Antibody data
- Antibody Data
- Antigen structure
- References [1]
- Comments [0]
- Validations
- Western blot [1]
- Other assay [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA5-38735 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Anti-ACRBP Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µg
- Concentration
- 1 mg/mL
- Storage
- -20°C
Submitted references Compartmentalization of the proteasome-interacting proteins during sperm capacitation.
Zigo M, Manaskova-Postlerova P, Jonakova V, Kerns K, Sutovsky P
Scientific reports 2019 Aug 29;9(1):12583
Scientific reports 2019 Aug 29;9(1):12583
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
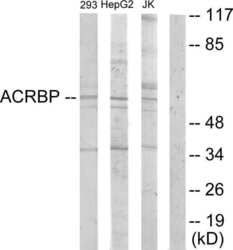
- Experimental details
- Western blot analysis of ACRBP in extracts from 293 cells, HepG2 cells and Jurkat cells using an ACRBP polyclonal antibody (Product # PA5-38735).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
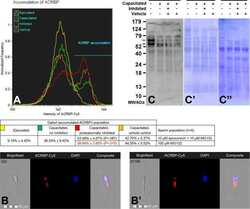
- Experimental details
- Figure 4 ( A ) Flow cytometric measurements of ACRBP accumulation (immature precursor protein and N-terminal part of the ACRBP degradation products were immunolabeled) during in vitro capacitation with proteasome permissive/inhibiting conditions (10 uM epoxomicin +10 uM MG 132 for mild inhibiting conditions, and 100 uM MG132 for strong inhibiting conditions) and vehicle controls, combined with epifluorescence imaging of ACRBP N-terminus in the sperm population with lower fluorescence intensity ( B ), and gated on sperm population with ACRBP accumulated in the midpiece region ( B '). Every flow cytometric run represents 10,000 events. Differences in proteasomal inhibition with statistical significance (P < 0.05), when compared to vehicle control, are highlighted in red. ( C ) Western blot detection of ACRBP in the ejaculated and capacitated spermatozoa under proteasome permissive/inhibiting conditions (100 muM MG132) including vehicle control, with highlighted (red arrows) 61 and 54 kDa doublet of the immature ACRBP precursor protein; ( C ') PVDF membrane stained with CBB after chemiluminescence detection shows comparable protein loads per lane, ( C "") residual gel after electrotransfer for protein normalization purpose. Proteins were extracted with 1% TrX-100, resolved on a 4-20% gradient gel under reducing conditions, and 20 mug of protein was loaded per single lane. Results are presented as mean +- SD of four independent replicates.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot