Antibody data
- Antibody Data
- Antigen structure
- References [5]
- Comments [0]
- Validations
- Western blot [1]
- Other assay [3]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 14-6223-63 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- MyD88 Polyclonal Antibody, eBioscience™
- Antibody type
- Polyclonal
- Antigen
- Other
- Description
- Description: The polyclonal antibody reacts with human and mouse MyD88; the antibody was raised against residues 279-296 of human MyD88 and recognizes human and mouse antigens. MyD88, a myeloid differentiation primary response gene, is expressed in a variety of tissues and functions as an adapter molecule in the IL-1 signaling pathway involved in the inflammatory responses induced by cytokines and LPS. MyD88 associates with and recruits IRAK to the IL-1 receptor. Dominant negative mutants of MyD88 attenuate IL-1R-mediated NF-kappaB activation. MyD88 also functions as a regulator molecule for IL-18 receptor and human Toll receptor family. Targeted disruption of the MyD88 gene results in loss of cellular responses to IL-1 and IL-18, and MyD88-deficient mice lack responses to LPS.
- Concentration
- 1 mg/mL
Submitted references HMGB1 facilitates hypoxia-induced vWF upregulation through TLR2-MYD88-SP1 pathway.
Human fetal membranes generate distinct cytokine profiles in response to bacterial Toll-like receptor and nod-like receptor agonists.
Microglial activation by Citrobacter koseri is mediated by TLR4- and MyD88-dependent pathways.
Peroxisome proliferator-activated receptor-alpha agonist fenofibrate regulates IL-12 family cytokine expression in the CNS: relevance to multiple sclerosis.
MyD88 predicts chemoresistance to paclitaxel in epithelial ovarian cancer.
Singh B, Biswas I, Bhagat S, Surya Kumari S, Khan GA
European journal of immunology 2016 Oct;46(10):2388-2400
European journal of immunology 2016 Oct;46(10):2388-2400
Human fetal membranes generate distinct cytokine profiles in response to bacterial Toll-like receptor and nod-like receptor agonists.
Hoang M, Potter JA, Gysler SM, Han CS, Guller S, Norwitz ER, Abrahams VM
Biology of reproduction 2014 Feb;90(2):39
Biology of reproduction 2014 Feb;90(2):39
Microglial activation by Citrobacter koseri is mediated by TLR4- and MyD88-dependent pathways.
Liu S, Kielian T
Journal of immunology (Baltimore, Md. : 1950) 2009 Nov 1;183(9):5537-47
Journal of immunology (Baltimore, Md. : 1950) 2009 Nov 1;183(9):5537-47
Peroxisome proliferator-activated receptor-alpha agonist fenofibrate regulates IL-12 family cytokine expression in the CNS: relevance to multiple sclerosis.
Xu J, Racke MK, Drew PD
Journal of neurochemistry 2007 Dec;103(5):1801-10
Journal of neurochemistry 2007 Dec;103(5):1801-10
MyD88 predicts chemoresistance to paclitaxel in epithelial ovarian cancer.
Silasi DA, Alvero AB, Illuzzi J, Kelly M, Chen R, Fu HH, Schwartz P, Rutherford T, Azodi M, Mor G
The Yale journal of biology and medicine 2006 Dec;79(3-4):153-63
The Yale journal of biology and medicine 2006 Dec;79(3-4):153-63
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
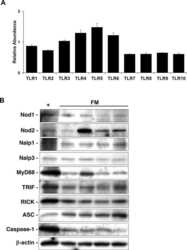
- Western blot was performed using Anti-MyD88 Polyclonal Antibody (Product # 14-6223-63) and a 35kDa band corresponding to MyD88 was observed in all cell lines and tissues tested. Whole cell extracts (30 µg lysate) of MOLT-4 (Lane 1), Jurkat (Lane 2), SK-BR-3 (Lane 3), HeLa (Lane 4), Mouse Spleen (Lane 5) and Mouse Lung (Lane 6) were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0322BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:1000 dilution) and detected by chemiluminescence with Goat anti-Rabbit IgG (H+L), Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
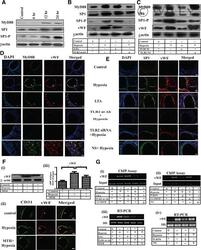
- Involvement of MyD88 and SP1 in TLR2-induced vWF upregulation in AH exposure. (A-G) Anti-TLR2 antibody or MTH was administered through tail vein of mice before AH exposure. Similarly, mice also were exposed to LTA through tail vein. In another set of experiment, Ttr2 gene was silenced in mice by injecting TLR2 siRNA intravascularly. Nonspecific (NS) control siRNA was used similarly in another group of mice. Mice were exposed to AH for 0 h (unexposed control) or 6, 12, 24 h. Bone marrow megakaryocytes were isolated at the indicated time point for evaluation. (A) MyD88, SP1, and SP1-P expression in megakaryocytes was carried out by immunoblotting at indicated time points. (B and C) Expression of MyD88, SP1, SP1-P, and vWF was analyzed by immunoblotting after respective treatment. (D and E) The expressions of vWF and MyD88 and vWF and SP1 in lung were analyzed by confocal microscopy (100x) after indicated treatments. Scale bar: 100 mum. The expression of vWF in megakaryocytes (Fi) by immunoblotting, (Fii) in lung by confocal microscopy (100x), and (Fiii) in plasma by ELISA after indicated treatment. (Gi and ii) ChIP assay and (Giii and iv) RT-PCR were performed in megakaryocytes after indicated treatment. beta-Actin and 18S RNA were used as loading control. (A-F) Data are shown as (F) mean +- SEM ( n = 3 mice) and (A-F) are representative of three independent experiments.* p < 0.05; one-way ANOVA.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
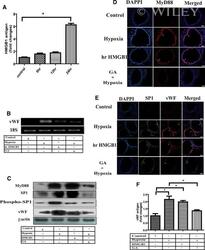
- HMGB1 acts as ligand of TLR2 in AH-induced vWF upregulation. (A-F) Mice were exposed to GA through tail vein before AH exposure. Similarly, mice were also treated with hr-HMGB through tail vain. Blood and lung were collected for indicated time for evaluation. (A) Plasma level of HMGB1 was determined by ELISA. (B) Expression of vWF mRNA in megakaryocytes was evaluated by RT-PCR after respective treatment. (C) Expression of MyD88, SP1, SP1-P, and vWF in megakaryocytes was evaluated by immunoblotting after respective treatment. (D) MyD88 (red) and (E) SP1 (green) expression was examined by immunofluorescence (100x) after respective treatment. (Scale bar: 100 mum.) (F) Plasma level of vWF was also measured by ELISA after indicated treatment. beta-Actin and 18S RNA were used as loading control. (A-F) Data are representative of three independent experiments. Data are presented as mean +- SEM ( n = 3 mice) and are from a single experiment. One-way ANOVA, * p < 0.05.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot