD, Wong CK, Lee WM, Han D, Cheng CY
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2015 Sep;29(9):3788-805
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2015 Sep;29(9):3788-805
12/15-Lipoxygenase-dependent ROS production is required for diet-induced endothelial barrier dysfunction.
Chattopadhyay R, Tinnikov A, Dyukova E, Singh NK, Kotla S, Mobley JA, Rao GN
Journal of lipid research 2015 Mar;56(3):562-77
Journal of lipid research 2015 Mar;56(3):562-77
Homoharringtonine increases intestinal epithelial permeability by modulating specific claudin isoforms in Caco-2 cell monolayers.
Watari A, Hashegawa M, Yagi K, Kondoh M
European journal of pharmaceutics and biopharmaceutics : official journal of Arbeitsgemeinschaft fur Pharmazeutische Verfahrenstechnik e.V 2015 Jan;89:232-8
European journal of pharmaceutics and biopharmaceutics : official journal of Arbeitsgemeinschaft fur Pharmazeutische Verfahrenstechnik e.V 2015 Jan;89:232-8
Deletion of the tyrosine phosphatase Shp2 in Sertoli cells causes infertility in mice.
Hu X, Tang Z, Li Y, Liu W, Zhang S, Wang B, Tian Y, Zhao Y, Ran H, Liu W, Feng GS, Shuai J, Wang H, Lu Z
Scientific reports 2015 Aug 12;5:12982
Scientific reports 2015 Aug 12;5:12982
Cytokines, polarity proteins, and endosomal protein trafficking and signaling-the sertoli cell blood-testis barrier system in vitro as a study model.
Xiao X, Wong EW, Lie PP, Mruk DD, Wong CK, Cheng CY
Methods in enzymology 2014;534:181-94
Methods in enzymology 2014;534:181-94
Peanut allergens alter intestinal barrier permeability and tight junction localisation in Caco-2 cell cultures.
Price DB, Ackland ML, Burks W, Knight MI, Suphioglu C
Cellular physiology and biochemistry : international journal of experimental cellular physiology, biochemistry, and pharmacology 2014;33(6):1758-77
Cellular physiology and biochemistry : international journal of experimental cellular physiology, biochemistry, and pharmacology 2014;33(6):1758-77
Differential effects of c-Src and c-Yes on the endocytic vesicle-mediated trafficking events at the Sertoli cell blood-testis barrier: an in vitro study.
Xiao X, Mruk DD, Wong EW, Lee WM, Han D, Wong CK, Cheng CY
American journal of physiology. Endocrinology and metabolism 2014 Oct 1;307(7):E553-62
American journal of physiology. Endocrinology and metabolism 2014 Oct 1;307(7):E553-62
rpS6 regulates blood-testis barrier dynamics through Akt-mediated effects on MMP-9.
Mok KW, Mruk DD, Cheng CY
Journal of cell science 2014 Nov 15;127(Pt 22):4870-82
Journal of cell science 2014 Nov 15;127(Pt 22):4870-82
Perfluorooctanesulfonate (PFOS) perturbs male rat Sertoli cell blood-testis barrier function by affecting F-actin organization via p-FAK-Tyr(407): an in vitro study.
Wan HT, Mruk DD, Wong CK, Cheng CY
Endocrinology 2014 Jan;155(1):249-62
Endocrinology 2014 Jan;155(1):249-62
Rai14 (retinoic acid induced protein 14) is involved in regulating f-actin dynamics at the ectoplasmic specialization in the rat testis*.
Qian X, Mruk DD, Cheng CY
PloS one 2013;8(4):e60656
PloS one 2013;8(4):e60656
Build them up and break them down: Tight junctions of cell lines expressing typical hepatocyte polarity with a varied repertoire of claudins.
Grosse B, Degrouard J, Jaillard D, Cassio D
Tissue barriers 2013 Oct 1;1(4):e25210
Tissue barriers 2013 Oct 1;1(4):e25210
Palladin is a regulator of actin filament bundles at the ectoplasmic specialization in adult rat testes.
Qian X, Mruk DD, Wong EW, Lie PP, Cheng CY
Endocrinology 2013 May;154(5):1907-20
Endocrinology 2013 May;154(5):1907-20
Rictor/mTORC2 regulates blood-testis barrier dynamics via its effects on gap junction communications and actin filament network.
Mok KW, Mruk DD, Lee WM, Cheng CY
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2013 Mar;27(3):1137-52
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2013 Mar;27(3):1137-52
Intercellular adhesion molecule-2 is involved in apical ectoplasmic specialization dynamics during spermatogenesis in the rat.
Xiao X, Cheng CY, Mruk DD
The Journal of endocrinology 2013 Jan;216(1):73-86
The Journal of endocrinology 2013 Jan;216(1):73-86
c-Yes regulates cell adhesion at the apical ectoplasmic specialization-blood-testis barrier axis via its effects on protein recruitment and distribution.
Xiao X, Mruk DD, Cheng CY
American journal of physiology. Endocrinology and metabolism 2013 Jan 15;304(2):E145-59
American journal of physiology. Endocrinology and metabolism 2013 Jan 15;304(2):E145-59
A survey of Sertoli cell differentiation in men after gonadotropin suppression and in testicular cancer.
Tarulli GA, Stanton PG, Loveland KL, Rajpert-De Meyts E, McLachlan RI, Meachem SJ
Spermatogenesis 2013 Jan 1;3(1):e24014
Spermatogenesis 2013 Jan 1;3(1):e24014
miR-199a-5p regulates urothelial permeability and may play a role in bladder pain syndrome.
Monastyrskaya K, Sánchez-Freire V, Hashemi Gheinani A, Klumpp DJ, Babiychuk EB, Draeger A, Burkhard FC
The American journal of pathology 2013 Feb;182(2):431-48
The American journal of pathology 2013 Feb;182(2):431-48
Low doses of celecoxib attenuate gut barrier failure during experimental peritonitis.
Short SS, Wang J, Castle SL, Fernandez GE, Smiley N, Zobel M, Pontarelli EM, Papillon SC, Grishin AV, Ford HR
Laboratory investigation; a journal of technical methods and pathology 2013 Dec;93(12):1265-75
Laboratory investigation; a journal of technical methods and pathology 2013 Dec;93(12):1265-75
Breast cancer resistance protein regulates apical ectoplasmic specialization dynamics stage specifically in the rat testis.
Qian X, Mruk DD, Wong EW, Cheng CY
American journal of physiology. Endocrinology and metabolism 2013 Apr 1;304(7):E757-69
American journal of physiology. Endocrinology and metabolism 2013 Apr 1;304(7):E757-69
NC1 domain of collagen α3(IV) derived from the basement membrane regulates Sertoli cell blood-testis barrier dynamics.
Wong EW, Cheng CY
Spermatogenesis 2013 Apr 1;3(2):e25465
Spermatogenesis 2013 Apr 1;3(2):e25465
Unique cell type-specific junctional complexes in vascular endothelium of human and rat liver sinusoids.
Géraud C, Evdokimov K, Straub BK, Peitsch WK, Demory A, Dörflinger Y, Schledzewski K, Schmieder A, Schemmer P, Augustin HG, Schirmacher P, Goerdt S
PloS one 2012;7(4):e34206
PloS one 2012;7(4):e34206
rpS6 Regulates blood-testis barrier dynamics by affecting F-actin organization and protein recruitment.
Mok KW, Mruk DD, Silvestrini B, Cheng CY
Endocrinology 2012 Oct;153(10):5036-48
Endocrinology 2012 Oct;153(10):5036-48
Filamin A is a regulator of blood-testis barrier assembly during postnatal development in the rat testis.
Su W, Mruk DD, Lie PP, Lui WY, Cheng CY
Endocrinology 2012 Oct;153(10):5023-35
Endocrinology 2012 Oct;153(10):5023-35
Regulation of the blood-testis barrier by coxsackievirus and adenovirus receptor.
Su L, Mruk DD, Cheng CY
American journal of physiology. Cell physiology 2012 Oct 15;303(8):C843-53
American journal of physiology. Cell physiology 2012 Oct 15;303(8):C843-53
The characterization of arachnoid cell transport II: paracellular transport and blood-cerebrospinal fluid barrier formation.
Lam CH, Hansen EA, Janson C, Bryan A, Hubel A
Neuroscience 2012 Oct 11;222:228-38
Neuroscience 2012 Oct 11;222:228-38
Differential effects of flavonoids on barrier integrity in human intestinal Caco-2 cells.
Noda S, Tanabe S, Suzuki T
Journal of agricultural and food chemistry 2012 May 9;60(18):4628-33
Journal of agricultural and food chemistry 2012 May 9;60(18):4628-33
Androgen initiates Sertoli cell tight junction formation in the hypogonadal (hpg) mouse.
McCabe MJ, Allan CM, Foo CF, Nicholls PK, McTavish KJ, Stanton PG
Biology of reproduction 2012 Aug;87(2):38
Biology of reproduction 2012 Aug;87(2):38
A marketed fermented dairy product containing Bifidobacterium lactis CNCM I-2494 suppresses gut hypersensitivity and colonic barrier disruption induced by acute stress in rats.
Agostini S, Goubern M, Tondereau V, Salvador-Cartier C, Bezirard V, Lévèque M, Keränen H, Theodorou V, Bourdu-Naturel S, Goupil-Feuillerat N, Legrain-Raspaud S, Eutamene H
Neurogastroenterology and motility 2012 Apr;24(4):376-e172
Neurogastroenterology and motility 2012 Apr;24(4):376-e172
Matrigel improves functional properties of primary human salivary gland cells.
Maria OM, Zeitouni A, Gologan O, Tran SD
Tissue engineering. Part A 2011 May;17(9-10):1229-38
Tissue engineering. Part A 2011 May;17(9-10):1229-38
Breast cancer cell migration is regulated through junctional adhesion molecule-A-mediated activation of Rap1 GTPase.
McSherry EA, Brennan K, Hudson L, Hill AD, Hopkins AM
Breast cancer research : BCR 2011 Mar 23;13(2):R31
Breast cancer research : BCR 2011 Mar 23;13(2):R31
c-Yes regulates cell adhesion at the blood-testis barrier and the apical ectoplasmic specialization in the seminiferous epithelium of rat testes.
Xiao X, Mruk DD, Lee WM, Cheng CY
The international journal of biochemistry & cell biology 2011 Apr;43(4):651-65
The international journal of biochemistry & cell biology 2011 Apr;43(4):651-65
Interleukin-1alpha is a regulator of the blood-testis barrier.
Lie PP, Cheng CY, Mruk DD
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2011 Apr;25(4):1244-53
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2011 Apr;25(4):1244-53
Evaluation of soluble junctional adhesion molecule-A as a biomarker of human brain endothelial barrier breakdown.
Haarmann A, Deiss A, Prochaska J, Foerch C, Weksler B, Romero I, Couraud PO, Stoll G, Rieckmann P, Buttmann M
PloS one 2010 Oct 21;5(10):e13568
PloS one 2010 Oct 21;5(10):e13568
Adjudin-mediated Sertoli-germ cell junction disassembly affects Sertoli cell barrier function in vitro and in vivo.
Su L, Cheng CY, Mruk DD
The international journal of biochemistry & cell biology 2010 Nov;42(11):1864-75
The international journal of biochemistry & cell biology 2010 Nov;42(11):1864-75
Gonadotropins regulate rat testicular tight junctions in vivo.
McCabe MJ, Tarulli GA, Meachem SJ, Robertson DM, Smooker PM, Stanton PG
Endocrinology 2010 Jun;151(6):2911-22
Endocrinology 2010 Jun;151(6):2911-22
Maternal glucocorticoid exposure alters tight junction protein expression in the brain of fetal sheep.
Sadowska GB, Malaeb SN, Stonestreet BS
American journal of physiology. Heart and circulatory physiology 2010 Jan;298(1):H179-88
American journal of physiology. Heart and circulatory physiology 2010 Jan;298(1):H179-88
Allogeneic human mesenchymal stem cells restore epithelial protein permeability in cultured human alveolar type II cells by secretion of angiopoietin-1.
Fang X, Neyrinck AP, Matthay MA, Lee JW
The Journal of biological chemistry 2010 Aug 20;285(34):26211-22
The Journal of biological chemistry 2010 Aug 20;285(34):26211-22
Disruption of the blood-testis barrier integrity by bisphenol A in vitro: is this a suitable model for studying blood-testis barrier dynamics?
Li MW, Mruk DD, Lee WM, Cheng CY
The international journal of biochemistry & cell biology 2009 Nov;41(11):2302-14
The international journal of biochemistry & cell biology 2009 Nov;41(11):2302-14
TGF-beta3 and TNFalpha perturb blood-testis barrier (BTB) dynamics by accelerating the clathrin-mediated endocytosis of integral membrane proteins: a new concept of BTB regulation during spermatogenesis.
Xia W, Wong EW, Mruk DD, Cheng CY
Developmental biology 2009 Mar 1;327(1):48-61
Developmental biology 2009 Mar 1;327(1):48-61
Conditional forebrain inactivation of nicastrin causes progressive memory impairment and age-related neurodegeneration.
Tabuchi K, Chen G, Südhof TC, Shen J
The Journal of neuroscience : the official journal of the Society for Neuroscience 2009 Jun 3;29(22):7290-301
The Journal of neuroscience : the official journal of the Society for Neuroscience 2009 Jun 3;29(22):7290-301
Drug transporter, P-glycoprotein (MDR1), is an integrated component of the mammalian blood-testis barrier.
Su L, Cheng CY, Mruk DD
The international journal of biochemistry & cell biology 2009 Dec;41(12):2578-87
The international journal of biochemistry & cell biology 2009 Dec;41(12):2578-87
Interleukin 1 alpha (IL1A) is a novel regulator of the blood-testis barrier in the rat.
Sarkar O, Mathur PP, Cheng CY, Mruk DD
Biology of reproduction 2008 Mar;78(3):445-54
Biology of reproduction 2008 Mar;78(3):445-54
Blood-testis barrier dynamics are regulated by testosterone and cytokines via their differential effects on the kinetics of protein endocytosis and recycling in Sertoli cells.
Yan HH, Mruk DD, Lee WM, Cheng CY
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2008 Jun;22(6):1945-59
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2008 Jun;22(6):1945-59
Regulation of testicular tight junctions by gonadotrophins in the adult Djungarian hamster in vivo.
Tarulli GA, Meachem SJ, Schlatt S, Stanton PG
Reproduction (Cambridge, England) 2008 Jun;135(6):867-77
Reproduction (Cambridge, England) 2008 Jun;135(6):867-77
JAM-A is both essential and inhibitory to development of hepatic polarity in WIF-B cells.
Braiterman LT, Heffernan S, Nyasae L, Johns D, See AP, Yutzy R, McNickle A, Herman M, Sharma A, Naik UP, Hubbard AL
American journal of physiology. Gastrointestinal and liver physiology 2008 Feb;294(2):G576-88
American journal of physiology. Gastrointestinal and liver physiology 2008 Feb;294(2):G576-88
Distribution of tight junction proteins in adult human salivary glands.
Maria OM, Kim JW, Gerstenhaber JA, Baum BJ, Tran SD
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2008 Dec;56(12):1093-8
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2008 Dec;56(12):1093-8
Coxsackie and adenovirus receptor (CAR) is a product of Sertoli and germ cells in rat testes which is localized at the Sertoli-Sertoli and Sertoli-germ cell interface.
Wang CQ, Mruk DD, Lee WM, Cheng CY
Experimental cell research 2007 Apr 15;313(7):1373-92
Experimental cell research 2007 Apr 15;313(7):1373-92
Laminin alpha 3 forms a complex with beta3 and gamma3 chains that serves as the ligand for alpha 6beta1-integrin at the apical ectoplasmic specialization in adult rat testes.
Yan HHN, Cheng CY
The Journal of biological chemistry 2006 Jun 23;281(25):17286-17303
The Journal of biological chemistry 2006 Jun 23;281(25):17286-17303
Adjudin-mediated junction restructuring in the seminiferous epithelium leads to displacement of soluble guanylate cyclase from adherens junctions.
Sarkar O, Xia W, Mruk DD
Journal of cellular physiology 2006 Jul;208(1):175-87
Journal of cellular physiology 2006 Jul;208(1):175-87
Dynamin II interacts with the cadherin- and occludin-based protein complexes at the blood-testis barrier in adult rat testes.
Lie PP, Xia W, Wang CQ, Mruk DD, Yan HH, Wong CH, Lee WM, Cheng CY
The Journal of endocrinology 2006 Dec;191(3):571-86
The Journal of endocrinology 2006 Dec;191(3):571-86
Tumor necrosis factor {alpha} reversibly disrupts the blood-testis barrier and impairs Sertoli-germ cell adhesion in the seminiferous epithelium of adult rat testes.
Li MW, Xia W, Mruk DD, Wang CQ, Yan HH, Siu MK, Lui WY, Lee WM, Cheng CY
The Journal of endocrinology 2006 Aug;190(2):313-29
The Journal of endocrinology 2006 Aug;190(2):313-29
The Rac activator Tiam1 controls tight junction biogenesis in keratinocytes through binding to and activation of the Par polarity complex.
Mertens AE, Rygiel TP, Olivo C, van der Kammen R, Collard JG
The Journal of cell biology 2005 Sep 26;170(7):1029-37
The Journal of cell biology 2005 Sep 26;170(7):1029-37
Development of tight junction molecules in blood vessels of germinal matrix, cerebral cortex, and white matter.
Ballabh P, Hu F, Kumarasiri M, Braun A, Nedergaard M
Pediatric research 2005 Oct;58(4):791-8
Pediatric research 2005 Oct;58(4):791-8
Disruption of Sertoli-germ cell adhesion function in the seminiferous epithelium of the rat testis can be limited to adherens junctions without affecting the blood-testis barrier integrity: an in vivo study using an androgen suppression model.
Xia W, Wong CH, Lee NP, Lee WM, Cheng CY
Journal of cellular physiology 2005 Oct;205(1):141-57
Journal of cellular physiology 2005 Oct;205(1):141-57
Junctional adhesion molecule 1 regulates epithelial cell morphology through effects on beta1 integrins and Rap1 activity.
Mandell KJ, Babbin BA, Nusrat A, Parkos CA
The Journal of biological chemistry 2005 Mar 25;280(12):11665-74
The Journal of biological chemistry 2005 Mar 25;280(12):11665-74
Knockdown of occludin expression leads to diverse phenotypic alterations in epithelial cells.
Yu AS, McCarthy KM, Francis SA, McCormack JM, Lai J, Rogers RA, Lynch RD, Schneeberger EE
American journal of physiology. Cell physiology 2005 Jun;288(6):C1231-41
American journal of physiology. Cell physiology 2005 Jun;288(6):C1231-41
Endocytosis of epithelial apical junctional proteins by a clathrin-mediated pathway into a unique storage compartment.
Ivanov AI, Nusrat A, Parkos CA
Molecular biology of the cell 2004 Jan;15(1):176-88
Molecular biology of the cell 2004 Jan;15(1):176-88
Expression and function of tight junctions in the crypt epithelium of human palatine tonsils.
Go M, Kojima T, Takano K, Murata M, Ichimiya S, Tsubota H, Himi T, Sawada N
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2004 Dec;52(12):1627-38
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2004 Dec;52(12):1627-38
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of Caco-2 cell lysates using Rabbit anti-JAM-A Polyclonal Antibody (Product # 36-1700).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of JAM1/CD321 showing staining in the membrane of paraffin-embedded human colon carcinoma tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a JAM1/CD321 polyclonal antibody (Product # 36-1700) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Immunocytochemical JAM-A tight junction staining in primary HBMEC is reduced under barrier-disturbing pro-inflammatory conditions. Cells were either left unstimulated or treated with TNF-alpha 10 ng/mL and IFN-gamma 100 U/mL for 24 h. A Immunocytochemical staining was performed with primary antibodies against JAM-A (M.Ab.F11), occludin and ZO-1. F-Actin was stained with Alexa Fluor 488 phalloidin. Representative for 5 independent experiments with different HBMEC preparations. B Analysis of paracellular barrier function by dextran 3000 transwell permeability assays. Mean and SD of 4 independent experiments in octuplicates with different HBMEC preparations. The two-tailed Mann-Whitney U test was used for statistical analysis. Bar = 25 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 In contrast to ICAM-1, JAM-A overall surface expression levels on HBMEC do not change upon pro-inflammatory stimulation. Flow cytometric analysis of Accutase(tm)-dissociated single cell suspensions of primary HBMEC after stimulation as in Figure 1 . JAM-A was stained with M.Ab.F11 and a rabbit polyclonal antibody from Zymed. Staining against ICAM-1 served as a positive control. In parallel, unstimulated and stimulated HBMEC were stained with matched isotype control antibodies. Filled histograms represent isotype stainings, open histograms JAM-A and ICAM-1 stainings. Representative for 5 independent experiments with different HBMEC preparations.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Pro-inflammatory stimulation of HBMEC induces JAM-A dissociation from the actin cytoskeleton. A Primary HBMEC were either left unstimulated or treated with TNF-alpha 10 ng/mL or IFN-gamma 100 IU/mL alone or in combination. Cell protein extracts were generated with a Nonidet-P40 based cell lysis buffer and subjected to Western blot analysis. JAM-A was stained with M.Ab.F11. Staining of the same, peroxidase-inactivated membranes with a rabbit antibody against beta-actin served as a loading control. B N-deglycosylation of JAM-A with increasing concentrations of PNGase F for 2 h. A rabbit polyclonal antibody against JAM-A (Zymed) was used for the detection of JAM-A. The asterisk represents N-glycosylated, the open circle N-declycosylated JAM-A. Representative experiments out of at least 5 independent experiments with different EC preparations for each subpanel of the figure are shown.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 HBMEC constitutively release sJAM-A, which is not altered under inflammatory or hypoxia/reoxygenation conditions. A Representative Western blot of a HBMEC whole cell protein extract, employing the goat anti-JAM-A antibody which was used as capture antibody for a self-developed sandwich ELISA (compare Figure 3 ). B Typical standard curve of the self-developed ELISA for JAM-A detection in culture supernatants and serum. C and D Concentrations of sJAM-A in culture supernatants of primary HBMEC ( C ) or hMCEC/D3 ( D ) which were either left unstimulated for 48 h, stimulated with TNF-alpha 10 ng/mL and IFN-gamma 100 U/mL for 48 h or subjected to 1% O 2 for 24 h followed by a reoxygenation period of 24 h. Please note: cell culture supernatants were concentrated by filter centrifugation before ELISA measurement, resulting in ~10-fold increased JAM-A concentration. Mean and SD of 3 independent experiments. The Kruskal-Wallis test was used for statistical analysis. n.s., not significant. E Comparative Western blot analysis of a whole cell protein extract and a TCA-precipitated protein extract from cell culture supernatant, using a rb polyclonal antibody (Zymed) for JAM-A detection. Representative of 3 independent experiments with different HBMEC preparations. The asterisk represents full-length JAM-A, open circles represent soluble JAM-A. F Comparative Western blot analysis of JAM-A in TCA-precipitated cell culture supernatants, using the same primary antibody as in E, after
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 Expression of JAM-family members in liver sinusoidal endothelial cells. (A) Immunofluorescent co-staining of rat liver cryosections with anti-JAM-A (green), anti-VE-cadherin (red), and anti-Stabilin-2 (blue) antibodies. (B) Immunofluorescent co-staining of human liver cryosections with anti-JAM-A (green), anti-CD32b (red), and anti-Stabilin-2 (blue) antibodies. Images were acquired using laser scanning confocal microscopy. Bars 11.9 um. (C) Quantitative reverse transcriptase-PCR with mRNA isolated from rat LSECs and rat LMECs (n indicates the number of samples analyzed, error bars represent SEM). Primers specific for JAM-A, JAM-B, JAM-C, and beta-Actin as normalizer were used.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4. JAM-A localization is disorganized in carcinoma in situ and becomes highly expressed in malignant seminoma. Confocal immunofluorescence of normal human testis ( A ), carcinoma in situ ( B and C ) and seminoma sections ( D ) detecting JAM-A (red, asterisks) and connexin 43 (green, gap junctions, squares). Inserts are negative controls. (Bar = 50 um).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Peritonitis is associated with gut barrier derangement (a) H&E-stained sections of ileum from mice 16 h after injection with NS, injection with 40 mg/kg LPS, sham operation, or CLP. Note abundant epithelial sloughing in animals injected with LPS or subjected to CLP. Data are representative of at least 9 animals in each group. (b) Transepithelial passage of FITC-dextran and bacteria during peritonitis. Mice were gavaged with FITC-dextran and E. coli 35354T prior to indicated treatments. Serum concentrations of FITC-dextran (top) and counts of viable E. coli in combined mesenteric lymph nodes and spleens (bottom) were determined 16 h post treatment. *, Significant differences (p = 12 in each group). (c) Localization of JAM-A (red) in ileal epithelium 16 h after indicated treatments. Note increased re-distribution of JAM-A from borders to intracellular space of enterocytes in LPS and CLP samples. (d) Localization of ZO-1 (red) in ileal epithelium 16 h after indicated treatments. Note increased redistribution of ZO-1 from TJ to intracellular space of enterocytes in LPS and CLP samples. DAPI-stained nuclei appear in blue. Bar=50 mum. All images are representative of at least 3 animals in each group.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Effects of Celecoxib on localization of TJ-associated proteins JAM-A and ZO-1 (a) JAM-A (red) after injection with NS or LPS, with or without indicated doses (mg/kg) of Celecoxib. (b) JAM-A (red) after sham operation or CLP, with or without indicated doses of Celecoxib. (c) ZO-1 (red) after injection with NS or LPS, with or without indicated doses of Celecoxib. (d) ZO-1 (red) after sham operation or CLP, with or without indicated doses of Celecoxib. All images show sections of ileal samples taken 16 h after peritonitis-inducing treatments and are representative of at least 3 animals in each group. DAPI-stained nuclei appear in blue. Bar=100 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
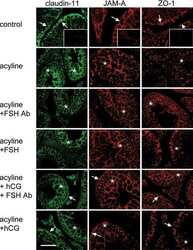
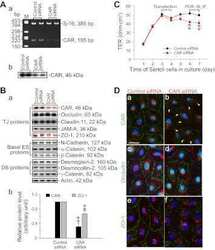
- Figure 3 BTB integrity was disrupted by Shp2 deletion. ( A ) BTB permeability was assayed with the biotin tracer (red) using confocal microscopy in two-week-old mice. Asterisk indicates the biotin tracer in the adluminal compartment of the seminiferous tubules. ( B ) Immunohistochemical staining of the BTB junction proteins Cx43, Claudin11 and JAMA in seminiferous tubules in two-week-old mice. ( C ) Altered BTB-related genes in the microarray gene database, and the protein levels of various genes were analyzed by Western blotting in testes from two-week-old mice. The full-length blots are presented in Supplemental Figure S7 . ( D ) The quantity of the cell junctions between primary Sertoli cells isolated from two-week-old mice was measured by transepithelial resistance (TER) assays. The experiments were repeated at least three times, and one representative result was shown. ( E ) Shp2 expression and Erk and Akt phosphorylation in primary Sertoli cells used in the TER assays. Tubulin was used as a loading control. The full-length blots are presented in Supplemental Figure S8 . Ad, adenovirus virus; f/f, Shp2 f/f cells; ko, SCSKO cells; Q79P, constitutively active Shp2 mutant. The values are expressed as the mean +- SEM. Statistical analysis was performed using Student's t -test. Asterisks denote the statistical significance, **P < 0.01; ***P < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Knockdown of Rai14 in the Sertoli cell epithelium with an established TJ-permeability barrier in vitro by RNAi disrupts actin filament organization and the TJ barrier. (A) Sertoli cells cultured alone on Matrigel-coated 12-well dishes for 2-day with an established TJ-permeability barrier were transfected with Rai14 siRNA duplexes (Rai14 RNAi) versus non-targeting control duplexes (Ctrl RNAi) at 100 nM using Ribojuice transfection medium for 24 hr, thereafter, cells were washed twice and cultured in F12/DMEM for 12 hr to allow recovery. Thereafter, cells were transfected again under the same conditions for another 24 hr. Thereafter, cells were rinsed with fresh F12/DMEM and cultured for an additional 12 hr before termination, and used to prepare lysates for immunoblotting using antibodies against several BTB-associated constituent or regulatory proteins. A knockdown of Rai14 by ~50% was noted in which the control was arbitrarily set at 1 against which statistical comparison was performed (B) without any apparent off-target effects (A). The findings shown herein are the results of 3 independent experiments excluding pilot experiments which were used to establish optimal experimental conditions, such as different concentrations of siRNA duplexes and Ribojuice. It was noted that we achieved only ~50-60% knockdown of Rai14 in several pilot experiments, unlike other target genes [ e.g ., Scribble, beta1-integrin, and P-glycoprotein] wherein we could silence the target gene
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Rai14 is an actin-binding protein in the rat testis. ( A ) A study by RT-PCR to confirm the expression of Rai14 in adult rat testis, Sertoli cells (SC, isolated from 20-day-old rat testes and cultured for 4-day), and germ cells (GC, isolated from adult rat testes and cultured for 16 hr). ( B ) Immunoblotting also confirmed the expression of Rai14 in the rat testis, Sertoli and germ cells, and the relative expression of Rai14 in SC vs. GC was shown in the histogram with n = 3 experiments in which the relative expression level of Rai14 in the testis was arbitrarily set at 1 so that the relative expression level between these samples can be compared. ( C ) The specificity of the anti-Rai14 antibody ( Table 1 ) was assessed by immunoblotting using lysates of GC (20 ug protein). ( D ) Using the specific anti-Rai14 antibody, Rai14 was shown to be an actin-binding protein by co-immunoprecipitation (Co-IP); however, Rai14 did not structurally interact with any of the BTB-associated proteins including several actin-binding and regulatory proteins ( e.g ., Arp3, drebrin E, Eps8) and vimentin (an intermediate filament-based constituent protein). However, Rai14 was found to structurally interact with an actin cross-linking protein palladin which is known to be involved in conferring actin filament bundles in other mammalian cells [48] . ( E ) Rai14 (red) was also shown to be an actin-binding protein by dual-labeled immunofluorescence analysis in which it co-localized with F-acti
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 1 Figure Intravitreal injection of melatonin improves retinal function and maintains iBRB integrity in diabetic rat retinas. (a) Representative ERG waveforms recorded from normal control and 8-week diabetic rats treated with or without melatonin. Statistical analysis of both a-wave (b) and b-wave (c) amplitudes among the three groups ( n = 15 eyes per group). (d) Fluorescent microscopic images of retinal flat-mounts in 8-week diabetic rats and normal control injected with 10 kDa FITC-dextran. Arrows indicate retinal vascular leakage. Scale bars, 100 mum. (e) Quantification of retinal vascular permeability by FITC-dextran ( n = 3 per group). The change of claudin-5 (f), ZO-1 (g), occludin (h), and JAM-A (i) expression was examined with Western blot in 8-week diabetic rat retinas with or without melatonin. Data were expressed as mean +- SEM ( n = 9 per group); p values by one-way ANOVA. * p < .05; ** p < .01; *** p < .001 compared with diabetes group. D, diabetes; ERG, electroretinogram; iBRB, blood-retinal barrier; M, diabetic rats treated with melatonin; N, normal control
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 5 Figure Melatonin decreased paracellular leakage and increased the tight junctions in glyoxal-treated hRMECs. (a) The FITC-dextran leakage was detected in glyoxal-treated hRMECs with or without melatonin ( n = 3 per group). Western blot analysis of occludin (b), ZO-1 (c), and JAM-A (d) in mean +- SEM ( n = 9-10 per group); p values by one-way ANOVA. * p < .05; ** p < .01; *** p < .001 compared with glyoxal group. (e) Representative images of ZO-1 (green) and claudin-5 (red) immunostainings in glyoxal-treated hRMECs with or without melatonin. The nuclei were counterstained with DAPI (blue). Scale bar = 5 mum. FITC-dextran is representative of three independent experiments. DAPI, 4',6-diamidino-2-phenylindole dihydrochloride; hRMECs, human retinal microvascular endothelial cells
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1. H. pylori disrupts the tight junction protein JAM-A in gastric epithelial cell lines, primary cells, and gastric biopsy specimens. (A) JAM-A immunofluorescence (green) in uninfected and H. pylori 26695-infected gastric cell lines MKN74, NCI-N87, and AGS-Ecad; scale bar, 10 mum; n = 3. (B) JAM-A immunofluorescence (red) in uninfected and H. pylori 26695-infected human primary gastric epithelial cells. A pancytokeratin (green) antibody was used to confirm the epithelial origin of the cells. Nuclei were counterstained with DAPI (blue); scale bar, 10 mum; n = 3; (C) JAM-A immunohistochemistry in human gastric biopsies specimens of H. pylori -infected and uninfected patients. Normal JAM-A pattern is a continuous staining circumscribing the apical region of epithelial cells (black arrowheads, upper panels). Altered JAM-A is the interruption of the belt pattern, or loss of expression (lower panels). Pictures were originally obtained with a 1000x magnification. (D) Frequency of JAM-A alterations in 52 gastric biopsy specimens according to the H. pylori colonization density (0, absent; 1, mild; 2, moderate; and 3 marked). The two-sided Mann-Whitney U test was used to determine statistical significance
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2. H. pylori cleaves the cytoplasmic domain of JAM-A. (A) Western blots of JAM-A in uninfected or H. pylori 26695-infected AGS-Ecad cells using antibodies against the cytoplasmic C-terminus and the extracellular N-terminus domain of the protein. Data are representative of n >= 4 experiments; (B) MALDI-TOF mass spectrometry analysis of N-terminus immunoprecipitated JAM-A (from H. pylori 26695-infected and non-infected samples) after separation in SDS-PAGE. Mass spectra were obtained after in gel tryptic digestion of excised bands and peptide peaks related to JAM-A cytoplasmic domains are indicated by arrows with the following correspondence m/z 1020.548 (VIYSQPSAR), m/z 1148.642 (KVIYSQPSAR), and m/z 1458.707 (SEGEFKQTSSFLV). This experiment was performed twice; (C) Protein coverage data from the uninfected and H. pylori -infected samples. Tryptic peptide fragments obtained are depicted in red in the full protein sequence of JAM-A. The JAM-A cytoplasmic domain is identified by the blue boxes. (D and E) MALDI-TOF mass spectrometry analyses of a synthetic peptide of the full cytoplasmic domain of JAM-A incubated with ultra-pure water (D) or with H. pylori 26695 sonicate (E). Results are representative of n >= 5 experiments, each with similar results. Arrows indicate the peak which corresponds to the full peptide (black), and the peaks obtained after incubation with H. pylori (blue, major pair of peptides; red, minor pair of peptides)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
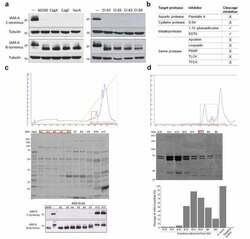
- Figure 5. Identification of the H. pylori factor that cleaves JAM-A. (A) Western blot of JAM-A in AGS-Ecad cells infected with H. pylori 60190, 60190CagA - , 60190CagE - , or 60190VacA - mutants, or with clinical isolates using antibodies to detect the C- or N-terminus of JAM-A; tubulin was used as loading control. (B) Screening of the type of protease involved in JAM-A cleavage with a panel of protease inhibitors. Cleavage was evaluated by MALDI-TOF/TOF analysis using a cytoplasmic domain JAM-A peptide ( 277 KVIYSQPSARSEGEFK 292 ) incubated with H. pylori 26695 lysates in the presence of the inhibitors. , cleavage inhibition; X, no cleavage inhibition. Experiments were performed n >= 2, with similar results. c) Upper panel , representative chromatogram obtained after separation of the 100 mM NaCl fraction from Q-Sepharose on SOURCE-15Q. Red boxes, fractions (A2 to A6) selected for size exclusion chromatography (SEC). Middle panel , Coomassie blue-stained SDS-PAGE gel of separated fractions. Bottom panel , detection of the H. pylori protease by western blot after incubation of the fractions with AGS-Ecad cell lysates. H. pylori 26695 and uninfected cell lysates were used as positive and negative controls, respectively. (D) Upper panel , representative chromatogram obtained from SEC after separation of fractions A2 to A6 obtained from SOURCE-15Q. Green box, fractions (A10 to B8) selected for the detection of cleavage activity; Red box, fraction selected for protein identificat
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 BmpR1a DeltaFoxL1+ mice displayed abnormalities in goblet cell vesicles and presented dysregulated mucus maturation and structural gene expression. ( A ) Relative mRNA levels of the colonocyte/secretory lineage specification Hes1 and Atoh1 , along with committed-secretory cell toward the goblet fate or maturation Gfi1 , Spdef and Klf4 . A significant decrease was observed for Klf4 mRNA levels in mutant mice while other mRNAs were not modulated between mutant and control mice ( N = 7-9). ( B ) Ultrastructural analysis of the goblet cells revealed vesicles without mucin diversity, disturbed morphology in BmpR1a DeltaFoxL1+ mice ( N = 4). ( C ) Vesicles pattern quantification revealed a significant increase in light grey vesicles in mutant goblet cells when compared to controls ( N = 3). ( D ) BmpR1a DeltaFoxL1+ mice showed a significant increase in Fut2 , Muc2 , Muc4 and Agr2 mRNA levels in mutant mice. ( E ) Ultrastructural analysis of the glycocalyx and the apical junctional complex (yellow bracket) revealed that the latter was not modified between both groups and showed loss of glycocalyx in BmpR1a DeltaFoxL1+ mice ( N = 4). ( F ) Immunofluorescence against ZO-1, claudin-1 and 2 as well as JAM-A revealed no modulation in the tight junction complex proteins between both groups ( N = 3). ( C ) 2-way ANOVA test. Data are presented as the mean +- SD. ( D ) Mann-Whitney test. N: Nucleus; Mu: Mucin vesicles; G: Glycocalyx; AJC: Apical junctional complex. Fold-change was n
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
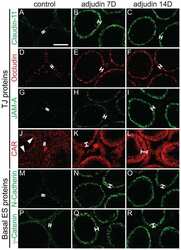
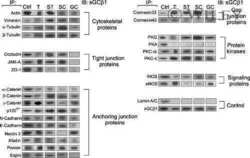
- 2 A, B : A study by immunoblotting to assess changes in TJ-associated proteins in the rat testis during androgen suppression-induced germ cell loss and its recovery. A: Immunoblotting results in which lysates of testes containing ~150 mug protein from each sample within an experimental group were resolved by SDS-PAGE, and the blots were probed with different primary antibodies. Immunoblot data were arranged into two groups: androgen depletion induced by TE implants and the two recovery groups. In the recovery phase [either via spontaneous recovery (SR) or under high T condition with 4 x 4 cm T implants (+T4)], the column labeled as 30/35D represents recovery under high T condition on day 30 or spontaneous recovery on day 35. The bottom part is the same blot as those shown above, but reprobed with an anti-actin antibody to assess equal protein loading. B: These are densitometrically-scanned data using immunoblots such as those shown in (A). All data were normalized against beta-actin to account for uneven protein loading and the level of a target protein in control rats was arbitrarily set at 1. Each data point is the mean +- SD of three rats. ns, not significantly different from control as determined by ANOVA; *, significantly different, P < 0.05; **, significantly different, P < 0.01.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Aroclor1254 accelerated junction protein endocytosis and ubiquitination via p38 MAPK. ( a ) Immunoblot analysis of endocytosed JAM-A, N-cadherin, and beta -catenin in SCs at different time points (0, 10, 30, 60 min) after cell surface biotinylation for 30 min in the presence of Aroclor1254 (or the mixture of Aroclor1254 and SB203580). The endocytosed proteins were pulled-down by UltraLink Immobilized NeutrAvidin Plus Resin after stripping residual biotin on cell membrane as described in 'Materials and methods' section. Total biotinylated proteins at 0 min without stripping were also detected as a positive control. Cell lysates without pull-down were also analyzed by immunoblot to confirm identical levels of the tested protein between groups with GAPDH served as the loading control. ( b ) Line and scatter graphs summarizing the result shown in a by calculating the percentage of endocytosed protein at each data point versus the total biotinylated protein. Each bar refers to mean+-S.D. of n =3 independent experiments using SCs cultured from different rats. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 The effects of Aroclor1254 on the barrier function of SC epithelium after inhibiting the caveolin-dependent endocytosis. The SCs were treated with or without Aroclor1254 in the presence or absence of CO as the regimen depicted in Supplementary Figure S2a . ( a and b ) The barrier function of SC epithelium measured by recording TER ( a ) or calculating the permeability of Na-F ( b ) across the cell monolayer. The Na-F permeability in the vehicle control on day 3 was arbitrarily set at 100%. CO treatment was found to partially resist the disruption of BTB by Aroclor1254. Each bar refers to mean+-S.D. of n =3 experiments using different batches of SCs. ** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 5 Re-establishment of BTB in the recipient testes after repaired SSC transplantation. A , B Distribution of N-cadherin and JAM-A (all in green) were found to be different in different tubules, depending on the status of spermatogenesis and the differentiation status of repaired SSCs. Abnormal distribution of these proteins in empty tubules devoid of germ cells (a and g) was obviously noted and gradually returned to normalcy as showed in normal wild type control testis (e and k) following the appearance of spermatocytes (b and h), round spermatids (c and i), and elongated spermatids (d and j). Spermatocytes were labeled by anti-SCP3 antibody (red), and round spermatids and elongated spermatids were identified by the corresponding nucleus shape (blue). There were no germ cells in panels a, a', f, g, g', and l. The boxed areas in a-d and g-j are magnified and shown in a'-d' and g'-j'. Scale bar = 50 mum in a-l and 10 mum in a'-d' and g'-j'
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA