Antibody data
- Antibody Data
- Antigen structure
- References [20]
- Comments [0]
- Validations
- Western blot [2]
- Other assay [10]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-604G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-GSK3B (Tyr216, Tyr279) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Storage
- -20°C
Submitted references Clinical and Preclinical Evidence for M(1) Muscarinic Acetylcholine Receptor Potentiation as a Therapeutic Approach for Rett Syndrome.
Disrupted-in-schizophrenia 1 enhances the quality of circadian rhythm by stabilizing BMAL1.
Differential Characterization of Temozolomide-Resistant Human Glioma Cells.
Beta-amyloid 1-42 monomers, but not oligomers, produce PHF-like conformation of Tau protein.
The identification of raft-derived tau-associated vesicles that are incorporated into immature tangles and paired helical filaments.
A reverse-phase protein microarray-based screen identifies host signaling dynamics upon Burkholderia spp. infection.
DISC1 regulates expression of the neurotrophin VGF through the PI3K/AKT/CREB pathway.
Tauopathy contributes to synaptic and cognitive deficits in a murine model for Alzheimer's disease.
Terminal hypothermic Tau.P301L mice have increased Tau phosphorylation independently of glycogen synthase kinase 3α/β.
Neurological characterization of mice deficient in GSK3α highlight pleiotropic physiological functions in cognition and pathological activity as Tau kinase.
Inhibition of JNK by a peptide inhibitor reduces traumatic brain injury-induced tauopathy in transgenic mice.
Transgenic expression of the amyloid-beta precursor protein-intracellular domain does not induce Alzheimer's Disease-like traits in vivo.
Developmental regulation of tau phosphorylation, tau kinases, and tau phosphatases.
Measuring GSK3 expression and activity in cells.
Glycogen synthase kinase-3 inactivation is not required for ischemic preconditioning or postconditioning in the mouse.
Interaction between ERK and GSK3beta mediates basic fibroblast growth factor-induced apoptosis in SK-N-MC neuroblastoma cells.
Effect of aged garlic extract on APP processing and tau phosphorylation in Alzheimer's transgenic model Tg2576.
Calcium-mediated transient phosphorylation of tau and amyloid precursor protein followed by intraneuronal amyloid-beta accumulation.
Akt contributes to neuroprotection by hypothermia against cerebral ischemia in rats.
Regulation and localization of tyrosine216 phosphorylation of glycogen synthase kinase-3beta in cellular and animal models of neuronal degeneration.
Smith M, Arthur B, Cikowski J, Holt C, Gonzalez S, Fisher NM, Vermudez SAD, Lindsley CW, Niswender CM, Gogliotti RG
Neurotherapeutics : the journal of the American Society for Experimental NeuroTherapeutics 2022 Jul;19(4):1340-1352
Neurotherapeutics : the journal of the American Society for Experimental NeuroTherapeutics 2022 Jul;19(4):1340-1352
Disrupted-in-schizophrenia 1 enhances the quality of circadian rhythm by stabilizing BMAL1.
Lee SB, Park J, Kwak Y, Park YU, Nhung TTM, Suh BK, Woo Y, Suh Y, Cho E, Cho S, Park SK
Translational psychiatry 2021 Feb 4;11(1):110
Translational psychiatry 2021 Feb 4;11(1):110
Differential Characterization of Temozolomide-Resistant Human Glioma Cells.
Lai SW, Huang BR, Liu YS, Lin HY, Chen CC, Tsai CF, Lu DY, Lin C
International journal of molecular sciences 2018 Jan 2;19(1)
International journal of molecular sciences 2018 Jan 2;19(1)
Beta-amyloid 1-42 monomers, but not oligomers, produce PHF-like conformation of Tau protein.
Manassero G, Guglielmotto M, Zamfir R, Borghi R, Colombo L, Salmona M, Perry G, Odetti P, Arancio O, Tamagno E, Tabaton M
Aging cell 2016 Oct;15(5):914-23
Aging cell 2016 Oct;15(5):914-23
The identification of raft-derived tau-associated vesicles that are incorporated into immature tangles and paired helical filaments.
Nishikawa T, Takahashi T, Nakamori M, Hosomi N, Maruyama H, Miyazaki Y, Izumi Y, Matsumoto M
Neuropathology and applied neurobiology 2016 Dec;42(7):639-653
Neuropathology and applied neurobiology 2016 Dec;42(7):639-653
A reverse-phase protein microarray-based screen identifies host signaling dynamics upon Burkholderia spp. infection.
Chiang CY, Uzoma I, Lane DJ, Memišević V, Alem F, Yao K, Kota KP, Bavari S, Wallqvist A, Hakami RM, Panchal RG
Frontiers in microbiology 2015;6:683
Frontiers in microbiology 2015;6:683
DISC1 regulates expression of the neurotrophin VGF through the PI3K/AKT/CREB pathway.
Rodríguez-Seoane C, Ramos A, Korth C, Requena JR
Journal of neurochemistry 2015 Nov;135(3):598-605
Journal of neurochemistry 2015 Nov;135(3):598-605
Tauopathy contributes to synaptic and cognitive deficits in a murine model for Alzheimer's disease.
Stancu IC, Ris L, Vasconcelos B, Marinangeli C, Goeminne L, Laporte V, Haylani LE, Couturier J, Schakman O, Gailly P, Pierrot N, Kienlen-Campard P, Octave JN, Dewachter I
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2014 Jun;28(6):2620-31
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2014 Jun;28(6):2620-31
Terminal hypothermic Tau.P301L mice have increased Tau phosphorylation independently of glycogen synthase kinase 3α/β.
Maurin H, Lechat B, Borghgraef P, Devijver H, Jaworski T, Van Leuven F
The European journal of neuroscience 2014 Jul;40(2):2442-53
The European journal of neuroscience 2014 Jul;40(2):2442-53
Neurological characterization of mice deficient in GSK3α highlight pleiotropic physiological functions in cognition and pathological activity as Tau kinase.
Maurin H, Lechat B, Dewachter I, Ris L, Louis JV, Borghgraef P, Devijver H, Jaworski T, Van Leuven F
Molecular brain 2013 May 25;6:27
Molecular brain 2013 May 25;6:27
Inhibition of JNK by a peptide inhibitor reduces traumatic brain injury-induced tauopathy in transgenic mice.
Tran HT, Sanchez L, Brody DL
Journal of neuropathology and experimental neurology 2012 Feb;71(2):116-29
Journal of neuropathology and experimental neurology 2012 Feb;71(2):116-29
Transgenic expression of the amyloid-beta precursor protein-intracellular domain does not induce Alzheimer's Disease-like traits in vivo.
Giliberto L, d'Abramo C, Acker CM, Davies P, D'Adamio L
PloS one 2010 Jul 16;5(7):e11609
PloS one 2010 Jul 16;5(7):e11609
Developmental regulation of tau phosphorylation, tau kinases, and tau phosphatases.
Yu Y, Run X, Liang Z, Li Y, Liu F, Liu Y, Iqbal K, Grundke-Iqbal I, Gong CX
Journal of neurochemistry 2009 Mar;108(6):1480-94
Journal of neurochemistry 2009 Mar;108(6):1480-94
Measuring GSK3 expression and activity in cells.
Cole AR, Sutherland C
Methods in molecular biology (Clifton, N.J.) 2008;468:45-65
Methods in molecular biology (Clifton, N.J.) 2008;468:45-65
Glycogen synthase kinase-3 inactivation is not required for ischemic preconditioning or postconditioning in the mouse.
Nishino Y, Webb IG, Davidson SM, Ahmed AI, Clark JE, Jacquet S, Shah AM, Miura T, Yellon DM, Avkiran M, Marber MS
Circulation research 2008 Aug 1;103(3):307-14
Circulation research 2008 Aug 1;103(3):307-14
Interaction between ERK and GSK3beta mediates basic fibroblast growth factor-induced apoptosis in SK-N-MC neuroblastoma cells.
Ma C, Bower KA, Chen G, Shi X, Ke ZJ, Luo J
The Journal of biological chemistry 2008 Apr 4;283(14):9248-56
The Journal of biological chemistry 2008 Apr 4;283(14):9248-56
Effect of aged garlic extract on APP processing and tau phosphorylation in Alzheimer's transgenic model Tg2576.
Chauhan NB
Journal of ethnopharmacology 2006 Dec 6;108(3):385-94
Journal of ethnopharmacology 2006 Dec 6;108(3):385-94
Calcium-mediated transient phosphorylation of tau and amyloid precursor protein followed by intraneuronal amyloid-beta accumulation.
Pierrot N, Santos SF, Feyt C, Morel M, Brion JP, Octave JN
The Journal of biological chemistry 2006 Dec 29;281(52):39907-14
The Journal of biological chemistry 2006 Dec 29;281(52):39907-14
Akt contributes to neuroprotection by hypothermia against cerebral ischemia in rats.
Zhao H, Shimohata T, Wang JQ, Sun G, Schaal DW, Sapolsky RM, Steinberg GK
The Journal of neuroscience : the official journal of the Society for Neuroscience 2005 Oct 19;25(42):9794-806
The Journal of neuroscience : the official journal of the Society for Neuroscience 2005 Oct 19;25(42):9794-806
Regulation and localization of tyrosine216 phosphorylation of glycogen synthase kinase-3beta in cellular and animal models of neuronal degeneration.
Bhat RV, Shanley J, Correll MP, Fieles WE, Keith RA, Scott CW, Lee CM
Proceedings of the National Academy of Sciences of the United States of America 2000 Sep 26;97(20):11074-9
Proceedings of the National Academy of Sciences of the United States of America 2000 Sep 26;97(20):11074-9
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
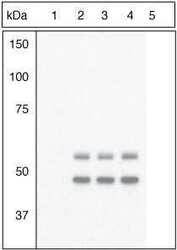
- Experimental details
- Figure Legend: Up-regulation and Antibody-Peptide Competition. Peptide Competition. Extracts of 3T3L1 cells stimulated with 100 nM insulin for 10 minutes were resolved by SDS-PAGE on a 10% Tris-glycine gel and transferred to PVDF. The membrane was blocked with a 5% BSA-TBST buffer for one hour at room temperature and either left untreated (1-4) or treated with lambda phosphatase (5), and then incubated with the GSK-3a (pY279)/ß (pY216) antibody for two hours at room temperature in a 1% BSA-TBST buffer, following its prior incubation with: the phosphopeptide immunogen (1), no peptide (2), the non-phosphopeptide corresponding to the phosphopeptide immunogen (3), or a generic phosphotyrosine-containing peptide (4). After washing, the membrane was incubated with goat F (ab')2 anti-rabbit IgG HRP conjugate (Product # ALI4404), and signals were detected using the Pierce SuperSignal method. The data show that only the phosphopeptide corresponding to GSK-3a (pY279)/ß (pY216) blocks the antibody signal, demonstrating the specificity of the antibody. The data also show that phosphatase stripping eliminates the signal, further verifying that the antibody is phospho-specific. (Product # 44-604G)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
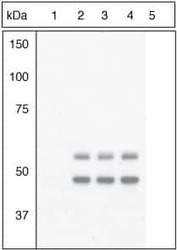
- Experimental details
- Figure Legend: Up-regulation and Antibody-Peptide Competition. Peptide Competition. Extracts of 3T3L1 cells stimulated with 100 nM insulin for 10 minutes were resolved by SDS-PAGE on a 10% Tris-glycine gel and transferred to PVDF. The membrane was blocked with a 5% BSA-TBST buffer for one hour at room temperature and either left untreated (1-4) or treated with lambda phosphatase (5), and then incubated with the GSK-3a (pY279)/ß (pY216) antibody for two hours at room temperature in a 1% BSA-TBST buffer, following its prior incubation with: the phosphopeptide immunogen (1), no peptide (2), the non-phosphopeptide corresponding to the phosphopeptide immunogen (3), or a generic phosphotyrosine-containing peptide (4). After washing, the membrane was incubated with goat F (ab')2 anti-rabbit IgG HRP conjugate (Product # ALI4404), and signals were detected using the Pierce SuperSignal™ method. The data show that only the phosphopeptide corresponding to GSK-3a (pY279)/ß (pY216) blocks the antibody signal, demonstrating the specificity of the antibody. The data also show that phosphatase stripping eliminates the signal, further verifying that the antibody is phospho-specific. (Product # 44-604G)
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
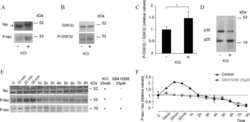
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
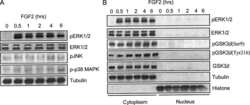
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
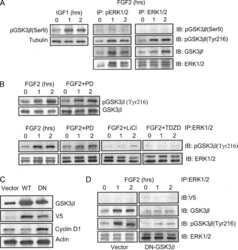
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
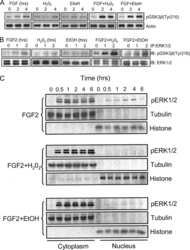
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
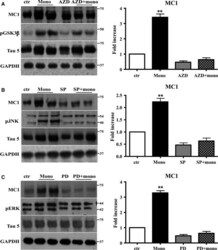
- Figure 6 The activation of JNK , ERK 1/2, and GSK is required to mediate the conformational change of Tau protein induced by Abeta1-42 monomers. Representative Western blot of brain extracts from control and pretreated or not with the GSK 3beta inhibitor AZD 1080, ERK inhibitor PD 98059, and JNK inhibitor SP 600125 before 3 h injection with Abeta1-42 Tau mice using MC 1 (A-C), pGSK 3beta (A), pJNK (B), and pERK 1/2 (C) antibodies for detection. An antibody raised against GAPDH or Tau5 served as loading control. Densitometric quantification shows that each pretreatment was followed by a complete inhibition of the pathway and by the complete reversion of Tau conformational change (A-C). The data are mean +- standard error of the mean ( SEM ), ** P < 0.01 vs. control by one-way ANOVA followed by Bonferroni post hoc test, n = 6 for each kinase.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
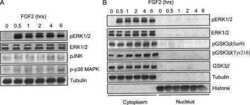
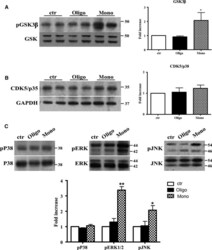
- Figure 4 Abeta1-42 monomers affect Tau phosphorylation through GSK 3beta, ERK 1/2, and JNK kinases activation. Representative Western blot of brain extracts from control (saline) and treated mice using pGSK 3beta (A), CDK 5/p35 (B), pP 38, pERK 1/2, and pJNK (C) antibodies for detection. Densitometric quantification shows an increase in the total protein level of pGSK 3beta induced by monomers (A), while CDK 5/p35 was not modified by any treatments (B). (A,B) An antibody raised against GAPDH was used as loading control. The data are mean +- standard error of the mean ( SEM ), * P < 0.05 vs. control by one-way ANOVA followed by Bonferroni post hoc test, n = 6 for each kinase. (C) pP 38, pERK 1/2, and pJNK levels were standardized against their respective total protein amount. Densitometric quantification shows an increase of ERK 1/2 and JNK activity due to monomers, while p38 was not involved by any treatments. The data are mean +- standard error of the mean ( SEM ), * P < 0.05; ** P < 0.01 vs. control by two-way ANOVA followed by Bonferroni post hoc test, n = 3 for each kinase.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
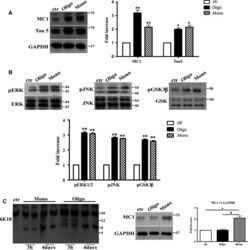
- Figure 5 After 4 days of treatment, Abeta1-42 oligomers and the monomers show a similar effect on Tau protein and phospho-kinases. Representative Western blot of brain extracts from control (saline) and treated mice using MC 1 and Tau5 (A), pERK 1/2, pJNK , and pGSK 3beta (B) antibodies for detection. (A) Densitometric quantification shows an increase in the total protein level of both MC 1 and Tau5 induced by both preparations. An antibody raised against GAPDH served as loading control. The data are mean +- standard error of the mean ( SEM ), * P < 0.05, ** P < 0.01 vs. control by one-way ANOVA followed by Bonferroni post hoc test, n = 3. (B) Densitometric quantification shows an increase of ERK 1/2 and JNK activity due to both monomers and oligomers treatments. pERK 1/2 and pJNK levels were standardized against their respective total protein amount. The data are mean +- SEM , ** P < 0.01 vs. control by two-way ANOVA followed by Bonferroni post hoc test, n = 3 for each kinase. (C) Representative Western blot of the aggregation state of Abeta1-42 in brain tissue of hT au mice injected after 3 h or 4 days with both preparations using the 6E10 antibody. As shown, the aggregation state of oligomeric preparation was different in the short and in the longer treatment: After 3 h, a band of approximately 12 Kda was observed, whereas after 4 days, this band was no more detectable. The data are mean +- SEM , ** P < 0.01 vs. control by two-way ANOVA followed by Bonferroni post hoc test
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
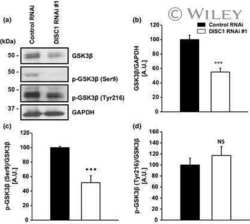
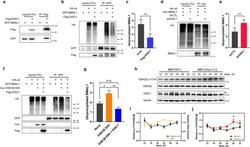
- Fig. 5 DISC1 inhibits BMAL1 ubiquitination in association with suppression of Y216 phosphorylation of GSK3beta. a Co-immunoprecipitation of DISC1 and BMAL1. Flag-DISC1 and GFP-BMAL1 were transfected in HEK293 cells and precipitated with Flag-DISC1. b Ubiquitination assay for BMAL1. HA-ubiquitin was transfected in HEK293 cells to assess the ubiquitination of BMAL1. GFP-BMAL1 was transfected with or without Flag-DISC1. c Quantification of ubiquitin level from ( b ) relative to precipitated BMAL1 level ( n = 5, biological replicates). d Ubiquitination assay of BMAL1 with DISC1 knockdown. HA-ubiquitin was transfected in HEK293 cells to assess the ubiquitination of BMAL1. myc-BMAL1 was transfected with shCTL or shDISC1 for knockdown of DISC1. e Quantification of ubiquitin level from ( d ) relative to precipitated BMAL1 level ( n = 4, biological replicates). f Ubiquitination assay of BMAL1 with GSK3beta S9A and DISC1. HA-ubiquitin was transfected in HEK293 cells to assess the ubiquitination of BMAL1. GFP-BMAL1 was transfected with myc-GSK3beta S9A and Flag-DISC1 according to each combination of constructs. g Quantification of ubiquitin level from ( f ) relative to precipitated BMAL1 level ( n = 4, biological replicates). h Western blot analysis of Y216 phosphorylation of GSK3beta in wild type and Disc1 knockout ( Disc1 -LI) MEFs. MEFs were treated with 1 muM of dexamethasone to synchronize the circadian cycle. Western blot of GSK3beta was conducted in different gel from others to a
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA