Antibody data
- Antibody Data
- Antigen structure
- References [2]
- Comments [0]
- Validations
- Immunocytochemistry [2]
- Immunoprecipitation [1]
- Immunohistochemistry [2]
- Flow cytometry [1]
- Chromatin Immunoprecipitation [2]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-362G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- STAT2 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 μL
- Storage
- -20°C
Submitted references Temporal regulation of interferon signalling in human EndoC-βH1 cells.
NS5 Sumoylation Directs Nuclear Responses That Permit Zika Virus To Persistently Infect Human Brain Microvascular Endothelial Cells.
Dhayal S, Leslie KA, Baity M, Akhbari P, Richardson SJ, Russell MA, Morgan NG
Journal of molecular endocrinology 2022 May 19;69(2):299-313
Journal of molecular endocrinology 2022 May 19;69(2):299-313
NS5 Sumoylation Directs Nuclear Responses That Permit Zika Virus To Persistently Infect Human Brain Microvascular Endothelial Cells.
Conde JN, Schutt WR, Mladinich M, Sohn SY, Hearing P, Mackow ER
Journal of virology 2020 Sep 15;94(19)
Journal of virology 2020 Sep 15;94(19)
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
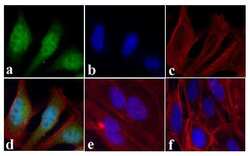
- Experimental details
- Immunofluorescent analysis of STAT2 was done on 70% confluent log phase HeLa cells treated with IFN-gamma (100 ng/mL for 30 min). The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton™ X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with STAT2 Rabbit polyclonal Antibody (Product # 44-362G) at 1:250 dilution in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Goat Anti-Rabbit IgG Secondary Antibody (Product # A-11008) at a dilution of 1:400 for 30 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing nuclear and cytoplasmic localization upon treatment. Panel e showing cytoplasmic localization in untreated HeLa cells. Panel f is a no primary antibody control. The images were captured at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
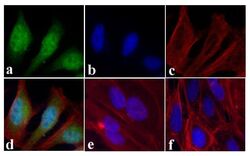
- Experimental details
- Immunofluorescent analysis of STAT2 was done on 70% confluent log phase HeLa cells treated with IFN-gamma (100 ng/mL for 30 min). The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton™ X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with STAT2 Rabbit polyclonal Antibody (Product # 44-362G) at 1:250 dilution in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Goat Anti-Rabbit IgG Secondary Antibody (Product # A-11008) at a dilution of 1:400 for 30 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing nuclear and cytoplasmic localization upon treatment. Panel e showing cytoplasmic localization in untreated HeLa cells. Panel f is a no primary antibody control. The images were captured at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
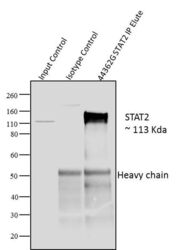
- Experimental details
- Immunoprecipitation of STAT2 was performed with 10 µl of the STAT2 Rabbit Polyclonal Antibody (Product # 44-362G) on cell extract from A431 cells using the Dynabeads® Protein A Immunoprecipitation Kit (10006D). Normal Rabbit IgG was used as a negative IP control. Subsequently, western blot analysis was performed using Novex®NuPAGE® 12 % Bis-Tris gel (Product # NP0341BOX), XCell SureLock Electrophoresis System (Product # EI0002), Novex® Sharp Pre-Stained Protein Standard (LC5800). Proteins were transferred using Pierce G2 Fast Blotter (Product # 62287) to a nitrocellulose and blocked with 5% skim milk for 1 hour at room temperature on a rocking platform. STAT2 was detected at 113 kDa usingSTAT2 Rabbit Polyclonal Antibody (Product # 44-362G) at 1:1000 dilution in 5% skim milk at 4°C overnight on a rocking platform. Goat Anti-Rabbit IgG - HRP Secondary Antibody (G21234) at 1:5000 dilution was used and chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106). Lane 1 represents 10% of the total cell extract (input), Lane 2 is the IP performed with Rabbit IgG and Lane 3 represents IP performed withSTAT2 Rabbit Polyclonal Antibody (Product # 44-362G).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
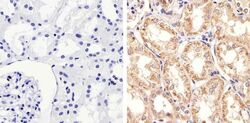
- Experimental details
- Immunohistochemistry analysis of STAT2 showing staining in the cytoplasm of paraffin-embedded human kidney tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a STAT2 Rabbit Polyclonal Antibody (Product # 44-362G) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
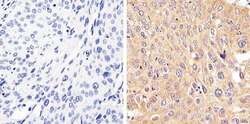
- Experimental details
- Immunohistochemistry analysis of STAT2 showing staining in the cytoplasm of paraffin-embedded human cervical carcinoma (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a STAT2 Rabbit Polyclonal Antibody (Product # 44-362G) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
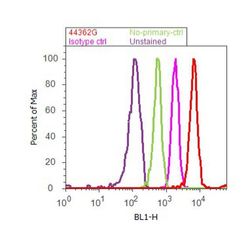
- Experimental details
- Flow cytometry analysis of STAT2 was done on Jurkat cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Tritonª X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with STAT2 Rabbit Polyclonal Antibody (44362G, red histogram) or with rabbit isotype control (pink histogram) at 3-5 µg/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor¨ 488 Goat Anti-Rabbit Secondary Antibody (A11008) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune¨ Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
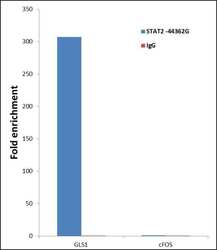
- Experimental details
- ChIP- qPCR analysis of STAT2 was performed with 10 µL of the STAT2 Rabbit polyclonal antibody (Product # 44-362G) on sheared chromatin from 2 million HeLa cells treated with 100 ng/mL of IFN Alpha for one hour using the MAGnify Chromatin Immunoprecipitation System (Product # 49-2024). Normal Rabbit IgG was used as a negative IP control. The purified DNA from each ChIP sample was analyzed by StepOnePlus Real-Time PCR System (Product # 4376600) with primers for the promoter of active GLS1 gene, used as positive control target, and the cFOS, used as negative control target. Data is presented as fold enrichment of the antibody signal versus the negative control IgG using the comparative CT method.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
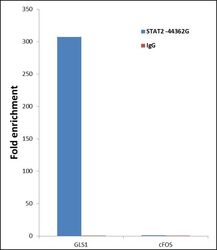
- Experimental details
- ChIP- qPCR analysis of STAT2 was performed with 10 µL of the STAT2 Rabbit polyclonal antibody (Product # 44-362G) on sheared chromatin from 2 million HeLa cells treated with 100 ng/mL of IFN Alpha for one hour using the MAGnify Chromatin Immunoprecipitation System (Product # 49-2024). Normal Rabbit IgG was used as a negative IP control. The purified DNA from each ChIP sample was analyzed by StepOnePlus Real-Time PCR System (Product # 4376600) with primers for the promoter of active GLS1 gene, used as positive control target, and the cFOS, used as negative control target. Data is presented as fold enrichment of the antibody signal versus the negative control IgG using the comparative CT method.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
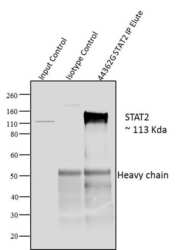
- Experimental details
- Immunoprecipitation of STAT2 was performed with 10 µl of the STAT2 Rabbit Polyclonal Antibody (Product # 44-362G) on cell extract from A431 cells using the Dynabeads? Protein A Immunoprecipitation Kit (10006D). Normal Rabbit IgG was used as a negative IP control. Subsequently, western blot analysis was performed using Novex?NuPAGE? 12 % Bis-Tris gel (Product # NP0341BOX), XCell SureLock Electrophoresis System (Product # EI0002), Novex? Sharp Pre-Stained Protein Standard (LC5800). Proteins were transferred using Pierce G2 Fast Blotter (Product # 62287) to a nitrocellulose and blocked with 5% skim milk for 1 hour at room temperature on a rocking platform. STAT2 was detected at 113 kDa usingSTAT2 Rabbit Polyclonal Antibody (Product # 44-362G) at 1:1000 dilution in 5% skim milk at 4°C overnight on a rocking platform. Goat Anti-Rabbit IgG - HRP Secondary Antibody (G21234) at 1:5000 dilution was used and chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106). Lane 1 represents 10% of the total cell extract (input), Lane 2 is the IP performed with Rabbit IgG and Lane 3 represents IP performed withSTAT2 Rabbit Polyclonal Antibody (Product # 44-362G).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
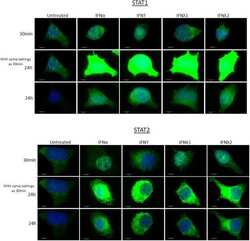
- Experimental details
- EndoC-betaH1 treated with either 1000 U/mL IFNalpha, 20 ng/mL IFNgamma or 200 ng/mL IFNy1 or IFNy2 for 30 min. Cells were fixed with 4% paraformaldehyde, permeabilised and stained with anti-STAT1 or anti-STAT2 antisera, as shown. Images were taken using a Leica DM4000 B LED Fluorescence microscope using exposure settings defined for the control conditions (upper and middle panels of each figure). In the lower panels of each figure, the exposure settings were adjusted to provide improved resolution of the subcellular localisation of each STAT isoform after incubation with interferons.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry