Antibody data
- Antibody Data
- Antigen structure
- References [58]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [18]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 12-0869-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD86 (B7-2) Monoclonal Antibody (IT2.2), PE, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The IT2.2 monoclonal antibody reacts with human CD86, an ~80 kDa surface receptor also known as B7-2. CD86 and CD80 are members of the B7 family of costimulatory molecules. CD86 is expressed at low levels on B cells, macrophages, and dendritic cells and is upregulated on B cells through a variety of surface stimuli including the BCR complex, CD40 and some cytokine receptors. In addition to CD80 (B7-1), CD86 is a counter-receptor for the T cell surface molecules CD28 and CD152 (CTLA-4). The interaction of CD86 with its ligands plays a critical role in T-B crosstalk, T cell costimulation, autoantibody production and Th2-mediated Ig production. The kinetics of upregulation of CD86 upon stimulation supports its major contribution during the primary phase of an immune response. Applications Reported: The IT2.2 antibody has been reported for use in flow cytometric analysis. Applications Tested: This IT2.2 antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0.5 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488-561 nm; Emission: 578 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Yellow dye
- Isotype
- IgG
- Antibody clone number
- IT2.2
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references SARS-CoV-2 Delta (B.1.617.2) variant replicates and induces syncytia formation in human induced pluripotent stem cell-derived macrophages.
WT1 Pulsed Human CD141+ Dendritic Cell Vaccine Has High Potential in Solid Tumor-Targeted Immunotherapy.
Interleukin 6 exacerbates the progression of warm autoimmune hemolytic anemia by influencing the activity and function of B cells.
M1 macrophage-derived exosomes and their key molecule lncRNA HOTTIP suppress head and neck squamous cell carcinoma progression by upregulating the TLR5/NF-κB pathway.
Glycerol monolaurate inhibition of human B cell activation.
TcpC Inhibits M1 but Promotes M2 Macrophage Polarization via Regulation of the MAPK/NF-κB and Akt/STAT6 Pathways in Urinary Tract Infection.
IL-10(-/-) Enhances DCs Immunity Against Chlamydia psittaci Infection via OX40L/NLRP3 and IDO/Treg Pathways.
Small-molecule inhibitors targeting Polycomb repressive complex 1 RING domain.
miR-155-5p regulates macrophage M1 polarization and apoptosis in the synovial fluid of patients with knee osteoarthritis.
Longitudinal profiling of respiratory and systemic immune responses reveals myeloid cell-driven lung inflammation in severe COVID-19.
Dendritic Cell Maturation Regulates TSPAN7 Function in HIV-1 Transfer to CD4(+) T Lymphocytes.
Breast cancer stem cell RNA-pulsed dendritic cells enhance tumor cell killing by effector T cells.
Mesenchymal Stem and Stromal Cells Harness Macrophage-Derived Amphiregulin to Maintain Tissue Homeostasis.
Antitumor immunity targeting fibroblast activation protein-α in a mouse Lewis lung carcinoma model.
Diffuse large B cell lymphoma-derived extracellular vesicles educate macrophages to promote tumours progression by increasing PGC-1β.
The N-Terminal Domain of cGAS Determines Preferential Association with Centromeric DNA and Innate Immune Activation in the Nucleus.
Intrathecal B Cells in MS Have Significantly Greater Lymphangiogenic Potential Compared to B Cells Derived From Non-MS Subjects.
Ginsenoside Rg3 Mitigates Atherosclerosis Progression in Diabetic apoE-/- Mice by Skewing Macrophages to the M2 Phenotype.
Schistosoma mansoni rSm29 Antigen Induces a Regulatory Phenotype on Dendritic Cells and Lymphocytes From Patients With Cutaneous Leishmaniasis.
A comprehensive characterization of aggravated aging-related changes in T lymphocytes and monocytes in end-stage renal disease: the iESRD study.
Dengue immune sera enhance Zika virus infection in human peripheral blood monocytes through Fc gamma receptors.
Functionalization-dependent effects of cellulose nanofibrils on tolerogenic mechanisms of human dendritic cells.
AG490 reverses phenotypic alteration of dendritic cells by bladder cancer cells.
Photodynamic therapy reduces the inhibitory effect of osteosarcoma cells on dendritic cells by upregulating HSP70.
Reshaping of the Dendritic Cell Chromatin Landscape and Interferon Pathways during HIV Infection.
PD-1 Blockade Promotes Emerging Checkpoint Inhibitors in Enhancing T Cell Responses to Allogeneic Dendritic Cells.
Role of Nuclear Factor (Erythroid-Derived 2)-Like 2 Signaling for Effects of Fumaric Acid Esters on Dendritic Cells.
Dexamethasone and Monophosphoryl Lipid A Induce a Distinctive Profile on Monocyte-Derived Dendritic Cells through Transcriptional Modulation of Genes Associated With Essential Processes of the Immune Response.
MALP-2, an agonist of TLR6, promotes the immune status without affecting the differentiation capacity of umbilical cord mesenchymal stem cells.
Abnormal phenotypic features of IgM+B cell subsets in patients with chronic hepatitis C virus infection.
Allostimulatory capacity of conditionally immortalized proximal tubule cell lines for bioartificial kidney application.
Modelling IRF8 Deficient Human Hematopoiesis and Dendritic Cell Development with Engineered iPS Cells.
Dendritic cell maturation, but not type I interferon exposure, restricts infection by HTLV-1, and viral transmission to T-cells.
Pentraxin-3 regulates the inflammatory activity of macrophages.
MicroRNA-720 suppresses M2 macrophage polarization by targeting GATA3.
An in vitro model of granuloma-like cell aggregates substantiates early host immune responses against Mycobacterium massiliense infection.
Pharmacological Inhibition of the Histone Lysine Demethylase KDM1A Suppresses the Growth of Multiple Acute Myeloid Leukemia Subtypes.
Antibodies to P-selectin glycoprotein ligand-1 block dendritic cell-mediated enterovirus 71 transmission and prevent virus-induced cells death.
Interferon beta and vitamin D synergize to induce immunoregulatory receptors on peripheral blood monocytes of multiple sclerosis patients.
Dendritic cell profile induced by Schistosoma mansoni antigen in cutaneous leishmaniasis patients.
Brugia malayi microfilariae induce a regulatory monocyte/macrophage phenotype that suppresses innate and adaptive immune responses.
M-CSF inhibits anti-HIV-1 activity of IL-32, but they enhance M2-like phenotypes of macrophages.
CX₃CR1⁺ mononuclear phagocytes support colitis-associated innate lymphoid cell production of IL-22.
Tumor necrosis factor receptor 1 associates with CD137 ligand and mediates its reverse signaling.
A Francisella tularensis live vaccine strain that improves stimulation of antigen-presenting cells does not enhance vaccine efficacy.
Leishmania major infection in humanized mice induces systemic infection and provokes a nonprotective human immune response.
Vitamin D suppression of endoplasmic reticulum stress promotes an antiatherogenic monocyte/macrophage phenotype in type 2 diabetic patients.
Penicillium marneffei-stimulated dendritic cells enhance HIV-1 trans-infection and promote viral infection by activating primary CD4+ T cells.
Optimal oligonucleotide sequences for TLR9 inhibitory activity in human cells: lack of correlation with TLR9 binding.
Novel role of plasmacytoid dendritic cells in humans: induction of interleukin-10-producing Treg cells by plasmacytoid dendritic cells in patients with rheumatoid arthritis responding to therapy.
Dendritic cells express hematopoietic prostaglandin D synthase and function as a source of prostaglandin D2 in the skin.
Immunological aspects of REIC/Dkk-3 in monocyte differentiation and tumor regression.
Polychromatic flow cytometric high-throughput assay to analyze the innate immune response to Toll-like receptor stimulation.
Enhanced efficacy of CTLA-4 fusion anti-caries DNA vaccines in gnotobiotic hamsters.
Clinical significance of costimulatory molecules CD80/CD86 expression in IgA nephropathy.
Functional expression of a costimulatory B7.2 (CD86) protein on human salivary gland epithelial cells that interacts with the CD28 receptor, but has reduced binding to CTLA4.
CD86 (B7-2) on human B cells. A functional role in proliferation and selective differentiation into IgE- and IgG4-producing cells.
B70 antigen is a second ligand for CTLA-4 and CD28.
Thaweerattanasinp T, Wanitchang A, Saenboonrueng J, Srisutthisamphan K, Wanasen N, Sungsuwan S, Jongkaewwattana A, Chailangkarn T
PeerJ 2023;11:e14918
PeerJ 2023;11:e14918
WT1 Pulsed Human CD141+ Dendritic Cell Vaccine Has High Potential in Solid Tumor-Targeted Immunotherapy.
Cho SY, Jeong SM, Jeon YJ, Yang SJ, Hwang JE, Yoo BM, Kim HS
International journal of molecular sciences 2023 Jan 12;24(2)
International journal of molecular sciences 2023 Jan 12;24(2)
Interleukin 6 exacerbates the progression of warm autoimmune hemolytic anemia by influencing the activity and function of B cells.
Zhao M, Chen L, Yang J, Zhang Z, Wang H, Shao Z, Liu X, Xing L
Scientific reports 2023 Aug 14;13(1):13231
Scientific reports 2023 Aug 14;13(1):13231
M1 macrophage-derived exosomes and their key molecule lncRNA HOTTIP suppress head and neck squamous cell carcinoma progression by upregulating the TLR5/NF-κB pathway.
Jiang H, Zhou L, Shen N, Ning X, Wu D, Jiang K, Huang X
Cell death & disease 2022 Feb 24;13(2):183
Cell death & disease 2022 Feb 24;13(2):183
Glycerol monolaurate inhibition of human B cell activation.
Fosdick MG, Loftus S, Phillips I, Zacharias ZR, Houtman JCD
Scientific reports 2022 Aug 5;12(1):13506
Scientific reports 2022 Aug 5;12(1):13506
TcpC Inhibits M1 but Promotes M2 Macrophage Polarization via Regulation of the MAPK/NF-κB and Akt/STAT6 Pathways in Urinary Tract Infection.
Fang J, Ou Q, Wu B, Li S, Wu M, Qiu J, Cen N, Hu K, Che Y, Ma Y, Pan J
Cells 2022 Aug 28;11(17)
Cells 2022 Aug 28;11(17)
IL-10(-/-) Enhances DCs Immunity Against Chlamydia psittaci Infection via OX40L/NLRP3 and IDO/Treg Pathways.
Li Q, Li X, Quan H, Wang Y, Qu G, Shen Z, He C
Frontiers in immunology 2021;12:645653
Frontiers in immunology 2021;12:645653
Small-molecule inhibitors targeting Polycomb repressive complex 1 RING domain.
Shukla S, Ying W, Gray F, Yao Y, Simes ML, Zhao Q, Miao H, Cho HJ, González-Alonso P, Winkler A, Lund G, Purohit T, Kim E, Zhang X, Ray JM, He S, Nikolaidis C, Ndoj J, Wang J, Jaremko Ł, Jaremko M, Ryan RJH, Guzman ML, Grembecka J, Cierpicki T
Nature chemical biology 2021 Jul;17(7):784-793
Nature chemical biology 2021 Jul;17(7):784-793
miR-155-5p regulates macrophage M1 polarization and apoptosis in the synovial fluid of patients with knee osteoarthritis.
Li GS, Cui L, Wang GD
Experimental and therapeutic medicine 2021 Jan;21(1):68
Experimental and therapeutic medicine 2021 Jan;21(1):68
Longitudinal profiling of respiratory and systemic immune responses reveals myeloid cell-driven lung inflammation in severe COVID-19.
Szabo PA, Dogra P, Gray JI, Wells SB, Connors TJ, Weisberg SP, Krupska I, Matsumoto R, Poon MML, Idzikowski E, Morris SE, Pasin C, Yates AJ, Ku A, Chait M, Davis-Porada J, Guo XV, Zhou J, Steinle M, Mackay S, Saqi A, Baldwin MR, Sims PA, Farber DL
Immunity 2021 Apr 13;54(4):797-814.e6
Immunity 2021 Apr 13;54(4):797-814.e6
Dendritic Cell Maturation Regulates TSPAN7 Function in HIV-1 Transfer to CD4(+) T Lymphocytes.
Perot BP, García-Paredes V, Luka M, Ménager MM
Frontiers in cellular and infection microbiology 2020;10:70
Frontiers in cellular and infection microbiology 2020;10:70
Breast cancer stem cell RNA-pulsed dendritic cells enhance tumor cell killing by effector T cells.
Sumransub N, Jirapongwattana N, Jamjuntra P, Thongchot S, Chieochansin T, Yenchitsomanus PT, Thuwajit P, Warnnissorn M, O-Charoenrat P, Thuwajit C
Oncology letters 2020 Mar;19(3):2422-2430
Oncology letters 2020 Mar;19(3):2422-2430
Mesenchymal Stem and Stromal Cells Harness Macrophage-Derived Amphiregulin to Maintain Tissue Homeostasis.
Ko JH, Kim HJ, Jeong HJ, Lee HJ, Oh JY
Cell reports 2020 Mar 17;30(11):3806-3820.e6
Cell reports 2020 Mar 17;30(11):3806-3820.e6
Antitumor immunity targeting fibroblast activation protein-α in a mouse Lewis lung carcinoma model.
Xie J, Yuan S, Peng L, Li H, Niu L, Xu H, Guo X, Yang M, Duan F
Oncology letters 2020 Jul;20(1):868-876
Oncology letters 2020 Jul;20(1):868-876
Diffuse large B cell lymphoma-derived extracellular vesicles educate macrophages to promote tumours progression by increasing PGC-1β.
Liu W, Zhu M, Wang H, Wang W, Lu Y
Scandinavian journal of immunology 2020 Feb;91(2):e12841
Scandinavian journal of immunology 2020 Feb;91(2):e12841
The N-Terminal Domain of cGAS Determines Preferential Association with Centromeric DNA and Innate Immune Activation in the Nucleus.
Gentili M, Lahaye X, Nadalin F, Nader GPF, Puig Lombardi E, Herve S, De Silva NS, Rookhuizen DC, Zueva E, Goudot C, Maurin M, Bochnakian A, Amigorena S, Piel M, Fachinetti D, Londoño-Vallejo A, Manel N
Cell reports 2019 Feb 26;26(9):2377-2393.e13
Cell reports 2019 Feb 26;26(9):2377-2393.e13
Intrathecal B Cells in MS Have Significantly Greater Lymphangiogenic Potential Compared to B Cells Derived From Non-MS Subjects.
Stein J, Xu Q, Jackson KC, Romm E, Wuest SC, Kosa P, Wu T, Bielekova B
Frontiers in neurology 2018;9:554
Frontiers in neurology 2018;9:554
Ginsenoside Rg3 Mitigates Atherosclerosis Progression in Diabetic apoE-/- Mice by Skewing Macrophages to the M2 Phenotype.
Guo M, Xiao J, Sheng X, Zhang X, Tie Y, Wang L, Zhao L, Ji X
Frontiers in pharmacology 2018;9:464
Frontiers in pharmacology 2018;9:464
Schistosoma mansoni rSm29 Antigen Induces a Regulatory Phenotype on Dendritic Cells and Lymphocytes From Patients With Cutaneous Leishmaniasis.
Lopes DM, Oliveira SC, Page B, Carvalho LP, Carvalho EM, Cardoso LS
Frontiers in immunology 2018;9:3122
Frontiers in immunology 2018;9:3122
A comprehensive characterization of aggravated aging-related changes in T lymphocytes and monocytes in end-stage renal disease: the iESRD study.
Chiu YL, Shu KH, Yang FJ, Chou TY, Chen PM, Lay FY, Pan SY, Lin CJ, Litjens NHR, Betjes MGH, Bermudez S, Kao KC, Chia JS, Wang G, Peng YS, Chuang YF
Immunity & ageing : I & A 2018;15:27
Immunity & ageing : I & A 2018;15:27
Dengue immune sera enhance Zika virus infection in human peripheral blood monocytes through Fc gamma receptors.
Li M, Zhao L, Zhang C, Wang X, Hong W, Sun J, Liu R, Yu L, Wang J, Zhang F, Jin X
PloS one 2018;13(7):e0200478
PloS one 2018;13(7):e0200478
Functionalization-dependent effects of cellulose nanofibrils on tolerogenic mechanisms of human dendritic cells.
Tomić S, Ilić N, Kokol V, Gruden-Movsesijan A, Mihajlović D, Bekić M, Sofronić-Milosavljević L, Čolić M, Vučević D
International journal of nanomedicine 2018;13:6941-6960
International journal of nanomedicine 2018;13:6941-6960
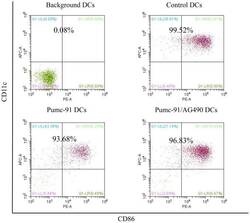
AG490 reverses phenotypic alteration of dendritic cells by bladder cancer cells.
Xiu W, Ma J, Lei T, Zhang M
Oncology letters 2018 Sep;16(3):2851-2856
Oncology letters 2018 Sep;16(3):2851-2856
Photodynamic therapy reduces the inhibitory effect of osteosarcoma cells on dendritic cells by upregulating HSP70.
Zhang F, Zhu Y, Fan G, Hu S
Oncology letters 2018 Oct;16(4):5034-5040
Oncology letters 2018 Oct;16(4):5034-5040
Reshaping of the Dendritic Cell Chromatin Landscape and Interferon Pathways during HIV Infection.
Johnson JS, Lucas SY, Amon LM, Skelton S, Nazitto R, Carbonetti S, Sather DN, Littman DR, Aderem A
Cell host & microbe 2018 Mar 14;23(3):366-381.e9
Cell host & microbe 2018 Mar 14;23(3):366-381.e9
PD-1 Blockade Promotes Emerging Checkpoint Inhibitors in Enhancing T Cell Responses to Allogeneic Dendritic Cells.
Stecher C, Battin C, Leitner J, Zettl M, Grabmeier-Pfistershammer K, Höller C, Zlabinger GJ, Steinberger P
Frontiers in immunology 2017;8:572
Frontiers in immunology 2017;8:572
Role of Nuclear Factor (Erythroid-Derived 2)-Like 2 Signaling for Effects of Fumaric Acid Esters on Dendritic Cells.
Hammer A, Waschbisch A, Knippertz I, Zinser E, Berg J, Jörg S, Kuhbandner K, David C, Pi J, Bayas A, Lee DH, Haghikia A, Gold R, Steinkasserer A, Linker RA
Frontiers in immunology 2017;8:1922
Frontiers in immunology 2017;8:1922
Dexamethasone and Monophosphoryl Lipid A Induce a Distinctive Profile on Monocyte-Derived Dendritic Cells through Transcriptional Modulation of Genes Associated With Essential Processes of the Immune Response.
García-González PA, Schinnerling K, Sepúlveda-Gutiérrez A, Maggi J, Mehdi AM, Nel HJ, Pesce B, Larrondo ML, Aravena O, Molina MC, Catalán D, Thomas R, Verdugo RA, Aguillón JC
Frontiers in immunology 2017;8:1350
Frontiers in immunology 2017;8:1350
MALP-2, an agonist of TLR6, promotes the immune status without affecting the differentiation capacity of umbilical cord mesenchymal stem cells.
Wu X, Xu L, Shen Y, Yu N, Zhang Y, Guo T
Experimental and therapeutic medicine 2017 Dec;14(6):5540-5546
Experimental and therapeutic medicine 2017 Dec;14(6):5540-5546
Abnormal phenotypic features of IgM+B cell subsets in patients with chronic hepatitis C virus infection.
Kong F, Feng B, Zhang H, Rao H, Wang J, Cong X, Wei L
Experimental and therapeutic medicine 2017 Aug;14(2):1846-1852
Experimental and therapeutic medicine 2017 Aug;14(2):1846-1852
Allostimulatory capacity of conditionally immortalized proximal tubule cell lines for bioartificial kidney application.
Mihajlovic M, van den Heuvel LP, Hoenderop JG, Jansen J, Wilmer MJ, Westheim AJF, Allebes WA, Stamatialis D, Hilbrands LB, Masereeuw R
Scientific reports 2017 Aug 2;7(1):7103
Scientific reports 2017 Aug 2;7(1):7103
Modelling IRF8 Deficient Human Hematopoiesis and Dendritic Cell Development with Engineered iPS Cells.
Sontag S, Förster M, Qin J, Wanek P, Mitzka S, Schüler HM, Koschmieder S, Rose-John S, Seré K, Zenke M
Stem cells (Dayton, Ohio) 2017 Apr;35(4):898-908
Stem cells (Dayton, Ohio) 2017 Apr;35(4):898-908
Dendritic cell maturation, but not type I interferon exposure, restricts infection by HTLV-1, and viral transmission to T-cells.
Rizkallah G, Alais S, Futsch N, Tanaka Y, Journo C, Mahieux R, Dutartre H
PLoS pathogens 2017 Apr;13(4):e1006353
PLoS pathogens 2017 Apr;13(4):e1006353
Pentraxin-3 regulates the inflammatory activity of macrophages.
Shiraki A, Kotooka N, Komoda H, Hirase T, Oyama JI, Node K
Biochemistry and biophysics reports 2016 Mar;5:290-295
Biochemistry and biophysics reports 2016 Mar;5:290-295
MicroRNA-720 suppresses M2 macrophage polarization by targeting GATA3.
Zhong Y, Yi C
Bioscience reports 2016 Aug;36(4)
Bioscience reports 2016 Aug;36(4)
An in vitro model of granuloma-like cell aggregates substantiates early host immune responses against Mycobacterium massiliense infection.
Je S, Quan H, Na Y, Cho SN, Kim BJ, Seok SH
Biology open 2016 Aug 15;5(8):1118-27
Biology open 2016 Aug 15;5(8):1118-27
Pharmacological Inhibition of the Histone Lysine Demethylase KDM1A Suppresses the Growth of Multiple Acute Myeloid Leukemia Subtypes.
McGrath JP, Williamson KE, Balasubramanian S, Odate S, Arora S, Hatton C, Edwards TM, O'Brien T, Magnuson S, Stokoe D, Daniels DL, Bryant BM, Trojer P
Cancer research 2016 Apr 1;76(7):1975-88
Cancer research 2016 Apr 1;76(7):1975-88
Antibodies to P-selectin glycoprotein ligand-1 block dendritic cell-mediated enterovirus 71 transmission and prevent virus-induced cells death.
Ren XX, Li C, Xiong SD, Huang Z, Wang JH, Wang HB
Virulence 2015;6(8):802-8
Virulence 2015;6(8):802-8
Interferon beta and vitamin D synergize to induce immunoregulatory receptors on peripheral blood monocytes of multiple sclerosis patients.
Waschbisch A, Sanderson N, Krumbholz M, Vlad G, Theil D, Schwab S, Mäurer M, Derfuss T
PloS one 2014;9(12):e115488
PloS one 2014;9(12):e115488
Dendritic cell profile induced by Schistosoma mansoni antigen in cutaneous leishmaniasis patients.
Lopes DM, Fernandes JS, Cardoso TM, Bafica AM, Oliveira SC, Carvalho EM, Araujo MI, Cardoso LS
BioMed research international 2014;2014:743069
BioMed research international 2014;2014:743069
Brugia malayi microfilariae induce a regulatory monocyte/macrophage phenotype that suppresses innate and adaptive immune responses.
O'Regan NL, Steinfelder S, Venugopal G, Rao GB, Lucius R, Srikantam A, Hartmann S
PLoS neglected tropical diseases 2014 Oct;8(10):e3206
PLoS neglected tropical diseases 2014 Oct;8(10):e3206
M-CSF inhibits anti-HIV-1 activity of IL-32, but they enhance M2-like phenotypes of macrophages.
Osman A, Bhuyan F, Hashimoto M, Nasser H, Maekawa T, Suzu S
Journal of immunology (Baltimore, Md. : 1950) 2014 Jun 1;192(11):5083-9
Journal of immunology (Baltimore, Md. : 1950) 2014 Jun 1;192(11):5083-9
CX₃CR1⁺ mononuclear phagocytes support colitis-associated innate lymphoid cell production of IL-22.
Longman RS, Diehl GE, Victorio DA, Huh JR, Galan C, Miraldi ER, Swaminath A, Bonneau R, Scherl EJ, Littman DR
The Journal of experimental medicine 2014 Jul 28;211(8):1571-83
The Journal of experimental medicine 2014 Jul 28;211(8):1571-83
Tumor necrosis factor receptor 1 associates with CD137 ligand and mediates its reverse signaling.
Moh MC, Lorenzini PA, Gullo C, Schwarz H
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2013 Aug;27(8):2957-66
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2013 Aug;27(8):2957-66
A Francisella tularensis live vaccine strain that improves stimulation of antigen-presenting cells does not enhance vaccine efficacy.
Schmitt DM, O'Dee DM, Horzempa J, Carlson PE Jr, Russo BC, Bales JM, Brown MJ, Nau GJ
PloS one 2012;7(2):e31172
PloS one 2012;7(2):e31172
Leishmania major infection in humanized mice induces systemic infection and provokes a nonprotective human immune response.
Wege AK, Florian C, Ernst W, Zimara N, Schleicher U, Hanses F, Schmid M, Ritter U
PLoS neglected tropical diseases 2012;6(7):e1741
PLoS neglected tropical diseases 2012;6(7):e1741
Vitamin D suppression of endoplasmic reticulum stress promotes an antiatherogenic monocyte/macrophage phenotype in type 2 diabetic patients.
Riek AE, Oh J, Sprague JE, Timpson A, de las Fuentes L, Bernal-Mizrachi L, Schechtman KB, Bernal-Mizrachi C
The Journal of biological chemistry 2012 Nov 9;287(46):38482-94
The Journal of biological chemistry 2012 Nov 9;287(46):38482-94
Penicillium marneffei-stimulated dendritic cells enhance HIV-1 trans-infection and promote viral infection by activating primary CD4+ T cells.
Qin Y, Li Y, Liu W, Tian R, Guo Q, Li S, Li H, Zhang D, Zheng Y, Wu L, Lan K, Wang J
PloS one 2011;6(11):e27609
PloS one 2011;6(11):e27609
Optimal oligonucleotide sequences for TLR9 inhibitory activity in human cells: lack of correlation with TLR9 binding.
Ashman RF, Goeken JA, Latz E, Lenert P
International immunology 2011 Mar;23(3):203-14
International immunology 2011 Mar;23(3):203-14
Novel role of plasmacytoid dendritic cells in humans: induction of interleukin-10-producing Treg cells by plasmacytoid dendritic cells in patients with rheumatoid arthritis responding to therapy.
Kavousanaki M, Makrigiannakis A, Boumpas D, Verginis P
Arthritis and rheumatism 2010 Jan;62(1):53-63
Arthritis and rheumatism 2010 Jan;62(1):53-63
Dendritic cells express hematopoietic prostaglandin D synthase and function as a source of prostaglandin D2 in the skin.
Shimura C, Satoh T, Igawa K, Aritake K, Urade Y, Nakamura M, Yokozeki H
The American journal of pathology 2010 Jan;176(1):227-37
The American journal of pathology 2010 Jan;176(1):227-37
Immunological aspects of REIC/Dkk-3 in monocyte differentiation and tumor regression.
Watanabe M, Kashiwakura Y, Huang P, Ochiai K, Futami J, Li SA, Takaoka M, Nasu Y, Sakaguchi M, Huh NH, Kumon H
International journal of oncology 2009 Mar;34(3):657-63
International journal of oncology 2009 Mar;34(3):657-63
Polychromatic flow cytometric high-throughput assay to analyze the innate immune response to Toll-like receptor stimulation.
Jansen K, Blimkie D, Furlong J, Hajjar A, Rein-Weston A, Crabtree J, Reikie B, Wilson C, Kollmann T
Journal of immunological methods 2008 Jul 31;336(2):183-92
Journal of immunological methods 2008 Jul 31;336(2):183-92
Enhanced efficacy of CTLA-4 fusion anti-caries DNA vaccines in gnotobiotic hamsters.
Zhang F, Li YH, Fan MW, Jia R, Xu QA, Guo JH, Yu F, Tian QW
Acta pharmacologica Sinica 2007 Aug;28(8):1236-42
Acta pharmacologica Sinica 2007 Aug;28(8):1236-42
Clinical significance of costimulatory molecules CD80/CD86 expression in IgA nephropathy.
Wu Q, Jinde K, Endoh M, Sakai H
Kidney international 2004 Mar;65(3):888-96
Kidney international 2004 Mar;65(3):888-96
Functional expression of a costimulatory B7.2 (CD86) protein on human salivary gland epithelial cells that interacts with the CD28 receptor, but has reduced binding to CTLA4.
Kapsogeorgou EK, Moutsopoulos HM, Manoussakis MN
Journal of immunology (Baltimore, Md. : 1950) 2001 Mar 1;166(5):3107-13
Journal of immunology (Baltimore, Md. : 1950) 2001 Mar 1;166(5):3107-13
CD86 (B7-2) on human B cells. A functional role in proliferation and selective differentiation into IgE- and IgG4-producing cells.
Jeannin P, Delneste Y, Lecoanet-Henchoz S, Gauchat JF, Ellis J, Bonnefoy JY
The Journal of biological chemistry 1997 Jun 20;272(25):15613-9
The Journal of biological chemistry 1997 Jun 20;272(25):15613-9
B70 antigen is a second ligand for CTLA-4 and CD28.
Azuma M, Ito D, Yagita H, Okumura K, Phillips JH, Lanier LL, Somoza C
Nature 1993 Nov 4;366(6450):76-9
Nature 1993 Nov 4;366(6450):76-9
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Staining of normal human peripheral blood cells with Mouse IgG2b kappa Isotype Control PE (Product # 12-4732-81) (blue histogram) or Anti-Human CD86 (B7-2) PE (purple histogram). Cells in the monocyte gate were used for analysis.
- Conjugate
- Yellow dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
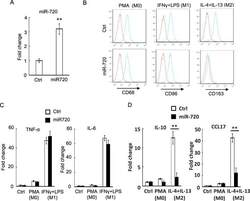
- Figure 2 miR-720 suppresses the expression of M2 phenotypes ( A ) Real-time PCR analysis of miR-720 expression in control vector or pLV- miR-720 -infected THP-1 cells. ( B ) Control or pLV- miR-720 -infected THP-1 cells were induced macrophage differentiation by PMA (M0) and then treated with IFN-gamma plus LPS (M1) or IL-4 plus IL-13 (M2) to induce polarization. The macrophage marker CD68, M1 marker CD86 and M2 marker CD163 were analysed by flow cytometry respectively. ( C ) Detection of M1 cytokine TNF-alpha and IL-6 or ( D ) M2 cytokine IL-10 and chemokine CCL17 by real-time PCR in THP-1 cells used in ( B ). Values are means +- S.D. ** P
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
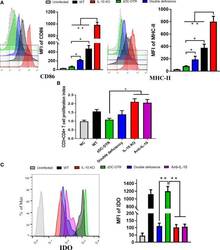
- Figure 5 DCs maturation, T cell proliferation, and IDO expression in the co-culture system. (A) Flow cytometry detection of CD86 and MHC-II DC surface markers; mean fluorescence intensity (MFI) was calculated at 72 hpi. CD86 and MHC-II were analyzed by pre-gated CD11c + cells, CD11c was gated by isotype. CD86 and MHC-II surface markers were significantly upregulated in the co-culture system of CD4 + T cells and IL-10 -/- DCs. Compared to the DCs from the zDC-DTR group, CD86 and MHC-II were significantly higher in the DD group. (B) Measurement of CD4 + T cell proliferation via BrdU assay in the co-culture system of CD4 + T cells and DCs at 72 h. Higher CD4 + T cell proliferation was observed in the IL-10 -/- and anti-IL-10 groups than the zDC-DTR group ( P
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
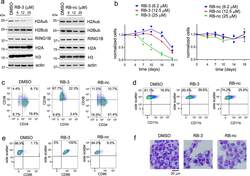
- Figure 3. Treatment with RB-3 leads to reduction in H2Aub and differentiation in TEX cells. a , Assessment of H2Aub, H2Bub, H3 and total H2A in TEX cells treated with RB-3 (left) or RB-nc (right) for 4 days. Representative gel of three independent experiments. b , Effect of RING1B inhibition on the growth of TEX cells treated with RB-3 and RB-nc. Representative data from two independent experiments. c, d, e, Flow-cytometry analysis of CD34 and CD38 ( c ), CD11b ( d ) and CD86 ( e ) expression in TEX cells treated with 25 muM RB-3 and 25 muM RB-nc for 21 days. Representative histograms of two independent experiments. f, Cell morphology of TEX cells after 21 days of treatment with 25 muM RB-3, 25 muM RB-nc or DMSO analyzed by Wright Giemsa staining. Representative slides are shown from two independent experiments.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
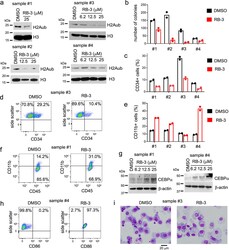
- Figure 6. RB-3 reduces global H2Aub levels and induces differentiation in primary AML patient samples. a, Western blot detection of H2Aub, and H3 in four primary AML patient samples treated with RB-3 for 12 days. Western blots were performed once due to limited amounts of primary samples. b , Colony counts from the second round of colony forming assay in primary AML patient samples treated with 25 muM RB-3 for 14 days. Data are two technical replicates from single experiment. c , Quantification of CD34 + cells in primary AML patient samples treated with 25 muM RB-3 for 12 days. Data are two technical replicates from single experiment. d, Representative FACS histograms showing the level of CD34 + cells for sample #3 treated with 25 muM RB-3 and 25 muM RB-nc for 12 days. e , Flow cytometry quantitation of CD11b + cells in primary AML samples treated with 25 muM RB-3 for 12 days. Data are two technical replicates from single experiment. f , Representative FACS histograms showing the level of CD11b + cells for sample #1 treated with 25 muM RB-3 and 25 muM RB-nc. g, Western blot detection of C/EBPalpha-p42 and beta-actin levels in AML samples treated with RB-3 for 12 days. Western blots were performed once. h , Representative FACS histograms showing CD86 level in sample #4 treated with 25 muM RB-3 and 25 muM RB-nc for 12 days. i , Cell morphology of AML cells analyzed by Wright-Giemsa Giemsa staining after 14 days treatment with 25 muM RB-3 or DMSO. Representative slides from sing
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
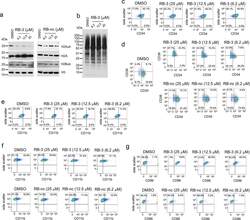
- Extended Data Fig. 6 Long-term effect of treatment of TEX cells with RB-3. a ) Western blot detection of H2Aub in TEX cells treated with increasing doses of RB-3 and RB-nc for 21 days. Representative blot out of two replicates. b ) Western blot detection of total cellular ubiquitination levels using ubiquitin specific antibody in TEX cells treated with RB-3 for 21 days. Representative blot out of three replicates. c ) Flow cytometry analysis of CD34 in TEX cells treated with RB-3 for 7 days. On day 7, cells were stained with APC/CY7-conjugated human anti-CD34 antibody and analyzed by FACS. Representative histograms of two independent experiments. d ) Flow cytometry analysis of CD34 and CD38 in TEX cells treated with RB-3 (top panel) and RB-nc (bottom panel) for 21 days. On day 21, cells were stained with APC/CY7-conjugated human CD34 and PE-conjugated CD38 antibodies and analyzed by FACS. Representative histograms of two independent experiments. e,f ) Flow cytometry analysis of myeloid differentiation marker CD11b/ ITGAM in TEX cells treated with RB-3 for 7 days ( e ) and with RB-3 ( f , top panel) and RB-nc ( f , bottom panel) for 21 days. Cells were stained with Pacific Blue human CD11b antibody and analyzed by FACS. Representative histograms of two independent experiments. g ) Flow cytometry analysis of dendritic cells differentiation marker CD86/B7-2 in TEX cells treated with RB-3 (top panel) and RB-nc (bottom panel) for 21 days. On day 21, cells were stained with Super B
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- The correlation between TAMs and HNSCC prognosis and the establishment of HOTTIP overexpression and knockdown exosomes in M1. A The infiltration levels of immune cells in HNSCC. B Prediction of overall survival according to high- and low-infiltration levels of M0, M1 and M2 macrophages. THP-1-derived M0, M1 and M2 macrophages were confirmed by flow cytometry using ( C ) CD14 plus CD86 and ( D ) CD14 plus CD163 ( n = 3). E The exosomes were extracted from three types of macrophages and confirmed by electronic microscopy and Western blot analysis using CD9 and CD63. F RT-qPCR was conducted to test the expression of HOTTIP in M0, M1 and M2 macrophages ( n = 3). RT-qPCR was conducted to test the expression of HOTTIP in wild type M1 macrophages, HOTTIP-overexpressed and HOTTIP-knockdown M1 macrophages ( G ) and their exosomes ( H ) ( n = 3). Data are presented as mean +- SD. Results were analyzed by One-way ANOVA with a post hoc t -test. Significance: * P < 0.05, ** P < 0.01, *** P < 0.001.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Cancer cells expressing HOTTIP and M1 exosomes reprogram the circulating monocytes into M1 phenotype. A-D Flow cytometry assay detected CD14 + CD86 + M1 and CD14 + CD163 + M2 phenotype in circulating blood of nude mice bearing with HOTTIP-knockdown Hep-2 cells ( n = 3). HOTTIP-knockdown Hep-2 cells suppressed the polarization of M1 monocytes ( A , C ) but induced the polarization of M2 ( B , D ). E - H Flow cytometry assay detected M1 and M2 phenotypes in the blood of tumor-bearing nude mice treated by M1 exosomes ( n = 3). M1 exosomes induced the polarization of M1 monocytes ( E , G ) but inhibited the polarization of M2 ( F , H ). Data are presented as mean +- SD. Results were analyzed by One-way ANOVA with a post hoc test. Significance: ns not significant, ** P < 0.01, *** P < 0.001.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 CFT073 wt infection significantly inhibits CD86 and iNOS but promotes CD206 and Arg-1 expression in K-macrophages from PN mouse models. ( A , B ) Flow cytometry analyses to examine the expression of CD86 and CD206 on K-macrophages. ( C ) FI of CD86 and CD206 were analyzed by FlowJo v10.0.7 software. FI in the control group was set as 1.0. ( D ) mRNA levels of iNOS and Arg-1 in K-macrophages were detected by qRT-PCR. ( E ) Protein levels of iNOS and Arg-1 in K-macrophages were detected by Western blot. ( F ) Gray-scale analyses of bands reflecting iNOS and Arg-1 protein levels in experiments as described in E. Gray scale in the control group was set as 1.0, Mean +- SD of three independent experiments are shown. *: p < 0.05; **: p < 0.01.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
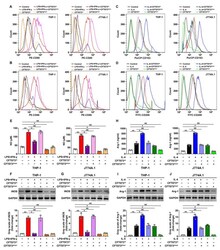
- Experimental details
- Figure 3 CFT073 wt suppresses M1 but promotes M2 macrophage polarization in vitro. ( A , B ) The expression of M1 surface markers CD80 and CD86 in THP-1 and J774A.1 was detected by flow cytometry. ( C , D ) The expression of M2 surface markers CD163 and CD206 in THP-1 and J774A.1 was analyzed by flow cytometry. ( E ) The production of NO in THP-1 and J774A.1 was measured by Griess method. ( F , G ) The iNOS protein levels and bands' gray-scale analyses in THP-1 and J774A.1, respectively. ( H ) The protein levels of Arg-1 in THP-1 and J774A.1 were detected by ELISA. ( I , J ) The protein levels of Arg-1 were detected by Western blot and bands' gray-scale analyses in THP-1 and J774A.1, respectively. Gray scale in the control group was set as 1.0. Mean +- SD of three independent experiments are shown. *: p < 0.05; **: p < 0.01. NS: not significant.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 2 Flow cytometry analysis of M1 and M2 surface markers (A) The expression of CD86 (M1 surface marker) and CD206 (M2 surface marker) on RAW264.7 cells were examined by flow cytometry. (B) Quantification of mean fluorescence intensity (MFI) of the surface markers ( n = 3, respectively). (C) The expression of CD86 and CD206 on THP-1 cells were examined by flow cytometry. (D) Quantification of mean fluorescence intensity (MFI) of the surface markers ( n = 3, respectively). The results are expressed as the mean values +- S.E.M. * p < 0.05, ** p < 0.01, *** p < 0.001.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 2. Percentage of purified DC samples in all experiments. DCs were labeled with the Non-DC Depletion Cocktail for negative selection. Next, DCs were labeled with DC Enrichment Cocktail for positive selection. The expression of CD11c and CD86 was tested by flow cytometery. The percentages of purified DC are presented in the top of each panel. DCs, dendritic cells; CD, cluster of differentiation.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1. The remnants of LM8 decreased the co-stimulatory molecules, and inhibited IL-12 and IL-6 levels, increased IL-10 levels, and inhibited the ability of DCs to stimulate T-cell proliferation. (A) The DCs were isolated following a 48-h co-culture with the remnants of LM8 cells. The cells were then labeled with antibodies against CD11c, MHC-2, CD40, CD86, CD80 and CCR7 for phenotypic analysis by flow cytometry. The numbers in the histograms indicate the geometric mean fluorescence intensity. (B) Following isolation from the co-culture system, the DCs were cultured for 12 h. Subsequently, the expression levels of IL-12, IL-6 and IL-10 in the supernatant were analyzed by ELISA. (C) CD4 + T cells from DO11.10 OVA 323-339 -specific (TCR-transgenic x C57BL/6) F1 hybrid mice were co-cultured with DCs or mDCs (control) in the presence of OVA peptides. Five days later, the total number of viable CD4 + T (CD4 + 7-AAD - ) cells in each well was measured by flow cytometry. Data represent one of at least three experiments with similar results. *P
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
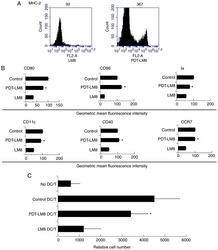
- Figure 2. PDT treatment partly reversed the effect of LM8 remnants on the phenotype of DCs and their ability to stimulate T cells proliferation. (A) The LM8 cells were pre-treated with PDT, and then labeled with antibodies against MHC-2, CD11c, CD40, CD86, CD80 and CCR7, for phenotypic analysis by flow cytometry. (B) The numbers in the histograms indicate the geometric mean fluorescence intensity. *P
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3. PDT treatment upregulated the HSP70 expression in LM8 cells and promoted upregulation of HSP70-activated DCs. (A) The LM8 cells with and without PDT pre-treatment were collected for RNA sequencing analysis. (B) The expression of HSP70, ATF3, Bcl-2, P53, P21, P16 and P27 was analyzed by reverse transcription-quantitative PCR. (C) LM8 cells were transfected with HSP70 small interfering RNA, and the DCs were then co-cultured with LM8 for 48 h, prior to being labeled with an antibody against CD86 for phenotypic analysis by flow cytometry. The numbers in the histograms indicate the geometric mean fluorescence intensity. Data represent one of at least three experiments with similar results. *P
- Conjugate
- Yellow dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry