Antibody data
- Antibody Data
- Antigen structure
- References [21]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [9]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 17-1979-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD197 (CCR7) Monoclonal Antibody (3D12), APC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 3D12 monoclonal antibody reacts with human CCR7, also known as EBI-1 and CD197. CCR7 is a member of the G-protein-coupled chemokine receptor family with seven membrane-spanning domains and functions as a receptor for 6Ckine/SLC (secondary lymphoid-tissue chemokine), CCL19 and CCL21. CCR7 has been shown to be internalized via clathrin-coated pits and the majority recycled back to the plasma membrane. CCR7 is expressed on T cells and can be used to distinguish populations of naive from central and effector memory T cells. CCR7 has been shown to play a role in migration of memory T cells to inflamed tissue. Expression of CCR7 is also found on DC's. During DC maturation CCR7 expression increases and is thought to be involved in a variety of functions: chemotaxis to the lymph node, cellular architecture, rate of endocytosis, survival and maturation. Expression of CCR7 on the cell surface can be down regulated upon ligand binding. Applications Reported: This 3D12 antibody has been reported for use in flow cytometric analysis. Applications Tested: This 3D12 antibody has been pre-titrated and tested by flow cytometric analysis of peripheral blood mononuclear cells. This can be used at 5 µL (0.125 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. It is recommended that the staining incubation time be increased to at least 45 minutes at 2-8°C for optimal staining. Excitation: 633-647 nm; Emission: 660 nm; Laser: Red Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Rat
- Isotype
- IgG
- Antibody clone number
- 3D12
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Alteration of the Immune Microenvironment in HBsAg and HBeAg Dual-Positive Pregnant Women Presenting a High HBV Viral Load.
Neuroimmune Consequences of eIF4E Phosphorylation on Chemotherapy-Induced Peripheral Neuropathy.
Longitudinal high-throughput TCR repertoire profiling reveals the dynamics of T-cell memory formation after mild COVID-19 infection.
Application of the chemokine-chemokine receptor axis increases the tumor-targeted migration ability of cytokine-induced killer cells in patients with colorectal cancer.
Single-Cell Profiling Defines Transcriptomic Signatures Specific to Tumor-Reactive versus Virus-Responsive CD4(+) T Cells.
Biochanin a Enhances the Defense Against Salmonella enterica Infection Through AMPK/ULK1/mTOR-Mediated Autophagy and Extracellular Traps and Reversing SPI-1-Dependent Macrophage (MΦ) M2 Polarization.
Clinically compliant spatial and temporal imaging of chimeric antigen receptor T-cells.
Oral Supplementation with Baker's Yeast Beta Glucan Is Associated with Altered Monocytes, T Cells and Cytokines following a Bout of Strenuous Exercise.
Role of Nuclear Factor (Erythroid-Derived 2)-Like 2 Signaling for Effects of Fumaric Acid Esters on Dendritic Cells.
The upregulation of LAG-3 on T cells defines a subpopulation with functional exhaustion and correlates with disease progression in HIV-infected subjects.
Baseline natural killer and T cell populations correlation with virologic outcome after regimen simplification to atazanavir/ritonavir alone (ACTG 5201).
Skewed distribution of IL-7 receptor-α-expressing effector memory CD8+ T cells with distinct functional characteristics in oral squamous cell carcinoma.
Secondary lymphoid organ homing phenotype of human myeloid dendritic cells disrupted by an intracellular oral pathogen.
Maintenance of intestinal Th17 cells and reduced microbial translocation in SIV-infected rhesus macaques treated with interleukin (IL)-21.
Expression of the memory marker CD45RO on helper T cells in macaques.
Vitamin D suppression of endoplasmic reticulum stress promotes an antiatherogenic monocyte/macrophage phenotype in type 2 diabetic patients.
Haemophilus ducreyi-induced interleukin-10 promotes a mixed M1 and M2 activation program in human macrophages.
Human CD8⁺ and CD4⁺ T cell memory to lymphocytic choriomeningitis virus infection.
Role of TLX1 in T-cell acute lymphoblastic leukaemia pathogenesis.
Proliferation and differentiation potential of human CD8+ memory T-cell subsets in response to antigen or homeostatic cytokines.
Two subsets of memory T lymphocytes with distinct homing potentials and effector functions.
Gao F, Wang H, Li X, Guo F, Yuan Y, Wang X, Zhang Y, Bai G
Journal of inflammation research 2021;14:5619-5632
Journal of inflammation research 2021;14:5619-5632
Neuroimmune Consequences of eIF4E Phosphorylation on Chemotherapy-Induced Peripheral Neuropathy.
Agalave NM, Mody PH, Szabo-Pardi TA, Jeong HS, Burton MD
Frontiers in immunology 2021;12:642420
Frontiers in immunology 2021;12:642420
Longitudinal high-throughput TCR repertoire profiling reveals the dynamics of T-cell memory formation after mild COVID-19 infection.
Minervina AA, Komech EA, Titov A, Bensouda Koraichi M, Rosati E, Mamedov IZ, Franke A, Efimov GA, Chudakov DM, Mora T, Walczak AM, Lebedev YB, Pogorelyy MV
eLife 2021 Jan 5;10
eLife 2021 Jan 5;10
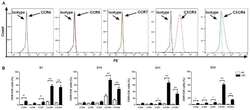
Application of the chemokine-chemokine receptor axis increases the tumor-targeted migration ability of cytokine-induced killer cells in patients with colorectal cancer.
Zou Y, Liang J, Li D, Fang J, Wang L, Wang J, Zhang J, Guo Q, Yan X, Tang H
Oncology letters 2020 Jul;20(1):123-134
Oncology letters 2020 Jul;20(1):123-134
Single-Cell Profiling Defines Transcriptomic Signatures Specific to Tumor-Reactive versus Virus-Responsive CD4(+) T Cells.
Magen A, Nie J, Ciucci T, Tamoutounour S, Zhao Y, Mehta M, Tran B, McGavern DB, Hannenhalli S, Bosselut R
Cell reports 2019 Dec 3;29(10):3019-3032.e6
Cell reports 2019 Dec 3;29(10):3019-3032.e6
Biochanin a Enhances the Defense Against Salmonella enterica Infection Through AMPK/ULK1/mTOR-Mediated Autophagy and Extracellular Traps and Reversing SPI-1-Dependent Macrophage (MΦ) M2 Polarization.
Zhao X, Tang X, Guo N, An Y, Chen X, Shi C, Wang C, Li Y, Li S, Xu H, Liu M, Wang Y, Yu L
Frontiers in cellular and infection microbiology 2018;8:318
Frontiers in cellular and infection microbiology 2018;8:318
Clinically compliant spatial and temporal imaging of chimeric antigen receptor T-cells.
Emami-Shahri N, Foster J, Kashani R, Gazinska P, Cook C, Sosabowski J, Maher J, Papa S
Nature communications 2018 Mar 14;9(1):1081
Nature communications 2018 Mar 14;9(1):1081
Oral Supplementation with Baker's Yeast Beta Glucan Is Associated with Altered Monocytes, T Cells and Cytokines following a Bout of Strenuous Exercise.
McFarlin BK, Venable AS, Carpenter KC, Henning AL, Ogenstad S
Frontiers in physiology 2017;8:786
Frontiers in physiology 2017;8:786
Role of Nuclear Factor (Erythroid-Derived 2)-Like 2 Signaling for Effects of Fumaric Acid Esters on Dendritic Cells.
Hammer A, Waschbisch A, Knippertz I, Zinser E, Berg J, Jörg S, Kuhbandner K, David C, Pi J, Bayas A, Lee DH, Haghikia A, Gold R, Steinkasserer A, Linker RA
Frontiers in immunology 2017;8:1922
Frontiers in immunology 2017;8:1922
The upregulation of LAG-3 on T cells defines a subpopulation with functional exhaustion and correlates with disease progression in HIV-infected subjects.
Tian X, Zhang A, Qiu C, Wang W, Yang Y, Qiu C, Liu A, Zhu L, Yuan S, Hu H, Wang W, Wei Q, Zhang X, Xu J
Journal of immunology (Baltimore, Md. : 1950) 2015 Apr 15;194(8):3873-82
Journal of immunology (Baltimore, Md. : 1950) 2015 Apr 15;194(8):3873-82
Baseline natural killer and T cell populations correlation with virologic outcome after regimen simplification to atazanavir/ritonavir alone (ACTG 5201).
McKinnon JE, Mailliard RB, Swindells S, Wilkin TJ, Borowski L, Roper JM, Bastow B, Kearney M, Wiegand A, Mellors JW, Rinaldo CR, A5201 study team
PloS one 2014;9(5):e95524
PloS one 2014;9(5):e95524
Skewed distribution of IL-7 receptor-α-expressing effector memory CD8+ T cells with distinct functional characteristics in oral squamous cell carcinoma.
Lee JJ, Yeh CY, Jung CJ, Chen CW, Du MK, Yu HM, Yang CJ, Lin HY, Sun A, Ko JY, Cheng SJ, Chang YL, Chia JS
PloS one 2014;9(1):e85521
PloS one 2014;9(1):e85521
Secondary lymphoid organ homing phenotype of human myeloid dendritic cells disrupted by an intracellular oral pathogen.
Miles B, Zakhary I, El-Awady A, Scisci E, Carrion J, O'Neill JC, Rawlings A, Stern JK, Susin C, Cutler CW
Infection and immunity 2014 Jan;82(1):101-11
Infection and immunity 2014 Jan;82(1):101-11
Maintenance of intestinal Th17 cells and reduced microbial translocation in SIV-infected rhesus macaques treated with interleukin (IL)-21.
Pallikkuth S, Micci L, Ende ZS, Iriele RI, Cervasi B, Lawson B, McGary CS, Rogers KA, Else JG, Silvestri G, Easley K, Estes JD, Villinger F, Pahwa S, Paiardini M
PLoS pathogens 2013;9(7):e1003471
PLoS pathogens 2013;9(7):e1003471
Expression of the memory marker CD45RO on helper T cells in macaques.
Valentine M, Song K, Maresh GA, Mack H, Huaman MC, Polacino P, Ho O, Cristillo A, Kyung Chung H, Hu SL, Pincus SH
PloS one 2013;8(9):e73969
PloS one 2013;8(9):e73969
Vitamin D suppression of endoplasmic reticulum stress promotes an antiatherogenic monocyte/macrophage phenotype in type 2 diabetic patients.
Riek AE, Oh J, Sprague JE, Timpson A, de las Fuentes L, Bernal-Mizrachi L, Schechtman KB, Bernal-Mizrachi C
The Journal of biological chemistry 2012 Nov 9;287(46):38482-94
The Journal of biological chemistry 2012 Nov 9;287(46):38482-94
Haemophilus ducreyi-induced interleukin-10 promotes a mixed M1 and M2 activation program in human macrophages.
Li W, Katz BP, Spinola SM
Infection and immunity 2012 Dec;80(12):4426-34
Infection and immunity 2012 Dec;80(12):4426-34
Human CD8⁺ and CD4⁺ T cell memory to lymphocytic choriomeningitis virus infection.
Kotturi MF, Swann JA, Peters B, Arlehamn CL, Sidney J, Kolla RV, James EA, Akondy RS, Ahmed R, Kwok WW, Buchmeier MJ, Sette A
Journal of virology 2011 Nov;85(22):11770-80
Journal of virology 2011 Nov;85(22):11770-80
Role of TLX1 in T-cell acute lymphoblastic leukaemia pathogenesis.
Riz I, Hawley TS, Johnston H, Hawley RG
British journal of haematology 2009 Apr;145(1):140-3
British journal of haematology 2009 Apr;145(1):140-3
Proliferation and differentiation potential of human CD8+ memory T-cell subsets in response to antigen or homeostatic cytokines.
Geginat J, Lanzavecchia A, Sallusto F
Blood 2003 Jun 1;101(11):4260-6
Blood 2003 Jun 1;101(11):4260-6
Two subsets of memory T lymphocytes with distinct homing potentials and effector functions.
Sallusto F, Lenig D, Förster R, Lipp M, Lanzavecchia A
Nature 1999 Oct 14;401(6754):708-12
Nature 1999 Oct 14;401(6754):708-12
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
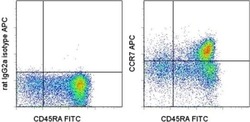
- Experimental details
- Staining of normal human peripheral blood cells with Anti-Human CD45RA FITC (Product # 11-0458-42) and Rat IgG2a K Isotype Control APC (Product # 17-4321-81) (left) or Anti-Human CD197 (CCR7) APC (right). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
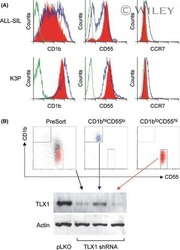
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
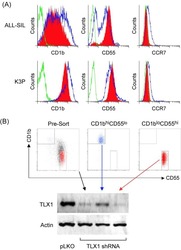
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 3. Expression levels of chemokine receptors on CIK cells generated from patients with CRC and healthy donors. (A) Analysis of the expression levels of CCR4, CCR5, CCR7, CXCR3 and CXCR4 on CIK cells via flow cytometry revealed that CXCR3 and CXCR4 had higher expression levels on CIK cells compared with isotype, expression profiles of CCR4, CCR5 and CCR7 did not show significant changes compared with isotype. (B) Dynamic changes of chemokine receptors CCR4, CCR5, CCR7, CXCR3 and CXCR4 of CIK cells on D7, 14, 21 and 28 were detected in patients with CRC and HD CIK cells via flow cytometry. The result revealed that expression levels of CXCR3 and CXCR4 were significantly higher on CIK cells cultured from CRC compared with HD at all the time points analyzed, while the expression levels of CCR4, CCR5 and CCR7 were significantly higher on CIK cells cultured from CRC compared with HD at different time points analyzed (CCR4 on D7 and D28; CCR5 on D28; CCR7 on D7, D21 and D28). All comparisons were performed using unpaired Student''s t-test with Welch''s correction. *P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Paclitaxel dysregulates CD4 + and CD8 + T-cell subpopulations in an eIF4E-dependent manner. (A) Gating strategy used for flow cytometry of lymphocytes from popliteal and inguinal lymph nodes. T-cells were separated from the whole lymphoid cells population using CD3. CD3 + cells were then gated for CD4 + T-cell and CD8 + T-cells. For each of these subsets, cells were further gated for CCR7 or CD25 and CD44. (B) Quantification of CD4 + T-cells from the CD3 population from male mice. (C) CCR7 + T-cells from isolated CD4 + population in males, ** p 0.0063. (D) CD4 + cells gated for CD25 and/or CD44 in males. (E) Quantification of CD4 + T-cells from the CD3 + population from female mice, * p 0.0101. (F) CCR7 + T-cells from isolated CD4 + population in female mice, * p 0.0250. (G) CD4 + cells gated for CD25 and/or CD44 in female mice, ** p 0.0011. (H) Quantification of CD8 + T-cells from the CD3 + population from male mice. (I) CCR7 + T-cells from isolated CD8 + population in males, * p 0.0433. (J) CD8 + cells gated for CD25 and/or CD44 in males, * p 0.0396. (K) Quantification of CD8 + T-cells from the CD3 population from female mice, * p 0.041. (L) CCR7 + T-cells from isolated CD8 + population in female mice, ** p 0.0097. (M) CD8 + cells gated for CD25 and/or CD44 in female mice. All data are presented as mean +- standard error of the mean, n = 3 mice each for male/female WT vehicle-treated and WT paclitaxel groups, n = 4 mice each for male/female eIF4E S209A vehicle-trea
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
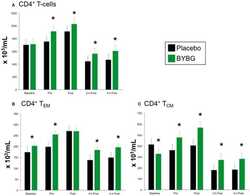
- Figure 5 Effect of BYBG on CD4+ T cells. CD4+ T cell concentrations were determined using peripheral blood mononuclear cells (PBMC) isolated from human whole blood samples. PBMCs were analyzed using flow cytometry to determine total ( A , CD3+/CD4+), T EM ( B , CCR7-/CD45RA-), and T CM ( C , CCR7+/CD45RA-) concentrations. Placebo (black bars) and BYBG (green bars) are represented separately and were statistically compared. Values represent the mean +- SEM. * indicates BYBG significantly different than placebo ( p < 0.05).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
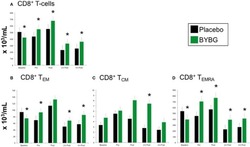
- Figure 6 Effect of BYBG on CD8+ T cells. CD8+ T cell concentrations were determined using peripheral blood mononuclear cells (PBMC) isolated from human whole blood samples. PBMCs were analyzed using flow cytometry to determine total ( A , CD3+/CD8+), T EM ( B , CCR7-/CD45RA-), and T CM ( C , CCR7+/CD45RA-), and T EMRA ( D , CCR7-/CD45RA+) concentrations. Placebo (black bars) and BYBG (green bars) are represented separately and were statistically compared. Values represent the mean +- SEM. * indicates BYBG significantly different than placebo ( p < 0.05).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
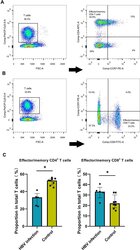
- Figure 2 HBV-related samples display higher proportion of effector/memory CD8+ T cells by flow cytometry assay. ( A ) Examples of CD3, CD4 and CCR7 staining to determine effector/memory CD4+ T cells. ( B ) Examples of CD3, CD8 and CCR7 staining to determine effector/memory CD8+ T cells. ( C ) The average proportion of effector/memory CD4+ T cells and CD8+ T cells in HBV and control groups (*p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
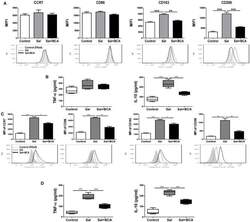
- Figure 7 BCA regulate MPhi polarization in vitro and in vivo . (A,B) Raw246.7 cells were treated with 4 mug/ml BCA for 2 h. Then, the cells were infected with Salmonella at MOI = 10:1 for 2 h. (A) The expression of surface markers (CCR7, CD86, CD163, and CD206) was determined by flow cytometry. The results are presented as MFI. (B) TNF-alpha and IL-10 in the supernatants were detected by ELISA. ** P < 0.01, *** P < 0.001. The data are representative of three experiments with similar results. (C,D) Mice were infected by intragastric administration of an overnight culture of Salmonella (10 5 bacteria in 0.1 ml PBS) through a gavage tube and then treated with 6.25 mg/kg BCA intragastrically by gavage daily (5 mice for each group). On the 5th day p.i., the mice were sacrificed and the peritoneal fluid was collected. (C) The expression of surface markers (CCR7, CD86, CD163, and CD206) in mouse peritoneal MPhis were determined by flow cytometry The results are presented as MFI. (D) TNF-alpha and IL-10 in ascitic fluid were detected by ELISA. * P < 0.05, ** P < 0.01, *** P < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
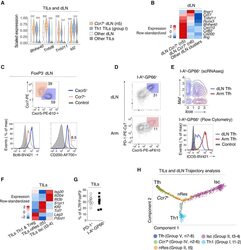
- Figure 4. Transcriptomic Continuum between TIL and dLN Tumor-Reactive Cells (A) Violin plots of differentially expressed genes across TIL group I Th1 and dLN group IV Ccr7 + (clusters t1-2 and n5, respectively, as shown in Figure 1A ), as well as all other TIL and dLN populations. Unpaired t-test; ***p < 0.001. (B) Heatmap shows row-standardized expression of differentially expressed genes across dLN Ccr7 + clusters (group IV n5-6) and other dLN clusters (Treg and Tfh clusters n1 and n7-8, respectively). (C) Flow cytometry contour plots of Cxcr5 versus Ccr7 in Foxp3 - dLN cells (top). Overlaid protein expression of Bcl6 and CD200 in Ccr7 + and Cxcr5 + dLN cells and naive CD4 + splenocytes from tumor-free control mice (bottom). Data are representative of 17 mice analyzed in three experiments. (D) Flow cytometry contour plots of Cxcr5 versus PD-1 in dLN and Arm cells. Data are representative of 10 mice analyzed in two experiments. (E) Contour plot of dLN (red, clusters n7-8) and Arm (blue) Tfh cell distribution according to scRNA-seq-detected normalized expression of Icos versus Maf (top).Overlaid protein expression of ICOS in dLN and Arm PD-1 + Cxcr5 + (Tfh) cells and naive CD4 + splenocytes from tumor-free control mice (bottom). (F) Heatmap shows row-standardized expression of differentially expressed genes across TIL Isc and nRes clusters (as defined in the text, group II t3-4 and t5,respectively) and all other TIL clusters (Th1 and Treg clusters t1-2 and t6-7, respectively)
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry