Antibody data
- Antibody Data
- Antigen structure
- References [16]
- Comments [0]
- Validations
- Western blot [1]
- Other assay [10]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-1144G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-VE-cadherin (Tyr658) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- This antibody has been negatively preadsorbed using a non-phosphopeptide corresponding to the site of phosphorylation to remove antibody that is reactive with non-phosphorylated VE-cadherin. The final product is generated by affinity chromatography using an VE-cadherin-derived peptide that is phosphorylated at tyrosine 658.
Submitted references The amyloid peptide β disrupts intercellular junctions and increases endothelial permeability in a NADPH oxidase 1-dependent manner.
Testisin/Prss21 deficiency causes increased vascular permeability and a hemorrhagic phenotype during luteal angiogenesis.
Direct reprogramming of human smooth muscle and vascular endothelial cells reveals defects associated with aging and Hutchinson-Gilford progeria syndrome.
P-Cresylsulfate, the Protein-Bound Uremic Toxin, Increased Endothelial Permeability Partly Mediated by Src-Induced Phosphorylation of VE-Cadherin.
c-Src controls stability of sprouting blood vessels in the developing retina independently of cell-cell adhesion through focal adhesion assembly.
Time-Variant SRC Kinase Activation Determines Endothelial Permeability Response.
Rap1B promotes VEGF-induced endothelial permeability and is required for dynamic regulation of the endothelial barrier.
The matrikine N-α-PGP couples extracellular matrix fragmentation to endothelial permeability.
Endothelial Gαq/11 is required for VEGF-induced vascular permeability and angiogenesis.
Novel role of CD47 in rat microvascular endothelium: signaling and regulation of T-cell transendothelial migration.
Identification of a high-mannose ICAM-1 glycoform: effects of ICAM-1 hypoglycosylation on monocyte adhesion and outside in signaling.
Phosphorylation of VE-cadherin is modulated by haemodynamic forces and contributes to the regulation of vascular permeability in vivo.
Endothelial CD47 promotes vascular endothelial-cadherin tyrosine phosphorylation and participates in T cell recruitment at sites of inflammation in vivo.
p120-Catenin prevents neutrophil transmigration independently of RhoA inhibition by impairing Src dependent VE-cadherin phosphorylation.
ICAM-1-mediated, Src- and Pyk2-dependent vascular endothelial cadherin tyrosine phosphorylation is required for leukocyte transendothelial migration.
ICAM-1-mediated, Src- and Pyk2-dependent vascular endothelial cadherin tyrosine phosphorylation is required for leukocyte transendothelial migration.
Tarafdar A, Wolska N, Krisp C, Schlüter H, Pula G
Redox biology 2022 Jun;52:102287
Redox biology 2022 Jun;52:102287
Testisin/Prss21 deficiency causes increased vascular permeability and a hemorrhagic phenotype during luteal angiogenesis.
Peroutka RJ, Buzza MS, Mukhopadhyay S, Johnson TA, Driesbaugh KH, Antalis TM
PloS one 2020;15(6):e0234407
PloS one 2020;15(6):e0234407
Direct reprogramming of human smooth muscle and vascular endothelial cells reveals defects associated with aging and Hutchinson-Gilford progeria syndrome.
Bersini S, Schulte R, Huang L, Tsai H, Hetzer MW
eLife 2020 Sep 8;9
eLife 2020 Sep 8;9
P-Cresylsulfate, the Protein-Bound Uremic Toxin, Increased Endothelial Permeability Partly Mediated by Src-Induced Phosphorylation of VE-Cadherin.
Chen SC, Huang SY, Wu CC, Hsu CF
Toxins 2020 Jan 21;12(2)
Toxins 2020 Jan 21;12(2)
c-Src controls stability of sprouting blood vessels in the developing retina independently of cell-cell adhesion through focal adhesion assembly.
Schimmel L, Fukuhara D, Richards M, Jin Y, Essebier P, Frampton E, Hedlund M, Dejana E, Claesson-Welsh L, Gordon E
Development (Cambridge, England) 2020 Apr 6;147(7)
Development (Cambridge, England) 2020 Apr 6;147(7)
Time-Variant SRC Kinase Activation Determines Endothelial Permeability Response.
Klomp JE, Shaaya M, Matsche J, Rebiai R, Aaron JS, Collins KB, Huyot V, Gonzalez AM, Muller WA, Chew TL, Malik AB, Karginov AV
Cell chemical biology 2019 Aug 15;26(8):1081-1094.e6
Cell chemical biology 2019 Aug 15;26(8):1081-1094.e6
Rap1B promotes VEGF-induced endothelial permeability and is required for dynamic regulation of the endothelial barrier.
Lakshmikanthan S, Sobczak M, Li Calzi S, Shaw L, Grant MB, Chrzanowska-Wodnicka M
Journal of cell science 2018 Jan 10;131(1)
Journal of cell science 2018 Jan 10;131(1)
The matrikine N-α-PGP couples extracellular matrix fragmentation to endothelial permeability.
Hahn CS, Scott DW, Xu X, Roda MA, Payne GA, Wells JM, Viera L, Winstead CJ, Bratcher P, Sparidans RW, Redegeld FA, Jackson PL, Folkerts G, Blalock JE, Patel RP, Gaggar A
Science advances 2015;1(3)
Science advances 2015;1(3)
Endothelial Gαq/11 is required for VEGF-induced vascular permeability and angiogenesis.
Sivaraj KK, Li R, Albarran-Juarez J, Wang S, Tischner D, Grimm M, Swiercz JM, Offermanns S, Wettschureck N
Cardiovascular research 2015 Oct 1;108(1):171-80
Cardiovascular research 2015 Oct 1;108(1):171-80
Novel role of CD47 in rat microvascular endothelium: signaling and regulation of T-cell transendothelial migration.
Martinelli R, Newton G, Carman CV, Greenwood J, Luscinskas FW
Arteriosclerosis, thrombosis, and vascular biology 2013 Nov;33(11):2566-76
Arteriosclerosis, thrombosis, and vascular biology 2013 Nov;33(11):2566-76
Identification of a high-mannose ICAM-1 glycoform: effects of ICAM-1 hypoglycosylation on monocyte adhesion and outside in signaling.
Scott DW, Dunn TS, Ballestas ME, Litovsky SH, Patel RP
American journal of physiology. Cell physiology 2013 Jul 15;305(2):C228-37
American journal of physiology. Cell physiology 2013 Jul 15;305(2):C228-37
Phosphorylation of VE-cadherin is modulated by haemodynamic forces and contributes to the regulation of vascular permeability in vivo.
Orsenigo F, Giampietro C, Ferrari A, Corada M, Galaup A, Sigismund S, Ristagno G, Maddaluno L, Koh GY, Franco D, Kurtcuoglu V, Poulikakos D, Baluk P, McDonald D, Grazia Lampugnani M, Dejana E
Nature communications 2012;3:1208
Nature communications 2012;3:1208
Endothelial CD47 promotes vascular endothelial-cadherin tyrosine phosphorylation and participates in T cell recruitment at sites of inflammation in vivo.
Azcutia V, Stefanidakis M, Tsuboi N, Mayadas T, Croce KJ, Fukuda D, Aikawa M, Newton G, Luscinskas FW
Journal of immunology (Baltimore, Md. : 1950) 2012 Sep 1;189(5):2553-62
Journal of immunology (Baltimore, Md. : 1950) 2012 Sep 1;189(5):2553-62
p120-Catenin prevents neutrophil transmigration independently of RhoA inhibition by impairing Src dependent VE-cadherin phosphorylation.
Alcaide P, Martinelli R, Newton G, Williams MR, Adam A, Vincent PA, Luscinskas FW
American journal of physiology. Cell physiology 2012 Aug 15;303(4):C385-95
American journal of physiology. Cell physiology 2012 Aug 15;303(4):C385-95
ICAM-1-mediated, Src- and Pyk2-dependent vascular endothelial cadherin tyrosine phosphorylation is required for leukocyte transendothelial migration.
Allingham MJ, van Buul JD, Burridge K
Journal of immunology (Baltimore, Md. : 1950) 2007 Sep 15;179(6):4053-64
Journal of immunology (Baltimore, Md. : 1950) 2007 Sep 15;179(6):4053-64
ICAM-1-mediated, Src- and Pyk2-dependent vascular endothelial cadherin tyrosine phosphorylation is required for leukocyte transendothelial migration.
Allingham MJ, van Buul JD, Burridge K
Journal of immunology (Baltimore, Md. : 1950) 2007 Sep 15;179(6):4053-64
Journal of immunology (Baltimore, Md. : 1950) 2007 Sep 15;179(6):4053-64
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
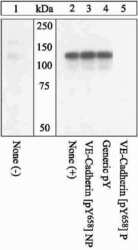
- Experimental details
- Extracts of HUVEC cells either untreated (1) or treated (2-5)with 100 ng/mL VEGF for 10. The membrane was blocked with a 5% BSA-TBST buffer for one hour at room temperature, then incubated with the VE-cadherin (pY658) (Product # 44-1144G).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
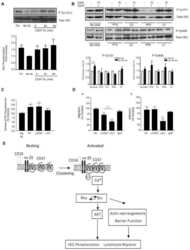
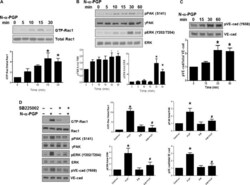
- Fig. 1 N-alpha-PGP activates endothelial cell signaling through CXCR2. ( A to C ) HUVECs were serum-starved for 2 hours before stimulation with N-alpha-PGP (0.5 mg/ml) for indicated times, and activation of Rac1 (A) and phosphorylation of PAK (pPAK) and ERK (pERK) (B) and VE-cadherin (pVE-cad) (C) were determined by Western blot. Shown are representative Western blots together with quantification. Bar graphs show means +- SEM ( n = 3). * P < 0.05 relative to time 0 by one-way analysis of variance (ANOVA) with Tukey post-test. ( D ) HUVECs were untreated or treated with N-alpha-PGP (0.5 mg/ml) (30 min) alone or after pretreatment with 200 nM SB225002, and Rac1 activity and phosphorylation of ERK, PAK, and VE-cadherin were determined by Western blot. Shown are representative Western blots together with quantification. Bar graphs show means +- SEM ( n = 3). * P < 0.05 relative to time 0, # P < 0.05 relative to PGP by one-way ANOVA with Tukey post-test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
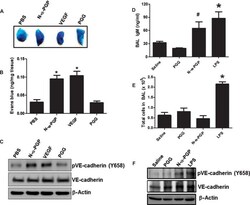
- Fig. 3 N-alpha-PGP induces skin and pulmonary microvascular permeability. ( A to C ) Mice ( n = 4 to 5 per group) were injected via the tail vein with Evans blue and then received abdominal subcutaneous injection of PBS alone, N-alpha-PGP (250 mug), PGG (250 mug), or VEGF (50 ng). Evans blue leak to the skin tissue was visually assessed (A) and quantified (B), and VE-cadherin phosphorylation after treatment was determined by Western blot (representative image) (C). * P < 0.05 versus PBS control by one-way ANOVA with Tukey post-test. ( D to F ) Mice ( n = 4 to 5 per group) were intraperitoneally injected with saline alone or containing N-alpha-PGP (250 mug), PGG (250 mug), LPS (75 mug) once a day for 4 days, and then the total IgM in BAL fluid was measured by immunoassay (D) and total BAL cell (E), and VE-cadherin phosphorylation in lung lysate was measured by Western blot (F). * P < 0.05 versus saline or PGG, # P < 0.05 versus PGG by one-way ANOVA with Tukey's multiple comparison post-test. All values represent means +- SEM.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
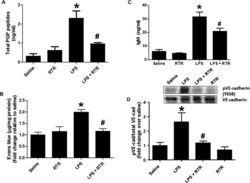
- Fig. 4 RTR attenuates LPS-induced pulmonary microvascular permeability. Mice were injected via the tail vein with 50 mul of PBS alone or containing 50 mug of RTR and then intraperitoneally administered with 75 mug of LPS (in 100 mul of PBS) once a day. ( A ) After 4 days of treatment, mice were sacrificed for serum measurements of PGP ( n = 6) or injected via the tail vein with Evans blue. ( B ) Evans blue leak to the lung was quantified and normalized to protein; data show fold change relative to saline ( n = 7 to 11). ( C ) IgM levels were measured in the BAL ( n = 4 to 6). ( D ) VE-cadherin phosphorylation in lung homogenates was assessed by Western blot. Representative image. * P < 0.05 versus saline control, # P < 0.05 relative to LPS for (A) to (C) by one-way ANOVA with Tukey post-test; # P < 0.05 relative to LPS by t test for (D). All values represent means +- SEM.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
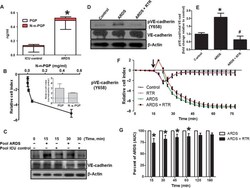
- Fig. 5 ARDS plasma induces endothelial activation, which is attenuated by RTR. ( A ) Plasma was collected from patients with ARDS and normal control, and plasma PGP and N-alpha-PGP levels were measured via electrospray ionization-liquid chromatography-tandem mass spectrometry. * P < 0.05 by t test between ARDS and normal for all PGP peptides ( n = 6). PMVECs were treated with N-alpha-PGP, and permeability was assessed (see fig. S4F). ( B ) Maximal changes in relative cell index as a function of N-alpha-PGP concentrations. Inset compares the effects of N-alpha-PGP and PGP (both at 1 ng/ml). ( C ) After 2 hours of serum starvation, HUVECs were treated with pooled ARDS (from three patients) or non-lung disease intensive care unit (ICU) patient ( n = 3) plasma at two different time points, and VE-cadherin phosphorylation was measured by Western blot. Representative image. HUVECs were treated as above for 15 min with plasma collected from individual ARDS with or without RTR (30 mug/ml), and changes in VE-cadherin phosphorylation were assessed. ( D ) Representative Western blot. ( E ) Quantitation. * P < 0.05 relative to control, # P < 0.05 relative to ARDS by one-way ANOVA with Tukey post-test ( n = 3). Measurements of cellular impedance were made in HUVECs over 75 min with ARDS plasma versus ARDS plasma + RTR. ( F ) Representative traces from one patient with four intraexperimental replicates. Arrow denotes addition of plasma. ( G ) Percent attenuation by RTR on ARDS plasma time-
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
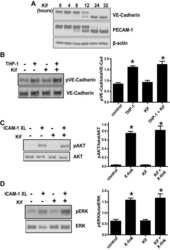
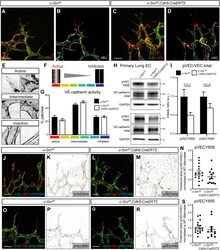
- Fig. 4. VEGFA-induced c-Src activation does not significantly alter VE-cadherin patterning. (A-D) High-magnification images of the sprouting front of retinas. B and D are heat maps of the VE-cadherin (VEC) morphology of retinas in A and C, respectively. Scale bars: 25 um. (E,F) Representative patches of VE-cadherin morphology, from active (warm colors) to intermediate to inactive (cool colors). (G) Quantification of A-D. The VE-cadherin morphology in each patch was manually classified using a scale from active (red, serrated line and bright interior) to inactive (blue, straight line and dark interior). n =4 (where n is the number of retinas, >150 patches quantified per retina). (H) Endothelial cells were isolated from lungs of c-Src flox/flox and c-Src flox/flox ; Cdh5-CreERT2 mice, and protein expression was assessed by western blot. (I) No significant decrease in p-VE-cadherin at either Y658 or Y685 was observed by western blot ( n >6 mice). (J-M) High-magnification images of the sprouting front of retinas immunostained for VE-cadherin and phospho-VE-cadherin Y658 (pVECY658). (K,M) Single-channel images of pVECY658 in J,L, respectively. Scale bars: 25 um. (N) Quantification of pVECY658 staining area normalized to total VEC immunostaining ( n >12, where n is the number of retinas). (O-R) High-magnification images of the sprouting front of retinas immunostained for VE-cadherin and phospho-VE-cadherin Y685 (pVECY685). Scale bars: 25 um. (P,R) Single-channel images of pVECY685
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
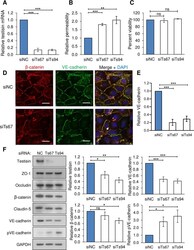
- Experimental details
- 10.1371/journal.pone.0234407.g004 Fig 4 Testisin is required for barrier function in HMEC-1 endothelial monolayers. A) Knockdown of testisin mRNA expression by siRNA silencing in HMEC-1 monolayers, assessed by qPCR analysis. Data is represented as mean +- SEM from 3 independent experiments. B) Assay of permeability of testisin silenced HMEC-1 monolayers compared to control cultures, 1 hour after the addition of FITC-albumin. Data is represented as mean +- SEM from 2-3 independent experiments. C) Cell viability of testisin silenced HMEC-1 monolayers compared to control cultures measured at the end of the assay by PrestoBlue assay. Data is represented as mean +- SEM from 3 independent experiments. D) Representative confocal images of VE-cadherin and beta-catenin expression in testisin siRNA silenced and control HMEC-1 monolayers grown in parallel transwell inserts along with the permeability assays, showing diminished staining and loss at cell-cell junctions (examples indicated by white arrows). Scale bar = 20 mum. E) Signal intensities for VE-cadherin were quantitated from EVOS images of 3 independent experiments performed in triplicate for each siRNA, using Image J. F) Immunoblotting and quantitation by densitometry of junctional proteins showing a significant decrease in total VE-cadherin expression and a significant increase in phospho(Tyr658)-VE cadherin in testisin silenced monolayers compared to control cultures. No significant differences in expression of the tight junc
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Abeta1-42 causes barrier function impairment and VE-cadherin phosphorylation in endothelial cells. HUVECs were cultured for 24 h to allow the consolidation of intercellular junctions and barrier function before incubation with 25 muM Abeta1-42 or scrambled Abeta1-42 (A). The barrier function was monitored by ECIS for 24 h (impedance measurement were performed at frequency 4000 Hz), after which the monolayer was damaged using a high intensity electrical injury (time 1 s, current 1400 muA). The impedance was measured for a further 24 h to assess cell monolayer and barrier function repair. A representative trace for the impedance within the 72 h of the experiments is shown (top panel) and the statistical analysis of data from 6 and 4 independent experiments (pre-wounding and post-wounding assessments, respectively) is shown in the bottom panel. The statistical significance of the difference was assessed by one-way ANOVA with Tukey post-test (*, p < 0.05; **, p < 0.01; ***, p < 0.001). The localisation of VE-cadherin following treatment with Abeta1-42 or scrambled control peptide was tested by immunocytochemistry (B). Following fixation with 4% w/v paraformaldehyde, VE-cadherin localisation was assessed by antibody staining (plus FITC-labelled secondary) and confocal imaging. VE-cadherin localisation at cell-cell junctions (magnified panel) was quantified by image analysis with FIJI software and the statistical significance of the difference was assessed (Mann Whitney non-paramet
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Vascular endothelial (VE)-cadherin tyrosine phosphorylation in endothelial monolayers treated with p-cresylsulfate (PCS). Human umbilical vein endothelial cell monolayers were treated with 0.1 or 0.2 mM PCS for 2 days. The control group was treated with medium only. Equal amounts (50 µg) of protein samples were loaded in duplicate gels and separated by SDS-PAGE. The protein levels of phosphorylated-VE-cadherin (pVE-cadherin), total VE-cadherin (VE-cadherin), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) were detected by western blotting. Following electrophoresis, the proteins were transferred to polyvinylidene fluoride membranes and detected by either anti-pVE-cadherin or anti-VE-cadherin antibodies via immunostaining on separate membranes. The levels of GAPDH in each membrane were also detected by anti-GAPDH antibodies. (a) Representative results. Lane 1: control sample; lanes 2-3: samples treated with PCS; lane 2: 0.1 mM PCS; and lane 3: 0.2 mM PCS. (b) Protein levels (mean +- SD, n = 3 per group) of pVE relative to VE.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot