Antibody data
- Antibody Data
- Antigen structure
- References [4]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-968G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-PKC lambda/iota (Thr557, Thr564) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 μL
- Storage
- -20°C
Submitted references Oncogenic PKC-ι activates Vimentin during epithelial-mesenchymal transition in melanoma; a study based on PKC-ι and PKC-ζ specific inhibitors.
Mature induced-pluripotent-stem-cell-derived human podocytes reconstitute kidney glomerular-capillary-wall function on a chip.
Two novel atypical PKC inhibitors; ACPD and DNDA effectively mitigate cell proliferation and epithelial to mesenchymal transition of metastatic melanoma while inducing apoptosis.
Regulation of polarized morphogenesis by protein kinase C iota in oncogenic epithelial spheroids.
Ratnayake WS, Apostolatos CA, Apostolatos AH, Schutte RJ, Huynh MA, Ostrov DA, Acevedo-Duncan M
Cell adhesion & migration 2018;12(5):447-463
Cell adhesion & migration 2018;12(5):447-463
Mature induced-pluripotent-stem-cell-derived human podocytes reconstitute kidney glomerular-capillary-wall function on a chip.
Musah S, Mammoto A, Ferrante TC, Jeanty SSF, Hirano-Kobayashi M, Mammoto T, Roberts K, Chung S, Novak R, Ingram M, Fatanat-Didar T, Koshy S, Weaver JC, Church GM, Ingber DE
Nature biomedical engineering 2017;1
Nature biomedical engineering 2017;1
Two novel atypical PKC inhibitors; ACPD and DNDA effectively mitigate cell proliferation and epithelial to mesenchymal transition of metastatic melanoma while inducing apoptosis.
Ratnayake WS, Apostolatos AH, Ostrov DA, Acevedo-Duncan M
International journal of oncology 2017 Nov;51(5):1370-1382
International journal of oncology 2017 Nov;51(5):1370-1382
Regulation of polarized morphogenesis by protein kinase C iota in oncogenic epithelial spheroids.
Linch M, Sanz-Garcia M, Rosse C, Riou P, Peel N, Madsen CD, Sahai E, Downward J, Khwaja A, Dillon C, Roffey J, Cameron AJ, Parker PJ
Carcinogenesis 2014 Feb;35(2):396-406
Carcinogenesis 2014 Feb;35(2):396-406
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
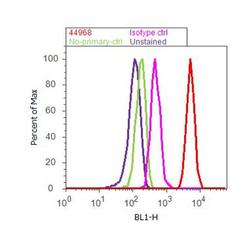
- Experimental details
- Flow cytometry analysis of PKC-iota [pT555]/ PKC lambda [pT563] was done on HeLa cells treated with PMA (200nM, 20 minutes). Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with PKC-iota [pT555]/ PKC lambda [pT563] Rabbit Polyclonal Antibody (44968G, red histogram) or with rabbit isotype control (pink histogram) at 3-5 ug/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Goat Anti-Rabbit Secondary Antibody (A11008) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
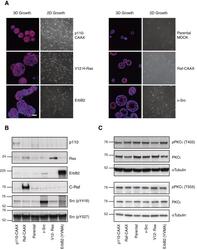
- Experimental details
- Fig. 1. Characterization of oncogenic MDCK cells. ( A ) MDCK cell variants cultured in Matrigel (3D) for 6 days alongside phase images of their growth in 2D. Phalloidin (red)-stained actin and Hoechst (blue)-identified nuclei. Scale bar represents 50 mum. Lysates of MDCK oncogenic variant cell lines in log phase growth were immunoblotted for ( B ) defining proteins and ( C ) phospho-PKCiota/total PKCiota.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
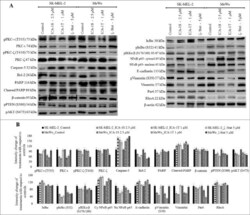
- Experimental details
- Figure 4. Effect of inhibitors (ICA-1S, ICA-1T and zeta-Stat) on aPKC expression, apoptosis, and signaling pathways related to EMT in melanoma cells. Expression of the protein levels of phosphorylated PKC-iota, total PKC-iota, phosphorylated PKC-zeta, total PKC-zeta, Caspase-3, cleaved PARP, total PARP, Bcl-2, beta-catenin, Vimentin, phosphorylated Vimentin, Par6, phosphorylated PTEN, RhoA, E-cadherin, phosphorylated AKT and NF-kappaB p65, IkappaB, phosphorylated IkappaB and phosphorylated IKKalpha/beta for the inhibitor treatments (2.5 uM of ICA-1S, 1 muM of ICA-1T and 5 muM of zeta-Stat) are shown in Fig. 4A . 40-80 ug of protein was loaded in to each well and beta-actin was used as the loading control in each Western blot. Fig. 4B represents the densitometry values for Western blots in Fig. 4A . Experiments ( N = 3) were performed in each trial and representative bands are shown.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Flow cytometry
Flow cytometry