Antibody data
- Antibody Data
- Antigen structure
- References [59]
- Comments [0]
- Validations
- Other assay [36]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 71-5800 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- beta Amyloid Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µg
- Concentration
- 0.25 mg/mL
- Storage
- -20°C
Submitted references Age-related metabolic and neurodegenerative changes in SAMP8 mice.
High-fat diet exacerbates cognitive decline in mouse models of Alzheimer's disease and mixed dementia in a sex-dependent manner.
Microglial transcription profiles in mouse and human are driven by APOE4 and sex.
CD4+ effector T cells accelerate Alzheimer's disease in mice.
Staining and Quantification of β-Amyloid Pathology in Transgenic Mouse Models of Alzheimer's Disease.
Aging Reduces Estradiol Protection Against Neural but Not Metabolic Effects of Obesity in Female 3xTg-AD Mice.
Cystatin C regulates the cytotoxicity of infection-induced endothelial-derived β-amyloid.
Increased transcription of transglutaminase 1 mediates neuronal death in in vitro models of neuronal stress and Aβ1-42-mediated toxicity.
mGluR5 Contribution to Neuropathology in Alzheimer Mice Is Disease Stage-Dependent.
URMC-099 facilitates amyloid-β clearance in a murine model of Alzheimer's disease.
Discrete Pools of Oligomeric Amyloid-β Track with Spatial Learning Deficits in a Mouse Model of Alzheimer Amyloidosis.
Quantification of Butyrylcholinesterase Activity as a Sensitive and Specific Biomarker of Alzheimer's Disease.
Age-dependent regulation of obesity and Alzheimer-related outcomes by hormone therapy in female 3xTg-AD mice.
Obesity Accelerates Alzheimer-Related Pathology in APOE4 but not APOE3 Mice.
Catalytically active tissue transglutaminase colocalises with Aβ pathology in Alzheimer's disease mouse models.
The blood clotting Factor XIIIa forms unique complexes with amyloid-beta (Aβ) and colocalizes with deposited Aβ in cerebral amyloid angiopathy.
Tissue transglutaminase-catalysed cross-linking induces Apolipoprotein E multimers inhibiting Apolipoprotein E's protective effects towards amyloid-beta-induced toxicity.
Prophylactic liraglutide treatment prevents amyloid plaque deposition, chronic inflammation and memory impairment in APP/PS1 mice.
Butyrylcholinesterase-knockout reduces brain deposition of fibrillar β-amyloid in an Alzheimer mouse model.
Early detection of cerebral glucose uptake changes in the 5XFAD mouse.
Lixisenatide, a drug developed to treat type 2 diabetes, shows neuroprotective effects in a mouse model of Alzheimer's disease.
Vascular pathology of 20-month-old hypercholesterolemia mice in comparison to triple-transgenic and APPSwDI Alzheimer's disease mouse models.
Genetic modulation of soluble Aβ rescues cognitive and synaptic impairment in a mouse model of Alzheimer's disease.
Liraglutide can reverse memory impairment, synaptic loss and reduce plaque load in aged APP/PS1 mice, a model of Alzheimer's disease.
A novel retro-inverso peptide inhibitor reduces amyloid deposition, oxidation and inflammation and stimulates neurogenesis in the APPswe/PS1ΔE9 mouse model of Alzheimer's disease.
Neuroprotective effects of D-Ala(2)GIP on Alzheimer's disease biomarkers in an APP/PS1 mouse model.
D-Ala2GIP facilitated synaptic plasticity and reduces plaque load in aged wild type mice and in an Alzheimer's disease mouse model.
Cholinergic degeneration is associated with increased plaque deposition and cognitive impairment in APPswe/PS1dE9 mice.
Tissue transglutaminase colocalizes with extracellular matrix proteins in cerebral amyloid angiopathy.
Protein restriction cycles reduce IGF-1 and phosphorylated Tau, and improve behavioral performance in an Alzheimer's disease mouse model.
Effects of NK-4 in a transgenic mouse model of Alzheimer's disease.
Evaluation of the effects of testosterone and luteinizing hormone on regulation of β-amyloid in male 3xTg-AD mice.
Butyrylcholinesterase is associated with β-amyloid plaques in the transgenic APPSWE/PSEN1dE9 mouse model of Alzheimer disease.
Effects of aromatase inhibition versus gonadectomy on hippocampal complex amyloid pathology in triple transgenic mice.
Val(8)GLP-1 rescues synaptic plasticity and reduces dense core plaques in APP/PS1 mice.
An anti-diabetes agent protects the mouse brain from defective insulin signaling caused by Alzheimer's disease- associated Aβ oligomers.
The effect of ageing on neurogenesis and oxidative stress in the APP(swe)/PS1(deltaE9) mouse model of Alzheimer's disease.
The diabetes drug liraglutide prevents degenerative processes in a mouse model of Alzheimer's disease.
Astroglial connexin immunoreactivity is specifically altered at β-amyloid plaques in β-amyloid precursor protein/presenilin1 mice.
Synaptic plasticity in the hippocampus of a APP/PS1 mouse model of Alzheimer's disease is impaired in old but not young mice.
Continuous and cyclic progesterone differentially interact with estradiol in the regulation of Alzheimer-like pathology in female 3xTransgenic-Alzheimer's disease mice.
Sex differences in β-amyloid accumulation in 3xTg-AD mice: role of neonatal sex steroid hormone exposure.
Receptor-associated protein (RAP) plays a central role in modulating Abeta deposition in APP/PS1 transgenic mice.
Selective estrogen receptor modulators differentially regulate Alzheimer-like changes in female 3xTg-AD mice.
Progesterone and estrogen regulate Alzheimer-like neuropathology in female 3xTg-AD mice.
Rodent A beta modulates the solubility and distribution of amyloid deposits in transgenic mice.
Androgens regulate the development of neuropathology in a triple transgenic mouse model of Alzheimer's disease.
Abeta-related angiitis: primary angiitis of the central nervous system associated with cerebral amyloid angiopathy.
Gradual alteration of mitochondrial structure and function by beta-amyloids: importance of membrane viscosity changes, energy deprivation, reactive oxygen species production, and cytochrome c release.
Glutamate receptor subunit 3 is modified by site-specific limited proteolysis including cleavage by gamma-secretase.
Function of beta-amyloid in cholesterol transport: a lead to neurotoxicity.
Patients with Alzheimer disease have lower levels of serum anti-amyloid peptide antibodies than healthy elderly individuals.
Rapid detection of protein aggregates in the brains of Alzheimer patients and transgenic mouse models of amyloidosis.
Rapid detection of protein aggregates in the brains of Alzheimer patients and transgenic mouse models of amyloidosis.
22R-Hydroxycholesterol protects neuronal cells from beta-amyloid-induced cytotoxicity by binding to beta-amyloid peptide.
Distinct binding sites in the structure of alpha 2-macroglobulin mediate the interaction with beta-amyloid peptide and growth factors.
Sporadic inclusion body myositis correlates with increased expression and cross-linking by transglutaminases 1 and 2.
Sporadic inclusion body myositis correlates with increased expression and cross-linking by transglutaminases 1 and 2.
Amyloid-beta deposition in skeletal muscle of transgenic mice: possible model of inclusion body myopathy.
Pačesová A, Holubová M, Hrubá L, Strnadová V, Neprašová B, Pelantová H, Kuzma M, Železná B, Kuneš J, Maletínská L
Aging 2022 Sep 16;14(18):7300-7327
Aging 2022 Sep 16;14(18):7300-7327
High-fat diet exacerbates cognitive decline in mouse models of Alzheimer's disease and mixed dementia in a sex-dependent manner.
Gannon OJ, Robison LS, Salinero AE, Abi-Ghanem C, Mansour FM, Kelly RD, Tyagi A, Brawley RR, Ogg JD, Zuloaga KL
Journal of neuroinflammation 2022 May 14;19(1):110
Journal of neuroinflammation 2022 May 14;19(1):110
Microglial transcription profiles in mouse and human are driven by APOE4 and sex.
Moser VA, Workman MJ, Hurwitz SJ, Lipman RM, Pike CJ, Svendsen CN
iScience 2021 Nov 19;24(11):103238
iScience 2021 Nov 19;24(11):103238
CD4+ effector T cells accelerate Alzheimer's disease in mice.
Machhi J, Yeapuri P, Lu Y, Foster E, Chikhale R, Herskovitz J, Namminga KL, Olson KE, Abdelmoaty MM, Gao J, Quadros RM, Kiyota T, Jingjing L, Kevadiya BD, Wang X, Liu Y, Poluektova LY, Gurumurthy CB, Mosley RL, Gendelman HE
Journal of neuroinflammation 2021 Nov 19;18(1):272
Journal of neuroinflammation 2021 Nov 19;18(1):272
Staining and Quantification of β-Amyloid Pathology in Transgenic Mouse Models of Alzheimer's Disease.
Christensen A, Pike CJ
Methods in molecular biology (Clifton, N.J.) 2020;2144:211-221
Methods in molecular biology (Clifton, N.J.) 2020;2144:211-221
Aging Reduces Estradiol Protection Against Neural but Not Metabolic Effects of Obesity in Female 3xTg-AD Mice.
Christensen A, Liu J, Pike CJ
Frontiers in aging neuroscience 2020;12:113
Frontiers in aging neuroscience 2020;12:113
Cystatin C regulates the cytotoxicity of infection-induced endothelial-derived β-amyloid.
Balczon R, Morrow KA, Leavesley S, Francis CM, Stevens TC, Agwaramgbo E, Williams C, Stevens RP, Langham G, Voth S, Cioffi EA, Weintraub SE, Stevens T
FEBS open bio 2020 Nov;10(11):2464-2477
FEBS open bio 2020 Nov;10(11):2464-2477
Increased transcription of transglutaminase 1 mediates neuronal death in in vitro models of neuronal stress and Aβ1-42-mediated toxicity.
Tripathy D, Migazzi A, Costa F, Roncador A, Gatto P, Fusco F, Boeri L, Albani D, Juárez-Hernández JL, Musio C, Colombo L, Salmona M, Wilhelmus MMM, Drukarch B, Pennuto M, Basso M
Neurobiology of disease 2020 Jul;140:104849
Neurobiology of disease 2020 Jul;140:104849
mGluR5 Contribution to Neuropathology in Alzheimer Mice Is Disease Stage-Dependent.
Abd-Elrahman KS, Hamilton A, Albaker A, Ferguson SSG
ACS pharmacology & translational science 2020 Apr 10;3(2):334-344
ACS pharmacology & translational science 2020 Apr 10;3(2):334-344
URMC-099 facilitates amyloid-β clearance in a murine model of Alzheimer's disease.
Kiyota T, Machhi J, Lu Y, Dyavarshetty B, Nemati M, Zhang G, Mosley RL, Gelbard HA, Gendelman HE
Journal of neuroinflammation 2018 May 5;15(1):137
Journal of neuroinflammation 2018 May 5;15(1):137
Discrete Pools of Oligomeric Amyloid-β Track with Spatial Learning Deficits in a Mouse Model of Alzheimer Amyloidosis.
Chiang ACA, Fowler SW, Reddy R, Pletnikova O, Troncoso JC, Sherman MA, Lesne SE, Jankowsky JL
The American journal of pathology 2018 Mar;188(3):739-756
The American journal of pathology 2018 Mar;188(3):739-756
Quantification of Butyrylcholinesterase Activity as a Sensitive and Specific Biomarker of Alzheimer's Disease.
Macdonald IR, Maxwell SP, Reid GA, Cash MK, DeBay DR, Darvesh S
Journal of Alzheimer's disease : JAD 2017;58(2):491-505
Journal of Alzheimer's disease : JAD 2017;58(2):491-505
Age-dependent regulation of obesity and Alzheimer-related outcomes by hormone therapy in female 3xTg-AD mice.
Christensen A, Pike CJ
PloS one 2017;12(6):e0178490
PloS one 2017;12(6):e0178490
Obesity Accelerates Alzheimer-Related Pathology in APOE4 but not APOE3 Mice.
Moser VA, Pike CJ
eNeuro 2017 May-Jun;4(3)
eNeuro 2017 May-Jun;4(3)
Catalytically active tissue transglutaminase colocalises with Aβ pathology in Alzheimer's disease mouse models.
Wilhelmus MM, de Jager M, Smit AB, van der Loo RJ, Drukarch B
Scientific reports 2016 Feb 3;6:20569
Scientific reports 2016 Feb 3;6:20569
The blood clotting Factor XIIIa forms unique complexes with amyloid-beta (Aβ) and colocalizes with deposited Aβ in cerebral amyloid angiopathy.
de Jager M, Boot MV, Bol JG, Brevé JJ, Jongenelen CA, Drukarch B, Wilhelmus MM
Neuropathology and applied neurobiology 2016 Apr;42(3):255-72
Neuropathology and applied neurobiology 2016 Apr;42(3):255-72
Tissue transglutaminase-catalysed cross-linking induces Apolipoprotein E multimers inhibiting Apolipoprotein E's protective effects towards amyloid-beta-induced toxicity.
de Jager M, Drukarch B, Hofstee M, Brevé J, Jongenelen CA, Bol JG, Wilhelmus MM
Journal of neurochemistry 2015 Sep;134(6):1116-28
Journal of neurochemistry 2015 Sep;134(6):1116-28
Prophylactic liraglutide treatment prevents amyloid plaque deposition, chronic inflammation and memory impairment in APP/PS1 mice.
McClean PL, Jalewa J, Hölscher C
Behavioural brain research 2015 Oct 15;293:96-106
Behavioural brain research 2015 Oct 15;293:96-106
Butyrylcholinesterase-knockout reduces brain deposition of fibrillar β-amyloid in an Alzheimer mouse model.
Reid GA, Darvesh S
Neuroscience 2015 Jul 9;298:424-35
Neuroscience 2015 Jul 9;298:424-35
Early detection of cerebral glucose uptake changes in the 5XFAD mouse.
Macdonald IR, DeBay DR, Reid GA, O'Leary TP, Jollymore CT, Mawko G, Burrell S, Martin E, Bowen CV, Brown RE, Darvesh S
Current Alzheimer research 2014;11(5):450-60
Current Alzheimer research 2014;11(5):450-60
Lixisenatide, a drug developed to treat type 2 diabetes, shows neuroprotective effects in a mouse model of Alzheimer's disease.
McClean PL, Hölscher C
Neuropharmacology 2014 Nov;86:241-58
Neuropharmacology 2014 Nov;86:241-58
Vascular pathology of 20-month-old hypercholesterolemia mice in comparison to triple-transgenic and APPSwDI Alzheimer's disease mouse models.
Hohsfield LA, Daschil N, Orädd G, Strömberg I, Humpel C
Molecular and cellular neurosciences 2014 Nov;63:83-95
Molecular and cellular neurosciences 2014 Nov;63:83-95
Genetic modulation of soluble Aβ rescues cognitive and synaptic impairment in a mouse model of Alzheimer's disease.
Fowler SW, Chiang AC, Savjani RR, Larson ME, Sherman MA, Schuler DR, Cirrito JR, Lesné SE, Jankowsky JL
The Journal of neuroscience : the official journal of the Society for Neuroscience 2014 Jun 4;34(23):7871-85
The Journal of neuroscience : the official journal of the Society for Neuroscience 2014 Jun 4;34(23):7871-85
Liraglutide can reverse memory impairment, synaptic loss and reduce plaque load in aged APP/PS1 mice, a model of Alzheimer's disease.
McClean PL, Hölscher C
Neuropharmacology 2014 Jan;76 Pt A:57-67
Neuropharmacology 2014 Jan;76 Pt A:57-67
A novel retro-inverso peptide inhibitor reduces amyloid deposition, oxidation and inflammation and stimulates neurogenesis in the APPswe/PS1ΔE9 mouse model of Alzheimer's disease.
Parthsarathy V, McClean PL, Hölscher C, Taylor M, Tinker C, Jones G, Kolosov O, Salvati E, Gregori M, Masserini M, Allsop D
PloS one 2013;8(1):e54769
PloS one 2013;8(1):e54769
Neuroprotective effects of D-Ala(2)GIP on Alzheimer's disease biomarkers in an APP/PS1 mouse model.
Faivre E, Hölscher C
Alzheimer's research & therapy 2013;5(2):20
Alzheimer's research & therapy 2013;5(2):20
D-Ala2GIP facilitated synaptic plasticity and reduces plaque load in aged wild type mice and in an Alzheimer's disease mouse model.
Faivre E, Hölscher C
Journal of Alzheimer's disease : JAD 2013;35(2):267-83
Journal of Alzheimer's disease : JAD 2013;35(2):267-83
Cholinergic degeneration is associated with increased plaque deposition and cognitive impairment in APPswe/PS1dE9 mice.
Laursen B, Mørk A, Plath N, Kristiansen U, Bastlund JF
Behavioural brain research 2013 Mar 1;240:146-52
Behavioural brain research 2013 Mar 1;240:146-52
Tissue transglutaminase colocalizes with extracellular matrix proteins in cerebral amyloid angiopathy.
de Jager M, van der Wildt B, Schul E, Bol JG, van Duinen SG, Drukarch B, Wilhelmus MM
Neurobiology of aging 2013 Apr;34(4):1159-69
Neurobiology of aging 2013 Apr;34(4):1159-69
Protein restriction cycles reduce IGF-1 and phosphorylated Tau, and improve behavioral performance in an Alzheimer's disease mouse model.
Parrella E, Maxim T, Maialetti F, Zhang L, Wan J, Wei M, Cohen P, Fontana L, Longo VD
Aging cell 2013 Apr;12(2):257-68
Aging cell 2013 Apr;12(2):257-68
Effects of NK-4 in a transgenic mouse model of Alzheimer's disease.
Ohta H, Arai S, Akita K, Ohta T, Fukuda S
PloS one 2012;7(1):e30007
PloS one 2012;7(1):e30007
Evaluation of the effects of testosterone and luteinizing hormone on regulation of β-amyloid in male 3xTg-AD mice.
Rosario ER, Carroll JC, Pike CJ
Brain research 2012 Jul 23;1466:137-45
Brain research 2012 Jul 23;1466:137-45
Butyrylcholinesterase is associated with β-amyloid plaques in the transgenic APPSWE/PSEN1dE9 mouse model of Alzheimer disease.
Darvesh S, Cash MK, Reid GA, Martin E, Mitnitski A, Geula C
Journal of neuropathology and experimental neurology 2012 Jan;71(1):2-14
Journal of neuropathology and experimental neurology 2012 Jan;71(1):2-14
Effects of aromatase inhibition versus gonadectomy on hippocampal complex amyloid pathology in triple transgenic mice.
Overk CR, Lu PY, Wang YT, Choi J, Shaw JW, Thatcher GR, Mufson EJ
Neurobiology of disease 2012 Jan;45(1):479-87
Neurobiology of disease 2012 Jan;45(1):479-87
Val(8)GLP-1 rescues synaptic plasticity and reduces dense core plaques in APP/PS1 mice.
Gengler S, McClean PL, McCurtin R, Gault VA, Hölscher C
Neurobiology of aging 2012 Feb;33(2):265-76
Neurobiology of aging 2012 Feb;33(2):265-76
An anti-diabetes agent protects the mouse brain from defective insulin signaling caused by Alzheimer's disease- associated Aβ oligomers.
Bomfim TR, Forny-Germano L, Sathler LB, Brito-Moreira J, Houzel JC, Decker H, Silverman MA, Kazi H, Melo HM, McClean PL, Holscher C, Arnold SE, Talbot K, Klein WL, Munoz DP, Ferreira ST, De Felice FG
The Journal of clinical investigation 2012 Apr;122(4):1339-53
The Journal of clinical investigation 2012 Apr;122(4):1339-53
The effect of ageing on neurogenesis and oxidative stress in the APP(swe)/PS1(deltaE9) mouse model of Alzheimer's disease.
Hamilton A, Holscher C
Brain research 2012 Apr 17;1449:83-93
Brain research 2012 Apr 17;1449:83-93
The diabetes drug liraglutide prevents degenerative processes in a mouse model of Alzheimer's disease.
McClean PL, Parthsarathy V, Faivre E, Hölscher C
The Journal of neuroscience : the official journal of the Society for Neuroscience 2011 Apr 27;31(17):6587-94
The Journal of neuroscience : the official journal of the Society for Neuroscience 2011 Apr 27;31(17):6587-94
Astroglial connexin immunoreactivity is specifically altered at β-amyloid plaques in β-amyloid precursor protein/presenilin1 mice.
Mei X, Ezan P, Giaume C, Koulakoff A
Neuroscience 2010 Nov 24;171(1):92-105
Neuroscience 2010 Nov 24;171(1):92-105
Synaptic plasticity in the hippocampus of a APP/PS1 mouse model of Alzheimer's disease is impaired in old but not young mice.
Gengler S, Hamilton A, Hölscher C
PloS one 2010 Mar 22;5(3):e9764
PloS one 2010 Mar 22;5(3):e9764
Continuous and cyclic progesterone differentially interact with estradiol in the regulation of Alzheimer-like pathology in female 3xTransgenic-Alzheimer's disease mice.
Carroll JC, Rosario ER, Villamagna A, Pike CJ
Endocrinology 2010 Jun;151(6):2713-22
Endocrinology 2010 Jun;151(6):2713-22
Sex differences in β-amyloid accumulation in 3xTg-AD mice: role of neonatal sex steroid hormone exposure.
Carroll JC, Rosario ER, Kreimer S, Villamagna A, Gentzschein E, Stanczyk FZ, Pike CJ
Brain research 2010 Dec 17;1366:233-45
Brain research 2010 Dec 17;1366:233-45
Receptor-associated protein (RAP) plays a central role in modulating Abeta deposition in APP/PS1 transgenic mice.
Xu G, Karch C, Li N, Lin N, Fromholt D, Gonzales V, Borchelt DR
PloS one 2008 Sep 8;3(9):e3159
PloS one 2008 Sep 8;3(9):e3159
Selective estrogen receptor modulators differentially regulate Alzheimer-like changes in female 3xTg-AD mice.
Carroll JC, Pike CJ
Endocrinology 2008 May;149(5):2607-11
Endocrinology 2008 May;149(5):2607-11
Progesterone and estrogen regulate Alzheimer-like neuropathology in female 3xTg-AD mice.
Carroll JC, Rosario ER, Chang L, Stanczyk FZ, Oddo S, LaFerla FM, Pike CJ
The Journal of neuroscience : the official journal of the Society for Neuroscience 2007 Nov 28;27(48):13357-65
The Journal of neuroscience : the official journal of the Society for Neuroscience 2007 Nov 28;27(48):13357-65
Rodent A beta modulates the solubility and distribution of amyloid deposits in transgenic mice.
Jankowsky JL, Younkin LH, Gonzales V, Fadale DJ, Slunt HH, Lester HA, Younkin SG, Borchelt DR
The Journal of biological chemistry 2007 Aug 3;282(31):22707-20
The Journal of biological chemistry 2007 Aug 3;282(31):22707-20
Androgens regulate the development of neuropathology in a triple transgenic mouse model of Alzheimer's disease.
Rosario ER, Carroll JC, Oddo S, LaFerla FM, Pike CJ
The Journal of neuroscience : the official journal of the Society for Neuroscience 2006 Dec 20;26(51):13384-9
The Journal of neuroscience : the official journal of the Society for Neuroscience 2006 Dec 20;26(51):13384-9
Abeta-related angiitis: primary angiitis of the central nervous system associated with cerebral amyloid angiopathy.
Scolding NJ, Joseph F, Kirby PA, Mazanti I, Gray F, Mikol J, Ellison D, Hilton DA, Williams TL, MacKenzie JM, Xuereb JH, Love S
Brain : a journal of neurology 2005 Mar;128(Pt 3):500-15
Brain : a journal of neurology 2005 Mar;128(Pt 3):500-15
Gradual alteration of mitochondrial structure and function by beta-amyloids: importance of membrane viscosity changes, energy deprivation, reactive oxygen species production, and cytochrome c release.
Aleardi AM, Benard G, Augereau O, Malgat M, Talbot JC, Mazat JP, Letellier T, Dachary-Prigent J, Solaini GC, Rossignol R
Journal of bioenergetics and biomembranes 2005 Aug;37(4):207-25
Journal of bioenergetics and biomembranes 2005 Aug;37(4):207-25
Glutamate receptor subunit 3 is modified by site-specific limited proteolysis including cleavage by gamma-secretase.
Meyer EL, Strutz N, Gahring LC, Rogers SW
The Journal of biological chemistry 2003 Jun 27;278(26):23786-96
The Journal of biological chemistry 2003 Jun 27;278(26):23786-96
Function of beta-amyloid in cholesterol transport: a lead to neurotoxicity.
Yao ZX, Papadopoulos V
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2002 Oct;16(12):1677-9
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2002 Oct;16(12):1677-9
Patients with Alzheimer disease have lower levels of serum anti-amyloid peptide antibodies than healthy elderly individuals.
Weksler ME, Relkin N, Turkenich R, LaRusse S, Zhou L, Szabo P
Experimental gerontology 2002 Jul;37(7):943-8
Experimental gerontology 2002 Jul;37(7):943-8
Rapid detection of protein aggregates in the brains of Alzheimer patients and transgenic mouse models of amyloidosis.
Xu G, Gonzales V, Borchelt DR
Alzheimer disease and associated disorders 2002 Jul-Sep;16(3):191-5
Alzheimer disease and associated disorders 2002 Jul-Sep;16(3):191-5
Rapid detection of protein aggregates in the brains of Alzheimer patients and transgenic mouse models of amyloidosis.
Xu G, Gonzales V, Borchelt DR
Alzheimer disease and associated disorders 2002 Jul-Sep;16(3):191-5
Alzheimer disease and associated disorders 2002 Jul-Sep;16(3):191-5
22R-Hydroxycholesterol protects neuronal cells from beta-amyloid-induced cytotoxicity by binding to beta-amyloid peptide.
Yao ZX, Brown RC, Teper G, Greeson J, Papadopoulos V
Journal of neurochemistry 2002 Dec;83(5):1110-9
Journal of neurochemistry 2002 Dec;83(5):1110-9
Distinct binding sites in the structure of alpha 2-macroglobulin mediate the interaction with beta-amyloid peptide and growth factors.
Mettenburg JM, Webb DJ, Gonias SL
The Journal of biological chemistry 2002 Apr 12;277(15):13338-45
The Journal of biological chemistry 2002 Apr 12;277(15):13338-45
Sporadic inclusion body myositis correlates with increased expression and cross-linking by transglutaminases 1 and 2.
Choi YC, Park GT, Kim TS, Sunwoo IN, Steinert PM, Kim SY
The Journal of biological chemistry 2000 Mar 24;275(12):8703-10
The Journal of biological chemistry 2000 Mar 24;275(12):8703-10
Sporadic inclusion body myositis correlates with increased expression and cross-linking by transglutaminases 1 and 2.
Choi YC, Park GT, Kim TS, Sunwoo IN, Steinert PM, Kim SY
The Journal of biological chemistry 2000 Mar 24;275(12):8703-10
The Journal of biological chemistry 2000 Mar 24;275(12):8703-10
Amyloid-beta deposition in skeletal muscle of transgenic mice: possible model of inclusion body myopathy.
Fukuchi K, Pham D, Hart M, Li L, Lindsey JR
The American journal of pathology 1998 Dec;153(6):1687-93
The American journal of pathology 1998 Dec;153(6):1687-93
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
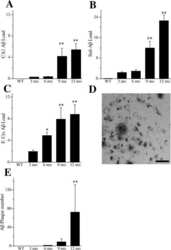
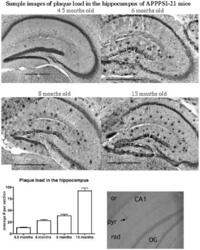
- Figure 10 Analysis of the plaque load shows an age-dependent increase of plaque numbers in the hippocampus of APPPS1 mice. A one-way ANOVA showed an overall difference of p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
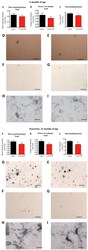
- Figure 10 Histological analysis of biomarkers in the brain . Top half of figure: effect of D-Ala2 glucose-dependent insulinotropic polypeptide (GIP) treatment on three hallmarks of Alzheimer's disease in 6-month-old APP/PS1 mice. ( A ) Quantification of beta-amyloid plaque, ( B ) of dense core plaque load in the cortex of APP/PS1 and, ( C ) of activated microglia (neuroinflammation) in the cortex of APP/PS1 mice. The percentage of stained cortex area was used as measurement for these three hallmarks. Data show mean +- standard error of the mean (SEM) of six mice per group (Student's unpaired t- test, * P < 0.05). Micrographs: The figures on the top row illustrate examples of the cortex of APP/PS1 mice injected with ( D ) saline and ( E ) D-Ala 2 GIP, immunohistologically stained against beta-amyloid (scale bar: 880 mum). The figures in the middle row illustrate example of cortex of APP/PS1 mice injected with ( F ) saline and ( G ) D-Ala 2 GIP, stained with Congo red (scale bar: 880 mum). The figures on the bottom row illustrate examples of cortex of APP/PS1 mice injected with ( H ) saline and ( I ) D-Ala 2 GIP, immunohistologically stained against Iba1 (scale bar: 80 mum). Bottom half of Figure: effect of D-Ala 2 GIP treatment on three hallmarks of Alzheimer's disease in 12-month-old APP/PS1 mice. ( A ) Quantification of beta-amyloid plaque, ( B ) of dense core plaque load in the cortex of APP/PS1 and, ( C ) of activated microglia (neuroinflammation) in the cor
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
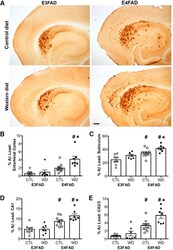
- Figure 3. Accumulation of beta-amyloid deposits assessed by immunohistochemistry in E3FAD and E4FAD mice across dietary treatments. A , Representative images of beta-amyloid immunoreactivity in entorhinal cortex and hippocampus in E3FAD and E4FAD males maintained on control and Western diets. Scale bar, 100 um. beta-Amyloid burden was quantified as immunoreactivity load in E3FAD and E4FAD mice in control and Western diets groups in ( B ) entorhinal cortex, and hippocampal subregions ( C ) subiculum, ( D ) CA1, and ( E ) CA2/3. Data are presented as mean (+-SEM) values; n = 7-11/group. E3FAD mice are shown as circles, E4FAD mice are shown as squares; control diet groups are indicated as open symbols, and Western diet groups as filled symbols. *, p < 0.05 relative to genotype-matched mice in control diet condition. #, p < 0.05 relative to E3FAD mice in same diet condition.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
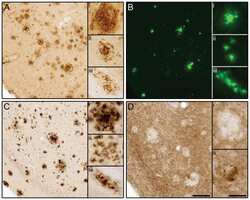
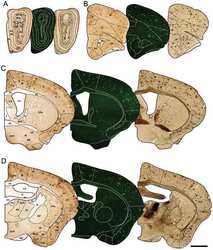
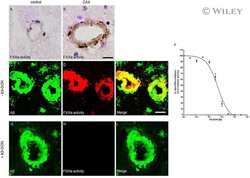
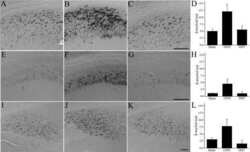
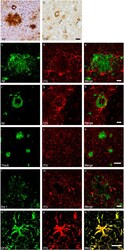
- Figure 1 Distribution of the anti-Abeta antibody and anti-tTG antibodies in sagittal whole brain sections of C57Bl6/J wild-type and APP23 mice brains. 27-months old APP23 mice demonstrated Abeta plaques ( a , black arrow) and vascular Abeta ( a , white arrow). Anti-tTG antibody immunoreactivity was observed in blood vessel walls ( b ) as well as neurons ( b , black arrow) and in glial cells ( b , black open arrow) in both 27-months old wild-type ( b ) and 27-months old APP23 mice (not shown). In APP23 mice, additional tTG antibody immunoreactivity was present in glial cells associated with Abeta plaques ( c-e ) and vascular Abeta ( f-h ) in 24/27-months old mice. Anti-tTG antibody immunoreactivity was associated with the majority of ThioS positive plaques ( i-k ). Double immunofluorescence of the anti-Iba-1 antibody or the anti-GFAP antibody with the anti-tTG antibody demonstrated absence of tTG in Iba-1 positive microglia ( l-n ), whereas tTG immunoreactivity colocalised with GFAP positive astrocytes ( o-q ). Scale bars: ( a ) 20 mum, ( b , f - h , l - q ) 15 mum, ( c - e ) 30 mum, ( i - k ) 60 mum. Abbreviations: Abeta = amyloid beta, GFAP = glial fibrillary acidic protein, Iba-1 = Ionized calcium binding adaptor molecule 1, ThioS = Thioflavin S, tTG = tissue transglutaminase.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
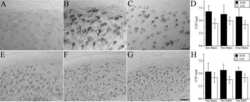
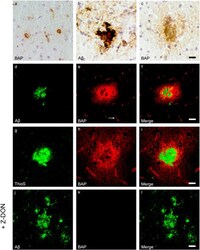
- Figure 3 Distribution of in situ TG activity in C57Bl6/J wild-type and APP23 mice. Serial sagittal whole brain sections of wild-type and APP23 mice were incubated with the TG substrate BAP or the anti-Abeta antibody and visualised using the DAB chromogen. The anti-Abeta antibody demonstrated Abeta plaques in 27-months old mice ( b ). BAP staining was found in the cerebral blood vessel walls of both 7-months old wild-type ( a ) and 27-months old APP23 mice ( c ). In addition, in APP23 mice BAP staining was present in Abeta plaque-like structures ( c ). Double immunofluorescence using the anti-Abeta antibody and BAP staining confirmed the presence of TG activity in Abeta plaques in 12-months old mice ( d-f ) as well as in cerebral vessel walls ( e , arrow). BAP staining was found in the majority of dense core plaques, although it was absent from the cores of these plaques, confirmed by double immunofluorescence of ThioS with BAP ( g-i ). Co-incubation of BAP with the selective tTG inhibitor Z-DON (100 muM) blocked the tTG-catalysed incorporation of BAP ( j-l ). Scale bars: ( c , d ) 36 mum, ( a , d - f , j -l) 30 mum, ( g - i ) 15 mum. Abbreviations: Abeta = amyloid beta, BAP = biotinylated 5-(biotinamido)-pentylamine, ThioS = Thioflavin S, tTG = tissue transglutaminase, Z-DON = Z-DON-Val-Pro-Leu-OMe.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
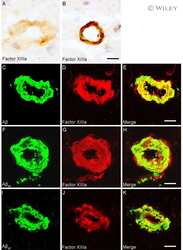
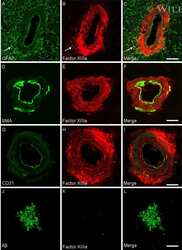
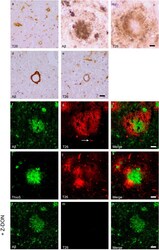
- Figure 4 Distribution of in situ tTG activity in sagittal whole brain sections in C57Bl6/J wild-type and APP23 mice brains. Sagittal serial brain sections of mice were incubated with the specific tTG substrate T26 or the anti-Abeta antibody and visualised using the DAB chromogen. The anti-Abeta antibody stained both plaques ( b ) and vascular Abeta ( d ) in 27-months old mice. T26 staining was present in cerebral blood vessel walls in both 27-months old wild-type ( a ) and APP23 mice ( g , arrow). In addition, in 27-months old APP23 mice, T26 stained both Abeta plaques ( c ) and in vascular Abeta ( e ). Double immunofluorescence of the anti-Abeta antibody with T26 staining demonstrated colocalisation of T26 with Abeta plaques ( f-h ). T26 staining colocalised with the majority of ThioS positive plaques, although T26 staining was absent from the dense cores of these plaques ( i-k ). Co-incubation of T26 with the selective tTG inhibitor Z-DON prevented the tTG-catalysed incorporation of T26 ( l-n ). Scale bars: ( a - c ) 20 mum, ( d - n ) 30 mum. Abbreviations: Abeta = amyloid beta, ThioS = Thioflavin S, tTG = tissue transglutaminase, Z-DON = Z-DON-Val-Pro-Leu-OMe.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Quantification of the percentage of anti-Abeta antibody positive and ThioS positive plaques with BAP or T26 staining was performed in 12-months old APP/PS1 mice and 24- and 27-months old APP23 mice. Double immunofluorescence was performed with an anti-Abeta antibody or ThioS with BAP or T26 resulting in double stainings Abeta/BAP, Abeta/T26, ThioS/BAP and ThioS/T26. Only well-defined anti-Abeta antibody positive or ThioS-positive plaques were counted. Well-defined plaques are marked with arrows, both for Abeta ( a ) and ThioS ( b ) staining (APP23 staining shown), whereas examples of plaques that were not taken into quantification are marked with asterisks ( a,b ). The percentages of anti-Abeta antibody positive and ThioS positive plaques positive for BAP or T26 are shown for both APP23 and APP/PS1 mice ( c ). Non-parametric Kruskal-Wallis testing demonstrated a significant higher percentage of ThioS positive plaques with BAP (77.3 +- 1.9% Mean +- SEM) or T26 (73.4 +- 5.2%) staining in APP23 mice compared to APP/PS1 mice where BAP and T26 staining were present in 50.5 +- 8.0% or 38.3 +- 8.1% of the ThioS positive plaques respectively (p = 0.02). In APP23, a trend increase of the percentage of Abeta plaques with T26 staining was present compared to APP/PS1 mice (66.1 +- 6.9% versus 35.5 +- 8.0% respectively, p = 0.06). The increased percentage of Abeta plaques with BAP staining in APP23 mice (61.0 +- 6.9%) compared to APP/PS1 mice (45.7 +- 7.8%) did not reach statisti
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
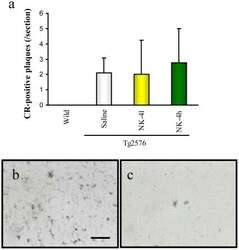
- Figure 8 Abeta deposition in brains of Tg2576 mice at 12 months of age. Coronal brain sections (cortex region) were stained by Congo Red (a) or anti-Abeta polyclonal antibody (b, c). The number of CR-positive plaques on the section was counted in cortex region (a). Typical images of Abeta-immunoreactive tangles of saline-treated Tg2576 mouse (b) and high-dose NK-4(500 ug/kg)-treated Tg2576 mouse (c). Bar represents 50 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
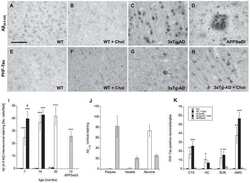
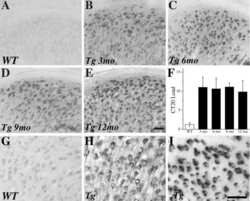
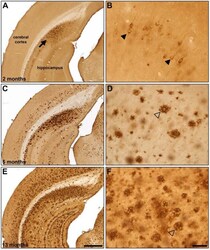
- Fig. (5) Representative beta-amyloid (Abeta) immunohistochemistry in 2 (A, B), 5 (C, D) and 13 (E, F) month old 5XFAD mice. Abeta plaque deposition is present in some areas at 2 months of age (A; arrow) and increases with age (C, E). Areas such as the cingulate cortex exhibit intraneuronal Abeta accumulation at 2 months of age (B; arrowheads) which progresses to Abeta plaque deposition at later ages (D, F; open arrowheads). Scale Bars: A, C, E = 500 um; B, D, F = 50 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
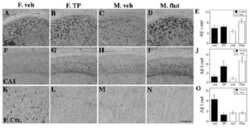
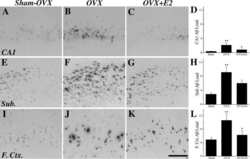
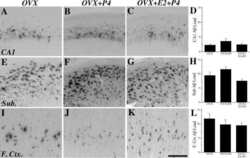
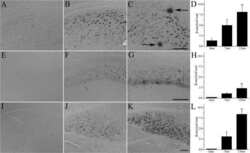
- FIGURE 5 Measures of Alzheimer-related neuropathology in early- and late-MA female 3xTg-AD mice. Representative pictures of beta-amyloid immunolabeling in the subiculum of the hippocampal formation from each treatment group in (A-D) early-MA and (G-J) late-MA female mice. beta-Amyloid load was measured in the subiculum of (E) early-MA and (K) late-MA. Tau-immunolabeled cells were counted in the subiculum and CA1 hippocampal regions in (F) early-MA and (L) late-MA mice. Significance is p < 0.05. D = significant main effect of diet; H = significant main effect of hormone treatment; X = significant interaction between diet and hormone; d = significant post hoc effect between diets, but within hormone treatment; h = significant post hoc effect between hormone treatment, within diet.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
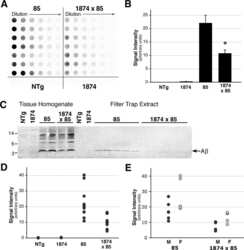
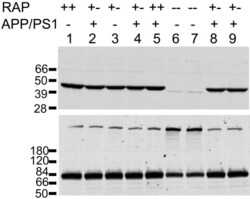
- Figure 4 Analysis of LRP and RAP levels in transgenic animals. Five [APPswe/PS1dE9](+/-)/RAP(+/-) mice (lanes 2, 4, 8, 9), one [APPswe/PS1dE9](-)/RAP(+/+) mouse (lane 1), one [APPswe/PS1dE9](-)/RAP(+/-) mouse (lane 3), one [APPswe/PS1dE9](+/-)RAP(+/+) mouse (lane 5) and two [APPswe/PS1dE9](-)/RAP(-/-) mice (lanes 6, 7) were used in this experiment. 100 ug total protein was loaded per lane. Upper panel - Immunoblot of tissue extract probed with antibody 4109 to RAP (1:1000). Lower panel - Immunoblot of tissue extract probed with antibody 377 to LRP (1:1000). The upper band (~600 kDa) is the full length LRP, the lower band is an 85 kDa fragment of mature, endoproteolytically cleaved, LRP.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
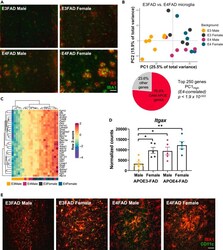
- Figure 2 Sex and APOE genotype drive amyloid pathology and microglial gene expression profiles in APOE-FAD mice (A) Representative images of immunohistochemical staining for amyloid-beta (red) and the microglial marker IBA1 (green) in male and female APOE3- and APOE4-FAD mice. Scale bar, 50 mum. (B) Samples separate by APOE and sex along PC1, with APOE3-FAD males clustering separately from mice of both sexes and APOE3-FAD females. A large proportion of the genes driving this separation (76.4%) are DAM-APOE genes. APOE3-FAD males are shown in yellow, APOE3-FAD females in gray, APOE4-FAD males in magenta, and APOE4-FAD females in teal. (C) Heatmap of the DAM-APOE genes associated with sample clustering along PC1. Gene expression levels were normalized, scaled, and centered and are displayed as Z scores. (D and E) Levels of the DAM-APOE marker, CD11c, are increased in APOE3-FAD females and in APOE4-FAD mice of both sexes, both at the (D) transcript and (E) protein levels. (D) Gene expression of Itgax in normalized counts; data are presented as mean (+-SEM) values; n = 5-7/group. *p < 0.05, **p < 0.01; one-way ANOVA with Tukey's multiple comparisons test correction. (E) Representative immunofluorescence images of microglia (IBA1, red) and CD11c (green) in the hippocampal subiculum of male and female APOE3- and APOE4-FAD mice. Scale bar, 25 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
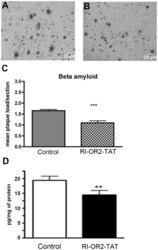
- Figure 5 RI-OR2-TAT reduces the beta-amyloid plaque load and levels of Abeta soluble oligomers in the brains of APP/PS1 transgenic mice. Representative images show amyloid deposits in the cortex region of 10 months old APP/PS1 mouse brains as shown by beta-amyloid immunostaining in animals treated with ( A ) 0.9% saline or ( B ) 100 nmol/kg RI-OR2-TAT in 0.9% saline. ( C ) Quantitative analysis shows a decrease in mean plaque load in the cortex of APP/PS1 mice treated with RI-OR2-TAT compared to animals treated with saline. ( D ) Levels of soluble Ass oligomers in these brains. Values represent mean +- SEM of 4 animals per group, where *** p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
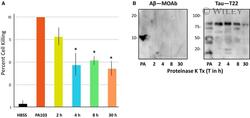
- 5 Fig. 5 Depletion of cytotoxic supernatant using Abeta antibody increases cell killing. (A) PMVECs were incubated with either HBSS, untreated PA103 supernatant, PA103 supernatant incubated with rabbit IgG followed by Protein A agarose beads (IgG Control), or supernatant that was immunodepleted using anti-Abeta or T22. The cells then were photographed at different time points and analyzed. As shown, by 10 h treatment, a time where cytotoxicity had not yet been initiated in untreated control PA103 supernatant, considerable cytotoxic activity was noted in the Abeta depleted supernatant. Bar = 50 mum. (B) Quantitation of cell killing in the five groups shown in Part A at different times following addition of supernatant; dark green = HBSS control, red = PA103 supernatant, yellow = IgG control treated supernatant, light blue = Abeta-depleted supernatant, and light green = T22 (tau)-depleted supernatant ( N = 3).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 6 Fig. Add back of eluted Abeta to Abeta-depleted supernatant restores cytoprotective characteristics. (A) PMVECs were treated for 10 h with either HBSS, untreated PA103 supernatant, supernatant that was immunodepleted using Abeta antibody, or Abeta-treated supernatant which was reconstituted with proteins that were eluted from protein A agarose beads used for immunodepletion. Bar = 50 mum. (B) Quantitation of cell killing at the 10 h time point in the four groups shown in Part A, and presented as +- SEM. Data were analyzed by one-way ANOVA followed by Tukey's post hoc analysis. N = 3, * P value of 0.002 compared to untreated PA103 supernatant.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 7 Fig. (A) Mass spectrometry of PA103 supernatant identified three potential regulators of beta amyloid toxicity. Identified peptides are highlighted. (B) Immunodepletion of cystatin C increases cytotoxicity of PA103 supernatant. PA103 supernatant was immunodepleted with antibodies against either cystatin C (4), gelsolin (5), or Apo J/clusterin (6). The depleted supernatants were then added to PMVECs and cells killing was assessed at 7 h after addition. HBSS (1), PA103 untreated supernatant (2), and Abeta-depleted supernatant (3) were used as controls. Heightened cytotoxicity was detected in the cystatin C-depleted supernatant. Bar = 50 mum. (C) Add back of isolated cystatin C to cystatin C-depleted supernatant partially rescues cytotoxic characteristics of PA103 supernatant. PMVECs were treated with either supernatant that was immune-depleted of cystatin C (1), untreated control PA103 supernatant (2), or anti-cystatin C-treated supernatant that was reconstituted with proteins that were eluted from agarose beads used for immunoprecipitation (3). Bar = 50 mum. (D) Direct quantitation of cell killing at 7 h after addition of control and immunodepleted supernatants, as well as a cystatin C-depleted supernatant that was reconstituted with cystatin C that was eluted from beads following immunodepletion (Add). HBSS and untreated PA103 supernatant served as controls. Data were analyzed by one-way ANOVA followed by Tukey's post hoc analysis. N = 3, +-SEM. * P value of 0.028 compared
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- 2 Fig. Analysis of Proteinase K-treated PA103 supernatant. (A) Supernatant obtained from PA103 infected PMVECs was treated with 100 ug*mL -1 Proteinase K for 0 (untreated control), 1, 4, 8 or 30 h and then added to PMVECs for 21 h. HBSS was added to one group of cells as a negative control. Cell killing was quantified as described, and presented as standard error of the mean (SEM). Data were analyzed by one-way ANOVA followed by Tukey's post hoc analysis. N = 3, * P values compared to PA103 control are 0.005, 0.008, and 0.002 for the 4, 8, and 30 h time points, respectively. (B) Immunoblot analyses of Proteinase K-treated supernatant using antibodies specific for Abeta (MOAb) and oligomeric tau (T22). Protease treatments were for either 1, 4, 8 or 30 h. Untreated PA103 supernatant (PA) was included as a control. Molecular weights are in kDa.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
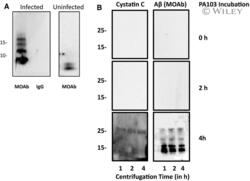
- 8 Fig. 8 Cystatin C is complexed with Abeta. (A) Co-immunoprecipitation of cystatin C and Abeta was assessed. Supernatant was collected from control cells that were cultured in HBSS for 4 h (Uninfected) and for cells that were infected with PA103 bacteria for 4 h (Infected). The supernatants were precipitated with either cystatin C antibody or control rabbit IgG. The samples were then analyzed by immunoblotting using MOAB anti-Abeta antibody. (B) Both Abeta and cystatin C were detected in pellets following ultracentrifugation of PA103 supernatant. Supernatants were collected either immediately after addition of PA103 bacteria (0 h) or after treatment with bacteria for 2 or 4 h. Each supernatant was centrifuged at 150 000 g for 1, 2, or 4 h and the pelleted material was analyzed using antibody against cystatin C and Abeta (MOAB antibody). Molecular weights are given in kDa.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Other assay
Other assay