16-0629-85
antibody from Invitrogen Antibodies
Targeting: SELL
CD62L, hLHRc, LAM-1, LAM1, Leu-8, LNHR, LSEL, Lyam-1, LYAM1, PLNHR
Antibody data
- Antibody Data
- Antigen structure
- References [17]
- Comments [0]
- Validations
- Flow cytometry [2]
- Other assay [7]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 16-0629-85 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD62L (L-Selectin) Monoclonal Antibody (DREG-56 (DREG56)), Functional Grade, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The DREG-56 monoclonal antibody reacts with human CD62L, a 76 kDa member of the selectin family. CD62L is expressed by neutrophils, monocytes, and subsets of T, B, and NK cells and binds a number of glycosylated, fucosylated, sulfated sialylated glycoproteins including CD34, glycam-1 and MAdCAM-1. These interactions mediate rolling of lymphocytes on activated endothelium at the sites of inflammation and homing of cells to the high endothelial venules (HEV) of peripheral lymphoid tissues. Applications Reported: The DREG-56 (DREG56) antibody has been reported for use in flow cytometric analysis. DREG-56 has also been reported in inhibition of binding to HEV. Applications Tested: The DREG-56 (DREG56) antibody has been tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at less than or equal to 1 µg per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest. Storage and handling: Use in a sterile environment. Filtration: 0.2 µm post-manufacturing filtered. Purity: Greater than 90%, as determined by SDS-PAGE. Endotoxin Level: Less than 0.001 ng/µg antibody, as determined by LAL assay. Aggregation: Less than 10%, as determined by HPLC.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- DREG-56 (DREG56)
- Vial size
- 500 μg
- Concentration
- 1 mg/mL
- Storage
- 4°C
Submitted references Induction of memory-like CD8+ T cells and CD4+ T cells from human naive T cells in culture.
Heterogeneity of human bone marrow and blood natural killer cells defined by single-cell transcriptome.
n-butanol extract from Folium isatidis inhibits the lipopolysaccharide-induced downregulation of CXCR1 and CXCR2 on human neutrophils.
Characteristic patterns of HLA presentation and T cell differentiation in adult-onset Still's disease.
Accelerated resolution of inflammation underlies sex differences in inflammatory responses in humans.
Blocking MHC class II on human endothelium mitigates acute rejection.
Peptide-induced immune regulation by a promiscuous and immunodominant CD4T-cell epitope of Timothy grass pollen: a role of Cbl-b and Itch in regulation.
Novel serial positive enrichment technology enables clinical multiparameter cell sorting.
Resolvin D1 limits polymorphonuclear leukocyte recruitment to inflammatory loci: receptor-dependent actions.
Image correlation microscopy for uniform illumination.
Comparison of gene expression profiles between human and mouse monocyte subsets.
Membrane mobility of beta2 integrins and rolling associated adhesion molecules in resting neutrophils.
Monitoring receptor-ligand interactions between surfaces by thermal fluctuations.
PILAR is a novel modulator of human T-cell expansion.
Wiskott-Aldrich syndrome protein is required for regulatory T cell homeostasis.
L-selectin serves as an E-selectin ligand on cultured human T lymphoblasts.
Identification of a human peripheral lymph node homing receptor: a rapidly down-regulated adhesion molecule.
Tokumoto Y, Araki Y, Narizuka Y, Mizuno Y, Ohshima S, Mimura T
Clinical and experimental immunology 2022 Jan 28;207(1):95-103
Clinical and experimental immunology 2022 Jan 28;207(1):95-103
Heterogeneity of human bone marrow and blood natural killer cells defined by single-cell transcriptome.
Yang C, Siebert JR, Burns R, Gerbec ZJ, Bonacci B, Rymaszewski A, Rau M, Riese MJ, Rao S, Carlson KS, Routes JM, Verbsky JW, Thakar MS, Malarkannan S
Nature communications 2019 Sep 2;10(1):3931
Nature communications 2019 Sep 2;10(1):3931
n-butanol extract from Folium isatidis inhibits the lipopolysaccharide-induced downregulation of CXCR1 and CXCR2 on human neutrophils.
Wu B, Wang L, Jiang L, Dong L, Xu F, Lu Y, Jin J, Wang Z, Liang G, Shan X
Molecular medicine reports 2018 Jan;17(1):179-185
Molecular medicine reports 2018 Jan;17(1):179-185
Characteristic patterns of HLA presentation and T cell differentiation in adult-onset Still's disease.
Jung JY, Choi B, Sayeed HM, Suh CH, Kim YW, Kim HA, Sohn S
International journal of immunopathology and pharmacology 2018 Jan-Dec;32:2058738418791284
International journal of immunopathology and pharmacology 2018 Jan-Dec;32:2058738418791284
Accelerated resolution of inflammation underlies sex differences in inflammatory responses in humans.
Rathod KS, Kapil V, Velmurugan S, Khambata RS, Siddique U, Khan S, Van Eijl S, Gee LC, Bansal J, Pitrola K, Shaw C, D'Acquisto F, Colas RA, Marelli-Berg F, Dalli J, Ahluwalia A
The Journal of clinical investigation 2017 Jan 3;127(1):169-182
The Journal of clinical investigation 2017 Jan 3;127(1):169-182
Blocking MHC class II on human endothelium mitigates acute rejection.
Abrahimi P, Qin L, Chang WG, Bothwell AL, Tellides G, Saltzman WM, Pober JS
JCI insight 2016;1(1)
JCI insight 2016;1(1)
Peptide-induced immune regulation by a promiscuous and immunodominant CD4T-cell epitope of Timothy grass pollen: a role of Cbl-b and Itch in regulation.
Till SJ, Raynsford EJ, Reynolds CJ, Quigley KJ, Grzybowska-Kowalczyk A, Saggar LR, Goldstone A, Maillere B, Kwok WW, Altmann DM, Durham SR, Boyton RJ
Thorax 2014 Apr;69(4):335-45
Thorax 2014 Apr;69(4):335-45
Novel serial positive enrichment technology enables clinical multiparameter cell sorting.
Stemberger C, Dreher S, Tschulik C, Piossek C, Bet J, Yamamoto TN, Schiemann M, Neuenhahn M, Martin K, Schlapschy M, Skerra A, Schmidt T, Edinger M, Riddell SR, Germeroth L, Busch DH
PloS one 2012;7(4):e35798
PloS one 2012;7(4):e35798
Resolvin D1 limits polymorphonuclear leukocyte recruitment to inflammatory loci: receptor-dependent actions.
Norling LV, Dalli J, Flower RJ, Serhan CN, Perretti M
Arteriosclerosis, thrombosis, and vascular biology 2012 Aug;32(8):1970-8
Arteriosclerosis, thrombosis, and vascular biology 2012 Aug;32(8):1970-8
Image correlation microscopy for uniform illumination.
Gaborski TR, Sealander MN, Ehrenberg M, Waugh RE, McGrath JL
Journal of microscopy 2010 Jan;237(1):39-50
Journal of microscopy 2010 Jan;237(1):39-50
Comparison of gene expression profiles between human and mouse monocyte subsets.
Ingersoll MA, Spanbroek R, Lottaz C, Gautier EL, Frankenberger M, Hoffmann R, Lang R, Haniffa M, Collin M, Tacke F, Habenicht AJ, Ziegler-Heitbrock L, Randolph GJ
Blood 2010 Jan 21;115(3):e10-9
Blood 2010 Jan 21;115(3):e10-9
Membrane mobility of beta2 integrins and rolling associated adhesion molecules in resting neutrophils.
Gaborski TR, Clark A Jr, Waugh RE, McGrath JL
Biophysical journal 2008 Nov 15;95(10):4934-47
Biophysical journal 2008 Nov 15;95(10):4934-47
Monitoring receptor-ligand interactions between surfaces by thermal fluctuations.
Chen W, Evans EA, McEver RP, Zhu C
Biophysical journal 2008 Jan 15;94(2):694-701
Biophysical journal 2008 Jan 15;94(2):694-701
PILAR is a novel modulator of human T-cell expansion.
Huarte E, Cubillos-Ruiz JR, Nesbeth YC, Scarlett UK, Martinez DG, Engle XA, Rigby WF, Pioli PA, Guyre PM, Conejo-Garcia JR
Blood 2008 Aug 15;112(4):1259-68
Blood 2008 Aug 15;112(4):1259-68
Wiskott-Aldrich syndrome protein is required for regulatory T cell homeostasis.
Humblet-Baron S, Sather B, Anover S, Becker-Herman S, Kasprowicz DJ, Khim S, Nguyen T, Hudkins-Loya K, Alpers CE, Ziegler SF, Ochs H, Torgerson T, Campbell DJ, Rawlings DJ
The Journal of clinical investigation 2007 Feb;117(2):407-18
The Journal of clinical investigation 2007 Feb;117(2):407-18
L-selectin serves as an E-selectin ligand on cultured human T lymphoblasts.
Jutila MA, Kurk S, Jackiw L, Knibbs RN, Stoolman LM
Journal of immunology (Baltimore, Md. : 1950) 2002 Aug 15;169(4):1768-73
Journal of immunology (Baltimore, Md. : 1950) 2002 Aug 15;169(4):1768-73
Identification of a human peripheral lymph node homing receptor: a rapidly down-regulated adhesion molecule.
Kishimoto TK, Jutila MA, Butcher EC
Proceedings of the National Academy of Sciences of the United States of America 1990 Mar;87(6):2244-8
Proceedings of the National Academy of Sciences of the United States of America 1990 Mar;87(6):2244-8
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
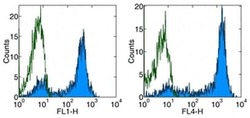
- Experimental details
- Staining of normal human peripheral blood cells with Anti-Human CD62L (L-Selectin) FITC (left) or APC (right).Appropriate isotype controls were used (open histogram).Cells in the lymphocyte population were used for analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
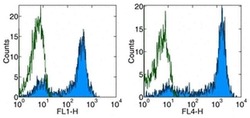
- Experimental details
- Staining of normal human peripheral blood cells with Anti-Human CD62L (L-Selectin) FITC (left) or APC (right).Appropriate isotype controls were used (open histogram).Cells in the lymphocyte population were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
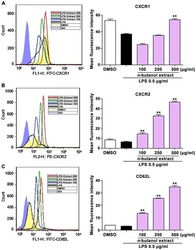
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
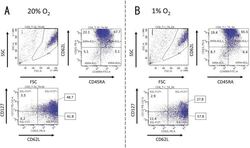
- Fig. 1. Activated CD8 + T cells in either normal or hypoxic culture. Naive CD8 + T cells derived from a healthy donor were cultured in human T-activator CD3/CD28 and IL-2 containing medium for 8 days. ( A ) in 20% O 2 condition and ( B ) in 1% O 2 condition. The 7-AAD-negative cells in the area gated as P1 in the FSC/SSC panel were considered as living cells. The expression pattern of CD45RA, CD62L, and CD127 of living cells were analyzed by FACS. The numbers on the FACS-plot panel mean the frequencies (%) of population of cells. We repeated this experiment four times.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
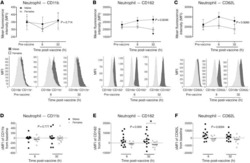
- Figure 5 Reduced inflammatory cell activation state in cantharidin-induced blister exudates in female compared with male healthy volunteers. Mean fluorescence intensity (MFI) of the expression molecules CD162, CD62L, and CD11b on ( A ) neutrophils, ( B ) inflammatory monocytes, and ( C ) CD4 + and CD8 + T cells in healthy male ( n = 16) and female ( n = 16) volunteers. Data are shown as mean +- SEM. Statistical significances determined using 2-way ANOVA, * P < 0.05, ** P < 0.01, *** P < 0.001, and **** P < 0.0001; followed by Sidak's post tests, # P < 0.05, ## P < 0.01, and #### P < 0.0001 comparing the sexes at each time point for all the panels.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
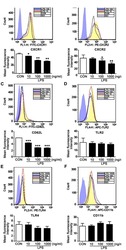
- Figure 2. Percentage of surface-stained cells presenting CD4+, CD8+, CCR7+, CD4+CCR7+, CD8+CCR7+, CD4+CD62L-, CD8+CD62L-, CD4+naive T cell, CD4+ effector memory T cell, CD4+ central memory T cells, CD8+ naive T cells, CD8+ effector memory T cells, and CD8+ central memory T cells in patients with AOSD, a patient with rheumatoid arthritis (RA), and a healthy control (HC). Results were obtained from 14 patients with AOSD, 20 RA patients, and 20 HCs. The horizontal line indicates the mean value for each group. The P value was determined by the Mann-Whitney U test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
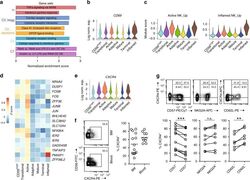
- Fig. 4 Active NK cells with a unique transcriptome profile. a Top two enriched gene sets (ranked by normalized enrichment score) of five different datasets from GSEA of the ""Inflamed NK"" cluster compared to the rest of the cells were plotted. b The expression of CD69 in the BM sample was shown as a violin plot. The y -axis represents log-normalized expression value. c Module score was calculated using up-regulated DEGs of ""Active NK"" (left) or ""Inflamed NK"" (right) cluster from BM sample and plotted via violin plots. d Up-regulated IEGs from ""Active NK"" cluster were plotted using heatmap of the BM sample. e The expression of CXCR4 in the BM sample was shown as a violin plot. The y -axis represents log-normalized expression value. f Percentage of CXCR4 + NK cells (gated on Lin - CD56 + cells) was evaluated via flow cytometry. g The expression of CXCR4 in CD57 +/- , CD62L +/- , or NKG2A +/- CD56 dim NK populations from BM was assessed via flow cytometry (top). Percentage of CXCR4 + cells within each population were quantified (bottom). n >= 6 from two to five independent experiments. Paired Student's t test was used for the statistical analysis. * P < 0.05; ** p < 0.01; *** p < 0.001; n.s. stands for ""not significant."" Source data for f and g are provided as a Source Data file. See also Supplementary Fig. 5
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry Blocking/Neutralizing
Blocking/Neutralizing