Antibody data
- Antibody Data
- Antigen structure
- References [1]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [1]
- Flow cytometry [1]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 38-8800 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- PDLIM5 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µg
- Concentration
- 0.25 mg/mL
- Storage
- -20°C
Submitted references Novel phospho-switch function of delta-catenin in dendrite development.
Baumert R, Ji H, Paulucci-Holthauzen A, Wolfe A, Sagum C, Hodgson L, Arikkath J, Chen X, Bedford MT, Waxham MN, McCrea PD
The Journal of cell biology 2020 Nov 2;219(11)
The Journal of cell biology 2020 Nov 2;219(11)
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
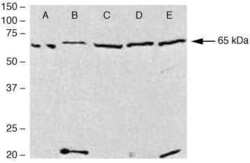
- Experimental details
- Western blot analysis of (A) rat heart homogenates, (B) PC-3, (C) LNCap-FGC, (D) Caco-2, (E) HT-29 cell lysates using Rb anti-ENH (Product # 38-8800)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
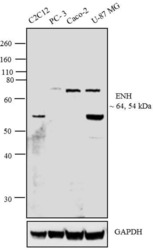
- Experimental details
- Western blot analysis was performed on whole cell extracts of C2C12 (Lane 1), PC-3 (Lane 2), Caco-2 (Lane 3) and U-87 MG (Lane 4). The blots were probed with Anti- ENH Rabbit Polyclonal Antibody (Product # 38-8800, 2 µg/mL) and detected by chemiluminescence using Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, HRP conj µgate (Product # A27036, 0.4 µg/mL, 1:2500 dilution). Bands of ~ 64 and 54 kDa corresponding to ENH were observed across cell lines tested. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane with iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was probed with the relevant primary and secondary Antibody using iBind™ Flex Western Starter Kit (Product # SLF2000S). Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
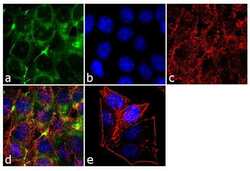
- Experimental details
- Immunofluorescence analysis of ENH was performed using 70% confluent log phase MCF-7 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with ENH Rabbit Polyclonal Antibody (Product # 38-8800) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conj µgate (Product # A27034) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing cytoplasmic and cell junctional localization. Panel e shows the no primary antibody control. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
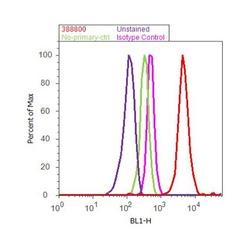
- Experimental details
- Flow cytometry analysis of ENH was done on MCF7 cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with ENH Rabbit Polyclonal Antibody (Product # 38-8800, red histogram) or with rabbit isotype control (pink histogram) at 3-5 µg/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Goat Anti-Rabbit Secondary Antibody (Product # A11008) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10, 000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
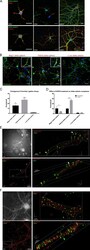
- Experimental details
- Figure 6. Delta-catenin interacts with Magi1 and Pdlim5 in developing hippocampal neurons. (A) Expression of endogenous delta-catenin (green), Magi1 (red, top), and Pdlim5 (red, bottom) in developing primary rat hippocampal neurons at 1 DIV, 7 DIV, and 28 DIV. Scale bars, 20 um. (B and C) Detection of direct associations between endogenous delta-catenin and the proteins Magi1 and Pdlim5 in developing hippocampal neurons. A PLA (Duolink), in combination with delta-catenin, Magi1, and Pdlim5 antibodies, was used to detect direct endogenous protein-protein interactions in 7-DIV hippocampal neurons. (B) Visualization of Magi1:delta-catenin (left) and Pdlim5:delta-catenin (center) interactions, as detected by the presence of red puncta. Insets show presence of puncta on/near small protrusions from the cell in several cases. Antibodies specific to delta-catenin and c-Jun were used in a negative control assay (right). Scale bars, 20 um in main panels and 5 um in zoomed insets. (C) Quantification of average puncta per neuron in each condition (delta-catenin+ Magi1: 16.01 [P = 0.0159]; Pdlim5: 37.73 [P = 0.0007]; c-Jun/Control: 2.393); n = 3 independent experiments. (D) Quantification of puncta/cell detected by a PLA (Duolink) in 7-DIV rat hippocampal neurons treated with DHPG. Cells were fixed immediately following treatment (no treatment versus 20 uM DHPG; 10 min) and subjected to the assay. DHPG treatment produced an increase in Pdlim5:delta-catenin puncta relative to no treatment
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
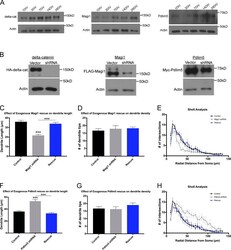
- Experimental details
- Figure S3. shRNA-mediated KD of delta-catenin, Magi1, and Pdlim5. (A) Immunoblots of delta-catenin, Magi1, and Pdlim5 from hippocampal neuron culture cell lysates from 1 DIV to 28 DIV. (B) Immunoblots of cell lysates from HEK293 cells coexpressing HA-delta-catenin, FLAG-Magi1, or Myc-Pdlim5 with either a control vector or the appropriate shRNA. We find that delta-catenin shRNA results in a near-complete loss of delta-catenin (~100% KD), while the Magi1 (~68% KD) and Pdlim5 (~60% KD) shRNAs produce slightly less complete KDs of their respective proteins. (C-E) Analysis of neurons expressing Magi1 shRNA compared with rescues (neurons expressing shRNA-KD and cDNA-overexpression constructs). (C) Expression of Flag-tagged Magi1 in Magi1 shRNA-expressing neurons resulted in a rescue of the shRNA dendrite length phenotype (control: 35.96 +- 1.56 um; shRNA: 21.72 +- 0.98 um; rescue: 33.94 +- 1.44 um, P < 0.0001). (D) Expression of Magi1 cDNA in combination with Magi1 shRNA did not produce a significant impact on dendrite density (control: 16.69 +- 1.26; shRNA: 17.92 +- 2.01; rescue: 18.22 +- 0.85 um, P = 0.7911). (E) Sholl analysis of Magi1 shRNA and rescue neurons reveals that rescue of the shRNA via expression of Magi1 results in a dendritic arborization pattern similar to that of controls. (F-H) Analysis of neurons expressing Pdlim5 shRNA compared with rescues (neurons expressing shRNA-KD and cDNA-overexpression constructs). (F) Expression of Myc-tagged Pdlim5 in Pdlim5 shRNA-expr
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot