Antibody data
- Antibody Data
- Antigen structure
- References [8]
- Comments [0]
- Validations
- Western blot [3]
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 35-6000 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Tyrosinase Monoclonal Antibody (T311)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Reactivity
- Human, Mouse
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- T311
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references Mechanism of action of 4-substituted phenols to induce vitiligo and antimelanoma immunity.
Absence of recognition of common melanocytic antigens by T cells isolated from the cerebrospinal fluid of a Vogt-Koyanagi-Harada patient.
Cultivation-dependent plasticity of melanoma phenotype.
Flow cytometry assessment of residual melanoma cells in tumor-infiltrating lymphocyte cultures.
T-cell immune function in tumor, skin, and peripheral blood of advanced stage melanoma patients: implications for immunotherapy.
Mitochondrial proteomics of the retinal pigment epithelium at progressive stages of age-related macular degeneration.
Immunophenotyping of melanomas for tyrosinase: implications for vaccine development.
Immunophenotyping of melanomas for tyrosinase: implications for vaccine development.
Kammeyer A, Willemsen KJ, Ouwerkerk W, Bakker WJ, Ratsma D, Pronk SD, Smit NPM, Luiten RM
Pigment cell & melanoma research 2019 Jul;32(4):540-552
Pigment cell & melanoma research 2019 Jul;32(4):540-552
Absence of recognition of common melanocytic antigens by T cells isolated from the cerebrospinal fluid of a Vogt-Koyanagi-Harada patient.
Abad S, Wieërs G, Colau D, Wildmann C, Delair E, Dhote R, Brézin AP, Kawakami Y, Coulie PG, van der Bruggen P
Molecular vision 2014;20:956-69
Molecular vision 2014;20:956-69
Cultivation-dependent plasticity of melanoma phenotype.
Kodet O, Dvořánková B, Krejčí E, Szabo P, Dvořák P, Štork J, Krajsová I, Dundr P, Smetana K Jr, Lacina L
Tumour biology : the journal of the International Society for Oncodevelopmental Biology and Medicine 2013 Dec;34(6):3345-55
Tumour biology : the journal of the International Society for Oncodevelopmental Biology and Medicine 2013 Dec;34(6):3345-55
Flow cytometry assessment of residual melanoma cells in tumor-infiltrating lymphocyte cultures.
Richards JO, Treisman J, Garlie N, Hanson JP, Oaks MK
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2012 May;81(5):374-81
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2012 May;81(5):374-81
T-cell immune function in tumor, skin, and peripheral blood of advanced stage melanoma patients: implications for immunotherapy.
Tjin EP, Konijnenberg D, Krebbers G, Mallo H, Drijfhout JW, Franken KL, van der Horst CM, Bos JD, Nieweg OE, Kroon BB, Haanen JB, Melief CJ, Vyth-Dreese FA, Luiten RM
Clinical cancer research : an official journal of the American Association for Cancer Research 2011 Sep 1;17(17):5736-47
Clinical cancer research : an official journal of the American Association for Cancer Research 2011 Sep 1;17(17):5736-47
Mitochondrial proteomics of the retinal pigment epithelium at progressive stages of age-related macular degeneration.
Nordgaard CL, Karunadharma PP, Feng X, Olsen TW, Ferrington DA
Investigative ophthalmology & visual science 2008 Jul;49(7):2848-55
Investigative ophthalmology & visual science 2008 Jul;49(7):2848-55
Immunophenotyping of melanomas for tyrosinase: implications for vaccine development.
Chen YT, Stockert E, Tsang S, Coplan KA, Old LJ
Proceedings of the National Academy of Sciences of the United States of America 1995 Aug 29;92(18):8125-9
Proceedings of the National Academy of Sciences of the United States of America 1995 Aug 29;92(18):8125-9
Immunophenotyping of melanomas for tyrosinase: implications for vaccine development.
Chen YT, Stockert E, Tsang S, Coplan KA, Old LJ
Proceedings of the National Academy of Sciences of the United States of America 1995 Aug 29;92(18):8125-9
Proceedings of the National Academy of Sciences of the United States of America 1995 Aug 29;92(18):8125-9
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on whole cell extracts (30 µg lysate) of A-375 (Lane 1), A-431 (Lane 2), B16-F10 (Lane 3), Caki-1 (Lane 4), Hep G2 (Lane 5) and COLO 205 (Lane 6). The blot was probed with Anti-Tyrosinase Mouse Monoclonal Antibody (Product # 35-6000, 2 µg/mL) and detected by chemiluminescence using Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, HRP conjµgate (Product # A28177, 0.4 µg/mL, 1:2500 dilution). A ~80 kDa band corresponding to Tyrosinase was observed across the cell lines tested. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane with iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was probed with the relevant primary and secondary Antibody using iBind™ Flex Western Starter Kit (Product # SLF2000S). Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
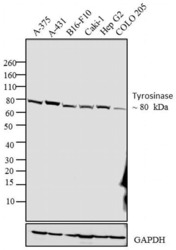
- Experimental details
- Western blot analysis was performed on whole cell extracts (30 µg lysate) of A-375 (Lane 1), A-431 (Lane 2), B16-F10 (Lane 3), Caki-1 (Lane 4), Hep G2 (Lane 5) and COLO 205 (Lane 6). The blot was probed with Anti-Tyrosinase Mouse Monoclonal Antibody (Product # 35-6000, 2 µg/mL) and detected by chemiluminescence using Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, HRP conjµgate (Product # A28177, 0.4 µg/mL, 1:2500 dilution). A ~80 kDa band corresponding to Tyrosinase was observed across the cell lines tested. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane with iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was probed with the relevant primary and secondary Antibody using iBind™ Flex Western Starter Kit (Product # SLF2000S). Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-Tyrosinase Monoclonal Antibody (T311) (Product # 35-6000) and an 80 kDa band corresponding to Tyrosinase was observed across all the cell lines and tissues tested. Whole cell extracts (30 µg lysate) of A-375 (Lane 1), A-431 (Lane 2), SK-MEL-31 (Lane 3), SK-MEL-5 (Lane 4), B16-F10 (Lane 5), HaCaT (Lane 6), Mouse Retina (Lane 7), Mouse Lung (Lane 8) or Mouse Spleen (Lane 9) were electrophoresed using NuPAGE™ 10% Bis-Tris Protein Gel (Product # NP0301BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:250) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177,1:4000) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
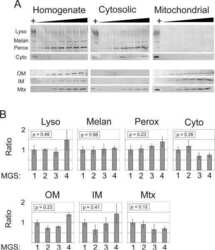
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 BP, PP, PhP, and TBP reduce tyrosinase protein levels, while the other phenols do not (a) Immunoblot showing tyrosinase protein levels (upper panel) in Mel 88.23 cells exposed for 72 hr to the indicated phenols (IC25 concentration) compared to untreated Mel 88.23 cells. Human keratinocytes (HaCaT cells) serve as negative control for tyrosinase expression. The two black bars indicate different tyrosinase forms (a mature 80-kDa and an immature 70-kDa form; Halaban, Cheng, Svedine, Aron, & Hebert, 2001 ). Actin immunostaining was performed as a loading control (lower panel). (b) Similar analyses as (a) for the effect of HOP, PP, PhP and TBP
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunohistochemistry
Immunohistochemistry