Antibody data
- Antibody Data
- Antigen structure
- References [11]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [13]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 48-0279-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD27 Monoclonal Antibody (O323), eFluor™ 450, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The O323 monoclonal antibody reacts with human CD27, a lymphocyte-specific member of the TNFR superfamily. CD27 is expressed by a subset of thymocytes and virtually all mature T cells and is upregulated upon T-cell stimulation. CD27 binds to CD70, and through this interaction, plays an important role in T cell-B cell interaction. Applications Reported: This O323 antibody has been reported for use in flow cytometric analysis. Applications Tested: This O323 antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0125 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. eFluor® 450 is an alternative to Pacific Blue®. eFluor® 450 emits at 445 nm and is excited with the Violet laser (405 nm). Please make sure that your instrument is capable of detecting this fluorochome. Excitation: 405 nm; Emission: 445 nm; Laser: Violet Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- O323
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Safety and feasibility of anti-CD19 CAR T cells with fully human binding domains in patients with B-cell lymphoma.
Expression of CD27 and CD28 on γδ T cells from the peripheral blood of patients with allergic rhinitis.
More than one antibody of individual B cells revealed by single-cell immune profiling.
Perturbed CD8(+) T cell immunity across universal influenza epitopes in the elderly.
Antigen receptor-redirected T cells derived from hematopoietic precursor cells lack expression of the endogenous TCR/CD3 receptor and exhibit specific antitumor capacities.
Maintenance of the EBV-specific CD8(+) TCRαβ repertoire in immunosuppressed lung transplant recipients.
Abnormal phenotypic features of IgM+B cell subsets in patients with chronic hepatitis C virus infection.
Immunological and clinical consequences of splenectomy in a multiple sclerosis patient treated with natalizumab.
Long-lived Plasmodium falciparum specific memory B cells in naturally exposed Swedish travelers.
Chronic exposure to Plasmodium falciparum is associated with phenotypic evidence of B and T cell exhaustion.
Long-term immunologically competent human peripheral lymphoid tissue cultures in a 3D bioreactor.
Brudno JN, Lam N, Vanasse D, Shen YW, Rose JJ, Rossi J, Xue A, Bot A, Scholler N, Mikkilineni L, Roschewski M, Dean R, Cachau R, Youkharibache P, Patel R, Hansen B, Stroncek DF, Rosenberg SA, Gress RE, Kochenderfer JN
Nature medicine 2020 Feb;26(2):270-280
Nature medicine 2020 Feb;26(2):270-280
Expression of CD27 and CD28 on γδ T cells from the peripheral blood of patients with allergic rhinitis.
Wang Q, Sun Q, Chen Q, Li H, Liu D
Experimental and therapeutic medicine 2020 Dec;20(6):224
Experimental and therapeutic medicine 2020 Dec;20(6):224
More than one antibody of individual B cells revealed by single-cell immune profiling.
Shi Z, Zhang Q, Yan H, Yang Y, Wang P, Zhang Y, Deng Z, Yu M, Zhou W, Wang Q, Yang X, Mo X, Zhang C, Huang J, Dai H, Sun B, Zhao Y, Zhang L, Yang YG, Qiu X
Cell discovery 2019;5:64
Cell discovery 2019;5:64
Perturbed CD8(+) T cell immunity across universal influenza epitopes in the elderly.
Nguyen THO, Sant S, Bird NL, Grant EJ, Clemens EB, Koutsakos M, Valkenburg SA, Gras S, Lappas M, Jaworowski A, Crowe J, Loh L, Kedzierska K
Journal of leukocyte biology 2018 Feb;103(2):321-339
Journal of leukocyte biology 2018 Feb;103(2):321-339
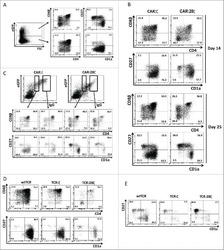
Antigen receptor-redirected T cells derived from hematopoietic precursor cells lack expression of the endogenous TCR/CD3 receptor and exhibit specific antitumor capacities.
Van Caeneghem Y, De Munter S, Tieppo P, Goetgeluk G, Weening K, Verstichel G, Bonte S, Taghon T, Leclercq G, Kerre T, Debets R, Vermijlen D, Abken H, Vandekerckhove B
Oncoimmunology 2017;6(3):e1283460
Oncoimmunology 2017;6(3):e1283460
Maintenance of the EBV-specific CD8(+) TCRαβ repertoire in immunosuppressed lung transplant recipients.
Nguyen TH, Bird NL, Grant EJ, Miles JJ, Thomas PG, Kotsimbos TC, Mifsud NA, Kedzierska K
Immunology and cell biology 2017 Jan;95(1):77-86
Immunology and cell biology 2017 Jan;95(1):77-86
Abnormal phenotypic features of IgM+B cell subsets in patients with chronic hepatitis C virus infection.
Kong F, Feng B, Zhang H, Rao H, Wang J, Cong X, Wei L
Experimental and therapeutic medicine 2017 Aug;14(2):1846-1852
Experimental and therapeutic medicine 2017 Aug;14(2):1846-1852
Immunological and clinical consequences of splenectomy in a multiple sclerosis patient treated with natalizumab.
Lee DH, Waschbisch A, Lämmer AB, Doerfler A, Schwab S, Linker RA
Journal of neuroinflammation 2013 Oct 9;10:123
Journal of neuroinflammation 2013 Oct 9;10:123
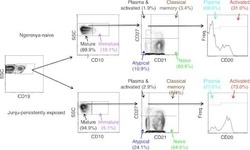
Long-lived Plasmodium falciparum specific memory B cells in naturally exposed Swedish travelers.
Ndungu FM, Lundblom K, Rono J, Illingworth J, Eriksson S, Färnert A
European journal of immunology 2013 Nov;43(11):2919-29
European journal of immunology 2013 Nov;43(11):2919-29
Chronic exposure to Plasmodium falciparum is associated with phenotypic evidence of B and T cell exhaustion.
Illingworth J, Butler NS, Roetynck S, Mwacharo J, Pierce SK, Bejon P, Crompton PD, Marsh K, Ndungu FM
Journal of immunology (Baltimore, Md. : 1950) 2013 Feb 1;190(3):1038-47
Journal of immunology (Baltimore, Md. : 1950) 2013 Feb 1;190(3):1038-47
Long-term immunologically competent human peripheral lymphoid tissue cultures in a 3D bioreactor.
Kuzin I, Sun H, Moshkani S, Feng C, Mantalaris A, Wu JH, Bottaro A
Biotechnology and bioengineering 2011 Jun;108(6):1430-40
Biotechnology and bioengineering 2011 Jun;108(6):1430-40
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
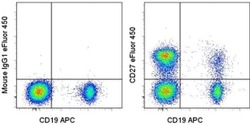
- Experimental details
- Staining of normal human peripheral blood cells with Anti-Human CD19 APC (Product # 17-0199-42) and Mouse IgG1 K Isotype Control eFluor® 450 (Product # 48-4714-82) (left) or Anti-Human CD27 eFluor® 450 (right). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
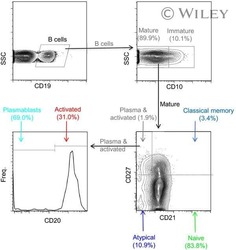
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
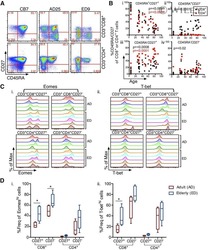
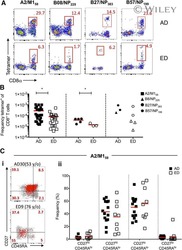
- Figure 4 Early detection of TRAV5-biased GLC-specific CD8 + T cells in patient Tx101. PBMC from patient Tx101 ( a ) at 87 days post transplant underwent tetramer-associated magnetic enrichment (TAME). Pre-enriched, post-enriched and the flowthrough fractions were collected and analyzed based on GLC-tetramer and anti-CD8 antibody staining. Percentages are based on CD3 + T cells. CD3 + T cells, GLC-tetramer + CD8 + and GLC-tetramer + CD8 low/- cells from the post-enriched fraction ( b ) were phenotypically analyzed based on CD45RA and CD27 expression. Pie charts of paired GLC-specific CD8 + TCRalphabeta clonotypes following TAME (d87) ( c ) or following in vitro expansion (d185, d299 and d472) ( d ) are shown. Pie chart area shaded in red represent TRAV5 clonotypes. Percentage of TCRalphabeta clonotypes shared compared to TAME (d87) (bar graph, left y axis) and SDI values (line graph, right y axis) are shown in e . Frequencies of GLC + CD8 + T cells following expansion and clonotype details are listed in f (ND; not determined).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
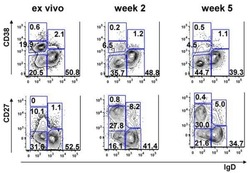
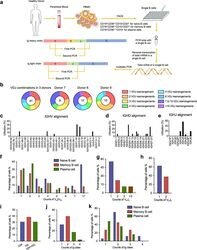
- Fig. 4 Sanger sequencing validation of multiple V(D)J recombination patterns in single B cells. a Sketching diagram of the single-cell sequencing procedure. We isolated peripheral blood from healthy donors and separated peripheral blood mononuclear cells (PBMCs). Naive B cells (CD19 + CD38 +/- CD10 - CD27 - ), memory B cells (CD19 + CD38 +/- CD10 - CD27 + ), and plasma cells (CD19 + CD38 +++ CD10 - ) were sorted by FACS. The total mRNA in single B cells was reverse transcribed, and the Ig heavy chain and light chain were amplified by multiplex PCR. FACS, fluorescence-activated cell sorting. b Proportions of cells expressing VDJ recombination patterns in single B cells from donor 7, donor 8, and donor 9. c - e Distribution and frequency of IGHV ( c ), IGHD ( d ), and IGHJ ( e ) segments in Ig germline genes in a single cell. f Proportions of single naive B cells, plasma cells, and memory B cells expressing one, two, or three V H DJ H segments. g , h Proportions of single B cells expressing one, two, or more V kappa J kappa segments ( g ) and V lamda J lamda segments ( h ). i Counts of single B cells expressing only Igkappa or Iglamda or expressing both Igkappa and Iglamda. j , k Proportions of naive B cells, plasma cells, and memory B cells expressing one, two, three, or four Ig classes
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
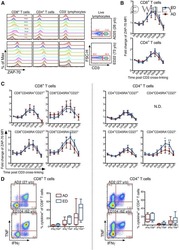
- Figure 3 Expression pattern of CD27 and CD28 on gammadelta T cells in AR. (A) Representative flow cytometry plots of the expression of CD27 and CD28 on gammadelta T cells among PBMCs from HC (n=12) and AR (n=14) subjects. (B-G) Quantification of the percentage of (B) CD27+, (C) CD28+, (D) CD27 + CD28 + , (E) CD27 + CD28 - , (F) CD27 - CD28 + and (G) CD27 - CD28 - gammadelta + T cells in PBMCs. Values are expressed as the mean +- standard deviation. AR, allergic rhinitis; HC, healthy controls; PBMCs, peripheral blood mononuclear cells.
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry