Antibody data
- Antibody Data
- Antigen structure
- References [13]
- Comments [0]
- Validations
- Western blot [3]
- Immunohistochemistry [8]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 13-1300 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- NEFH Monoclonal Antibody (RMdO-20)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- This antibody reacts with 200 kDa subunit protein of human neurofilament. It specifically recognizes a nonphosphorylated epitope in the tail domain of NF-H. Dephosphorylation of NF-H will increase immunoreactivity with this antibody. This antibody is suitable for immunohistochemical staining of Bouin's and alcohol-fixed paraffin-embedded or frozen tissue sections. To stain, incubate 30-60 minutes at room temperature or overnight at 4°C.
- Reactivity
- Human, Mouse, Rat, Bovine, Hamster, Rabbit
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- RMdO-20
- Vial size
- 200 µg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references Platelet-rich plasma promotes peripheral nerve regeneration after sciatic nerve injury.
Unique Neural Circuit Connectivity of Mouse Proximal, Middle, and Distal Colon Defines Regional Colonic Motor Patterns.
Application of immune enhanced organoids in modeling personalized Merkel cell carcinoma research.
Gene therapy via canalostomy approach preserves auditory and vestibular functions in a mouse model of Jervell and Lange-Nielsen syndrome type 2.
3D Culture Method for Alzheimer's Disease Modeling Reveals Interleukin-4 Rescues Aβ42-Induced Loss of Human Neural Stem Cell Plasticity.
Sustained release of neurotrophin-3 via calcium phosphate-coated sutures promotes axonal regeneration after spinal cord injury.
Cerebral and spinal cord tanycytic ependymomas in a young adult with a mutation in the NF2 gene.
Evolution of neuronal and endothelial transcriptomes in primates.
Glutamate receptor expression and chronic glutamate toxicity in rat motor cortex.
Double immunolabeling of central nervous system atypical teratoid/rhabdoid tumors.
Conversion of myoblasts to physiologically active neuronal phenotype.
Mouse embryos cloned from brain tumors.
Role of the sensory neuron cytoskeleton in second messenger signaling for inflammatory pain.
Wang SL, Liu XL, Kang ZC, Wang YS
Neural regeneration research 2023 Feb;18(2):375-381
Neural regeneration research 2023 Feb;18(2):375-381
Unique Neural Circuit Connectivity of Mouse Proximal, Middle, and Distal Colon Defines Regional Colonic Motor Patterns.
Nestor-Kalinoski A, Smith-Edwards KM, Meerschaert K, Margiotta JF, Rajwa B, Davis BM, Howard MJ
Cellular and molecular gastroenterology and hepatology 2022;13(1):309-337.e3
Cellular and molecular gastroenterology and hepatology 2022;13(1):309-337.e3
Application of immune enhanced organoids in modeling personalized Merkel cell carcinoma research.
Forsythe SD, Erali RA, Laney P, Sivakumar H, Li W, Skardal A, Soker S, Votanopoulos KI
Scientific reports 2022 Aug 16;12(1):13865
Scientific reports 2022 Aug 16;12(1):13865
Gene therapy via canalostomy approach preserves auditory and vestibular functions in a mouse model of Jervell and Lange-Nielsen syndrome type 2.
Wu X, Zhang L, Li Y, Zhang W, Wang J, Cai C, Lin X
Nature communications 2021 Jan 29;12(1):697
Nature communications 2021 Jan 29;12(1):697
3D Culture Method for Alzheimer's Disease Modeling Reveals Interleukin-4 Rescues Aβ42-Induced Loss of Human Neural Stem Cell Plasticity.
Papadimitriou C, Celikkaya H, Cosacak MI, Mashkaryan V, Bray L, Bhattarai P, Brandt K, Hollak H, Chen X, He S, Antos CL, Lin W, Thomas AK, Dahl A, Kurth T, Friedrichs J, Zhang Y, Freudenberg U, Werner C, Kizil C
Developmental cell 2018 Jul 2;46(1):85-101.e8
Developmental cell 2018 Jul 2;46(1):85-101.e8
Sustained release of neurotrophin-3 via calcium phosphate-coated sutures promotes axonal regeneration after spinal cord injury.
Hanna A, Thompson DL, Hellenbrand DJ, Lee JS, Madura CJ, Wesley MG, Dillon NJ, Sharma T, Enright CJ, Murphy WL
Journal of neuroscience research 2016 Jul;94(7):645-52
Journal of neuroscience research 2016 Jul;94(7):645-52
Cerebral and spinal cord tanycytic ependymomas in a young adult with a mutation in the NF2 gene.
Kuga Y, Ohnishi H, Kodama Y, Takakura S, Hayashi M, Yagi R, Fukutome K, Matsushima K, Okamoto K, Taomoto K, Takahashi H
Neuropathology : official journal of the Japanese Society of Neuropathology 2014 Aug;34(4):406-13
Neuropathology : official journal of the Japanese Society of Neuropathology 2014 Aug;34(4):406-13
Evolution of neuronal and endothelial transcriptomes in primates.
Giger T, Khaitovich P, Somel M, Lorenc A, Lizano E, Harris LW, Ryan MM, Lan M, Wayland MT, Bahn S, Pääbo S
Genome biology and evolution 2010 Jul 12;2:284-92
Genome biology and evolution 2010 Jul 12;2:284-92
Glutamate receptor expression and chronic glutamate toxicity in rat motor cortex.
Young KC, McGehee DS, Brorson JR
Neurobiology of disease 2007 Apr;26(1):78-85
Neurobiology of disease 2007 Apr;26(1):78-85
Double immunolabeling of central nervous system atypical teratoid/rhabdoid tumors.
Bouffard JP, Sandberg GD, Golden JA, Rorke LB
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2004 Jun;17(6):679-83
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2004 Jun;17(6):679-83
Conversion of myoblasts to physiologically active neuronal phenotype.
Watanabe Y, Kameoka S, Gopalakrishnan V, Aldape KD, Pan ZZ, Lang FF, Majumder S
Genes & development 2004 Apr 15;18(8):889-900
Genes & development 2004 Apr 15;18(8):889-900
Mouse embryos cloned from brain tumors.
Li L, Connelly MC, Wetmore C, Curran T, Morgan JI
Cancer research 2003 Jun 1;63(11):2733-6
Cancer research 2003 Jun 1;63(11):2733-6
Role of the sensory neuron cytoskeleton in second messenger signaling for inflammatory pain.
Dina OA, McCarter GC, de Coupade C, Levine JD
Neuron 2003 Aug 14;39(4):613-24
Neuron 2003 Aug 14;39(4):613-24
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of neurofilament heavy chain was performed by loading 25 µg of mouse brain tissue lysate per well onto a polyacrylamide gel. Proteins were transferred to a PVDF membrane and blocked. NF-H was detected at ~180-220 kD using a neurofilament heavy chain antibody (Product # 13-1300) at a dilution of 2 µg/mL in blocking buffer overnight at 4C, followed by a HRP-labeled secondary antibody for 1 hour at room temperature and detection with a chemiluminescent substrate.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
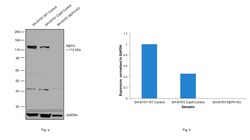
- Experimental details
- Knockout of NEFH was achieved by CRISPR-Cas9 genome editing using LentiArray™ Lentiviral sgRNA (Product # A32042, AssayID CRISPR819005_LV) and LentiArray Cas9 Lentivirus (Product # A32064). Western blot analysis of NEFH was performed by loading 20 µg of SH-SY5Y wild type (Lane 1), SH-SY5Y CAS9 (Lane 2), SH-SY5Y NEFH KO (Lane 3) whole cell extracts. The blot was probed with Anti-NEFH Monoclonal Antibody (RMdO-20)(Product # 13-1300) using 0.5 µg/mL dilution and Goat anti-Mouse IgG (H+L), Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177). Loss of signal upon CRISPR mediated knockout (KO) confirms that the antibody is specific to NEFH. An uncharacterized band was observed at ~42 kDa.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-NEFH Monoclonal Antibody (RMdO-20) (Product # 13-1300) and a 150 kDa band corresponding to NEFH was observed across SH-SY5Y and IMR-32. Whole cell extracts (30 µg lysate) of SH-SY5Y (Lane 1), IMR-32 (Lane 2), LNCaP (Lane 3) and MCF7 (Lane 4) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0321BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1 µg/mL) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of the neurofilament heavy chain in paraffin-embedded mouse brain tissue (right) compared to a negative control without primary antibody (left). Tissue sections were deparaffinized with xylene, and rehydrated with ethanol. To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were washed with water and PBS, and then blocked in 0.3% BSA for 30 min at room temperature. Tissues were then probed with a neurofilament heavy chain monoclonal antibody (Product # 13-1300) in 0.3% BSA at a dilution of 1:20 for 1 hour at 37°C. Tissues were then incubated with a Goat anti-Mouse IgG (H+L) Secondary Antibody, DyLight 488 conjugate for 1 hour at 37°C (green). Nuclei (blue) were stained with DAPI. Images were taken at 40X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of the neurofilament heavy chain showing staining in the filaments of paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Neurofilament H chain monoclonal antibody (Product # 13-1300) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of the neurofilament heavy chain showing staining in the filaments of paraffin-embedded rat brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Neurofilament H chain monoclonal antibody (Product # 13-1300) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of the neurofilament heavy chain in paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). Tissue sections were deparaffinized with xylene, and rehydrated with ethanol. To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were washed with water and PBS, and then blocked in 0.3% BSA for 30 min at room temperature. Tissues were then probed with a neurofilament heavy chain monoclonal antibody (Product # 13-1300) in 0.3% BSA at a dilution of 1:20 for 1 hour at 37°C. Tissues were then incubated with a Goat anti-Mouse IgG (H+L) Secondary Antibody, DyLight 488 conjugate for 1 hour at 37°C (green). Nuclei (blue) were stained with DAPI. Images were taken at 40X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of the neurofilament heavy chain in paraffin-embedded rat brain tissue (right) compared to a negative control without primary antibody (left). Tissue sections were deparaffinized with xylene, and rehydrated with ethanol. To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were washed with water and PBS, and then blocked in 0.3% BSA for 30 min at room temperature. Tissues were then probed with a neurofilament heavy chain monoclonal antibody (Product # 13-1300) in 0.3% BSA at a dilution of 1:20 for 1 hour at 37°C. Tissues were then incubated with a Goat anti-Mouse IgG (H+L) Secondary Antibody, DyLight 488 conjugate for 1 hour at 37°C (green). Nuclei (blue) were stained with DAPI. Images were taken at 40X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of the neurofilament heavy chain in paraffin-embedded mouse brain tissue (right) compared to a negative control without primary antibody (left). Tissue sections were deparaffinized with xylene, and rehydrated with ethanol. To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were washed with water and PBS, and then blocked in 0.3% BSA for 30 min at room temperature. Tissues were then probed with a neurofilament heavy chain monoclonal antibody (Product # 13-1300) in 0.3% BSA at a dilution of 1:20 for 1 hour at 37°C. Tissues were then incubated with a Goat anti-Mouse IgG (H+L) Secondary Antibody, DyLight 488 conjugate for 1 hour at 37°C (green). Nuclei (blue) were stained with DAPI. Images were taken at 40X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of the neurofilament heavy chain showing staining in the filaments of paraffin-embedded mouse brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Neurofilament H chain monoclonal antibody (Product # 13-1300) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of the neurofilament heavy chain in frozen sections of human cortex. Tissues were probed with a neurofilament heavy chain monoclonal antibody (Product # 13-1300, red) at a dilution of 1:20, then incubated with a Alexa Fluor 594 Goat anti-Mouse IgG (H+L) Secondary Antibody. Nuclei (blue) were stained with DAPI. Data courtesy of Min Sun Kim at Seoul National University, Korea.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Effects of PRP on morphological recovery of regenerated nerves in rabbits with sciatic nerve injury . (A) Images of myelinated nerve fibers using toluidine blue staining. The percentage of myelinated nerve fibers in the medium- and high-concentration PRP groups was higher than that in the model and low-concentration PRP groups. Black arrows indicate myelinated nerve fibers. Scale bars: 20 mum. (B) Immunofluorescent staining of the NEFH (NF; green, stained by fluorescein isothiocyanate, white arrows). The presence of neurofilaments in the medium- and high-concentration PRP groups was significantly higher than that in the model and low-concentration PRP groups. Scale bars: 20 mum. (C) Myelin sheaths of regenerated nerves observed by transmission electron microscopy. The white arrows indicate myelinated nerve fibers. Scale bars: 2 mum. (D) Lamellar structure of myelin sheaths. The thickness of the myelin sheath in the medium- and high-concentration PRP groups was greater than that in the model and low-concentration PRP groups. Scale bars: 100 nm. (E-G) Quantitative results of the percentage of myelinated nerve fibers (E), integral optical density of the neurofilament (F), and thickness of the myelin sheath (G). Data in E and F are presented as means +- SD ( n = 7) and were analyzed by Tukey's multiple comparison test. Data in G are presented as median and 95% confidence interval ( n = 7) and were analyzed by Tukey's multiple comparison test. ** P < 0.01, *** P < 0.001, **** P
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA