Antibody data
- Antibody Data
- Antigen structure
- References [11]
- Comments [0]
- Validations
- Immunocytochemistry [1]
- Other assay [4]
Submit
Validation data
Reference
Comment
Report error
- Product number
- OMA1-03132 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Amyloid Precursor Protein Monoclonal Antibody (mAbP2-1)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- OMA1-03132 detects amyloid precursor protein (APP) from human and monkey tissues. This antibody is specific for native, non-denatured protein, and does not cross-react with mouse, rat APP or other APP homologs. OMA1-03132 has been successfully used in Western blot (non-reducing conditions), immunocytochemistry, immunoprecipitation and ELISA procedures. This antibody does not work in in IHC on paraffin embedded tissues nor does it recognize APP if the samples are boiled in Laemmli (SDS) sample buffer containing a reducing agent such as DTT or ß-ME. Can be used in immunoprecipitation utilizing 25 µg/mL of OMA1-03132 linked sepharose. OMA1-03132 antigen is the native, secreted form of human APP. The epitope recognized by OMA1-03132 maps to residues 104-118 of APP
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- mAbP2-1
- Vial size
- 50 µg
- Concentration
- 1.0 mg/mL
- Storage
- -80° C, Avoid Freeze/Thaw Cycles
Submitted references Pulse-Chase Proteomics of the App Knockin Mouse Models of Alzheimer's Disease Reveals that Synaptic Dysfunction Originates in Presynaptic Terminals.
Pre-plaque conformational changes in Alzheimer's disease-linked Aβ and APP.
Aminomethyl-Derived Beta Secretase (BACE1) Inhibitors: Engaging Gly230 without an Anilide Functionality.
Aβ accumulation causes MVB enlargement and is modelled by dominant negative VPS4A.
Chemoproteomic profiling reveals that cathepsin D off-target activity drives ocular toxicity of β-secretase inhibitors.
Discovery of a series of efficient, centrally efficacious BACE1 inhibitors through structure-based drug design.
Utilizing structures of CYP2D6 and BACE1 complexes to reduce risk of drug-drug interactions with a novel series of centrally efficacious BACE1 inhibitors.
Spirocyclic sulfamides as β-secretase 1 (BACE-1) inhibitors for the treatment of Alzheimer's disease: utilization of structure based drug design, WaterMap, and CNS penetration studies to identify centrally efficacious inhibitors.
Identification of novel N-terminal fragments of amyloid precursor protein in cerebrospinal fluid.
X11alpha modulates secretory and endocytic trafficking and metabolism of amyloid precursor protein: mutational analysis of the YENPTY sequence.
X11alpha modulates secretory and endocytic trafficking and metabolism of amyloid precursor protein: mutational analysis of the YENPTY sequence.
Hark TJ, Rao NR, Castillon C, Basta T, Smukowski S, Bao H, Upadhyay A, Bomba-Warczak E, Nomura T, O'Toole ET, Morgan GP, Ali L, Saito T, Guillermier C, Saido TC, Steinhauser ML, Stowell MHB, Chapman ER, Contractor A, Savas JN
Cell systems 2021 Feb 17;12(2):141-158.e9
Cell systems 2021 Feb 17;12(2):141-158.e9
Pre-plaque conformational changes in Alzheimer's disease-linked Aβ and APP.
Klementieva O, Willén K, Martinsson I, Israelsson B, Engdahl A, Cladera J, Uvdal P, Gouras GK
Nature communications 2017 Mar 13;8:14726
Nature communications 2017 Mar 13;8:14726
Aminomethyl-Derived Beta Secretase (BACE1) Inhibitors: Engaging Gly230 without an Anilide Functionality.
Butler CR, Ogilvie K, Martinez-Alsina L, Barreiro G, Beck EM, Nolan CE, Atchison K, Benvenuti E, Buzon L, Doran S, Gonzales C, Helal CJ, Hou X, Hsu MH, Johnson EF, Lapham K, Lanyon L, Parris K, O'Neill BT, Riddell D, Robshaw A, Vajdos F, Brodney MA
Journal of medicinal chemistry 2017 Jan 12;60(1):386-402
Journal of medicinal chemistry 2017 Jan 12;60(1):386-402
Aβ accumulation causes MVB enlargement and is modelled by dominant negative VPS4A.
Willén K, Edgar JR, Hasegawa T, Tanaka N, Futter CE, Gouras GK
Molecular neurodegeneration 2017 Aug 23;12(1):61
Molecular neurodegeneration 2017 Aug 23;12(1):61
Chemoproteomic profiling reveals that cathepsin D off-target activity drives ocular toxicity of β-secretase inhibitors.
Zuhl AM, Nolan CE, Brodney MA, Niessen S, Atchison K, Houle C, Karanian DA, Ambroise C, Brulet JW, Beck EM, Doran SD, O'Neill BT, Am Ende CW, Chang C, Geoghegan KF, West GM, Judkins JC, Hou X, Riddell DR, Johnson DS
Nature communications 2016 Oct 11;7:13042
Nature communications 2016 Oct 11;7:13042
Discovery of a series of efficient, centrally efficacious BACE1 inhibitors through structure-based drug design.
Butler CR, Brodney MA, Beck EM, Barreiro G, Nolan CE, Pan F, Vajdos F, Parris K, Varghese AH, Helal CJ, Lira R, Doran SD, Riddell DR, Buzon LM, Dutra JK, Martinez-Alsina LA, Ogilvie K, Murray JC, Young JM, Atchison K, Robshaw A, Gonzales C, Wang J, Zhang Y, O'Neill BT
Journal of medicinal chemistry 2015 Mar 26;58(6):2678-702
Journal of medicinal chemistry 2015 Mar 26;58(6):2678-702
Utilizing structures of CYP2D6 and BACE1 complexes to reduce risk of drug-drug interactions with a novel series of centrally efficacious BACE1 inhibitors.
Brodney MA, Beck EM, Butler CR, Barreiro G, Johnson EF, Riddell D, Parris K, Nolan CE, Fan Y, Atchison K, Gonzales C, Robshaw AE, Doran SD, Bundesmann MW, Buzon L, Dutra J, Henegar K, LaChapelle E, Hou X, Rogers BN, Pandit J, Lira R, Martinez-Alsina L, Mikochik P, Murray JC, Ogilvie K, Price L, Sakya SM, Yu A, Zhang Y, O'Neill BT
Journal of medicinal chemistry 2015 Apr 9;58(7):3223-52
Journal of medicinal chemistry 2015 Apr 9;58(7):3223-52
Spirocyclic sulfamides as β-secretase 1 (BACE-1) inhibitors for the treatment of Alzheimer's disease: utilization of structure based drug design, WaterMap, and CNS penetration studies to identify centrally efficacious inhibitors.
Brodney MA, Barreiro G, Ogilvie K, Hajos-Korcsok E, Murray J, Vajdos F, Ambroise C, Christoffersen C, Fisher K, Lanyon L, Liu J, Nolan CE, Withka JM, Borzilleri KA, Efremov I, Oborski CE, Varghese A, O'Neill BT
Journal of medicinal chemistry 2012 Nov 8;55(21):9224-39
Journal of medicinal chemistry 2012 Nov 8;55(21):9224-39
Identification of novel N-terminal fragments of amyloid precursor protein in cerebrospinal fluid.
Portelius E, Brinkmalm G, Tran A, Andreasson U, Zetterberg H, Westman-Brinkmalm A, Blennow K, Ohrfelt A
Experimental neurology 2010 Jun;223(2):351-8
Experimental neurology 2010 Jun;223(2):351-8
X11alpha modulates secretory and endocytic trafficking and metabolism of amyloid precursor protein: mutational analysis of the YENPTY sequence.
King GD, Perez RG, Steinhilb ML, Gaut JR, Turner RS
Neuroscience 2003;120(1):143-54
Neuroscience 2003;120(1):143-54
X11alpha modulates secretory and endocytic trafficking and metabolism of amyloid precursor protein: mutational analysis of the YENPTY sequence.
King GD, Perez RG, Steinhilb ML, Gaut JR, Turner RS
Neuroscience 2003;120(1):143-54
Neuroscience 2003;120(1):143-54
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of Amyloid Precursor Protein was performed using 70% confluent log phase PC-3 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with Amyloid Precursor Protein Monoclonal Antibody (mAbP2-1) (Product # OMA1-03132) at 1:200 dilution in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766), (1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b:Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing Golgi apparatus and vesicles localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 60 magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Additional file 6: Figure S5. Abeta1-42 increases the diameter of LAMP-1 positive vesicles in N2a cells. Confocal images of exogenously added monomeric Abeta1-42 incubated for different time points, ranging from 15 min to 48 h, in N2a cells. 3D-rendering with Imaris from confocal z-stack. Colocalization of OC labelling and LAMP1 labelling can be seen from 45 min of Abeta treatment. The last image is from a single focal plane showing OC labelling inside an enlarged LAMP1-positive structure as well as OC labelling that appears to localize at the cell surface. Scale bar 10 mum. (PDF 13678 kb)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
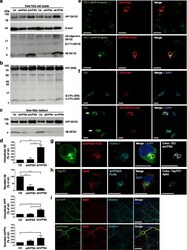
- Experimental details
- Fig. 4 DnVPS4A causes increased accumulation and reduced secretion of Abeta42. Expression of control plasmid (ctr), wtVPS4A (wtVPS4) or the ATP hydrolysis deficient mutant dnVPS4A (dnVPS4) in Swe N2a cells (A-H) or wt primary mouse neurons (I) for 24 h. a Representative Western blot of APP, SDS-stable oligomeric Abeta species, beta-CTFs and monomeric Abeta in cell lysate probed with antibody 6E10, and beta-actin for protein normalization. b Western blot analysis of full length APP, beta-CTFs and alpha-CTFs in cell lysate probed with the C-terminal specific APP antibody 369. c Western blot analysis of secreted alphaAPP (sAPPalpha) and Abeta in cell medium with antibody 6E10. d Densitometric quantification of A and C demonstrates that expression of dnVPS4, but not wtVPS4, increases intracellular Abeta compared to ctr. On the other hand, both wtVPS4A and dnVPS4A reduces secreted Abeta compared to ctr, although to a greater extent with dnVPS4A. DnVPS4 increases both cellular and secreted APP. Values are normalized against actin and expressed as percentage of control, n > 3; * p < 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001. e Cellular full length APP is increased in cells transfected with dnVPS4A-FLAG (lower panel) compared to control plasmid Ctr-FLAG ( upper panel ). Scale bar 25 mum. f Antibody OC labelling of fibrillar oligomers and fibrils is increased in cells transfected with dnVPS4A ( arrows ) compared to control plasmid. Scale bar 25 mum. g Antibody OC labelling in
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry