Antibody data
- Antibody Data
- Antigen structure
- References [35]
- Comments [0]
- Validations
- Other assay [28]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-344 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- beta Amyloid (1-42) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- Reacts with human Abeta42. No significant cross-reactivity to Abeta40 has been observed. Previous lots have shown no cross reactivity to Abeta43. Cross-reactivity against Abeta42 from mouse, rat, pig, cow, sheep, dog, rabbit, frog and polar bear (100% homology) is expected but has not been tested. The antibody has been used in ELISA. Previous lots of this antibody have been used in dot blots, RIA, and other related assays.
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 25 µg
- Concentration
- 1 mg/mL
- Storage
- -20°C
Submitted references A Preliminary Study of Cu Exposure Effects upon Alzheimer's Amyloid Pathology.
Oral Immunization with Soybean Storage Protein Containing Amyloid-β 4-10 Prevents Spatial Learning Decline.
Beta-amyloid induces apoptosis of neuronal cells by inhibition of the Arg/N-end rule pathway proteolytic activity.
Alleviation of Neuropathology by Inhibition of Monoacylglycerol Lipase in APP Transgenic Mice Lacking CB2 Receptors.
Protective effects of 7,8-dihydroxyflavone on neuropathological and neurochemical changes in a mouse model of Alzheimer's disease.
The Bri2 and Bri3 BRICHOS Domains Interact Differently with Aβ(42) and Alzheimer Amyloid Plaques.
Comparative Analysis of Cortical Microinfarcts and Microbleeds using 3.0-Tesla Postmortem Magnetic Resonance Images and Histopathology.
Perinatal Choline Supplementation Reduces Amyloidosis and Increases Choline Acetyltransferase Expression in the Hippocampus of the APPswePS1dE9 Alzheimer's Disease Model Mice.
Evidence of Tau Hyperphosphorylation and Dystrophic Microglia in the Common Marmoset.
Magnetic immunoassay platform based on the planar frequency mixing magnetic technique.
The Transient Receptor Potential Melastatin 2 (TRPM2) Channel Contributes to β-Amyloid Oligomer-Related Neurotoxicity and Memory Impairment.
Closed head injury in an age-related Alzheimer mouse model leads to an altered neuroinflammatory response and persistent cognitive impairment.
Neuropathological and biochemical assessments of an Alzheimer's disease patient treated with the γ-secretase inhibitor semagacestat.
Synaptic and cognitive improvements by inhibition of 2-AG metabolism are through upregulation of microRNA-188-3p in a mouse model of Alzheimer's disease.
Increased mtDNA mutations with aging promotes amyloid accumulation and brain atrophy in the APP/Ld transgenic mouse model of Alzheimer's disease.
Genetic suppression of transgenic APP rescues Hypersynchronous network activity in a mouse model of Alzeimer's disease.
Combination therapy in a transgenic model of Alzheimer's disease.
Clinical and neuropathological findings in a patient with familial Alzheimer disease showing a mutation in the PSEN1 gene.
Amyloid precursor protein (APP) contributes to pathology in the SOD1(G93A) mouse model of amyotrophic lateral sclerosis.
Intraneuronal Aβ detection in 5xFAD mice by a new Aβ-specific antibody.
Hyperphosphorylated tau in young and middle-aged subjects.
Abnormal accumulation of autophagic vesicles correlates with axonal and synaptic pathology in young Alzheimer's mice hippocampus.
Histochemical localization of caldesmon in the CNS and ganglia of the mouse.
Locus ceruleus controls Alzheimer's disease pathology by modulating microglial functions through norepinephrine.
The actin-binding protein caldesmon is in spleen and lymph nodes predominately expressed by smooth-muscle cells, reticular cells, and follicular dendritic cells.
Amyloid-beta42 is preferentially accumulated in muscle fibers of patients with sporadic inclusion-body myositis.
Osteopetrotic (op/op) mice have reduced microglia, no Abeta deposition, and no changes in dopaminergic neurons.
Upregulation of tPA/plasminogen proteolytic system in the periphery of amyloid deposits in the Tg2576 mouse model of Alzheimer's disease.
Intraneuronal Abeta immunoreactivity is not a predictor of brain amyloidosis-beta or neurofibrillary degeneration.
Intraneuronal beta-amyloid aggregates, neurodegeneration, and neuron loss in transgenic mice with five familial Alzheimer's disease mutations: potential factors in amyloid plaque formation.
Locus ceruleus degeneration promotes Alzheimer pathogenesis in amyloid precursor protein 23 transgenic mice.
Focal glial activation coincides with increased BACE1 activation and precedes amyloid plaque deposition in APP[V717I] transgenic mice.
Acute treatment with the PPARgamma agonist pioglitazone and ibuprofen reduces glial inflammation and Abeta1-42 levels in APPV717I transgenic mice.
Decreased beta-amyloid1-42 and increased tau levels in cerebrospinal fluid of patients with Alzheimer disease.
A pathogenic presenilin-1 deletion causes abberrant Abeta 42 production in the absence of congophilic amyloid plaques.
Pilozzi A, Yu Z, Carreras I, Cormier K, Hartley D, Rogers J, Dedeoglu A, Huang X
Biomolecules 2020 Mar 6;10(3)
Biomolecules 2020 Mar 6;10(3)
Oral Immunization with Soybean Storage Protein Containing Amyloid-β 4-10 Prevents Spatial Learning Decline.
Kawarabayashi T, Terakawa T, Takahashi A, Hasegawa H, Narita S, Sato K, Nakamura T, Seino Y, Hirohata M, Baba N, Ueda T, Harigaya Y, Kametani F, Maruyama N, Ishimoto M, St George-Hyslop P, Shoji M
Journal of Alzheimer's disease : JAD 2019;70(2):487-503
Journal of Alzheimer's disease : JAD 2019;70(2):487-503
Beta-amyloid induces apoptosis of neuronal cells by inhibition of the Arg/N-end rule pathway proteolytic activity.
Kechko OI, Petrushanko IY, Brower CS, Adzhubei AA, Moskalev AA, Piatkov KI, Mitkevich VA, Makarov AA
Aging 2019 Aug 24;11(16):6134-6152
Aging 2019 Aug 24;11(16):6134-6152
Alleviation of Neuropathology by Inhibition of Monoacylglycerol Lipase in APP Transgenic Mice Lacking CB2 Receptors.
Zhang J, Chen C
Molecular neurobiology 2018 Jun;55(6):4802-4810
Molecular neurobiology 2018 Jun;55(6):4802-4810
Protective effects of 7,8-dihydroxyflavone on neuropathological and neurochemical changes in a mouse model of Alzheimer's disease.
Aytan N, Choi JK, Carreras I, Crabtree L, Nguyen B, Lehar M, Blusztajn JK, Jenkins BG, Dedeoglu A
European journal of pharmacology 2018 Jun 5;828:9-17
European journal of pharmacology 2018 Jun 5;828:9-17
The Bri2 and Bri3 BRICHOS Domains Interact Differently with Aβ(42) and Alzheimer Amyloid Plaques.
Dolfe L, Tambaro S, Tigro H, Del Campo M, Hoozemans JJM, Wiehager B, Graff C, Winblad B, Ankarcrona M, Kaldmäe M, Teunissen CE, Rönnbäck A, Johansson J, Presto J
Journal of Alzheimer's disease reports 2018 Feb 16;2(1):27-39
Journal of Alzheimer's disease reports 2018 Feb 16;2(1):27-39
Comparative Analysis of Cortical Microinfarcts and Microbleeds using 3.0-Tesla Postmortem Magnetic Resonance Images and Histopathology.
Niwa A, Ii Y, Shindo A, Matsuo K, Ishikawa H, Taniguchi A, Takase S, Maeda M, Sakuma H, Akatsu H, Hashizume Y, Tomimoto H
Journal of Alzheimer's disease : JAD 2017;59(3):951-959
Journal of Alzheimer's disease : JAD 2017;59(3):951-959
Perinatal Choline Supplementation Reduces Amyloidosis and Increases Choline Acetyltransferase Expression in the Hippocampus of the APPswePS1dE9 Alzheimer's Disease Model Mice.
Mellott TJ, Huleatt OM, Shade BN, Pender SM, Liu YB, Slack BE, Blusztajn JK
PloS one 2017;12(1):e0170450
PloS one 2017;12(1):e0170450
Evidence of Tau Hyperphosphorylation and Dystrophic Microglia in the Common Marmoset.
Rodriguez-Callejas JD, Fuchs E, Perez-Cruz C
Frontiers in aging neuroscience 2016;8:315
Frontiers in aging neuroscience 2016;8:315
Magnetic immunoassay platform based on the planar frequency mixing magnetic technique.
Kim CB, Lim EG, Shin SW, Krause HJ, Hong H
Biosensors & bioelectronics 2016 Sep 15;83:293-9
Biosensors & bioelectronics 2016 Sep 15;83:293-9
The Transient Receptor Potential Melastatin 2 (TRPM2) Channel Contributes to β-Amyloid Oligomer-Related Neurotoxicity and Memory Impairment.
Ostapchenko VG, Chen M, Guzman MS, Xie YF, Lavine N, Fan J, Beraldo FH, Martyn AC, Belrose JC, Mori Y, MacDonald JF, Prado VF, Prado MA, Jackson MF
The Journal of neuroscience : the official journal of the Society for Neuroscience 2015 Nov 11;35(45):15157-69
The Journal of neuroscience : the official journal of the Society for Neuroscience 2015 Nov 11;35(45):15157-69
Closed head injury in an age-related Alzheimer mouse model leads to an altered neuroinflammatory response and persistent cognitive impairment.
Webster SJ, Van Eldik LJ, Watterson DM, Bachstetter AD
The Journal of neuroscience : the official journal of the Society for Neuroscience 2015 Apr 22;35(16):6554-69
The Journal of neuroscience : the official journal of the Society for Neuroscience 2015 Apr 22;35(16):6554-69
Neuropathological and biochemical assessments of an Alzheimer's disease patient treated with the γ-secretase inhibitor semagacestat.
Roher AE, Maarouf CL, Kokjohn TA, Whiteside CM, Kalback WM, Serrano G, Belden C, Liebsack C, Jacobson SA, Sabbagh MN, Beach TG
American journal of neurodegenerative disease 2014;3(3):115-33
American journal of neurodegenerative disease 2014;3(3):115-33
Synaptic and cognitive improvements by inhibition of 2-AG metabolism are through upregulation of microRNA-188-3p in a mouse model of Alzheimer's disease.
Zhang J, Hu M, Teng Z, Tang YP, Chen C
The Journal of neuroscience : the official journal of the Society for Neuroscience 2014 Nov 5;34(45):14919-33
The Journal of neuroscience : the official journal of the Society for Neuroscience 2014 Nov 5;34(45):14919-33
Increased mtDNA mutations with aging promotes amyloid accumulation and brain atrophy in the APP/Ld transgenic mouse model of Alzheimer's disease.
Kukreja L, Kujoth GC, Prolla TA, Van Leuven F, Vassar R
Molecular neurodegeneration 2014 May 2;9:16
Molecular neurodegeneration 2014 May 2;9:16
Genetic suppression of transgenic APP rescues Hypersynchronous network activity in a mouse model of Alzeimer's disease.
Born HA, Kim JY, Savjani RR, Das P, Dabaghian YA, Guo Q, Yoo JW, Schuler DR, Cirrito JR, Zheng H, Golde TE, Noebels JL, Jankowsky JL
The Journal of neuroscience : the official journal of the Society for Neuroscience 2014 Mar 12;34(11):3826-40
The Journal of neuroscience : the official journal of the Society for Neuroscience 2014 Mar 12;34(11):3826-40
Combination therapy in a transgenic model of Alzheimer's disease.
Aytan N, Choi JK, Carreras I, Kowall NW, Jenkins BG, Dedeoglu A
Experimental neurology 2013 Dec;250:228-238
Experimental neurology 2013 Dec;250:228-238
Clinical and neuropathological findings in a patient with familial Alzheimer disease showing a mutation in the PSEN1 gene.
Niwa A, Matsuo K, Shindo A, Yata K, Shiraishi T, Tomimoto H
Neuropathology : official journal of the Japanese Society of Neuropathology 2013 Apr;33(2):199-203
Neuropathology : official journal of the Japanese Society of Neuropathology 2013 Apr;33(2):199-203
Amyloid precursor protein (APP) contributes to pathology in the SOD1(G93A) mouse model of amyotrophic lateral sclerosis.
Bryson JB, Hobbs C, Parsons MJ, Bosch KD, Pandraud A, Walsh FS, Doherty P, Greensmith L
Human molecular genetics 2012 Sep 1;21(17):3871-82
Human molecular genetics 2012 Sep 1;21(17):3871-82
Intraneuronal Aβ detection in 5xFAD mice by a new Aβ-specific antibody.
Youmans KL, Tai LM, Kanekiyo T, Stine WB Jr, Michon SC, Nwabuisi-Heath E, Manelli AM, Fu Y, Riordan S, Eimer WA, Binder L, Bu G, Yu C, Hartley DM, LaDu MJ
Molecular neurodegeneration 2012 Mar 16;7:8
Molecular neurodegeneration 2012 Mar 16;7:8
Hyperphosphorylated tau in young and middle-aged subjects.
Elobeid A, Soininen H, Alafuzoff I
Acta neuropathologica 2012 Jan;123(1):97-104
Acta neuropathologica 2012 Jan;123(1):97-104
Abnormal accumulation of autophagic vesicles correlates with axonal and synaptic pathology in young Alzheimer's mice hippocampus.
Sanchez-Varo R, Trujillo-Estrada L, Sanchez-Mejias E, Torres M, Baglietto-Vargas D, Moreno-Gonzalez I, De Castro V, Jimenez S, Ruano D, Vizuete M, Davila JC, Garcia-Verdugo JM, Jimenez AJ, Vitorica J, Gutierrez A
Acta neuropathologica 2012 Jan;123(1):53-70
Acta neuropathologica 2012 Jan;123(1):53-70
Histochemical localization of caldesmon in the CNS and ganglia of the mouse.
Köhler CN
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2011 May;59(5):504-17
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2011 May;59(5):504-17
Locus ceruleus controls Alzheimer's disease pathology by modulating microglial functions through norepinephrine.
Heneka MT, Nadrigny F, Regen T, Martinez-Hernandez A, Dumitrescu-Ozimek L, Terwel D, Jardanhazi-Kurutz D, Walter J, Kirchhoff F, Hanisch UK, Kummer MP
Proceedings of the National Academy of Sciences of the United States of America 2010 Mar 30;107(13):6058-63
Proceedings of the National Academy of Sciences of the United States of America 2010 Mar 30;107(13):6058-63
The actin-binding protein caldesmon is in spleen and lymph nodes predominately expressed by smooth-muscle cells, reticular cells, and follicular dendritic cells.
Köhler CN
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2010 Feb;58(2):183-93
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2010 Feb;58(2):183-93
Amyloid-beta42 is preferentially accumulated in muscle fibers of patients with sporadic inclusion-body myositis.
Vattemi G, Nogalska A, King Engel W, D'Agostino C, Checler F, Askanas V
Acta neuropathologica 2009 May;117(5):569-74
Acta neuropathologica 2009 May;117(5):569-74
Osteopetrotic (op/op) mice have reduced microglia, no Abeta deposition, and no changes in dopaminergic neurons.
Kondo Y, Lemere CA, Seabrook TJ
Journal of neuroinflammation 2007 Dec 20;4:31
Journal of neuroinflammation 2007 Dec 20;4:31
Upregulation of tPA/plasminogen proteolytic system in the periphery of amyloid deposits in the Tg2576 mouse model of Alzheimer's disease.
Lee JY, Kweon HS, Cho E, Lee JY, Byun HR, Kim DH, Kim YH, Han PL, Koh JY
Neuroscience letters 2007 Aug 9;423(1):82-7
Neuroscience letters 2007 Aug 9;423(1):82-7
Intraneuronal Abeta immunoreactivity is not a predictor of brain amyloidosis-beta or neurofibrillary degeneration.
Wegiel J, Kuchna I, Nowicki K, Frackowiak J, Mazur-Kolecka B, Imaki H, Wegiel J, Mehta PD, Silverman WP, Reisberg B, Deleon M, Wisniewski T, Pirttilla T, Frey H, Lehtimäki T, Kivimäki T, Visser FE, Kamphorst W, Potempska A, Bolton D, Currie JR, Miller DL
Acta neuropathologica 2007 Apr;113(4):389-402
Acta neuropathologica 2007 Apr;113(4):389-402
Intraneuronal beta-amyloid aggregates, neurodegeneration, and neuron loss in transgenic mice with five familial Alzheimer's disease mutations: potential factors in amyloid plaque formation.
Oakley H, Cole SL, Logan S, Maus E, Shao P, Craft J, Guillozet-Bongaarts A, Ohno M, Disterhoft J, Van Eldik L, Berry R, Vassar R
The Journal of neuroscience : the official journal of the Society for Neuroscience 2006 Oct 4;26(40):10129-40
The Journal of neuroscience : the official journal of the Society for Neuroscience 2006 Oct 4;26(40):10129-40
Locus ceruleus degeneration promotes Alzheimer pathogenesis in amyloid precursor protein 23 transgenic mice.
Heneka MT, Ramanathan M, Jacobs AH, Dumitrescu-Ozimek L, Bilkei-Gorzo A, Debeir T, Sastre M, Galldiks N, Zimmer A, Hoehn M, Heiss WD, Klockgether T, Staufenbiel M
The Journal of neuroscience : the official journal of the Society for Neuroscience 2006 Feb 1;26(5):1343-54
The Journal of neuroscience : the official journal of the Society for Neuroscience 2006 Feb 1;26(5):1343-54
Focal glial activation coincides with increased BACE1 activation and precedes amyloid plaque deposition in APP[V717I] transgenic mice.
Heneka MT, Sastre M, Dumitrescu-Ozimek L, Dewachter I, Walter J, Klockgether T, Van Leuven F
Journal of neuroinflammation 2005 Oct 7;2:22
Journal of neuroinflammation 2005 Oct 7;2:22
Acute treatment with the PPARgamma agonist pioglitazone and ibuprofen reduces glial inflammation and Abeta1-42 levels in APPV717I transgenic mice.
Heneka MT, Sastre M, Dumitrescu-Ozimek L, Hanke A, Dewachter I, Kuiperi C, O'Banion K, Klockgether T, Van Leuven F, Landreth GE
Brain : a journal of neurology 2005 Jun;128(Pt 6):1442-53
Brain : a journal of neurology 2005 Jun;128(Pt 6):1442-53
Decreased beta-amyloid1-42 and increased tau levels in cerebrospinal fluid of patients with Alzheimer disease.
Sunderland T, Linker G, Mirza N, Putnam KT, Friedman DL, Kimmel LH, Bergeson J, Manetti GJ, Zimmermann M, Tang B, Bartko JJ, Cohen RM
JAMA 2003 Apr 23-30;289(16):2094-103
JAMA 2003 Apr 23-30;289(16):2094-103
A pathogenic presenilin-1 deletion causes abberrant Abeta 42 production in the absence of congophilic amyloid plaques.
Steiner H, Revesz T, Neumann M, Romig H, Grim MG, Pesold B, Kretzschmar HA, Hardy J, Holton JL, Baumeister R, Houlden H, Haass C
The Journal of biological chemistry 2001 Mar 9;276(10):7233-9
The Journal of biological chemistry 2001 Mar 9;276(10):7233-9
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
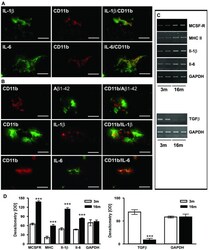
- Figure 2 Characterisation of microglial inflammation. ( A ) Representative confocal immunohistochemistry of APP [V717I] mice revealed that Il-1beta and Il-6 colocalized with activated CD11b-positive microglial cells at 3 month. ( B ) At 16 month CD11b positive cells were almost exculsively detected in close proximity to Abeta1-42 positive plaques. At this time point, CD11b-positive and plaque associated microglia were also found to be colocalized with Il-1beta and Il-6. ( C ) RT-PCR analysis was performed with frontal cortex brain lysates and is being displayed from two single animals at each age (3 and 16 month) for Il-1beta, Il-6, MCSF-R, MHCII and TGFbeta-1 and showed increased gene transcription at 16 months. ( D ) Densitometry of PCR products of APP [V717I] mice at 3 (open bars) and 16 months (filled bars) for the indicated inflammatory molecule. RT-PCR for GAPDH served as control. (n = 6, ANOVA followed by a TUKEY test, ***p < 0.001. Bar graphs in A-B are = 25 mum).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
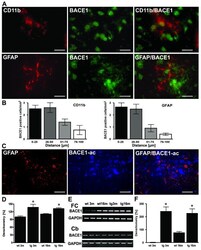
- Figure 4 Sites of focal and early inflammation show BACE1 upregulation in neurons. ( A ) Representative confocal immunostaining of CD11b positive microglia and BACE1 and GFAP and BACE1 in 3 month old APP transgenics showed that BACE positive neurons were found close to focally activated microglia cells in 3 month old APP [V717I] mice. ( B ) Quantitation of the number of BACE1 positive cells in relation to the distance to CD11b or GFAP positive cells. ( C ) Representative image of focal GFAP expression, BACE1 activity and overlay in APP [V717I] mice at 3 m of age. ( D ) Measurement of BACE1-activity was calculated as percentage of 3 month old controls (wt 3 m) and showed that enzyme activity was already elevated in APP [V717I] mice at 3 month (tg 3 m) (n = 5, ANOVA followed by a TUKEY test, *p < 0.05). ( E ) RT-PCR detection of BACE1 mRNA levels of cortical (frontal cortex, FC) and cerebellar (Cb) lysates from wild type controls (wt), APPV [7171I] (tg) mice at 3 (3 m) and 16 months (16 m). ( F ) Densitometrical analysis and quantitation of BACE1 mRNA levels of frontal cortex lysates of APP [V717I] transgenic and controls at the respective age (n = 6, ANOVA followed by a TUKEY test, *p < 0.05). Bar graphs are = 50 mum for CD11b/neuN and GFAP/BACE and = 25 mum for CD11b/BACE1).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
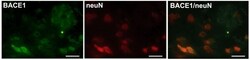
- Figure 5 Expression of BACE1 in neurons in APP [V717I] transgenic mice at 3 month of age. Representative confocal immunostaining of BACE1 positive cells and neuN positive neurons in the cortex of 3 month old APP [V717I] mice. Bar graphs are = 50 mum for BACE1, neuN and BACE1/neuN.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
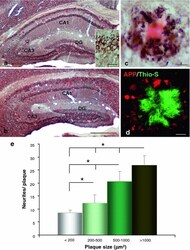
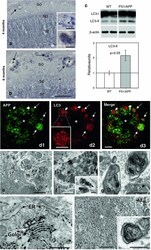
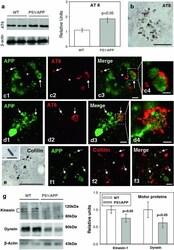
- Fig. 1 Early plaque-associated neuritic dystrophy pathology in PS1/APP hippocampus. a and b APP-immunolabeled sections counterstained with Congo red for fibrillar amyloid deposits at 4 ( a ) and 6 ( b ) months of age showing the early occurrence of the neuritic pathology. The number of neuritic plaques progressively increases with age. APP-positive dystrophic neurites arise from glutamatergic neurons since the human APP transgene is exclusively expressed by principal neurons as shown in the inset . c A representative neuritic plaque formed by a core of congophilic fibrillar amyloid surrounded by numerous APP-positive dystrophic neurites. d Confocal image showing APP-positive dystrophic neurites ( red ) around a plaque stained with thioflavin-S ( green ). e Stereological quantification of the dystrophic neurites around plaques. The number of dystrophies/plaque increased with the size of plaque (mum 2 ). Data are expressed as mean +- SD, * p < 0.05. Scale bars a and b 500 mum, inset 25 mum, c and d 10 mum. CA1 - CA3 subfields of the hippocampus proper, DG dentate gyrus
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 3 Autophagic compartments within dystrophic neurites are sites of APP processing. a and b Immunohistochemistry for the autophagosome marker LC3 in CA1 subfield of PS1/APP mice hippocampus. At 4 months ( a ), immunoreactivity was concentrated in pyramidal cell somata (stratum pyramidale) and their apical dendrites (stratum radiatum). LC3-positive dystrophic neurites were surrounded amyloid plaques ( upper inset in a ) and were punctate in nature (the lower inset shows a high magnification image of the LC3-positive dystrophy circled in the upper inset ). At 6 months ( b ), the immunoreaction was mainly associated with dystrophic neurites around plaques ( arrows ) as shown here for stratum oriens ( SO ). c Quantitative immunoblot analysis of LC3-II form in the hippocampus of 6-month-old PS1/APP and WT mice. PS1/APP mice had significantly higher LC3-II levels than age-matched WT mice ( n = 6, p < 0.05). d1 - d3 Confocal images of double immunofluorescence labeling for APP ( green ) and LC3 ( red ); APP-positive dystrophic neurites around an amyloid plaque ( asterisk ) displayed the LC3 marker ( long arrows ); some LC3-positive dystrophic neurites were negative for APP ( short arrows ) indicating the non-glutamatergic origin of these aberrant neurites. e Dystrophic neurites ( encircled ) around amyloid plaques ( asterisk ) were APP immunopositive as revealed by electron microscopy using silver-enhanced immunogold labeling. f Higher magnification of a dystrophic neurite immuno
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
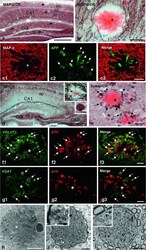
- Fig. 4 Dystrophic neurites are of axonal origin. a MAP-2 (dendritic marker) and Congo red staining in the hippocampus of 6-month-old PS1/APP mice ( arrows indicate amyloid plaques). b No dystrophic neurites immunopositive for MAP-2 were seen around amyloid plaques, as illustrated here for a Congo red stained amyloid plaque ( asterisk ) embedded in a MAP2-positive neuropil in the radiatum layer. c1 - c3 Confocal double immunofluorescence labeling for MAP-2 ( red ) and APP ( green ) revealed no colocalization of the two markers ( asterisk indicates an amyloid plaque). d Strong overlapping in the location of hippocampal axonal pathways and amyloid plaques as shown here using neurofilament (NF) immunolabeling and Congo red staining. Axonal dystrophies were observed around congophilic plaques as shown in the larger inset , and a detail of an axonal swelling is shown in the smaller inset. e Neuritic dystrophies around plaques ( asterisk ) were immunopositive for synaptophysin ( arrows ). f1 - f3 Confocal laser double immunofluorescence labeling for APP ( red ) and VGLUT1 ( green ) shows extensive colocalization ( arrows ) of the two markers indicating the axonal glutamatergic nature of APP-positive dystrophic neurites around plaques ( asterisk ). g1 - g3 Double-label immunofluorescence confocal laser microscopy for APP ( red ) and VGAT ( green ) shows the existence of GABAergic axonal dystrophies (VGAT-positive and APP-negative; arrows ) around amyloid plaques ( asterisk ). h-j Tra
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
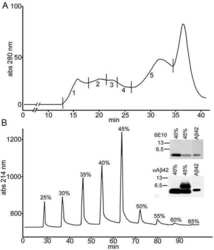
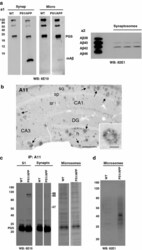
- Fig. 7 Monomeric Abeta accumulates in isolated synaptosomes whereas Abeta oligomers are present in the plaque periphery and microsomes. a The Abeta content were analyzed by 6E10 immunoprecipitation and Western blots (16%-Tris-Tricine-SDS-PAGE) in synaptosomal and microsomal fractions from 6-month-old WT and PS1/APP mice ( a1 ) or by 82E1 Western blots after urea-SDS-PAGE (16%) in PS1/APP synaptosomal fractions ( a2 ). As shown, the monomeric Abeta was predominantly enriched in the synaptosomal fractions ( a1 ). From the different Abeta peptides, the Abeta42 was the predominant form found in synaptosomes ( a2 ). b Oligomeric Abeta peptides were localized at the periphery of the plaques by immunohistochemistry with the A11 antibody. Inset shows a higher magnification of an immunostained amyloid plaque. c The presence of oligomeric Abeta in extracellular soluble fractions ( S1 ), synaptosomes and microsomes was assessed by immunoprecipitation experiments, using the A11 antibody, and Western blots (6E10) using 12%-Tris-Glycine-SDS-PAGE. In agreement with a putative plaque origin, Abeta oligomers (57, 90 and 96 kDa) were identified in S1 fractions. d Representative Western blot of WT and PS1/APP microsomal fractions using the mAb 82E1. As shown, Abeta oligomers (ranging Mr 30-60 kDa) were clearly identified using this antibody. CA1 - CA3 subfields of the hippocampus proper, DG dentate gyrus. Scale bars b 250 mum ( inset 25 mum)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
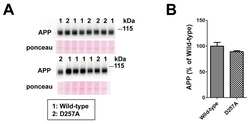
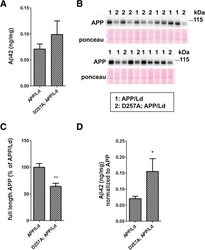
- Figure 1 PolgA D257A mutation increases normalized Abeta42 levels in APP / Ld mice. (A) Hemi-brain homogenates of ~12 month-old APP/Ld and D257A; APP/Ld mice were prepared and Abeta42 levels were measured using a human Abeta42-specific ELISA. Values are expressed in nanograms per milligram of total brain protein. Note that the raw Abeta42 levels are not significantly elevated in D257A; APP/Ld mice (n = 10), but that an upward trend exists, as compared to APP/Ld mice (n = 12). (B) 15 mug/lane of the same mouse brain homogenates in (A) were used for immunoblot analysis of full-length transgenic human APP/Ld protein using the anti-human APP monoclonal antibody 6E10. Ponceau S staining was used as a loading control. The numbers on immunoblot correspond to specific genotypes (as denoted in boxed legend key) of individual brain homogenates that were randomly loaded. (C) Full-length APP/Ld immunosignals in (B) were quantified by phosphorimager, normalized to Ponceau S staining intensities per lane, and expressed as percentage of the mean APP/Ld immunosignal. Note that levels of APP/Ld are significantly reduced in the D257A; APP/Ld mice compared to APP/Ld mice (mean +- SEM; **p < 0.01; Student's t-test). (D) Raw Abeta42 ELISA values (ng/mg) from (A) were normalized to the mean transgenic APP/Ld protein levels for each genotype as determined in (C) . Note that Abeta42 levels normalized to APP/Ld are significantly increased in the D257A; APP/Ld mice compared to APP/Ld mice (mean +
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
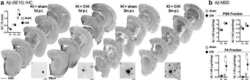
- Figure 2 PolgA D257A mutation increases amyloid plaque pathology in APP / Ld mouse brains. (A) Hemi-brains of the same mice from Figure 1 were fixed, sectioned coronally, and co-stained with anti-Abeta42 specific antibody (green) and thiazine red for beta-sheet amyloid and imaged by immunofluorescence microscopy. (A) Representative coronal brain sections of APP/Ld (left) and D257A; APP/Ld mice (right). Both APP/Ld and D257A; APP/Ld mice exhibit diffuse (green box) and dense-core (red box) plaques. The insets show high-magnification images of plaques in green and red boxes. Dense-core plaques are positive for both Abeta42 and thiazine red, while diffuse plaques are only Abeta42-immunopositive. In addition to the cortex, D257A; APP/Ld mice form plaques in other brain regions such as the thalamus (for example see red arrow) Blue = DAPI. Scale bar: 500 mum (25 mum in insets). (B) The amyloid plaque density (number of Abeta42-positive plaques per cm 2 ) was determined from representative brain sections with equivalent rostral-caudal location from D257A; APP/Ld (n = 10) and APP/Ld (n = 12) mice. (C) Plaque densities in (B) were normalized to human transgenic APP protein levels as determined by APP immunoblot analysis (Figure 1 C). Note that normalized Abeta42 plaque density is significantly increased in D257A; APP/Ld mice compared to APP/Ld mice (mean +- SEM; *p < 0.05; Student's t-test).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
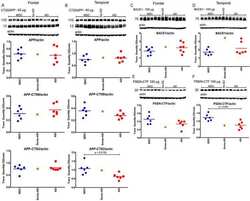
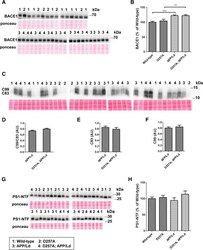
- Figure 3 PolgA D257A mutation does not affect levels of Abeta generating enzymes in APP/Ld mice. 15 mug of brain homogenates from D257A; APP/Ld, D257A, APP/Ld, and wild type mice were loaded per lane and subjected to immunoblot analysis. Immunoblot signals were normalized to the Ponceau S staining intensity for a given lane. The numbers on the immunoblot correspond to mice with specific genotypes as denoted in the legend key (box). (A) BACE1 protein runs at ~68 kDa. (B) BACE1 immunosignals in (A) were expressed as the percentage of the mean wild-type mouse BACE1 immunosignal. Note that BACE1 levels are significantly higher in APP/Ld monogenic and D257A; APP/Ld bigenic mice compared to D257A and wild type non-transgenic mice. BACE1 levels are not altered by the presence of the D257A mutation (mean +- SEM; **p < 0.01; ***p < 0.001; One-way ANOVA followed by Newman-Keuls multiple-comparisons post hoc test). (C) APP processing was determined by measuring levels of both APP C-terminal fragments (C83 & C99) via immunoblot using a rabbit monoclonal antibody that recognizes the C-terminus of APP. Note however, that these C-terminal fragments were below the level of immunoblot detection in non-transgenic APP mouse brains. (D) The ratio of C99:C83, which is directly proportional to the fraction of APP cleaved by BACE1, is not altered by the presence of the D257A mutation. Separate quantification of C83 (E) and C99 (F) , normalized to Ponceau S, also show that alpha- and beta-secretase
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 5 Cytoskeletal pathology in young PS1/APP hippocampus. a Quantitative Western blotting using AT8 antibody in the hippocampus of 6-month-old PS1/APP and WT mice; PS1/APP mice had significantly higher AT8 levels than to age-matched WT mice ( n = 6, p < 0.05). b The presence of AT8-positive structures around plaques ( asterisk ) was observed from 4 months of age. c1 - c4 and d1 - d4. Double-label immunofluorescence confocal laser microscopy of APP ( green ) and AT8 ( red ) in 6-month-old transgenic mice showing the presence of phospho-tau in some APP-positive dystrophic neurites. e Immunohistochemistry for cofilin revealed the presence of actin-cofilin rods around plaques ( asterisk ) from 4 months of age. A higher magnification detail of a rod is shown in the inset . f1 - f3 Confocal laser microscopy with double APP/cofilin labeling revealed the presence of cofilin within some APP-positive neurites. g Quantitative Western blotting for heavy chain kinesin-1 and dynein in 6-month-old PS1/APP and WT mice hippocampus. PS1/APP mice were found to have significantly lower kinesin-1 and dynein levels than age-matched WT mice ( n = 8, p < 0.05). Scale bars b 100 mum, c1 - c3 5 mum, c4 2.5 mum, d1 - d3 7.5 mum, d4 2.5 mum, e 50 mum ( inset 7 mum), f1 - f3 7.5 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
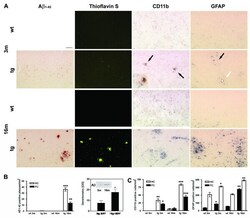
- Figure 1 Comparison of Abeta deposition, micro- and astroglial activation. ( A ) Representative detection of Abeta1-42 immunostaining, Thioflavin-S histochemistry, microglial (CD11b) and astroglial activation (GFAP) in APP [V717I] mice and non-transgenic controls of the identical genetic background at 3 (3 m) and 16 (16 m) months (Bar graph = 50 mum (Abeta1-42, Thioflavin-S), = 25 mum (CD11b, GFAP)) Focal microglial activation is indicated by black arrows. Focal astroglial activation within the parenchyma by black arrows and at the side of of a brain vessel by a white arrow ( B ). Quantification of hippocampal (HC, open bar) and cortical (FC, filled bar) Abeta1-42-positive plaques of APP [V717I] mice at 3 and 16 months (tg 3 m, tg 16 m) (n = 12, ANOVA followed by a TUKEY test, **p < 0.01, ***p < 0.001.) and total Abeta detection by immunoprecipitation/western blot and subsequent quantification by densitometry (n = 3, Students t-test, *p < 0.05). ( C ) Quantification of CD11b positive, activated microglia (see insert, arrows) and GFAP positive astrocytes in the hippocampus (HC, open bar) and frontal cortex (FC, filled bars) (n = 12, ANOVA followed by a TUKEY test, *p < 0.05, **p < 0.01,***p < 0.001).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
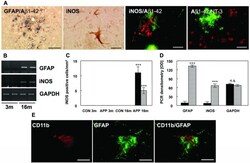
- Figure 3 Astrocytic iNOS expression and plaque associated nitrotyrosine. ( A ) Costaining of Abeta1-42 and GFAP at 16 months detected activated astrocytes nearby Abeta plaques. Astrocytic iNOS and confocal staining of iNOS (red) and Abeta1-42 (green) or nitrotyrosine (red) and Abeta1-42 (green). ( B ) RT-PCR for GFAP and iNOS in APP [V717I] mice at 3 (3 m) and 16 months (16 m) of age. ( C ) Quantification of iNOS-positive astrocytes in the hippocampus (HC, black bar) and frontal cortex (FC, hatched bars) of APP transgenic mice (tg) and wild type controls (wt) at 3 and 16 months. ( D ) Densitometry of GFAP, iNOS and GAPDH mRNA from APPV [7171I] mice at 3 (black bars) and 16 months (hatched bars). ( E ) Confocal staining of CD11b positive microglia and GFAP labelled astrocytes showed that both cells were located in close neighbouring in APP [V717I] mice at 3 month of age. (n = 6, ANOVA followed by a TUKEY test, n.s. = non significant, ***p < 0.001). Bar graph = 50 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
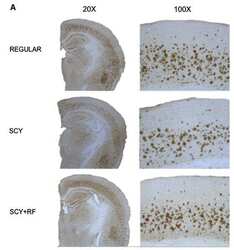
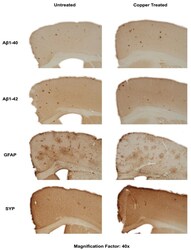
- Figure 3 Dietary Cu exposure enhanced cerebral Abeta amyloid pathology in APP/PS1 transgenic mice. Representative coronal brain sections (at the anterior commissure level) from APP/PS1 transgenic mice on regular and Cu-enhanced diets for 24 days. They were immunostained for Abeta1-42 and Abeta1-40, illustrating the size, number and distribution of Abeta amyloid plaques. The immunostaining for reactive astrogliosis marker- GFAP and negative staining for SYP, a marker for neuronal synaptic activity, were also performed.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
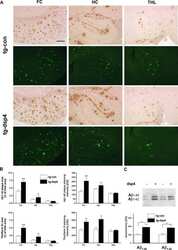
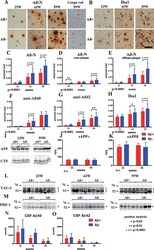
- Fig.6 Staining of Abeta+ or Abeta- treated brains with Abeta-N and Congo red (A), microglial marker Iba1 (B) in Abeta+ or Abeta- treated mouse brains, quantification of Abeta burden (C-G), Iba1 (H), western blot of AbetaPP and CTFs (I), ELISA of sAbetaPP alpha (J), sAbetaPPbeta (K), western blotting of total tau (L), phosphorylated tau (M), CSF Abeta 40 (N), and Abeta 42 (O) measured by ELISA. A) Abeta-immunoreactive load using Abeta-N was suppressed in the Abeta+ treated group compared with that of the Abeta- group. Most Abeta staining showed diffuse plaques that are not stained with Congo red. B) Microgliosis was weaker in the Abeta+ treated group than that in the Abeta- treated group. Bar represents 100 mum in A and 200 mum in B. C) The area labeled by Abeta-N was significantly suppressed in the Abeta+ treated group compared with that in the Abeta- treated group ( p < 0.0001). D, E) The area occupied by diffuse plaques was more than 3-fold of that by core plaques. Both plaques were suppressed by Abeta+ treatment (D, p < 0.0001; E, p < 0.0001). Large part of the Abeta decrease by immunization consisted of diffuse plaques. Abeta burdens detected by anti-Abeta 40 (F) and anti-Abeta 42 (G) were significantly decreased in the Abeta+ treated group (F, p < 0.001; G, p < 0.0001). H) The area of Iba1 staining in Abeta+ treated mice was smaller than that in Abeta- treated mice ( p < 0.0001). The numbers of mice analyzed: 23 weeks ( n = 9 for Abeta+, n = 11 for Abeta-), 43 weeks ( n
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
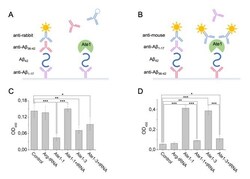
- Figure 3 Interaction of Abeta 42 , tRNA, and Ate1. Schematic representation of ELISA assay with immobilized anti-Abeta1-17 antibodies ( A ) or anti-Abeta36-42 antibodies ( B ). ( C ) Detection of tRNA and Ate1 ability to interact with C-terminus of Abeta. ( D ) Same as (C) but with N-terminal region of Abeta. OD 450 - optical density measured at 450 nm. Each value is the mean +- SD of at least four independent experiments; *p < 0.04, **p < 0.01, ***p < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
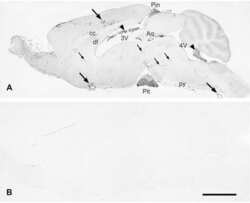
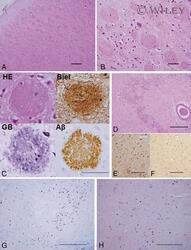
- 1 Photomicrographs showing histopathology (A-D, G and H) and immunohistochemistry of Alzheimer's pathology (C, E and F). Large, round non-cored plaques were densely packed throughout most of the cortical layers in the temporal lobe by HE staining (A-C). With Bielschowsky and modified Gallyas-Braak staining, cotton wool plaques (CWP) were weakly argyrophilic and contained only a few thin neurites (C). CWP were immunopositive with anti-Abeta 42 antibody (C). CWP interrupted and distorted the granular cell layer of the dentate gyrus by HE staining (D). Immunohistochemical staining by both RD3 (E) and (F) revealed positive NFT and neuropil threads (NT). In the substantia nigra, pigmented nerve cells were relatively preserved in KB staining (G), but a moderate number of NFT and NT were seen in modified Gallyas-Braak staining (H). Scale bars in A, D, E and F: 200 um; in B and C: 50 um and in G and H: 500 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
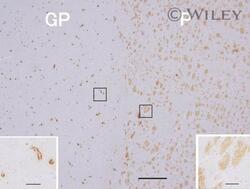
- 3 Photomicrographs showing immunohistochemistry of Abeta 42 in the lenticular nucleus. Putamen (P) contained densely packed diffuse plaques but relatively little capillary CAA, whereas the neighboring lateral segment of the globus pallidus (GP) showed abundant capillary CAA but no plaques. Scale bar: 500 um. The insets show higher-magnification views with scale bars: 50 um.
 Explore
Explore Validate
Validate Learn
Learn ELISA
ELISA Other assay
Other assay