Antibody data
- Antibody Data
- Antigen structure
- References [0]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [29]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 16-0037-81 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD3 Monoclonal Antibody (OKT3), Functional Grade, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The OKT3 monoclonal antibody reacts with an epitope on the epsilon-subunit within the human CD3 complex. The OKT3 antibody has been reported to have potent immunosuppressive properties in vivo and has been proven effective in the treatment of renal, heart and liver allograft rejection. The CD3 subunits, gamma, delta, and epsilon chains, are required for proper assembly, trafficking and surface expression of the TCR complex. CD3 is expressed by thymocytes in a developmentally regulated manner and by all mature T cells. Crosslinking of TCR initiates an intracellular biochemical pathway resulting in cellular activation and proliferation. Applications Reported: The OKT3 antibody has been reported for use in flow cytometric analysis. OKT3 has also been reported for in vitro activation of T cells. Applications Tested: The OKT3 antibody has been tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at less than or equal to 0.25 µg per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest. Storage and handling: Use in a sterile environment. Filtration: 0.2 µm post-manufacturing filtered. Purity: Greater than 90%, as determined by SDS-PAGE. Endotoxin Level: Less than 0.001 ng/µg antibody, as determined by LAL assay. Aggregation: Less than 10%, as determined by HPLC.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- OKT3
- Vial size
- 50 µg
- Concentration
- 1 mg/mL
- Storage
- 4° C
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Staining of normal human peripheral blood cells with 0.125 µg of Mouse IgG2a K Isotype Control Purified (Product # 16-4724-85) (open histogram) or Anti-Human CD3 Functional Grade Purified (filled histogram) followed by Anti-Mouse IgG FITC (Product # 11-4011-85).Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
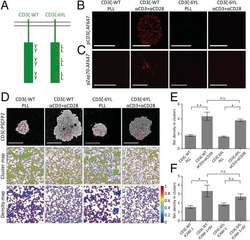
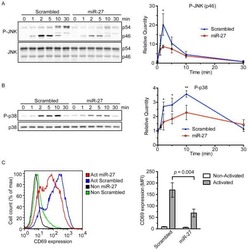
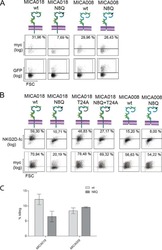
- Figure 9 IL-6 produced by PBLs depleted for CD4 + , CD8 + , HLA-DR + or CD56 + /CD16 + cells. (A) Freshly purified PBLs stained for CD4, CD8, CD3, HLA-DR, CD16, CD56. Depletion controls for PBLs depleted for CD4 + , CD8 + , HLA-DR + or CD16 + /CD56 + cells. PBLs and depletions were stimulated for 7 days with combinations of IL-12 (4 ng/mL), IL-15 (10 ng/mL), IL-18 (40 ng/mL) and TL1A (100 ng/mL) and stained for CD4, CD8, CD3, HLA-DR, CD16 and CD56 expression. (B) IL-6 production after 7 days by PBLs and depletions stimulated with IL-12, IL-15, IL-18 and TL1A as described above. Error bars represent the SEM of two measurements. Statistically significant differences by t-test: *** = p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
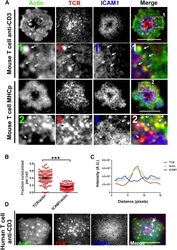
- Figure 2. F-actin foci co-localize with TCR MC and not ICAM1. ( A ) Freshly isolated mouse AND CD4 T cells were incubated with lipid bilayer reconstituted with Alexa568 tagged anti-CD3 (TCR, red) and Alexa647-ICAM1 (blue), for 2 min at 37degC. Post incubation, cells were fixed and stained for F-actin using Alexa488-phalloidin (green), and imaged using TIRF microscopy. The region marked 1 in the 'merge' panel is magnified to clearly show the co-localization of actin foci with TCR-containing MCs. Lower panels: AND mouse CD4 T cell blasts exhibit F-actin enrichment at TCR MCs sites. AND mouse CD4 T cell blasts were labeled with Alexa568-H57 Fab (TCR), and incubated with bilayer reconstituted with MHCp and Alexa405-ICAM1 for 2 min at 37degC, fixed and stained for F-actin. Region marked 2 in the 'merge' image is further magnified to show the overlap between TCR and F-actin (arrows). The insets 1 and 2 in both MHC-activated and anti-CD3-activated mouse T cells are contrasted differently from the original 'merge' image to highlight the TCR and actin distribution. ( B ) Quantitation of the fraction of TCR or ICAM1 localized with F-actin foci. AND CD4 T blasts were incubated with antigen containing bilayer for 2 min, as described above and the images acquired were processed for colocalization assessment as described in 'Materials and methods' section. Each point represents fraction of total synaptic TCR or ICAM1 associated with F-actin foci in a
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
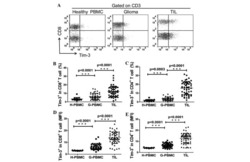
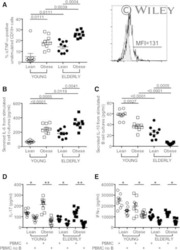
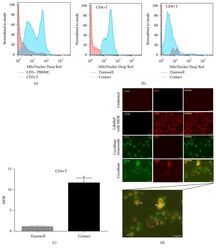
- Figure 7 UC-MSCs transferred mitochondria to activate T cells. PBMCs from SLE patients were labeled with carboxyfluorescein succinimidyl amino ester (CFSE) and treated with anti-CD3/CD28 antibodies for two days. Then they were cocultured with UC-MSCs for 12 h, which had been prelabeled with respiratory mitochondrion specific probe Mitotracker Deep Red (MDR). PBMCs were cultured with UC-MSCs through transwell as control. Then PBMCs were stained with anti-CD3, anti-CD4, or anti-CD8 dye and detected for MDR fluorescence with flow cytometry. Fluorescence microscopy was carried out similarly with anti-CD3/28 stimulation for 12 h and then cocultured for 6 h. All experiments were performed in triplicate. (a) T cells (CD3 positive) rather than non-T cells (CD3 negative) got MDR staining. (b-d) T cells cultured with UC-MSCs directly rather than through transwell got transferred mitochondria ( n = 3). Arrows indicated transferred mitochondria within lymphocytes. * p < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
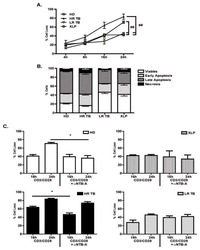
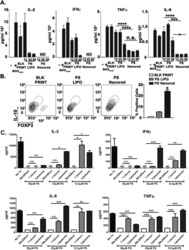
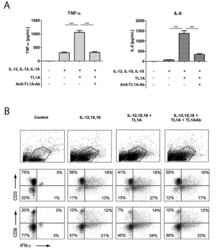
- Figure 2 TL1A induces IL-6 and TNF-alpha. Freshly purified PBMCs were incubated with IL-12 (2 ng/mL), IL-15 (10 ng/mL), IL-18 (10 ng/mL), TL1A (100 ng/mL) and TL1AAb (1 ug/mL, blocking antibody). Extra IL-15 (2 ng/mL) was added on day 3. ( A ) After 6 days, supernatants were collected and different cytokines were measured by bead-based ELISA. Error bars represent the SEM of eight measurements. Statistically significant differences are indicated by ***(t-test, P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
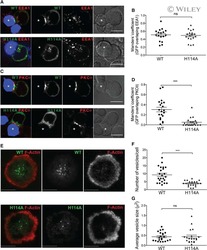
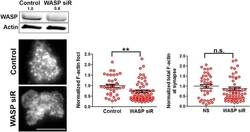
- Figure 1--figure supplement 3. WASP silencing causes a reduction in F-actin foci. Mouse CD4 T cell blasts were electroporated with WASP siRNAs, or with non-targeting sequences (control). The extent of depletion of protein levels was estimated using western blots. ~40% of protein depletion was achieved using WASP siRNAs (relative densitometry value mentioned on top of gel lanes). Cells electroporated with WASP siRNA exhibited significantly fewer F-actin foci. AND T cell blasts electroporated with siRNA sequences, as described above, were incubated with bilayer containing anti-CD3 and ICAM1 for 2 min, followed with fixation and staining with Alexa488-phalloidin (left panels), and imaged using TIRF microscopy. The graphs show quantitation of total F-actin (right graph) or F-actin foci (left graph) per cell, in control or WASP siRNA-treated cells. n1 = 35, n2 = 60, p = 0.238 (right) , p = 0.0025 (left graph). DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
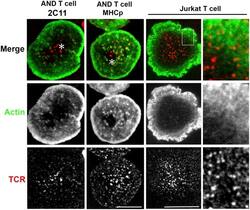
- Figure 2--figure supplement 4. Lack of F-actin foci in the cSMAC of primary T cells and the Jurkat T cell line. AND CD4 T cell blasts were either incubated with bilayers containing Alexa568 tagged anti-CD3 and ICAM1 (far left panels) or were labeled with Alexa568-H57 Fab and incubated with MHCp/ICAM1 bilayers (center left panels), for 2 min. Cells were fixed and stained for F-actin using Alexa488-phalloidin (middle row, green) and visualized using spinning disc confocal microscopy. Each image is a maximum intensity projection of the bottom three planes that show high intensity signals from the bilayer. Note that TCR MCs (bottom row, red) in the F-actin depleted central zone of the cell (asterisk) exhibit no significant co-localization with actin foci. This phenomenon where central microclusters lack F-actin foci was observed in >90% cells exhibiting well-defined cSMAC (number of experiments >3). F-actin foci were not detected in Jurkat T cells (center right panel, and far right insets). Jurkat T cells were activated on bilayer containing Aexa568 tagged anti-CD3 (TCR, red, bottom row) and ICAM1 for 2 min and were fixed and stained with Alexa488-phalloidin (Actin, green, middle row). As highlighted in insets, visible F-actin enrichment is missing from the TCR MCs in these cells. Scale bars, 5 um (AND T cell) and 10 um (Jurkat T cell). DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
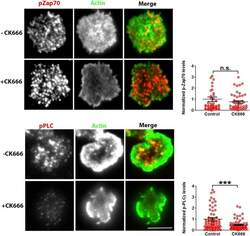
- Figure 6--figure supplement 2. Effect of CK666 on TCR-induced Zap70 and PLCgamma phosphorylation in human T cells. Freshly purified human CD4 T cells were treated with DMSO (control) or CK666, and incubated with bilayer reconstituted with anti-CD3 and ICAM1 for 2 min, in presence of the inhibitor. Cells were then fixed and stained with Alexa488 phalloidin (Actin, green) and either of anti-phospho Zap70 (upper panels) or anti-phospho-PLCgamma1 (Lower panels) antibodies, and subsequently imaged using TIRFM. The graph represents phospho-Zap70 (upper graph) or phospho-PLCgamma1 levels at synapse, normalized to the mean value in control cells. For upper graph, n1 = 49 n2 = 47, p = 0.976; for the lower graph, n1 = 78, n2 = 67, p = 0.004. DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6--figure supplement 1. CK666 treatment leads to loss of foci-associated PLCgamma1 and phospho-HS1 from the synapse. ( A ) Images from Figure 4E were analyzed for phospho-PLCgamma1 and F-actin foci chance level co-localization, after pixel shifts of phospho-PLCgamma1 images by 5 pixels, as performed earlier, in Figure 3--figure supplement 1 . Note that both in DMSO (control) as well as CK666 treated cells, a shift of 5 pixels (x and y) in phospho-PLCgamma1 image results in significant reduction in co-localization, indicating that loss of overlap in CK666 treatment is not just due to reduction in total phospho-PLCgamma1 levels. ( B ) Characterization of the effect of CK666 vs Cytochalasin D (CytoD) on total synaptic F-actin, F-actin foci, phospho-Zap70 and phospho-PLCgamma1 levels. Mouse CD4 T cells were treated with 100 uM CK666 or 5 uM CytoD for indicated time duration, were activated on anti-CD3 and ICAM1 containing surface for 2 min in the presence of inhibitors, fixed and processed for immunofluorescence of the indicated molecules. Note that, while both CK666 as well as CytoD treatments lead to loss of F-actin foci, CytoD treatment causes a greater reduction in total F-actin and phospho-Zap70 levels. For the left graph, for total actin and F-actin foci, n1 = 123, n2 = 118, n3 = 100, n4 = 87; for phospho-Zap70, n1 = 49, n2 = 47, n3 = 53, n4 = 42; for phospho-PLCgamma1, n1 = 78, n2 = 67, n3 = 48, n4 = 42. For total phospho-Zap70, p > 0.90 , and for the rest of the da
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
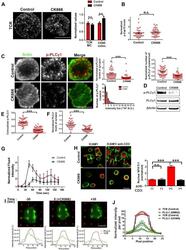
- Figure 6. Arp2/3 inhibition leads to defective TCR-distal signaling. ( A ) Formation of TCR MCs or CD45 exclusion does not require F-actin foci. AND CD4 T cell blasts labeled with Alexa568-H57 Fab (to assess TCR clustering), or Alexa488-CD45 Fab (for CD45 exclusion) at 40C, were then incubated with bilayers containing ICAM1 and MHCp for 2 min, fixed (to assess TCR clustering) or visualized live (for CD45 exclusion) using TIRF microscopy. The images were processed to assess TCR clustering, or CD45 and TCR colocalization using rank filter based filtering, as described in 'Materials and methods' section. For the bars showing TCR cluster intensities per cell, n1 = 45, n2 = 28, p = 0.6; for CD45 co-localization, n1 = 26, n2 = 13, p = 0.09 . ( B ) Phosphorylation of TCR-proximal molecule Zap70 is not reduced in the cells treated with CK666. T cells were treated with DMSO or CK666 for 10 min, then incubated with surface containing ICAM1/anti-CD3 for 3 min, and processed for Y319-phospho-Zap70 and imaged using TIRF. The images were quantified to obtain the synaptic levels phospho-Zap70, and plotted as normalized to mean value of the 'control' cells. In the graph shown here, n1 = 34, n2 = 38, p = 0.16 . ( C ) Synaptic phospho-PLCgamma1 levels are reduced in cells lacking F-actin foci. DMSO (control, top panel) or CK666 (bottom panel) treated AND CD4 T cell blasts were incubated with bilayer containing anti-CD3 and ICAM1 for 2 min, fixed and stained with Alexa488-ph
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
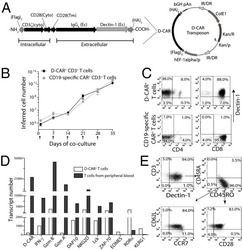
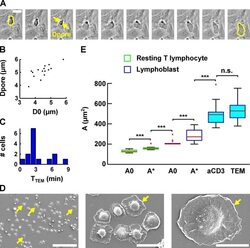
- FIGURE 7: Evolution of lymphoblast membrane surface area during transendothelial migration and cell spreading. (A) Time lapse of a lymphoblast transmigrating between human aortic endothelial cells. Scale bar, 20 mum. Images were taken every 15 s. The projected surface area S proj is represented in yellow before (leftmost image) and after (rightmost image) transendothelial migration. The pore diameter, D pore , is estimated by taking the image in which the lymphoblast width is identical above and below the pore (yellow arrows). (B) D pore during transendothelial migration as a function of the lymphoblast's projected diameter before transendothelial migration (computed using D 0 = S proj /2pi, and D 0 is an equivalent diameter for a sphere whose projected area is S proj ). (C) Histogram of the duration of transmigration. The mean duration is 3 +- 2 min (mean +- SD). (D) Scanning electron microscopy images of lymphoblasts spreading on a substrate coated with anti-CD3 plus anti-CD28 activating antibodies. Scale bars, 100 mum, 20 mum, 10 mum (left to right). Yellow arrows indicate spread cells. (E) Boxplots of the apparent membrane surface area of T-lymphocytes under both passive (white-filled box) and active (blue-filled box) deformations. The bottom and top of the box indicate the 25th and 75th percentiles, respectively. Red plus signs indicate outliers. From left to right, resting T-lymphocytes initially (column 1, A 0 , n = 14) and at rupture (column 2, A *, n = 14) aspir
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
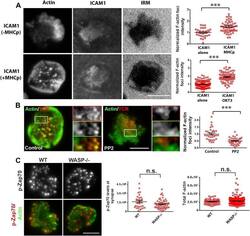
- Figure 3. Relationship of F-actin foci to TCR engagement and TCR proximal signaling. ( A ) AND mouse CD4 T cell blasts were incubated with lipid bilayer containing Alexa405-ICAM1 and MHCp, or bilayer containing Alexa405-ICAM1 alone (ICAM1) at 37degC for 2 min. The cells were fixed and stained with Alexa488-phalloidin (Actin) and imaged using TIRF microscopy. The images in the right panel are reflection (interference reflection microscopy - IRM) images in the two conditions, showing cell-bilayer contact area. Actin images were further high-pass filtered using a rank-filter based subtraction method (see 'Materials and methods') to reveal spatially localized actin features within each cell, under both incubation conditions. The graph (top right) shows average intensity of actin features (F-actin foci) per cell. n1 = 31, n2 = 53, p < 0.0001 . The bottom graph shows foci induction in human CD4 T cells. Primary human CD4 T cells were incubated with bilayer containing Alexa405-ICAM1 alone, or both Alexa405-ICAM1 and Alexa568 tagged anti-CD3 at 37degC for 2 min, and were then fixed and stained for F-actin using Alexa488-phalloidin, for imaging using TIRF microscopy. The graph shows integrated intensity of actin spots per cell, each point on the graph represents a single cell. n1 = 117, n = 79, p < 0.0001 ( B ) Formation of TCR MC associated F-actin foci requires SFK signaling. AND T cell blasts were treated with PP2 for 10 min at 37degC and were labeled with Alexa568-H57 Fab. This
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
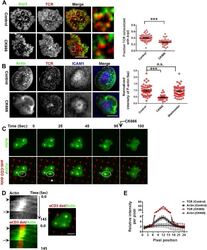
- Figure 5. F-actin foci require activity of the Arp2/3 complex. ( A ) Arp2/3 complex is localized at TCR MCs. DMSO (Control, top) or CK666 treated-(bottom) AND mouse T cell blasts were labeled with Alexa568-H57 Fab and were incubated with bilayer containing ICAM1 and MHCp for 2 min at 37degC, and then fixed and immunostained with anti-Arp3 antibody. The TIRF images show relative distribution of Arp3 (left, green) and TCR (middle, red) in the TIRF plane. Note that while Arp3 distribution overlaps with MCs in control cells, MCs are significantly devoid of Arp3 in CK666-treated cells (areas from 'merge' panels further magnified in insets). The graph (far right) shows the fraction co-localization of total synaptic TCR with Arp3. n1 = 54, n2 = 41, p < 0.0001 . In this experiment, the mean intensity of Arp3 in synapse was 3.46 (+-0.022) x 10 6 for control cells, and 2.38 (+-0.015) x 10 6 for CK666 treated cells (data not shown), and it was repeated twice with similar results. ( B ) TCR MCs associated F-actin enrichment is generated by Arp2/3 complex activation. AND T cell blasts were treated with DMSO alone (control), or 100 muM CK666, or 100 muM blebbistatin for 10 min at 37degC, were labeled with Alexa568-H57 Fab (TCR, red), and incubated with Alexa405-ICAM1 (ICAM1, blue) and MHCp-containing bilayer in the presence of the specific inhibitors at 37degC for 2 min. Cells were subsequently fixed, stained with Alexa488-phalloidin and imaged using TIRF microscopy. Note that
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
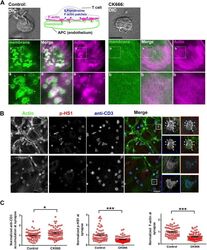
- Figure 7--figure supplement 1. ILP F-actin and signaling in T-EC immunological synapse is dependent on Arp2/3 activity. ( A ) Visualization of loss of F-actin in ILPs following CK666 treatment. T cells were incubated with membrane-YFP (green) expressing endothelial cells in the presence of DMSO (control, left) or CK666 (right) for 5 min, then fixed, permeabilized and stained with Alexa594-phalloidin ('Actin', pink). Note that F-actin rich protrusions are missing at the CK666-treated T cell interface. Scale bar, 5 um. ( B ) Loss of ILPs correlated with reduced phospho-HS1 at the T cell-endothelial cell synaptic interface. T cells were incubated with endothelial cell layer, as described in 'Materials and methods' for 5 min and were then fixed and stained with Alexa594-phalloidin ('Actin', green), phospho-HS1 (red) and anti-CD3 (blue). Cells were then imaged using confocal microscopy. The insets show single T cells marked in the 'Merge' image. Scale bar, 10 um. ( C ) The intensity of the indicated proteins was then analyzed in the synaptic plane without and with CK666 treatment. n1 = 58, n2 = 71, p1 = 0.031, p2, p3 < 0.0001 . DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
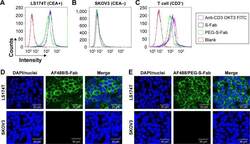
- Figure 4 PEG-S-Fab can bind CEA on tumor cells and CD3 + on T cells. Notes: Flow cytometry analysis of PEG-S-Fab and S-Fab on CEA-positive LS174T cells ( A ), CEA-negative SKOV3 cells ( B ), and CD3 + T cells ( C ). The positive control anti-CD3 antibody OKT3 was used for T-cell flow cytometry. Confocal microscopy of immunofluorescence staining as described in the ""Materials and methods"" section. S-Fab ( D ) and PEG-S-Fab ( E ) on LS174T cells (upper panel) and SKOV3 cells (lower panel), respectively. The scale bar represents 30 mum. AF488, stained by the goat antihuman IgG (H + L)-AlexaFluor 488 antibody; LS174T, human colorectal cell line LS174T; SKOV3, human ovarian cancer cell line SKOV3. Abbreviations: CEA, carcinoembryonic antigen; DAPI, 4',6-diamidino-2-phenylindole; IgG, immunoglobulin G; PEG, polyethylene glycol; PEG-S-Fab, PEGylated S-Fab; S-Fab, single-domain antibody-linked Fab; FITC, fluoresceine isothiocyanate.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
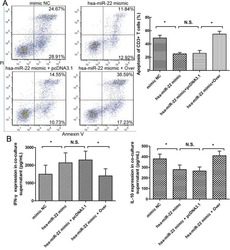
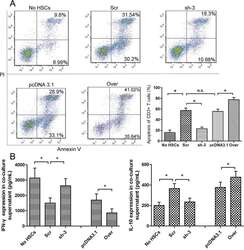
- Figure 4 The expression of galectin-1 in HSCs promotes HSC-induced T cell apoptosis and Th1/Th2 cytokine balance skewing A. Flow cytometry (annexin V-FITC apoptosis detection) analyses to detect T cell apoptosis in CD3+ T cells, cultured alone or co-cultured with HSCs subjected to different pre-treatments (cell transfection for galectin-1 knockdown and overexpression: sh-3 group versus Scr group; Over group versus pcDNA3.1 group) for 48 hours at a ratio of 10:1 (T:HSC), B. ELISA showing the levels of cytokines (IFN-gamma and IL-10) in the supernatant. Data are shown as the means (+- SD) of triplicates (n = 7). *P < 0.05. NC, negative control group; Scr, non-targeting scrambled sequence group; sh, small hairpin RNA sequence transfection group; pcDNA3.1, negative control group; Over, galectin-1 overexpression group; No HSCs, T cells cultured alone.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Cell surface expression of SAIL in CLL, AML and MM patient samples and normal BMMC and PBMC controls. ( a ) Three CLL specimens analyzed by flow cytometry. CLL cells were identified as CD19/CD5 double-positive cells. The histograms present SAIL (filled) and isotype control (open) staining in the live-cell and the CLL population. ( b ) Flow cytometry analysis of three AML specimens. SAIL expression is assessed in live-cells, CD33-positive and CD34-positive cells. ( c ) Flow cytometry analysis of three MM specimens. CD38 high cells with CD56 expression were gated for MM cells. SAIL expression is assessed in the live-cell and the MM population. ( d and e ) Flow cytometry analysis of SAIL expression in BMMC ( d ) and PBMC ( e ) via co-staining with CD19, CD3, CD14, CD56, CD33, CD34 and a cocktail of lineage (LN) markers. Numbers in histograms are median-fluorescence-intensity fold-change values relative to the isotype control. Three and two representative examples are shown for the tumor and normal samples, respectively.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
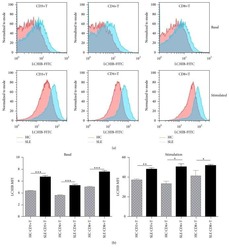
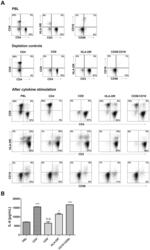
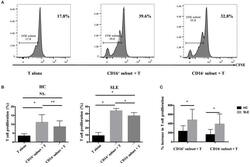
- Figure 6 CD16 + monocytes promoted T-cell proliferation in SLE . CFSE-labeled CD4 + T cells isolated from freshly collected SLE blood or blood bank collected HC blood buffy coat were cocultured with CD16 + and CD16 - monocytes for 60 h in the presence of anti-CD3 (1 mug/mL) and anti-CD 28 (1 mug/mL) antibodies and M-CSF (50 ng/mL). CFSE histograms depicted the number of events ( y -axis) and the fluorescence intensity ( x -axis), with proliferating cells displaying a progressive loss in fluorescence intensity following cell division, indicative of proliferating cells. (A) Representative histogram plots from a patient with SLE displayed the frequencies of CFSE low T cells in monocyte/T-cell cocultures and T cells cultured alone. (B) Percentages of CFSE low T cells in CD4 + T cells after coculture with each monocyte subset from five healthy donors and six patients with SLE. (C) The percentage increase in CFSE low T cells in cocultures of monocytes and T cells compared with CD4 + T cells cultured alone was compared between five healthy individuals and six patients with SLE. Data were expressed as mean +- SD and analyzed by non-parametric paired t test and Mann-Whitney U test. * P < 0.05, ** P < 0.01; NS, no significance.
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry