Antibody data
- Antibody Data
- Antigen structure
- References [17]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [6]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 12-9994-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Perforin Monoclonal Antibody (dG9 (delta G9)), PE, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The dG9 antibody clone reacts with human perforin (pore-forming protein, pfp). Perforin is one of the cytolytic mediators present in the cytoplasmic granules of cytotoxic T lymphocytes (CTL) and natural killer cells (NK). Perforin is involved in the killing function by CTLs and NKs and has an important role in the immune response against tumors and virus infections. Applications Reported: This dG9 (delta G9) antibody has been reported for use in intracellular staining followed by flow cytometric analysis. Applications Tested: This dG9 (delta G9) antibody has been pre-titrated and tested by intracellular staining and flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0.06 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488-561 nm; Emission: 578 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human, Porcine
- Host
- Mouse
- Conjugate
- Yellow dye
- Isotype
- IgG
- Antibody clone number
- dG9 (delta G9)
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references CD8+ T cell-Dependent Remodeling of the Tumor Microenvironment Overcomes Chemoresistance.
Rapamycin improves Graves' orbitopathy by suppressing CD4+ cytotoxic T lymphocytes.
LINC01123 promotes immune escape by sponging miR-214-3p to regulate B7-H3 in head and neck squamous-cell carcinoma.
PD-1-Expressing SARS-CoV-2-Specific CD8(+) T Cells Are Not Exhausted, but Functional in Patients with COVID-19.
NK and T Cell Differentiation at the Maternal-Fetal Interface in Sows During Late Gestation.
Human Thymic CD10(+) PD-1(+) Intraepithelial Lymphocyte Precursors Acquire Interleukin-15 Responsiveness at the CD1a(-) CD95(+) CD28(-) CCR7(-) Developmental Stage.
Interleukin-24 Regulates T Cell Activity in Patients With Colorectal Adenocarcinoma.
A Bispecific Antibody to Link a TRAIL-Based Antitumor Approach to Immunotherapy.
Zoledronic acid boosts γδ T-cell activity in children receiving αβ(+) T and CD19(+) cell-depleted grafts from an HLA-haplo-identical donor.
Enhancing adoptive cancer immunotherapy with Vγ2Vδ2 T cells through pulse zoledronate stimulation.
Blocking the recruitment of naive CD4(+) T cells reverses immunosuppression in breast cancer.
Peritoneal carcinomatosis of colorectal cancer is characterized by structural and functional reorganization of the tumor microenvironment inducing senescence and proliferation arrest in cancer cells.
Added effects of dexamethasone and mesenchymal stem cells on early Natural Killer cell activation.
Reprogramming tumor-infiltrating dendritic cells for CD103+ CD8+ mucosal T-cell differentiation and breast cancer rejection.
Signal transducer and activator of transcription 3 (STAT3) mutations underlying autosomal dominant hyper-IgE syndrome impair human CD8(+) T-cell memory formation and function.
DOCK8 deficiency impairs CD8 T cell survival and function in humans and mice.
Virulence of Toxoplasma gondii is associated with distinct dendritic cell responses and reduced numbers of activated CD8+ T cells.
Lao L, Zeng W, Huang P, Chen H, Jia Z, Wang P, Huang D, Chen J, Nie Y, Yang L, Wu W, Liu J
Cancer immunology research 2023 Mar 1;11(3):320-338
Cancer immunology research 2023 Mar 1;11(3):320-338
Rapamycin improves Graves' orbitopathy by suppressing CD4+ cytotoxic T lymphocytes.
Zhang M, Chong KK, Chen ZY, Guo H, Liu YF, Kang YY, Li YJ, Shi TT, Lai KK, He MQ, Ye K, Kahaly GJ, Shi BY, Wang Y
JCI insight 2023 Feb 8;8(3)
JCI insight 2023 Feb 8;8(3)
LINC01123 promotes immune escape by sponging miR-214-3p to regulate B7-H3 in head and neck squamous-cell carcinoma.
Li H, Yang Z, Yang X, Zhang F, Wang J, Wu Z, Wanyan C, Meng Q, Gao W, Yang X, Wei J
Cell death & disease 2022 Feb 3;13(2):109
Cell death & disease 2022 Feb 3;13(2):109
PD-1-Expressing SARS-CoV-2-Specific CD8(+) T Cells Are Not Exhausted, but Functional in Patients with COVID-19.
Rha MS, Jeong HW, Ko JH, Choi SJ, Seo IH, Lee JS, Sa M, Kim AR, Joo EJ, Ahn JY, Kim JH, Song KH, Kim ES, Oh DH, Ahn MY, Choi HK, Jeon JH, Choi JP, Kim HB, Kim YK, Park SH, Choi WS, Choi JY, Peck KR, Shin EC
Immunity 2021 Jan 12;54(1):44-52.e3
Immunity 2021 Jan 12;54(1):44-52.e3
NK and T Cell Differentiation at the Maternal-Fetal Interface in Sows During Late Gestation.
Stas MR, Koch M, Stadler M, Sawyer S, Sassu EL, Mair KH, Saalmüller A, Gerner W, Ladinig A
Frontiers in immunology 2020;11:582065
Frontiers in immunology 2020;11:582065
Human Thymic CD10(+) PD-1(+) Intraepithelial Lymphocyte Precursors Acquire Interleukin-15 Responsiveness at the CD1a(-) CD95(+) CD28(-) CCR7(-) Developmental Stage.
Billiet L, Goetgeluk G, Bonte S, De Munter S, De Cock L, Pille M, Ingels J, Jansen H, Weening K, Van Nieuwerburgh F, Kerre T, Taghon T, Leclercq G, Vandekerckhove B
International journal of molecular sciences 2020 Nov 20;21(22)
International journal of molecular sciences 2020 Nov 20;21(22)
Interleukin-24 Regulates T Cell Activity in Patients With Colorectal Adenocarcinoma.
Zhang Y, Liu Y, Xu Y
Frontiers in oncology 2019;9:1401
Frontiers in oncology 2019;9:1401
A Bispecific Antibody to Link a TRAIL-Based Antitumor Approach to Immunotherapy.
Satta A, Grazia G, Caroli F, Frigerio B, Di Nicola M, Raspagliesi F, Mezzanzanica D, Zaffaroni N, Gianni AM, Anichini A, Figini M
Frontiers in immunology 2019;10:2514
Frontiers in immunology 2019;10:2514
Zoledronic acid boosts γδ T-cell activity in children receiving αβ(+) T and CD19(+) cell-depleted grafts from an HLA-haplo-identical donor.
Bertaina A, Zorzoli A, Petretto A, Barbarito G, Inglese E, Merli P, Lavarello C, Brescia LP, De Angelis B, Tripodi G, Moretta L, Locatelli F, Airoldi I
Oncoimmunology 2017;6(2):e1216291
Oncoimmunology 2017;6(2):e1216291
Enhancing adoptive cancer immunotherapy with Vγ2Vδ2 T cells through pulse zoledronate stimulation.
Nada MH, Wang H, Workalemahu G, Tanaka Y, Morita CT
Journal for immunotherapy of cancer 2017;5:9
Journal for immunotherapy of cancer 2017;5:9
Blocking the recruitment of naive CD4(+) T cells reverses immunosuppression in breast cancer.
Su S, Liao J, Liu J, Huang D, He C, Chen F, Yang L, Wu W, Chen J, Lin L, Zeng Y, Ouyang N, Cui X, Yao H, Su F, Huang JD, Lieberman J, Liu Q, Song E
Cell research 2017 Apr;27(4):461-482
Cell research 2017 Apr;27(4):461-482
Peritoneal carcinomatosis of colorectal cancer is characterized by structural and functional reorganization of the tumor microenvironment inducing senescence and proliferation arrest in cancer cells.
Seebauer CT, Brunner S, Glockzin G, Piso P, Ruemmele P, Schlitt HJ, Geissler EK, Fichtner-Feigl S, Kesselring R
Oncoimmunology 2016;5(12):e1242543
Oncoimmunology 2016;5(12):e1242543
Added effects of dexamethasone and mesenchymal stem cells on early Natural Killer cell activation.
Michelo CM, Fasse E, van Cranenbroek B, Linda K, van der Meer A, Abdelrazik H, Joosten I
Transplant immunology 2016 Jul;37:1-9
Transplant immunology 2016 Jul;37:1-9
Reprogramming tumor-infiltrating dendritic cells for CD103+ CD8+ mucosal T-cell differentiation and breast cancer rejection.
Wu TC, Xu K, Banchereau R, Marches F, Yu CI, Martinek J, Anguiano E, Pedroza-Gonzalez A, Snipes GJ, O'Shaughnessy J, Nishimura S, Liu YJ, Pascual V, Banchereau J, Oh S, Palucka K
Cancer immunology research 2014 May;2(5):487-500
Cancer immunology research 2014 May;2(5):487-500
Signal transducer and activator of transcription 3 (STAT3) mutations underlying autosomal dominant hyper-IgE syndrome impair human CD8(+) T-cell memory formation and function.
Ives ML, Ma CS, Palendira U, Chan A, Bustamante J, Boisson-Dupuis S, Arkwright PD, Engelhard D, Averbuch D, Magdorf K, Roesler J, Peake J, Wong M, Adelstein S, Choo S, Smart JM, French MA, Fulcher DA, Cook MC, Picard C, Durandy A, Tsumura M, Kobayashi M, Uzel G, Casanova JL, Tangye SG, Deenick EK
The Journal of allergy and clinical immunology 2013 Aug;132(2):400-11.e9
The Journal of allergy and clinical immunology 2013 Aug;132(2):400-11.e9
DOCK8 deficiency impairs CD8 T cell survival and function in humans and mice.
Randall KL, Chan SS, Ma CS, Fung I, Mei Y, Yabas M, Tan A, Arkwright PD, Al Suwairi W, Lugo Reyes SO, Yamazaki-Nakashimada MA, Garcia-Cruz Mde L, Smart JM, Picard C, Okada S, Jouanguy E, Casanova JL, Lambe T, Cornall RJ, Russell S, Oliaro J, Tangye SG, Bertram EM, Goodnow CC
The Journal of experimental medicine 2011 Oct 24;208(11):2305-20
The Journal of experimental medicine 2011 Oct 24;208(11):2305-20
Virulence of Toxoplasma gondii is associated with distinct dendritic cell responses and reduced numbers of activated CD8+ T cells.
Tait ED, Jordan KA, Dupont CD, Harris TH, Gregg B, Wilson EH, Pepper M, Dzierszinski F, Roos DS, Hunter CA
Journal of immunology (Baltimore, Md. : 1950) 2010 Aug 1;185(3):1502-12
Journal of immunology (Baltimore, Md. : 1950) 2010 Aug 1;185(3):1502-12
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
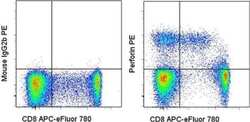
- Experimental details
- Staining of normal human peripheral blood cells with Anti-Human CD8 APC-eFluor® 780 (Product # 47-0088-42) followed by fixation and permeabilization with the Foxp3 Staining Buffers (Product # 00-5523-00) and intracellular staining with Mouse IgG2b K Isotype Control PE (Product # 12-4732-81) (left) or Anti-Human Perforin PE (right). Cells in the lymphocyte gate were used for analysis.
- Conjugate
- Yellow dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
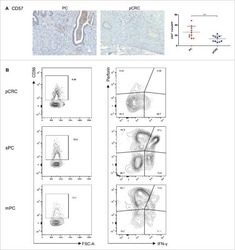
- Experimental details
- Figure 3 Proliferation with IL-15 induces cytotoxic mediators in CD10 + PD-1 + IELps. ( A ) fold expansion of TCRgammadelta + , conventional and IELp cells cultured for 8 days under various conditions (n = 2); ( B ) IFN-gamma, granzyme B and perforin production by unstimulated TCRgammadelta + , conventional and IELp cells incubated for 5 days in the presence of IL-7, IL-15 or IL-15 + IL-12 + IL-18. IFN-gamma, granzyme B, and perforin production measured via intracellular flow cytometry. Bottom: Cytokine production in IELps, plotted for the first and sixth/last generation ( Figure S5 ; mean +- SEM, n = 3); ( C ) flow cytometric analysis of IFN-gamma, granzyme B and perforin production by TCRgammadelta + cells and IELps proliferated for 6 days with IL-15, followed by stimulation with IL-12 + IL-18 for 48 h or with PMA + ionomycin for 4 h (mean +- SEM, n = 3). Dunnett's multiple comparisons test was used to assess statistically significant differences in cytokine production between the different populations and different interleukins. p -value < 0.05 (*), p < 0.01 (**), and p < 0.001 (***).
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
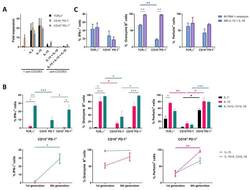
- Main image
- Experimental details
- Fig. 4 Upregulation of LINC01123 or B7-H3 or downregulation of miR-214-3p induces dysfunction of CD8 + T cells. CD8 + T cells were isolated from PBMCs using immunomagnetic beads. After activation and amplification, CD8 + T cells were cocultured with different CAL-27 cell treatment groups. A Expression of TNF-alpha, IFN-gamma, perforin, and granzyme B in CD8 + T-cell population was analyzed with flow cytometry. B Statistical analysis of the results from flow cytometry of CD8 + T cells. C Expression levels of TNF-alpha, IFN-gamma, perforin, and granzyme B in CAL-27-CD8 + T cocultured supernatants, as detected by ELISA. D CCK8 cytotoxicity test of CD8 + T cells. E ELISA was used to assess the immune activity of CD8 + T cells after recombinant human B7-H3 treatment. * P < 0.05 compared with cells without treatment. The experiment was repeated three times.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
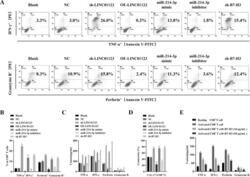
- Experimental details
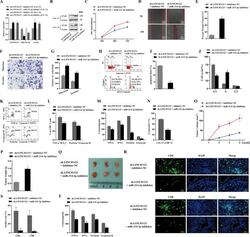
- Fig. 6 The function of LINC01123 in HNSCC can be reversed by miR-214-3p silencing. HNSCC cells were transfected with sh-LINC01123 in the presence of inhibitor-NC or miR-214-3p inhibitor. A Relative expression levels of LINC01123, miR-214-3p, and B7-H3 were determined by qRT-PCR. B B7-H3 protein expression was determined by western blot analysis. C CAL-27 cell viability was measured using a CCK8 assay. D Migration of CAL-27 cells was determined by scratch wound-healing assay. E Statistical results of the scratch-healing test. F Migration and invasion of CAL-27 cells, as determined by transwell migration and invasion assay. G Number of migration and invasion cells. H Apoptosis rate and cell-cycle stage were detected by flow cytometry. I Statistical analysis of apoptosis results. J Statistical analysis of cell cycle stage results. K CD8 + T cells were cocultured with CAL-27 cells that had been transfected with plasmids, and the expression of TNF-alpha, IFN-gamma, perforin, and granzyme B in the CD8 + T cells was analyzed by flow cytometry. L Statistical analysis of CD8 + T-cell flow cytometry results. M After different treatments, CAL-27 cells were cocultured with CD8 + T cells, and the expression levels of TNF-alpha, IFN-gamma, perforin, and granzyme B in coculture supernatants were detected by ELISA. N Killing effect of CD8 + T cells on CAL-27 cells, as assessed by CCK8. O Tumor-volume growth curves of the sh-LINC01123 + inhibitor-NC group and sh-LINC01123 + miR-214-3p-inhibit
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
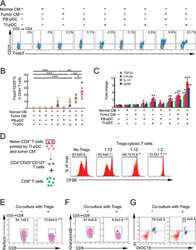
- Figure 3 Naive CD4 + T cells are converted to functional Tregs by tumor-infiltrating DCs and tumor conditioned medium (CM). (A-C) Naive CD4 + T cells from peripheral blood of patients with invasive breast carcinoma were co-cultured with or without autologous pDCs isolated from tumor (TI) or peripheral blood (PB) for 9 days in the presence or absence of 30% CM from autologous tumor slices or adjacent normal tissue slices. (A , B) Non-adherent cells from co-cultures were stained for CD3, CD4, CD25 and intracellular Foxp3, and analyzed by flow cytometry. Representative plots of gated CD3 + CD4 + cells (A) and quantification of percentage of Foxp3 + CD25 + cells among CD3 + CD4 + cells (B) are shown (mean +- SEM, n = 19; * P < 0.05, ** P < 0.01, *** P < 0.001 by Student's t -test). (C) Expression of Treg-associated genes, assessed by qRT-PCR normalized to GAPDH , in sorted CD4 + T cells, relative to expression in cultures without DCs or CM (mean +- SEM, n = 19; * P < 0.05, ** P < 0.01, *** P < 0.001 compared with naive CD4 + T cells cultured alone by Student's t -test). (D-G) Effect of naive CD4 + T cell-derived Tregs, obtained by co-culture with TI pDCs and tumor CM as above, on function of autologous tumor-specific CD8 + T cells. Tumor-specific CD8 + T cells were generated for each subject by stimulating autologous PB CD8 + T cells with autologous tumor lysate-pulsed autologous DCs. Tregs were recovered from co-cultures by magnetic sorting. (D) CFSE-labeled CD8 + T ce
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
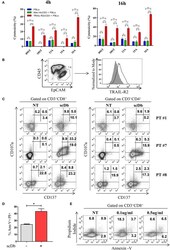
- Figure 7 Treatment of ex vivo cells derived from ovarian cancer patients. (A) Primary ovarian carcinoma cytotoxicity assay after 4 and 16 h of treatment with TRAIL-R2xCD3 or Mec14xCD3 scDbs and pre-activated healthy donors' PBLs. 09ST and 10ST: short-term ovarian cancer cell lines established from biopsies; 13A, 15A, and 16A: cells isolated from ascitic fluid of ovarian cancer patients. The graphs show the percentage of direct cell lysis as the mean +- SD of three wells for treatment. Statistical analysis by one-way ANOVA followed by Tukey's post-test. *** p < 0.001; ns: not significant. (B) Representative staining for CD45 and EpCAM (left) and TRAIL-R2 expression in EpCAM + tumor cells (right) in an ascitic fluid from an ovarian cancer patient. (C) Flow cytometry expression of the surface activation markers CD107a and CD137 on CD8 + (left) and CD4 + (right) T lymphocytes of ascites samples from three ovarian cancer patients, treated or not with scDb (0.5 mug/mL, 18 h). (D) Annexin-V/propidium iodide apoptosis assay of tumor cells (EpCAM + /CD45 - ) treated or not with 0.5 mug/mL scDb for 48 h. Statistical analysis by Student T -test. * p < 0.05. (E) Annexin-V/propidium iodide apoptosis assay on CD8 + T cells treated as in (D) .
- Conjugate
- Yellow dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry