PA5-16734
antibody from Invitrogen Antibodies
Targeting: DMD
BMD, DXS142, DXS164, DXS206, DXS230, DXS239, DXS268, DXS269, DXS270, DXS272, MRX85
Antibody data
- Antibody Data
- Antigen structure
- References [11]
- Comments [0]
- Validations
- Immunohistochemistry [1]
- Other assay [4]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA5-16734 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Dystrophin Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 500 µL
- Concentration
- 0.2 mg/mL
- Storage
- 4° C
Submitted references Evaluation of vitamin A status on myogenic gene expression and muscle fiber characteristics.
A Novel Mutation in N-Terminal Actin-Binding Domain of the DMD Gene Presenting Becker Muscular Dystrophy as Recurrent Exertional Rhabdomyolysis: A Case Report.
Muscle phenotype of a rat model of Duchenne muscular dystrophy.
Co-Transplantation of Bone Marrow-MSCs and Myogenic Stem/Progenitor Cells from Adult Donors Improves Muscle Function of Patients with Duchenne Muscular Dystrophy.
Acute and chronic resistance training downregulates select LINE-1 retrotransposon activity markers in human skeletal muscle.
Targeted Expression of Catalase to Mitochondria Protects Against Ischemic Myopathy in High-Fat Diet-Fed Mice.
The first exon duplication mouse model of Duchenne muscular dystrophy: A tool for therapeutic development.
Recovery of altered neuromuscular junction morphology and muscle function in mdx mice after injury.
Recovery of diaphragm function following mechanical ventilation in a rodent model.
Obscurin is required for ankyrinB-dependent dystrophin localization and sarcolemma integrity.
Variable phenotype of del45-55 Becker patients correlated with nNOSμ mislocalization and RYR1 hypernitrosylation.
Wellmann KB, Kim J, Urso PM, Smith ZK, Johnson BJ
Journal of animal science 2021 Mar 1;99(3)
Journal of animal science 2021 Mar 1;99(3)
A Novel Mutation in N-Terminal Actin-Binding Domain of the DMD Gene Presenting Becker Muscular Dystrophy as Recurrent Exertional Rhabdomyolysis: A Case Report.
Lee JM
Annals of Indian Academy of Neurology 2020 Jan-Feb;23(1):123-125
Annals of Indian Academy of Neurology 2020 Jan-Feb;23(1):123-125
Muscle phenotype of a rat model of Duchenne muscular dystrophy.
Iyer SR, Xu S, Shah SB, Lovering RM
Muscle & nerve 2020 Dec;62(6):757-761
Muscle & nerve 2020 Dec;62(6):757-761
Co-Transplantation of Bone Marrow-MSCs and Myogenic Stem/Progenitor Cells from Adult Donors Improves Muscle Function of Patients with Duchenne Muscular Dystrophy.
Klimczak A, Zimna A, Malcher A, Kozlowska U, Futoma K, Czarnota J, Kemnitz P, Bryl A, Kurpisz M
Cells 2020 Apr 30;9(5)
Cells 2020 Apr 30;9(5)
Acute and chronic resistance training downregulates select LINE-1 retrotransposon activity markers in human skeletal muscle.
Romero MA, Mobley CB, Mumford PW, Roberson PA, Haun CT, Kephart WC, Healy JC, Beck DT, Young KC, Martin JS, Lockwood CM, Roberts MD
American journal of physiology. Cell physiology 2018 Mar 1;314(3):C379-C388
American journal of physiology. Cell physiology 2018 Mar 1;314(3):C379-C388
Targeted Expression of Catalase to Mitochondria Protects Against Ischemic Myopathy in High-Fat Diet-Fed Mice.
Ryan TE, Schmidt CA, Green TD, Spangenburg EE, Neufer PD, McClung JM
Diabetes 2016 Sep;65(9):2553-68
Diabetes 2016 Sep;65(9):2553-68
The first exon duplication mouse model of Duchenne muscular dystrophy: A tool for therapeutic development.
Vulin A, Wein N, Simmons TR, Rutherford AM, Findlay AR, Yurkoski JA, Kaminoh Y, Flanigan KM
Neuromuscular disorders : NMD 2015 Nov;25(11):827-34
Neuromuscular disorders : NMD 2015 Nov;25(11):827-34
Recovery of altered neuromuscular junction morphology and muscle function in mdx mice after injury.
Pratt SJP, Shah SB, Ward CW, Kerr JP, Stains JP, Lovering RM
Cellular and molecular life sciences : CMLS 2015 Jan;72(1):153-64
Cellular and molecular life sciences : CMLS 2015 Jan;72(1):153-64
Recovery of diaphragm function following mechanical ventilation in a rodent model.
Bruells CS, Bergs I, Rossaint R, Du J, Bleilevens C, Goetzenich A, Weis J, Wiggs MP, Powers SK, Hein M
PloS one 2014;9(1):e87460
PloS one 2014;9(1):e87460
Obscurin is required for ankyrinB-dependent dystrophin localization and sarcolemma integrity.
Randazzo D, Giacomello E, Lorenzini S, Rossi D, Pierantozzi E, Blaauw B, Reggiani C, Lange S, Peter AK, Chen J, Sorrentino V
The Journal of cell biology 2013 Feb 18;200(4):523-36
The Journal of cell biology 2013 Feb 18;200(4):523-36
Variable phenotype of del45-55 Becker patients correlated with nNOSμ mislocalization and RYR1 hypernitrosylation.
Gentil C, Leturcq F, Ben Yaou R, Kaplan JC, Laforet P, Pénisson-Besnier I, Espil-Taris C, Voit T, Garcia L, Piétri-Rouxel F
Human molecular genetics 2012 Aug 1;21(15):3449-60
Human molecular genetics 2012 Aug 1;21(15):3449-60
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
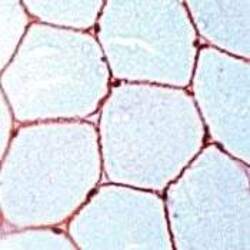
- Experimental details
- Formalin-fixed, paraffin-embedded human skeletal muscle stained with Dystrophin antibody using peroxidase-conjugate and AEC chromogen. Note cell membrane staining of muscle fibers.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 8 Dystrophin expression at mRNA and protein level. ( A ) Four weeks after the cellular therapy, mRNA for dystrophin gene expression in Patient 1 and Patient 3 was assessed at 20% (RQ = 0.206) and at 27% (RQ = 0.265), respectively, of healthy controls (*** p < 0.001). ( B ) In Patient 1, six months after the co-transplantation of BM-MSCs and myogenic stem/progenitor cells, about 15% of small myofibres expressed dystrophin (arrow). Myofibres expressing dystrophin (arrow) were present in the tissue samples taken from Patent 2 at 4 weeks after cellular therapy (EnVision TM FLEX/HRP detection system staining with HRP magenta substrate chromogen).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 The timeline of clinical information, results of the ischemic forearm exercise test, IHC using dystrophin C-terminal antibody, and genetic analysis. (a) Clinical complaints and the results of the blood test corresponding to symptoms are depicted depending on the age. (b) Exercise-associated lactate and ammonia production is identified. (c) Mildly increased fiber size variation and increased number of fibers with internal nuclei are seen in hematoxylin and eosin stain. Absence of immune activity against dystrophin C-terminal antibody. Scale bar, 100 mum. (d) The mutation c.119T > A (p.Leu40His, NM_004006.2) in the DMD gene is confirmed by Sanger sequence in both patient and mother. AST, aspartate transaminase, U/L; ALT, alanine transaminase, U/L; CK, creatine kinase; U/L; myoglobin, ng/mL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 2 FIGURE A, The top panel shows representative curves of maximal isometric torque (200 ms in duration) before injury (pre) and after injury (post) for quadriceps (QUAD) and TA muscle of WT and Dmd-KO rats. The bottom panels show representative trace recordings of torque from lengthening (eccentric) contractions. Muscles were stimulated for 200 ms to induce a peak isometric contraction before lengthening by movement of knee (QUAD) or ankle (TA) in the opposite direction of muscle's action at that joint. B, Scatter plots describing muscle mass, maximal isometric torque, and susceptibility to injury for TA (top) and Quad (bottom) in WT and Dmd-KO rats. * = Significance at P < .05 [Color figure can be viewed at wileyonlinelibrary.com ]
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 1 FIGURE T 2 -weighted images show the thigh and leg muscles in cross-sections (axial, transverse plane) in WT and Dmd-KO rats. As noted in the Methods, imaging was performed prior to any contractile testing. Dmd-KO rat muscles have increased T 2 signal, heterogeneity, and localized hyper-intensities, which have been linked to muscle damage, inflammation, and fatty infiltration. Bar graphs show heterogeneity quantification of the MRI. H&E micrographs on the left show stitched images of the entire muscle for quadriceps and TA (muscle shown are from the untested side to eliminate effects of contractile testing/injury). H&E micrographs on the right show higher power representative images of muscle tissue in cross section. WT muscles show congruent muscle fibers with relatively similar size, while Dmd-KO muscle show variability in fiber size, inflammation (yellow arrows), and centrally nucleated fibers (green arrows), a marker of ongoing muscle degeneration/regeneration. The immunofluorescent micrographs show labeling for dystrophin (green) and nuclei (blue), with absence of dystrophin in Dmd-KO muscles. A.U. = arbitrary units; * = significance at P < .05 [Color figure can be viewed at wileyonlinelibrary.com ]
 Explore
Explore Validate
Validate Learn
Learn Immunohistochemistry
Immunohistochemistry