Antibody data
- Antibody Data
- Antigen structure
- References [13]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [1]
- Other assay [7]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-1145G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-VE-cadherin (Tyr731) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Storage
- -20°C
Submitted references GRP78 translocation to the cell surface and O-GlcNAcylation of VE-Cadherin contribute to ER stress-mediated endothelial permeability.
Time-Variant SRC Kinase Activation Determines Endothelial Permeability Response.
Deubiquitinase function of A20 maintains and repairs endothelial barrier after lung vascular injury.
Rap1B promotes VEGF-induced endothelial permeability and is required for dynamic regulation of the endothelial barrier.
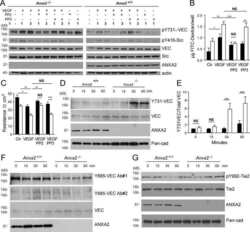
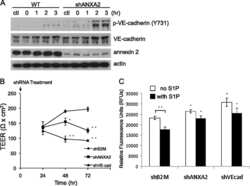
Annexin A2 supports pulmonary microvascular integrity by linking vascular endothelial cadherin and protein tyrosine phosphatases.
Pyk2 phosphorylation of VE-PTP downstream of STIM1-induced Ca(2+) entry regulates disassembly of adherens junctions.
Endothelial Gαq/11 is required for VEGF-induced vascular permeability and angiogenesis.
Novel role of CD47 in rat microvascular endothelium: signaling and regulation of T-cell transendothelial migration.
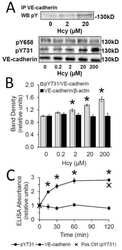
Metabotropic glutamate receptor 5 mediates phosphorylation of vascular endothelial cadherin and nuclear localization of β-catenin in response to homocysteine.
Annexin 2 regulates endothelial morphogenesis by controlling AKT activation and junctional integrity.
ICAM-1-mediated, Src- and Pyk2-dependent vascular endothelial cadherin tyrosine phosphorylation is required for leukocyte transendothelial migration.
ICAM-1-mediated, Src- and Pyk2-dependent vascular endothelial cadherin tyrosine phosphorylation is required for leukocyte transendothelial migration.
Tyrosine phosphorylation of VE-cadherin prevents binding of p120- and beta-catenin and maintains the cellular mesenchymal state.
Lenin R, Nagy PG, Jha KA, Gangaraju R
Scientific reports 2019 Jul 25;9(1):10783
Scientific reports 2019 Jul 25;9(1):10783
Time-Variant SRC Kinase Activation Determines Endothelial Permeability Response.
Klomp JE, Shaaya M, Matsche J, Rebiai R, Aaron JS, Collins KB, Huyot V, Gonzalez AM, Muller WA, Chew TL, Malik AB, Karginov AV
Cell chemical biology 2019 Aug 15;26(8):1081-1094.e6
Cell chemical biology 2019 Aug 15;26(8):1081-1094.e6
Deubiquitinase function of A20 maintains and repairs endothelial barrier after lung vascular injury.
Soni D, Wang DM, Regmi SC, Mittal M, Vogel SM, Schlüter D, Tiruppathi C
Cell death discovery 2018;4:60
Cell death discovery 2018;4:60
Rap1B promotes VEGF-induced endothelial permeability and is required for dynamic regulation of the endothelial barrier.
Lakshmikanthan S, Sobczak M, Li Calzi S, Shaw L, Grant MB, Chrzanowska-Wodnicka M
Journal of cell science 2018 Jan 10;131(1)
Journal of cell science 2018 Jan 10;131(1)
Annexin A2 supports pulmonary microvascular integrity by linking vascular endothelial cadherin and protein tyrosine phosphatases.
Luo M, Flood EC, Almeida D, Yan L, Berlin DA, Heerdt PM, Hajjar KA
The Journal of experimental medicine 2017 Sep 4;214(9):2535-2545
The Journal of experimental medicine 2017 Sep 4;214(9):2535-2545
Pyk2 phosphorylation of VE-PTP downstream of STIM1-induced Ca(2+) entry regulates disassembly of adherens junctions.
Soni D, Regmi SC, Wang DM, DebRoy A, Zhao YY, Vogel SM, Malik AB, Tiruppathi C
American journal of physiology. Lung cellular and molecular physiology 2017 Jun 1;312(6):L1003-L1017
American journal of physiology. Lung cellular and molecular physiology 2017 Jun 1;312(6):L1003-L1017
Endothelial Gαq/11 is required for VEGF-induced vascular permeability and angiogenesis.
Sivaraj KK, Li R, Albarran-Juarez J, Wang S, Tischner D, Grimm M, Swiercz JM, Offermanns S, Wettschureck N
Cardiovascular research 2015 Oct 1;108(1):171-80
Cardiovascular research 2015 Oct 1;108(1):171-80
Novel role of CD47 in rat microvascular endothelium: signaling and regulation of T-cell transendothelial migration.
Martinelli R, Newton G, Carman CV, Greenwood J, Luscinskas FW
Arteriosclerosis, thrombosis, and vascular biology 2013 Nov;33(11):2566-76
Arteriosclerosis, thrombosis, and vascular biology 2013 Nov;33(11):2566-76
Metabotropic glutamate receptor 5 mediates phosphorylation of vascular endothelial cadherin and nuclear localization of β-catenin in response to homocysteine.
Beard RS Jr, Reynolds JJ, Bearden SE
Vascular pharmacology 2012 Mar-Apr;56(3-4):159-67
Vascular pharmacology 2012 Mar-Apr;56(3-4):159-67
Annexin 2 regulates endothelial morphogenesis by controlling AKT activation and junctional integrity.
Su SC, Maxwell SA, Bayless KJ
The Journal of biological chemistry 2010 Dec 24;285(52):40624-34
The Journal of biological chemistry 2010 Dec 24;285(52):40624-34
ICAM-1-mediated, Src- and Pyk2-dependent vascular endothelial cadherin tyrosine phosphorylation is required for leukocyte transendothelial migration.
Allingham MJ, van Buul JD, Burridge K
Journal of immunology (Baltimore, Md. : 1950) 2007 Sep 15;179(6):4053-64
Journal of immunology (Baltimore, Md. : 1950) 2007 Sep 15;179(6):4053-64
ICAM-1-mediated, Src- and Pyk2-dependent vascular endothelial cadherin tyrosine phosphorylation is required for leukocyte transendothelial migration.
Allingham MJ, van Buul JD, Burridge K
Journal of immunology (Baltimore, Md. : 1950) 2007 Sep 15;179(6):4053-64
Journal of immunology (Baltimore, Md. : 1950) 2007 Sep 15;179(6):4053-64
Tyrosine phosphorylation of VE-cadherin prevents binding of p120- and beta-catenin and maintains the cellular mesenchymal state.
Potter MD, Barbero S, Cheresh DA
The Journal of biological chemistry 2005 Sep 9;280(36):31906-12
The Journal of biological chemistry 2005 Sep 9;280(36):31906-12
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
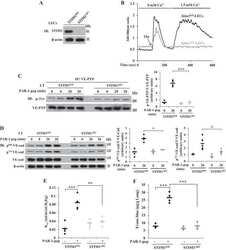
- VE-Cadherin was immunoprecipitated with a VE-Cadherin (pY731) Polyclonal Antibody, Rabbit from HUVEC cells either untreated (1) or treated (2-5) with 100 ng/mL VEGF for 10 minutes. Proteins were resolved by SDS-PAGE on a 10% polyacrylamide gel and transferred to PVDF.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on membrane enriched extracts (30 µg lysate) of HeLa (Lane 1) and HeLa treated with VEGF 100 ng/mL 10 min (Lane 2). The blot was probed with Anti- Phospho-VE-cadherin (Tyr731 ) Polyclonal Antibody (Product # 44-1145G,1:500 dilution) and detected by chemiluminescence using Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, HRP conjugate (Product # A27036, 0.25 µg/mL, 1:4000 dilution). A 74 kDa band corresponding to Phospho-VE-cadherin (Tyr731 ) (Fig. a) was observed in the cell line tested which is also shown in the histogram (Fig. b).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of Phospho-VE-cadherin (Tyr731) was performed using 70% confluent log phase HUVEC cells treated with 100 ng/mL VEGF for 10 min. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Phospho-VE-cadherin (Tyr731) Polyclonal Antibody (Product # 44-1145G) at 1:100 dilution in 0.1% BSA, incubated overnight at 4 degree Celsius and then labeled with Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing plasma membrane localization upon VEGF treatment. Panel e shows untreated cells without any staining. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
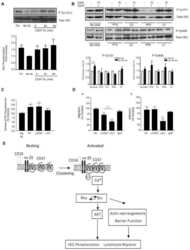
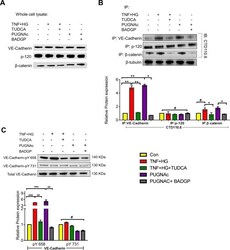
- Figure 6 HRECs exposed to TNF-alpha, and high glucose for 24 h demonstrated alterations in VE-Cadherin and defective complex formation. The whole cell lysates were immunoprecipitated with indicated antibodies, and the immunocomplexes were analyzed for O-GlcNAcylation using anti-O-GlcNAc antibody (clone CTD110.6) ( A ). Whole cell lysates were loaded and probed for specific antibodies as loading controls. Immunoblots of phospho-VE-Cadherin (pY658 and pY731) and total VE-Cadherin from whole cell lysates ( B ). beta-tubulin served as an internal control. Data represent Mean +- SEM from 3 independent experiments performed in duplicates. ***p < 0.001; **p < 0.01; *p < 0.05, # p > 0.05.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot