Antibody data
- Antibody Data
- Antigen structure
- References [1]
- Comments [0]
- Validations
- Western blot [2]
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-1064G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-PTEN (Ser385) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Storage
- -20°C
Submitted references Loss of NDRG2 expression activates PI3K-AKT signalling via PTEN phosphorylation in ATLL and other cancers.
Nakahata S, Ichikawa T, Maneesaay P, Saito Y, Nagai K, Tamura T, Manachai N, Yamakawa N, Hamasaki M, Kitabayashi I, Arai Y, Kanai Y, Taki T, Abe T, Kiyonari H, Shimoda K, Ohshima K, Horii A, Shima H, Taniwaki M, Yamaguchi R, Morishita K
Nature communications 2014 Feb 26;5:3393
Nature communications 2014 Feb 26;5:3393
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Peptide Competition and Mutant Analysis. Lysates prepared from HEK293 cells transiently transfected with WT PTEN (1, 3-6) or mutant S385A (2) were resolved by SDS-PAGE on a 10% polyacrylamide gel and transferred to PVDF. Membranes were either left untreated (1-5) or treated with lambda phosphatase (6), blocked with a 3% Milk-TBST buffer for one hour at room temperature, and incubated with PTEN (pS385) antibody for two hours at room temperature in a 3% Milk-TBST buffer, following prior incubation with: no peptide (1, 2, 6), the non-phosphopeptide corresponding to the immunogen (3), a generic phosphoserine-containing peptide (4), or, the phosphopeptide immunogen (5). After washing, membranes were incubated with goat F (ab)2 anti-rabbit IgG HRP conjugate (Product # ALI4404) and bands were detected using the Pierce SuperSignal method.The data show that only the peptide corresponding to PTEN (pS385) blocks the antibody signal. Moreover, the signal is lost in the S385A single mutant (kindly provided by Dr. A. Hall). The data also show that phosphatase stripping eliminates the signal, verifying that the antibody is phospho-specific.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
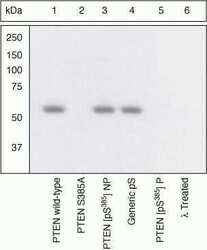
- Experimental details
- Peptide Competition and Mutant Analysis. Lysates prepared from HEK293 cells transiently transfected with WT PTEN (1, 3-6) or mutant S385A (2) were resolved by SDS-PAGE on a 10% polyacrylamide gel and transferred to PVDF. Membranes were either left untreated (1-5) or treated with lambda phosphatase (6), blocked with a 3% Milk-TBST buffer for one hour at room temperature, and incubated with PTEN (pS385) antibody for two hours at room temperature in a 3% Milk-TBST buffer, following prior incubation with: no peptide (1, 2, 6), the non-phosphopeptide corresponding to the immunogen (3), a generic phosphoserine-containing peptide (4), or, the phosphopeptide immunogen (5). After washing, membranes were incubated with goat F (ab’)2 anti-rabbit IgG HRP conjugate (Product # ALI4404) and bands were detected using the Pierce SuperSignal™ method.The data show that only the peptide corresponding to PTEN (pS385) blocks the antibody signal. Moreover, the signal is lost in the S385A single mutant (kindly provided by Dr. A. Hall). The data also show that phosphatase stripping eliminates the signal, verifying that the antibody is phospho-specific.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 The PI3K-AKT signalling pathway is activated in ATLL cells. ( a ) A western blot analysis of NDRG2, PTEN, phosphorylated PTEN (p-PTEN) (Ser370, Ser380/Thr382/Thr383 and Ser385), non-p-PTEN (Ser380/Thr382/Thr383), SHIP1, AKT, p-AKT (Ser473 and Thr308) and Tax was performed in primary leukaemic cells from acute-type ATLL patients. The CD4 + T lymphocytes from healthy volunteers (CD4 + T lymph) served as the controls. No expression of Tax was detected in CD4 + T lymphocytes from healthy volunteers or ATLL cells from acute-type ATLL patients. The graph shows relative band intensity of p-PTEN (Ser380/Thr382/Thr383) and non-p-PTEN (Ser380/Thr382/Thr383). Each p-PTEN or non-p-PTEN value was divided by the total PTEN value for a given sample. The data are expressed as the mean value+-s.d. (* P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Decreased expression of NDRG2 and the enhanced phosphorylation of PTEN are involved in the activation of PI3K-AKT. ( a ) Western blots analysis of stably transfected KK1 cells. Note that the amount of NDRG2 protein in both MOLT4 and NDRG2-transfected KK1 cells was found to be similar. The data are representative of three experiments. ( b ) The proliferation rates of KK1-NDRG2, KK1-Mock, and parental KK1 cells. The mean+-s.d. is shown; * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 NDRG2 recruits PP2A to PTEN leading to its dephosphorylation at pSer380/pThr382/pThr383. ( a ) KK1-NDRG2 cell lysates were incubated with a pSer380/pThr382/pThr383 PTEN phosphopeptide in the presence or absence of different concentrations of OA. The amount of released phosphate was quantitated using the malachite green assay. The mean+-s.d. is shown; ** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 Downregulation of NDRG2 is associated with enhanced phosphorylation of PTEN-Ser380/Thr382/Thr383 and enhanced activation of PI3K-AKT in various cancers. ( a ) Bisulfite genomic sequencing of the NDRG2 promoter region in the KLM1 (pancreatic cancer), SKOV3 (ovarian cancer), HeLa (cervical cancer), HepG2 (hepatic cancer) and KatoIII (gastric cancer) cell lines. PCR products amplified from bisulfite-treated genomic DNA were subcloned, and ten clones in each cell line were sequenced. Open circles indicate unmethylated CpGs (Thy) and filled circles indicate methylated CpGs (Cyt). The region sequenced spans from -396 bp to -133 bp. ( b ) SKOV3, HepG2 and KLM1 cells were cultured with 10 muM 5-aza-dC for 72 h, with 1.2 muM TSA for 48 h, or with 1.2 muM of TSA for 48 h, followed by 10 muM of 5-aza-dC for 24 h. After treatments, total RNA was extracted and quantitative RT-PCR was performed with NDRG2 and beta-actin. The relative amounts of mRNA were normalized against beta-actin mRNA and expressed relative to the mRNA abundance in untreated cells. The mean+-s.d. is shown; * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 NDRG2 -deficient mice are susceptible to tumour formation. ( a ) Western blots of 3-month-old adult tissues of WT mice. The data are representative of two experiments. ( b ) Western blots of various tissues of 3-month-old NDRG2 -deficient mice. The data are representative of two experiments. ( c ) The co-immunoprecipitation of PTEN and NDRG2 in frontal cortex homogenates from 3-month-old WT mice. The data are representative of three experiments. ( d ) Whole-cell lysates from WT, NDRG2 +/- and NDRG2 -/- embryonic fibroblasts were subjected to western blotting. ( e ) The proliferation rates of WT, NDRG2 +/- , and NDRG2 -/- embryonic fibroblasts. The mean+-s.d. is shown; * P
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot