Antibody data
- Antibody Data
- Antigen structure
- References [3]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 25-0419-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD41a Monoclonal Antibody (HIP8), PE-Cyanine7, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The HIP8 monoclonal antibody reacts with the human CD41 molecule, the integrin alpha IIb also known as platelet GPIIb. CD41 non-covalently associates with integrin beta3 (GPIIIa, CD61) and is expressed by megakaryocytes and platelets. The CD41/CD61 complex is a receptor for fibronectin, fibrinogen, von Willebrand factor, vitronectin and thrombospondin and mediates platelets aggregation. HIP8 blocks platelet aggregation.
- Antibody clone number
- HIP8
- Concentration
- 5 µL/Test
Submitted references Platelet-Derived GARP Induces Peripheral Regulatory T Cells-Potential Impact on T Cell Suppression in Patients with Melanoma-Associated Thrombocytosis.
Mitochondrial DNA in the tumour microenvironment activates neutrophils and is associated with worse outcomes in patients with advanced epithelial ovarian cancer.
EDAG promotes the expansion and survival of human CD34+ cells.
Zimmer N, Krebs FK, Zimmer S, Mitzel-Rink H, Kumm EJ, Jurk K, Grabbe S, Loquai C, Tuettenberg A
Cancers 2020 Dec 5;12(12)
Cancers 2020 Dec 5;12(12)
Mitochondrial DNA in the tumour microenvironment activates neutrophils and is associated with worse outcomes in patients with advanced epithelial ovarian cancer.
Singel KL, Grzankowski KS, Khan ANMNH, Grimm MJ, D'Auria AC, Morrell K, Eng KH, Hylander B, Mayor PC, Emmons TR, Lénárt N, Fekete R, Környei Z, Muthukrishnan U, Gilthorpe JD, Urban CF, Itagaki K, Hauser CJ, Leifer C, Moysich KB, Odunsi K, Dénes Á, Segal BH
British journal of cancer 2019 Jan;120(2):207-217
British journal of cancer 2019 Jan;120(2):207-217
EDAG promotes the expansion and survival of human CD34+ cells.
Zhao K, Zheng WW, Dong XM, Yin RH, Gao R, Li X, Liu JF, Zhan YQ, Yu M, Chen H, Ge CH, Ning HM, Yang XM, Li CY
PloS one 2018;13(1):e0190794
PloS one 2018;13(1):e0190794
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
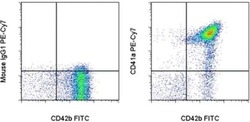
- Experimental details
- Staining of human platelets with Anti-Human CD42b FITC (Product # 11-0429-42) and Mouse IgG1 K Isotype Control PE-Cyanine7 (Product # 25-4714-80) (left) or Anti-Human CD41a PE-Cyanine7 (right). Total events were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
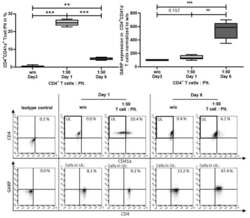
- Experimental details
- Figure A2 Platelets bound to T cells upon activation in coculture. CD4 + CD25 - T cells were cocultured with or without platelets at the ratio of 1:50 and stimulated with 0.5 ug/mL anti-CD3 mAb and 1.0 ug/mL anti-CD28 mAb for 6 days. At day 1 and 6, platelet-T cell conjugates (CD41a + CD4 + double-positive cells) were analyzed via flow cytometry. For assessment of GARP expression, only CD4 + CD41a- cells were included in the analysis (indicated by the pregating on the upper left (UL)). GARP expression was normalized to the untreated (w/o) control. Dot plots show one representative result of five independent experiments. Isotype controls are shown ( n = 5, box and whiskers, medians + min/max, ** p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
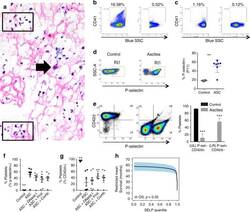
- Experimental details
- Fig. 3 Ovarian cancer ascites induces rapid platelet activation and aggregation that is partially abrogated by DNase and protease treatments. a - g Ascites were collected from patients with newly diagnosed advanced EOC and 500 g supernatants (ASC) were used. a Floating aggregates were collected from ascites before centrifugation and analysed by H&E. Abundant neutrophils (black box) and a sparse number of tumour cells (black arrow) embedded in fibrin deposits (pink filaments) were identified. b , c CD41 + PMP were measured in ascites supernatants by modified flow cytometry ( n = 2). Variability exists between different patients (B, left) 16.58% and (C, left) 1.16% CD41a + . In parallel, samples were 0.1 um-filtered to remove microparticles as a specificity control ( b , c , right panels). d - g . Platelets were isolated from peripheral blood of healthy donors and murine cardiac puncture. d Donor platelets were exposed to Tyrode's buffer with 1 mM CaCl 2 (negative control) or ascites supernatants ( n = 7) in the presence of 1 mM CaCl 2 for 30 minutes prior to staining for flow cytometry. Representative density plots show increased number of P-selectin + platelets (P11 gate) after exposure to ascites supernatants, which is quantified to the right (**, p < 0.01 ). E) Naive murine platelets were exposed to the same ascites supernatants ( n = 7), resulting in increased P-selectin expression and loss of CD42d from the surface of platelets within 15 minutes. The proportion of platele
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry