Antibody data
- Antibody Data
- Antigen structure
- References [7]
- Comments [0]
- Validations
- Immunocytochemistry [2]
- Immunohistochemistry [3]
- Flow cytometry [1]
- Chromatin Immunoprecipitation [2]
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-376G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-STAT1 (Tyr701) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 μL
- Storage
- -20°C
Submitted references Protective effect of suppressor of cytokine signalling 1-based therapy in experimental abdominal aortic aneurysm.
PTPN14 aggravates inflammation through promoting proteasomal degradation of SOCS7 in acute liver failure.
MicroRNA-325-3p Facilitates Immune Escape of Mycobacterium tuberculosis through Targeting LNX1 via NEK6 Accumulation to Promote Anti-Apoptotic STAT3 Signaling.
Comprehensive systems biology analysis of a 7-month cigarette smoke inhalation study in C57BL/6 mice.
Common γ-chain cytokine signaling is required for macroautophagy induction during CD4+ T-cell activation.
Transcriptional profiling and targeted proteomics reveals common molecular changes associated with cigarette smoke-induced lung emphysema development in five susceptible mouse strains.
Effects of leukemia inhibitory factor on 3T3-L1 adipocytes.
Bernal S, Lopez-Sanz L, Jimenez-Castilla L, Prieto I, Melgar A, La Manna S, Martin-Ventura JL, Blanco-Colio LM, Egido J, Gomez-Guerrero C
British journal of pharmacology 2021 Feb;178(3):564-581
British journal of pharmacology 2021 Feb;178(3):564-581
PTPN14 aggravates inflammation through promoting proteasomal degradation of SOCS7 in acute liver failure.
Fu B, Yin S, Lin X, Shi L, Wang Y, Zhang S, Zhao Q, Li Z, Yang Y, Wu H
Cell death & disease 2020 Sep 25;11(9):803
Cell death & disease 2020 Sep 25;11(9):803
MicroRNA-325-3p Facilitates Immune Escape of Mycobacterium tuberculosis through Targeting LNX1 via NEK6 Accumulation to Promote Anti-Apoptotic STAT3 Signaling.
Fu B, Xue W, Zhang H, Zhang R, Feldman K, Zhao Q, Zhang S, Shi L, Pavani KC, Nian W, Lin X, Wu H
mBio 2020 Jun 2;11(3)
mBio 2020 Jun 2;11(3)
Comprehensive systems biology analysis of a 7-month cigarette smoke inhalation study in C57BL/6 mice.
Ansari S, Baumer K, Boué S, Dijon S, Dulize R, Ekroos K, Elamin A, Foong C, Guedj E, Hoeng J, Ivanov NV, Krishnan S, Leroy P, Martin F, Merg C, Peck MJ, Peitsch MC, Phillips B, Schlage WK, Schneider T, Talikka M, Titz B, Vanscheeuwijck P, Veljkovic E, Vihervaara T, Vuillaume G, Woon CQ
Scientific data 2016 Jan 5;3:150077
Scientific data 2016 Jan 5;3:150077
Common γ-chain cytokine signaling is required for macroautophagy induction during CD4+ T-cell activation.
Botbol Y, Patel B, Macian F
Autophagy 2015;11(10):1864-77
Autophagy 2015;11(10):1864-77
Transcriptional profiling and targeted proteomics reveals common molecular changes associated with cigarette smoke-induced lung emphysema development in five susceptible mouse strains.
Cabanski M, Fields B, Boue S, Boukharov N, DeLeon H, Dror N, Geertz M, Guedj E, Iskandar A, Kogel U, Merg C, Peck MJ, Poussin C, Schlage WK, Talikka M, Ivanov NV, Hoeng J, Peitsch MC
Inflammation research : official journal of the European Histamine Research Society ... [et al.] 2015 Jul;64(7):471-86
Inflammation research : official journal of the European Histamine Research Society ... [et al.] 2015 Jul;64(7):471-86
Effects of leukemia inhibitory factor on 3T3-L1 adipocytes.
Hogan JC, Stephens JM
The Journal of endocrinology 2005 Jun;185(3):485-96
The Journal of endocrinology 2005 Jun;185(3):485-96
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
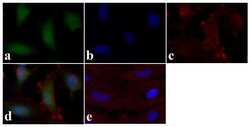
- Experimental details
- Immunofluorescence analysis of STAT1 (pY701) was done on 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with STAT1 (pY701) Rabbit polyclonal Antibody (Product # 44-376G) at 2 µg/mL in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Goat Anti-Rabbit IgG Secondary Antibody (Product # A-11008) at a dilution of 1:400 for 30 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing cytoplasmic and nuclear localization. Panel e shows no primary antibody control. The images were captured at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
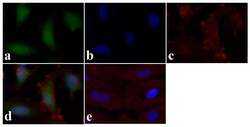
- Experimental details
- Immunofluorescence analysis of STAT1 (pY701) was done on 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with STAT1 (pY701) Rabbit polyclonal Antibody (Product # 44-376G) at 2 µg/mL in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Goat Anti-Rabbit IgG Secondary Antibody (Product # A-11008) at a dilution of 1:400 for 30 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing cytoplasmic and nuclear localization. Panel e shows no primary antibody control. The images were captured at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
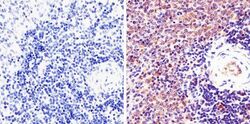
- Experimental details
- Immunohistochemistry analysis of STAT1 (pY701) showing staining in the cytoplasm and nucleus of paraffin-embedded mouse spleen tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a STAT1 (pY701) Rabbit Polyclonal Antibody (Product # 44-376G) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
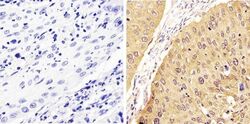
- Experimental details
- Immunohistochemistry analysis of STAT1 (pY701) showing staining in the cytoplasm and nucleus of paraffin-embedded human cervical carcinoma (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a STAT1 (pY701) Rabbit Polyclonal Antibody (Product # 44-376G) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
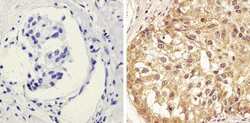
- Experimental details
- Immunohistochemistry analysis of STAT1 (pY701) showing staining in the cytoplasm and nucleus of paraffin-embedded human breast carcinoma (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a STAT1 (pY701) Rabbit Polyclonal Antibody (Product # 44-376G) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
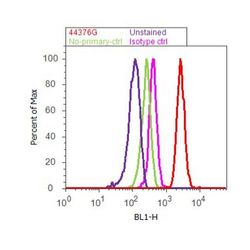
- Experimental details
- Flow cytometry analysis of STAT1 [pY701] was done on HeLa cells treated with IFN gamma (100ng/ml, 30 minutes). Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Tritonª X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with STAT1 [pY701] Rabbit Polyclonal Antibody (44376G, red histogram) or with rabbit isotype control (pink histogram) at 3-5 µg/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor¨ 488 Goat Anti-Rabbit Secondary Antibody (A11008) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune¨ Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
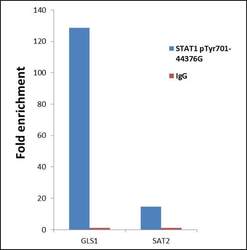
- Experimental details
- ChIP- qPCR analysis of STAT1 (pTyr701) was performed with 10 µL of the STAT1 (pTyr701) Rabbit polyclonal antibody (Product # 44-376G) on sheared chromatin from 2 million HeLa cells treated with 50 ng/mL of IFN Gamma for one hour using the MAGnify™ Chromatin Immunoprecipitation System (Product # 49-2024). Normal Rabbit IgG was used as a negative IP control. The purified DNA from each ChIP sample was analyzed by StepOnePlus™ Real-Time PCR System (Product # 4376600) with primers for the promoter of active GLS1 gene, used as positive control target, and the inactive SAT2, used as negative control target. Data is presented as fold enrichment of the antibody signal versus the negative control IgG using the comparative CT method.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
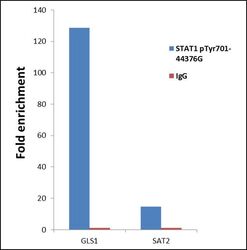
- Experimental details
- ChIP- qPCR analysis of STAT1 (pTyr701) was performed with 10 µL of the STAT1 (pTyr701) Rabbit polyclonal antibody (Product # 44-376G) on sheared chromatin from 2 million HeLa cells treated with 50 ng/mL of IFN Gamma for one hour using the MAGnify Chromatin Immunoprecipitation System (Product # 49-2024). Normal Rabbit IgG was used as a negative IP control. The purified DNA from each ChIP sample was analyzed by StepOnePlus Real-Time PCR System (Product # 4376600) with primers for the promoter of active GLS1 gene, used as positive control target, and the inactive SAT2, used as negative control target. Data is presented as fold enrichment of the antibody signal versus the negative control IgG using the comparative CT method.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
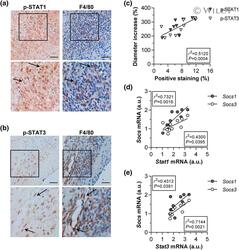
- 1 FIGURE JAK/STAT pathway activation in experimental AAA. (a, b) Immunohistochemical detection of p-STAT1 (a) and p-STAT3 (b) and colocalization with macrophages (F4/80) in aortic sections from the elastase-induced AAA mouse model. Shown are representative images (scale bars, 50 mum) and expanded views of the rectangular areas. Arrows indicate positive cells. (c) Pearson's correlation analysis of p-STAT1 and p-STAT3 positive immunostaining (expressed as percentage of total area) with the aortic diameter increment in AAA lesions. Positive correlation of Socs1/3 expression levels with Stat1 (d) and Stat3 (e) in aortic tissue from AAA model ( n = 10 mice). Real-time PCR values normalized to 18S rRNA are expressed as arbitrary units (a.u.). Pearson's r and P values for the correlations are given within the figures
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
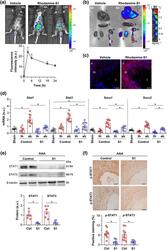
- 2 FIGURE In vivo targeting of JAK/STAT pathway in experimental AAA. (a-c) Biodistribution of S1 peptide in the AAA mouse model. Seven days after elastase perfusion, rhodamine labelled S1 peptide was injected intraperitoneally and allowed to target for 3, 6, 18 and 24 h. (a) Representative in vivo IVIS images of whole mouse after S1 injection (6 and 18 h) and summary of relative fluorescence intensities along the time ( n = 3 animals). Statistical analysis was not performed on these data. (b) ex vivo imaging of mouse tissues at 6 h of S1 injection and in vitro fluorescence signal of serial dilutions of peptide in 96-well plates. (c) Representative confocal images (scale bar, 25 mum) showing distribution of fluorescent S1 peptide in abdominal aortic sections at 6 h post-injection. The orientation of aorta is described by l, lumen; m, media; a, adventitia. (d) Quantitative real-time PCR analysis of STAT1/3 and SOCS1/3 mRNA expression in thoracic (th) and abdominal (ab) aortic tissue from elastase-perfused mice (Control, n = 10; S1, n = 9), using abdominal aorta from saline-perfused mice (sham, n = 8) as reference group. PCR values normalized to 18S rRNA are expressed as arbitrary units (a.u.). (e) Western blot analysis of STAT1/3 proteins and beta-tubulin (loading control) in mouse abdominal aorta from elastase-perfused mice after 14 days of treatment with either vehicle (control; Ctrl, n = 6) or SOCS1-derived peptide (S1, n = 6). Representative blots and summary of normalized q
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
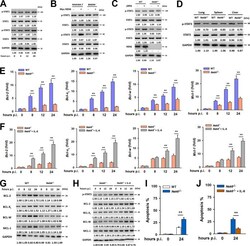
- FIG 5 NEK6 inhibits apoptosis through the activation of STAT3. (A) Immunoblot analysis of the phosphorylated STAT1 (p-STAT1), STAT1, phosphorylated STAT3 (p-STAT3), and STAT3 in RAW 264.7 cells stimulated with 10 mug/mL of gamma-irradiated M. tuberculosis . (B) Immunoblot analysis of the p-STAT1, STAT1, p-STAT3, and STAT3 in RAW 264.7 cells and BMDMs transfected with Myc-NEK6. (C) BMDMs from WT and NEK6-deficient ( Nek6 -/- ) mice were stimulated with 10 mug/mL of gamma-irradiated M. tuberculosis for 24 h. The expressions of p-STAT1, STAT1, p-STAT3, STAT3, and NEK6 were analyzed by Western blotting. (D) Immunoblot analysis of the p-STAT3 and STAT3 in lungs, spleens, and livers from WT and Nek6 -/- mice at 7 dpi. (E and G) The expression levels of BCL-2, BCL-X L , BCL-W, and MCL-1 in 10 mug/mL of gamma-irradiated M. tuberculosis -stimulated BMDMs from WT and Nek6 -/- mice were calculated by qRT-PCR at the indicated times (E) and by Western blotting (G). (F and H) BMDMs from Nek6 -/- mice were pretreated with 20 ng/mL IL-6 for 4 h h, then cells were stimulated with 10 mug/mL of gamma-irradiated M. tuberculosis . The expression levels of BCL-2, BCL-X L , BCL-W, and MCL-1 were calculated by qRT-PCR (F) and Western blotting (H) at the indicated times. (I) BMDMs from WT and Nek6 -/- mice were stimulated with 10 mug/mL of gamma-irradiated M. tuberculosis for 24 h, and the apoptosis rates were detected by flow cytometry. (J) BMDMs from Nek6 -/- mice were pretreated with 20 ng/mL of I
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
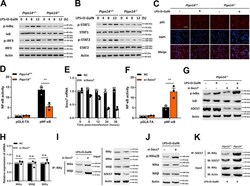
- Experimental details
- Fig. 4 SOCS7 blocks the NF-kappaB pathway by interfering with IKK complex assembly. a , b Wild-type, and PTPN14-deficient Hep G2 cells were stimulated with 1 mug/mL LPS and 5 mM D-GalN. Cell lysates were collected at time points as indicated and western blotting was used to detect the phosphorylation levels of IkappaB, IRF-3 ( a ), and STAT1, STAT2 ( b ). c Wild-type and PTPN14-deficient Hep G2 cells were stimulated with 1 mug/mL LPS and 5 mM D-GalN for 8 h. The p65 location was identified by immunofluorescence. Scale bar = 50 mum. d The activation of the NF-kappaB pathway in wild-type and PTPN14-deficient BMDMs. e The knockdown efficiency of Socs7 in PTPN14-deficient BMDMs stimulated with 1 mug/mL LPS and 5 mM D-GalN. f , g PTPN14-deficient BMDMs were transfected with Socs7 siRNA and were stimulated with 1 mug/mL LPS and 5 mM D-GalN for 24 h. The activation of the NF-kappaB pathway ( f ) and the phosphorylation level of IkappaB ( g ) were detected. h PTPN14-deficient BMDMs were transfected with Socs7 siRNA and were stimulated with 1 mug/mL LPS and 5 mM D-GalN for 24 h. The mRNA levels of IKKalpha, IKKbeta, and IKKgamma were tested using qRT-PCR. i , j PTPN14-deficient BMDMs were transfected with Socs7 siRNA and were stimulated with 1 mug/mL LPS and 5 mM D-GalN for 24 h. The interaction between IKKgamma and IKKalpha or IKKbeta ( h ) and the phosphorylation level of IKKalpha/beta ( i ) were detected. k Wild-type and PTPN14-deficient BMDMs were stimulated with 1 mug/mL LPS and
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry