Antibody data
- Antibody Data
- Antigen structure
- References [28]
- Comments [0]
- Validations
- Western blot [1]
- Flow cytometry [1]
- Other assay [35]
Submit
Validation data
Reference
Comment
Report error
- Product number
- AHO0502 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- FAK Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Recombinant full-length protein
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 500 µg
- Concentration
- 1 mg/mL
- Storage
- -20°C
Submitted references Increased Stiffness Downregulates Focal Adhesion Kinase Expression in Pancreatic Cancer Cells Cultured in 3D Self-Assembling Peptide Scaffolds.
Mechanical Loading Disrupts Focal Adhesion Kinase Activation in Mandibular Fibrochondrocytes During Murine Temporomandibular Joint Osteoarthritis.
Dexamethasone accelerates muscle regeneration by modulating kinesin-1-mediated focal adhesion signals.
EB1 Restricts Breast Cancer Cell Invadopodia Formation and Matrix Proteolysis via FAK.
Effects of Atypical Protein Kinase C Inhibitor (DNDA) on Lung Cancer Proliferation and Migration by PKC-ι/FAK Ubiquitination Through the Cbl-b Pathway.
Focal adhesion ribonucleoprotein complex proteins are major humoral cancer antigens and targets in autoimmune diseases.
Cell adhesion-induced transient interaction of ADAM15 with poly(A) binding protein at the cell membrane colocalizes with mRNA translation.
Periostin overexpression in collecting ducts accelerates renal cyst growth and fibrosis in polycystic kidney disease.
Fibronectin in cell adhesion and migration via N-glycosylation.
Low-intensity pulsed ultrasound promotes cell motility through vinculin-controlled Rac1 GTPase activity.
A Short Region of Connexin43 Reduces Human Glioma Stem Cell Migration, Invasion, and Survival through Src, PTEN, and FAK.
The microRNA-200/Zeb1 axis regulates ECM-dependent β1-integrin/FAK signaling, cancer cell invasion and metastasis through CRKL.
Focal adhesion kinase-dependent focal adhesion recruitment of SH2 domains directs SRC into focal adhesions to regulate cell adhesion and migration.
MicroRNA miR-125a-3p modulates molecular pathway of motility and migration in prostate cancer cells.
LKB1 represses focal adhesion kinase (FAK) signaling via a FAK-LKB1 complex to regulate FAK site maturation and directional persistence.
microRNA-125a-3p reduces cell proliferation and migration by targeting Fyn.
Secreted Hsp90 is a novel regulator of the epithelial to mesenchymal transition (EMT) in prostate cancer.
Enhanced integrin mediated signaling and cell cycle progression on fibronectin mimetic peptide amphiphile monolayers.
Perisynaptic chondroitin sulfate proteoglycans restrict structural plasticity in an integrin-dependent manner.
Comparative analysis of novel and conventional Hsp90 inhibitors on HIF activity and angiogenic potential in clear cell renal cell carcinoma: implications for clinical evaluation.
Integrin activation promotes axon growth on inhibitory chondroitin sulfate proteoglycans by enhancing integrin signaling.
Mesenchymal/stromal gene expression signature relates to basal-like breast cancers, identifies bone metastasis and predicts resistance to therapies.
Extracellular matrix-induced transforming growth factor-beta receptor signaling dynamics.
Dissection of the osteogenic effects of laminin-332 utilizing specific LG domains: LG3 induces osteogenic differentiation, but not mineralization.
Quantitative multicolor compositional imaging resolves molecular domains in cell-matrix adhesions.
Focal adhesion kinase signaling pathways regulate the osteogenic differentiation of human mesenchymal stem cells.
Activation of FAK is necessary for the osteogenic differentiation of human mesenchymal stem cells on laminin-5.
Analyzing FAK and Pyk2 in early integrin signaling events.
Betriu N, Andreeva A, Alonso A, Semino CE
Biomedicines 2022 Jul 29;10(8)
Biomedicines 2022 Jul 29;10(8)
Mechanical Loading Disrupts Focal Adhesion Kinase Activation in Mandibular Fibrochondrocytes During Murine Temporomandibular Joint Osteoarthritis.
Reed DA, Zhao Y, Han M, Mercuri LG, Miloro M
Journal of oral and maxillofacial surgery : official journal of the American Association of Oral and Maxillofacial Surgeons 2021 Oct;79(10):2058.e1-2058.e15
Journal of oral and maxillofacial surgery : official journal of the American Association of Oral and Maxillofacial Surgeons 2021 Oct;79(10):2058.e1-2058.e15
Dexamethasone accelerates muscle regeneration by modulating kinesin-1-mediated focal adhesion signals.
Lin JW, Huang YM, Chen YQ, Chuang TY, Lan TY, Liu YW, Pan HW, You LR, Wang YK, Lin KH, Chiou A, Kuo JC
Cell death discovery 2021 Feb 17;7(1):35
Cell death discovery 2021 Feb 17;7(1):35
EB1 Restricts Breast Cancer Cell Invadopodia Formation and Matrix Proteolysis via FAK.
Chanez B, Ostacolo K, Badache A, Thuault S
Cells 2021 Feb 13;10(2)
Cells 2021 Feb 13;10(2)
Effects of Atypical Protein Kinase C Inhibitor (DNDA) on Lung Cancer Proliferation and Migration by PKC-ι/FAK Ubiquitination Through the Cbl-b Pathway.
BommaReddy RR, Patel R, Smalley T, Acevedo-Duncan M
OncoTargets and therapy 2020;13:1661-1676
OncoTargets and therapy 2020;13:1661-1676
Focal adhesion ribonucleoprotein complex proteins are major humoral cancer antigens and targets in autoimmune diseases.
Atsumi S, Katoh H, Komura D, Hashimoto I, Furuya G, Koda H, Konishi H, Suzuki R, Yamamoto A, Yuba S, Abe H, Rino Y, Oshima T, Ushiku T, Fukayama M, Seto Y, Ishikawa S
Communications biology 2020 Oct 16;3(1):588
Communications biology 2020 Oct 16;3(1):588
Cell adhesion-induced transient interaction of ADAM15 with poly(A) binding protein at the cell membrane colocalizes with mRNA translation.
Böhm BB, Fehrl Y, Janczi T, Schneider N, Burkhardt H
PloS one 2018;13(9):e0203847
PloS one 2018;13(9):e0203847
Periostin overexpression in collecting ducts accelerates renal cyst growth and fibrosis in polycystic kidney disease.
Raman A, Parnell SC, Zhang Y, Reif GA, Dai Y, Khanna A, Daniel E, White C, Vivian JL, Wallace DP
American journal of physiology. Renal physiology 2018 Dec 1;315(6):F1695-F1707
American journal of physiology. Renal physiology 2018 Dec 1;315(6):F1695-F1707
Fibronectin in cell adhesion and migration via N-glycosylation.
Hsiao CT, Cheng HW, Huang CM, Li HR, Ou MH, Huang JR, Khoo KH, Yu HW, Chen YQ, Wang YK, Chiou A, Kuo JC
Oncotarget 2017 Sep 19;8(41):70653-70668
Oncotarget 2017 Sep 19;8(41):70653-70668
Low-intensity pulsed ultrasound promotes cell motility through vinculin-controlled Rac1 GTPase activity.
Atherton P, Lausecker F, Harrison A, Ballestrem C
Journal of cell science 2017 Jul 15;130(14):2277-2291
Journal of cell science 2017 Jul 15;130(14):2277-2291
A Short Region of Connexin43 Reduces Human Glioma Stem Cell Migration, Invasion, and Survival through Src, PTEN, and FAK.
Jaraíz-Rodríguez M, Tabernero MD, González-Tablas M, Otero A, Orfao A, Medina JM, Tabernero A
Stem cell reports 2017 Aug 8;9(2):451-463
Stem cell reports 2017 Aug 8;9(2):451-463
The microRNA-200/Zeb1 axis regulates ECM-dependent β1-integrin/FAK signaling, cancer cell invasion and metastasis through CRKL.
Ungewiss C, Rizvi ZH, Roybal JD, Peng DH, Gold KA, Shin DH, Creighton CJ, Gibbons DL
Scientific reports 2016 Jan 5;6:18652
Scientific reports 2016 Jan 5;6:18652
Focal adhesion kinase-dependent focal adhesion recruitment of SH2 domains directs SRC into focal adhesions to regulate cell adhesion and migration.
Wu JC, Chen YC, Kuo CT, Wenshin Yu H, Chen YQ, Chiou A, Kuo JC
Scientific reports 2015 Dec 18;5:18476
Scientific reports 2015 Dec 18;5:18476
MicroRNA miR-125a-3p modulates molecular pathway of motility and migration in prostate cancer cells.
Ninio-Many L, Grossman H, Levi M, Zilber S, Tsarfaty I, Shomron N, Tuvar A, Chuderland D, Stemmer SM, Ben-Aharon I, Shalgi R
Oncoscience 2014;1(4):250-261
Oncoscience 2014;1(4):250-261
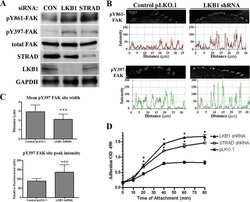
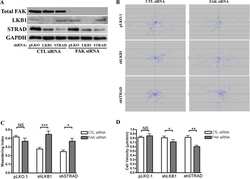
LKB1 represses focal adhesion kinase (FAK) signaling via a FAK-LKB1 complex to regulate FAK site maturation and directional persistence.
Kline ER, Shupe J, Gilbert-Ross M, Zhou W, Marcus AI
The Journal of biological chemistry 2013 Jun 14;288(24):17663-74
The Journal of biological chemistry 2013 Jun 14;288(24):17663-74
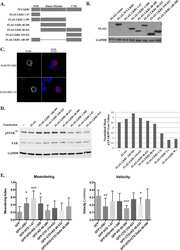
microRNA-125a-3p reduces cell proliferation and migration by targeting Fyn.
Ninio-Many L, Grossman H, Shomron N, Chuderland D, Shalgi R
Journal of cell science 2013 Jul 1;126(Pt 13):2867-76
Journal of cell science 2013 Jul 1;126(Pt 13):2867-76
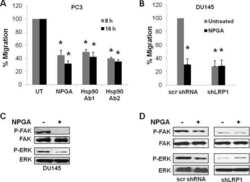
Secreted Hsp90 is a novel regulator of the epithelial to mesenchymal transition (EMT) in prostate cancer.
Hance MW, Dole K, Gopal U, Bohonowych JE, Jezierska-Drutel A, Neumann CA, Liu H, Garraway IP, Isaacs JS
The Journal of biological chemistry 2012 Nov 2;287(45):37732-44
The Journal of biological chemistry 2012 Nov 2;287(45):37732-44
Enhanced integrin mediated signaling and cell cycle progression on fibronectin mimetic peptide amphiphile monolayers.
Shroff K, Pearce TR, Kokkoli E
Langmuir : the ACS journal of surfaces and colloids 2012 Jan 24;28(3):1858-65
Langmuir : the ACS journal of surfaces and colloids 2012 Jan 24;28(3):1858-65
Perisynaptic chondroitin sulfate proteoglycans restrict structural plasticity in an integrin-dependent manner.
Orlando C, Ster J, Gerber U, Fawcett JW, Raineteau O
The Journal of neuroscience : the official journal of the Society for Neuroscience 2012 Dec 12;32(50):18009-17, 18017a
The Journal of neuroscience : the official journal of the Society for Neuroscience 2012 Dec 12;32(50):18009-17, 18017a
Comparative analysis of novel and conventional Hsp90 inhibitors on HIF activity and angiogenic potential in clear cell renal cell carcinoma: implications for clinical evaluation.
Bohonowych JE, Peng S, Gopal U, Hance MW, Wing SB, Argraves KM, Lundgren K, Isaacs JS
BMC cancer 2011 Dec 15;11:520
BMC cancer 2011 Dec 15;11:520
Integrin activation promotes axon growth on inhibitory chondroitin sulfate proteoglycans by enhancing integrin signaling.
Tan CL, Kwok JC, Patani R, Ffrench-Constant C, Chandran S, Fawcett JW
The Journal of neuroscience : the official journal of the Society for Neuroscience 2011 Apr 27;31(17):6289-95
The Journal of neuroscience : the official journal of the Society for Neuroscience 2011 Apr 27;31(17):6289-95
Mesenchymal/stromal gene expression signature relates to basal-like breast cancers, identifies bone metastasis and predicts resistance to therapies.
Marchini C, Montani M, Konstantinidou G, Orrù R, Mannucci S, Ramadori G, Gabrielli F, Baruzzi A, Berton G, Merigo F, Fin S, Iezzi M, Bisaro B, Sbarbati A, Zerani M, Galiè M, Amici A
PloS one 2010 Nov 30;5(11):e14131
PloS one 2010 Nov 30;5(11):e14131
Extracellular matrix-induced transforming growth factor-beta receptor signaling dynamics.
Garamszegi N, Garamszegi SP, Samavarchi-Tehrani P, Walford E, Schneiderbauer MM, Wrana JL, Scully SP
Oncogene 2010 Apr 22;29(16):2368-80
Oncogene 2010 Apr 22;29(16):2368-80
Dissection of the osteogenic effects of laminin-332 utilizing specific LG domains: LG3 induces osteogenic differentiation, but not mineralization.
Klees RF, Salasznyk RM, Ward DF, Crone DE, Williams WA, Harris MP, Boskey A, Quaranta V, Plopper GE
Experimental cell research 2008 Feb 15;314(4):763-73
Experimental cell research 2008 Feb 15;314(4):763-73
Quantitative multicolor compositional imaging resolves molecular domains in cell-matrix adhesions.
Zamir E, Geiger B, Kam Z
PloS one 2008 Apr 2;3(4):e1901
PloS one 2008 Apr 2;3(4):e1901
Focal adhesion kinase signaling pathways regulate the osteogenic differentiation of human mesenchymal stem cells.
Salasznyk RM, Klees RF, Williams WA, Boskey A, Plopper GE
Experimental cell research 2007 Jan 1;313(1):22-37
Experimental cell research 2007 Jan 1;313(1):22-37
Activation of FAK is necessary for the osteogenic differentiation of human mesenchymal stem cells on laminin-5.
Salasznyk RM, Klees RF, Boskey A, Plopper GE
Journal of cellular biochemistry 2007 Feb 1;100(2):499-514
Journal of cellular biochemistry 2007 Feb 1;100(2):499-514
Analyzing FAK and Pyk2 in early integrin signaling events.
Bernard-Trifilo JA, Lim ST, Hou S, Schlaepfer DD, Ilic D
Current protocols in cell biology 2006 Apr;Chapter 14:Unit 14.7
Current protocols in cell biology 2006 Apr;Chapter 14:Unit 14.7
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- CRISPR-Cas9 mediated genome editing ofFAK (as confirmed by next generation sequencing) was achieved by using LentiArray™ Lentiviral sgRNA (Product # A32042, AssayID CRISPR627416_LV) and LentiArray Cas9 Lentivirus (Product # A32064). Fig (a) Western blot analysis of FAK was performed by loading 30 µg of A549 wild type (Lane 1), A549 Cas9 (Lane 2) and A549 Cas9 cells transduced with FAK Lentiviral sgRNA (Lane 3) membrane enriched extracts. The samples were electrophoresed using NuPAGE™ 3 to 8%, Tris-Acetate, 1.0 mm, Mini Protein Gel (Product # EC6695BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with Anti-FAK Polyclonal Antibody (Product # AHO0502) using 1:1000 dilution and Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177 1:5000 dilution).Chemiluminescent detection was performed using SuperSignal™ West Dura Extended Duration Substrate (Product # 34076). A reduced signal in sgRNA transduced cells using the LentiArray™ CRISPR product line confirms that antibody is specific toFAK (Fig (b)).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Flow cytometry analysis of FAK was done on A549 cells treated with EGF (200ng/mL, 10 minutes). Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with FAK Rabbit Polyclonal Antibody (AHO0502, red histogram) or with rabbit isotype control (pink histogram) at 3-5 ug/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Goat Anti-Rabbit Secondary Antibody (A11008) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
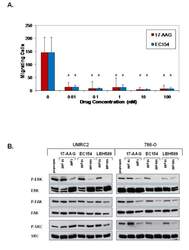
- Figure 6 17-AAG and EC154 suppress UMRC2 cell motility and differentially modulate ERK, FAK, and src activation . ( a ) Serum starved UMRC2 cells were plated in transwell Boyden chambers for 16 h with a serum gradient of 0.1% (top) to 10% (bottom) in the presence of serially diluted concentrations of the indicated Hsp90 inhibitors. Migrating cells were fixed with 3.7% formaldehyde, stained with Crystal Violet and counted, with standard deviation shown. All drug treatments significantly inhibited migration in both cell types (*), as determined by ANOVA and Student's t- test ( p < 0.05). ( b ) UMRC2 and 786-O cells were treated for 20 h with the indicated doses of inhibitors and phosphorylated and total ERK, FAK, and src were analyzed by SDS-PAGE and Western blot.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
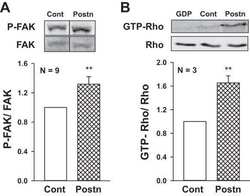
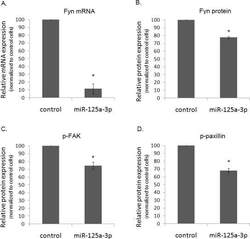
- Figure 4 The effect of miR-125a-3p on hallmark genes in prostate cancer PC3 cells, transfected with miR-125a-3p or with empty vector (control), were cultured for 48 hours. Cells were lysed and, (A) their Fyn mRNA expression was analyzed by Real Time PCR. Proteins were analyzed by WB with specific antibodies against: (B) Fyn, (C) phospho-FAK (p-FAK) and (D) phospho-paxillin (p-paxillin), as well as against their loading control proteins (actin, general FAK and general paxillin, respectively). The experiment was repeated 3 times. Intensity of bands was analyzed using the image J software and the ratio between each protein and its control was plotted. The bars are mean+SD. (*) - Significantly different from control value (p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
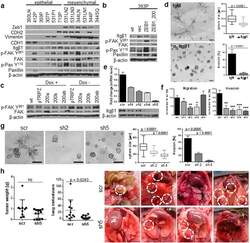
- Figure 3 Integrin beta1 is required for the invasion & metastasis of the murine mesenchymal cell lines. ( a ) Western blot analysis of FAK pathway activation in the mesenchymal vs epithelial cells lines of the murine cell line panel, and in cell lines stably expressing Zeb1 ( b ). ( c ) Western blot of FAK activation in the human H157 cells with inducible miR-200ab expression. ( d ) 393P_ZEB1 cells grown in Matrigel/Collagen I (1.75 mg/ml) and treated with an ITGbeta1-blocking antibody or IgM control for 7 days. ( e ) Quantitative RT-PCR and Western blot of 393P_ZEB1 cells after ITGbeta1-shRNA knockdown. ( f ) In vitro migration and invasion assay for the Itgbeta1 shRNA cells. *p < 0.02, **p < 0.002, ***p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 5 CRKL regulates FAK/Src complex formation at focal adhesions and the invasive/metastatic phenotype. ( a ) qRT-PCR and Western blot ( b ) analysis of 344SQ_shCRKL knockdown cells showed a decrease in FAK signaling. ( c ) CRKL knockdown decreased adhesion to fibronectin and 2D Transwell migration/invasion ( d ). ( e ) Immunofluorescent staining and biochemical fractionation ( f ) of CRKL knockdown cells for activated Src Y 418 , CRKL Y 207 , PaxY 118 , and p-FAK Y 861 in the focal adhesion complex at the membrane. An average of 20-30 cells was counted for the presence of focal adhesions and data are presented per cell. Scale bar is 200 mum. *p < 0.005, **p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
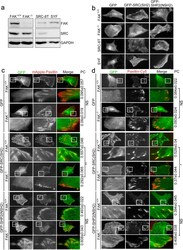
- Figure 3 Focal adhesion kinase expression controls the abundance of SRC_SH2 domain within focal adhesions. ( a ) Total cell lysate from FAK +/+ , FAK -/- , SRC-8T (SRC +/+ /YES +/+ /FYN +/+ ), and SYF (SRC -/- /YES -/- /FYN -/- ) cells were analyzed by Western blotting to detect FAK, SRC and GAPDH. ( b ) TIRFM images of FAK +/+ , FAK -/- , SRC-8T, and SYF cells expressing GFP, GFP-SRC(SH2) or GFP-SHP2(NSH2). Scale bar, 10 mum. ( c ) TIRFM images of FAK +/+ and FAK -/- cells co-transfected with mApple-paxillin (red) and pGFP-C1, pGFP-SRC(SH2) or pGFP-SHP2(NSH2) (green). Bar, 10 mum. The 20 mum x 20 mum areas indicated in the upper images are magnified in the images below. Values indicate Pearson's correlation coefficients (PC) of images of mApple-paxillin and GFP, GFP-SRC(SH2) or GFP-SHP2(NSH2) in FAK +/+ or FAK -/- cells. Data are means +- s.e.m. (GFP: n = 3 FAK +/+ cells, n = 4 FAK -/- cells; GFP-SRC(SH2): n = 5 FAK +/+ cells, n = 5 FAK -/- cells; GFP-SHP2(NSH2): n = 3 FAK +/+ cells, n 9 FAK -/- cells). ** p < 0.01; NS, no significance. ( d ) TIRFM images of FAK +/+ and FAK -/- cells transfected with pGFP-C1, pGFP-SRC(SH2) or pGFP-SHP2(NSH2) (green) and immunostained for paxillin (red). Bar, 10 mum. The 20 mum x 20 mum areas indicated in the upper images are magnified in the images below. Values indicate PC of images of paxillin-Cy5 and GFP, GFP-SRC(SH2) or GFP-SHP2(NSH2) in FAK +/+ or FAK -/- cells. Data are means +- s.e.m. (GFP: n = 4 FAK +/+ cells, n = 3 FAK -/- cell
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
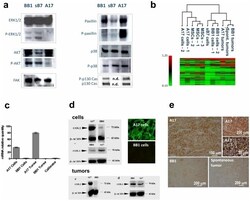
- Figure 1 Signal transduction-related profile of A17. A17 cells exhibit a clearly different pattern of expression and activation (phosphorylation) of key signal transduction molecules compared to syngeneic epithelial cell lines (BB1 and sB7) ( a ). ERK1/2 or Akt proved to be constitutively expressed but not phosphorylated in A17 cells. In contrast, P-ERK1/2 and P-Akt were found in both BB1 and sB7 cells, where they are presumably recruited downstream of the HER-2/neu signaling pathway. Furthermore, whereas both epithelial and A17 cells proved to express Focal Adhesion Kinase (FAK), we found an increased phosphorylation of paxillin and p130 Cas in A17 cells, which is in line with the constitutively motile phenotype of these mesenchymal cells. p-38 was expressed and activated in both epithelial and A17 cells. Microarray analysis restricted to 96 signal transduction-related genes show the A17 cell profile to be more related to that of MSCs than that of syngenic epithelial cells (BB1). However, the A17 tumor profile was shown to be more correlated to that of epithelial tumors than that of epithelial or mesenchymal cells ( b ). The differential expression of COX-2 in A17 compared to epithelial cells was confirmed at the transcriptional level through quantitative real-time PCR ( Figure 1c ). Western Blot analysis confirmed the differential expression of COX-2, but not of COX-1, in A17 cells and tumors comapared to BB1 cells and tumors ( figure 1d ). Differential expression of COX-2
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 5 Cell adhesion and migration in response to the N-glycosylation of human and porcine fibronectin (A) Cells were pre-incubated with serum-free medium containing H 2 O (control), serum-free medium containing 0.5 mM EDTA, or culture medium containing 2 mM MnCl 2 before plating on the indicated concentrations of fibronectin [FN] treated with (+) or without (-) PNGase F. Cells were plated on [FN] for 10 min and then their attachment was measured. Percentage of cells remaining attached on [FN] was shown. Data are the mean +- s.e.m. (n = number of independent experiments). * p < 0.05; ** p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Focal adhesion kinase activity, but not SRC activity, positively regulates the abundance of SRC_SH2 domain within focal adhesions. ( a ) Total cell lysate from untreated U2OS cells (-) or cells treated with FAKi (focal adhesion kinase inhibitor; 50 muM, 1 h), PP2 (SRC inhibitor; 10 muM, 1 h) or SU6656 (SRC inhibitor; 5 muM, 1 h) were analyzed by Western blotting to detect pY118-paxillin, pY31-paxillin, SRC, FAK, paxillin and GAPDH. ( b ) U2OS cells transfected with pGFP-C1, pGFP-SRC(SH2) or pGFP-SHP2(NSH2) were untreated (-) or treated with FAKi or PP2, and immunostained to detect paxillin. TIRFM images of GFP, GFP-SRC(SH2) or GFP-SHP2(NSH2) (green) and paxillin (red). Bar, 10 mum. The 20 mum x 20 mum areas indicated in the upper images are magnified in the images below. Values indicate Pearson's correlation coefficients (PC) of images of paxillin-Cy5 and GFP, GFP-SRC(SH2) or GFP-SHP2(NSH2) in U2OS cells untreated (-) or treated with FAKi or PP2. Data are means +- s.e.m. (GFP: n = 3 cells (-), n = 4 cells (FAKi), n = 6 cells (PP2); GFP-SRC(SH2): n = 7 cells (-), n = 6 cells (FAKi), n = 7 cells (PP2); GFP-SHP2(NSH2): n = 5 cells (-), n = 5 cells (FAKi), n = 4 cells (PP2)). ** p < 0.01; NS, no significance. ( c ) Time-lapse TIRFM images of U2OS cells co-transfected with mApple-paxillin (red) and pGFP-C1, pGFP-SRC(SH2) or pGFP-SHP2(NSH2) (green) during FAKi or PP2 treatment. The time is shown in hour. Bar, 10 mum. ( d ) The ratio of the average density (intensity per mu
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
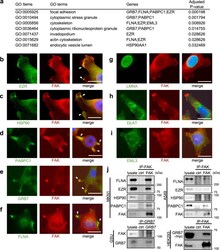
- Fig. 3 Focal adhesion-related protein complexes are major and common humoral antigens in tumor microenvironments. a GO enrichment analysis of the eight identified protein antigens. b - i Fluorescent immunocytochemistry of the identified protein antigens (green) along with focal adhesion kinase (FAK) (red) onto a human gastric cancer cell line MKN1. The blue color represents Hoechst nuclear staining. The white bars indicate 20 mum. The white arrowheads indicate representative hot spots of colocalization between protein antigens and focal adhesions (only in b - f ). Similar colocalizations were reproducibly observed in all analyzed cells with formations of visible focal adhesions (only in b - f ). The same immunocytochemical staining was also performed onto another human gastric cancer cell line GSU, obtaining similar results (Supplementary Fig. 4 ). j Co-immunoprecipitation experiments of the FAK complex. The upper and lower panels at the left side show co-IP using anti-FAK antibody for MKN1 and anti-GRB7 antibody for GSU, respectively, followed by immunoblots using the indicated antibodies. The right-side panels show co-IP using anti-FAK antibody for A549, a lung adenocarcinoma cell line, and HEK293 transfected with GRB7-myc/His construct, respectively, followed by immunoblots using the indicated antibodies. Co-IP experiments were conducted at least twice.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
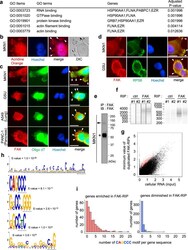
- Fig. 5 The identified humoral antigens in the focal adhesion complexes are ribonucleoproteins (RNPs) colocalized with ribosomal translation machinery. a GO enrichment analysis of the five protein antigens correlated with focal adhesion complexes. b Fluorescent staining of single-strand nucleotides using acridine orange in MKN1 cells. The blue color represents Hoechst nuclear staining. DIC, differential interference contrast image. c Fluorescent dual staining of anti-FAK antibody (red) and Cy5-conjugated oligo-dT probe (green) in human gastric cancer cells MKN1 and GSU, a lung adenocarcinoma cell A549, and a pancreatic cancer cell PANC-1. The blue color represents Hoechst staining. The white bars indicate 20 mum. d Fluorescent immunocytochemistry of FAK (red) and a ribosome marker (ribosomal protein S6 [RPS6]) (green). The blue represents nuclear staining. The white bars indicate 20 mum. b - d White arrowheads indicate representative colocalization signals, and the similar colocalizations were reproducibly observed in all analyzed cells with formations of visible focal adhesions. e The validity of the anti-FAK antibody in our RIP procedure was confirmed by immunoblots. The arrowhead and asterisk represent the FAK protein and heavy chain of the anti-FAK rabbit antibody, respectively. f The left and right panels show electropherograms of the precipitated RNA and constructed cDNA sequencing library, respectively, of FAK-RIP experiments in biological duplicates (#1 and #2). g Plot
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
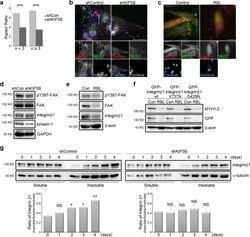
- Experimental details
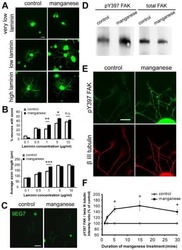
- Fig. 4 Kinesin-1 regulates integrin beta1 activation and myotube morphology. a Effects of kinesin-1 expression on morphological changes of myotubes. C2C12 cells expressing non-silenced (shCon) or Kinesin-1-silenced (shKIF5B) shRNAs were treated with differentiation medium for 5 days and immunostained for MYH1/2 and DAPI. The MYH1/2 + cells were grouped based on the number ( n ) of nucleus in order to calculate the aspect ratio, namely the ratio of cell's longest length to the cell's shortest length. Data are mean +- s.e.m (shCon with =3 nuclei, n = 45 MYH1/2 + cells; shKIF5B with =3 nuclei, n = 23 MYH1/2 + cells). *** p < 0.001. b TIRF microscopy images of immunolocalized paxillin (to visualize FAs; red), 9EG7 (to visualize activated integrin beta1; green), phalloidin (to visualize F-actin; blue), and DAPI (to visualize nucleus; gray) in non-silenced (shControl) and kinesin-1-silenced (shKIF5B) C2C12 cells treated with differentiation medium for 5 days. Scale bar, 30 mum. c TIRF microscopy images of immunolocalized paxillin (to visualize FAs; red), 9EG7 (to visualize activated integrin beta1; green), and DAPI (to visualize nucleus; blue) in C2C12 cells, treated with differentiation medium containing DMSO (Control) or 40 uM RBL for 5 days. Scale bar, 30 mum. d Western blot analysis of cell lysates obtained from non-silenced (shCon) and kinesin-1-silenced (shKIF5B) C2C12 cells, treate
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
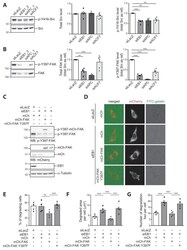
- Experimental details
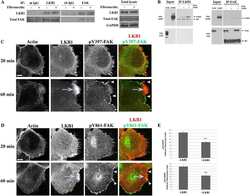
- Figure 4 EB1 restricts invadopodia formation via inhibition of FAK activity. ( A , B ) MDA-MB-231 cells were transfected with a control siRNA (siLacZ) or siRNA directed against EB1 (siEB1_2), APC (siAPC_1) and ACF7 (siACF7). Levels of Src, activated Src (p-Y416-Src), FAK and FAK phosphorylated on Tyr397 (p-Y397-FAK) were analyzed by Western blotting using specific antibodies. ( A ) Total Src and p-Y416-Src levels are represented as the mean +- SEM of five independent experiments. The value of each individual experiment is reported. A representative Western blot is shown (left). ( B ) Total FAK and p-Y397-FAK levels are represented as the mean +- SEM of five independent experiments. The value of each individual experiment is reported. A representative Western blot is shown (left). Src was used as a loading control and as a reference to quantify p-Y416-Src, total FAK, and p-Y397-FAK levels. The paired t -test was used to determine significant differences. *** p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
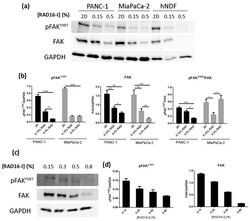
- Experimental details
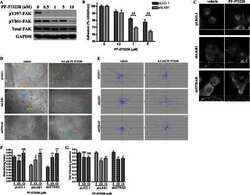
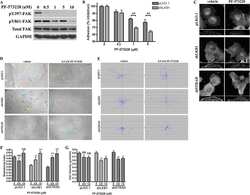
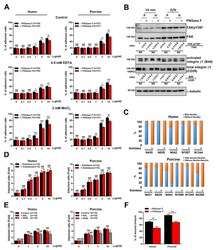
- Figure 4 pFAK Y397 and FAK expression in cancer cell lines and normal fibroblasts cultured in RAD16-I hydrogels. ( a ) Western blot bands of pFAK Y397 and total FAK in PANC-1, MiaPaCa-2 and hNDF cells cultured in 2D and 0.15% (soft) and 0.5% (stiff) RAD16-I gels; ( b ) densitometry of bands shown in ( a ); ( c ) Western blot bands of pFAK Y397 and total FAK in PANC-1 cells cultured in 3D RAD16-I hydrogels of increased stiffness (from 0.15% to 0.8% RAD16-I, representing a stiffness range of 100-8500 Pa); ( d ) densitometry of bands shown in ( c ). GAPDH was used as loading control. Statistical differences are indicated as * for p value < 0.05, ** for p value < 0.01, *** for p value < 0.001 and **** for p value < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
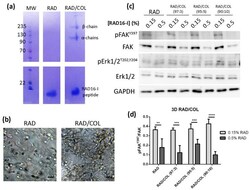
- Experimental details
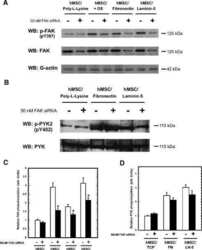
- Cell culture in RAD16-I matrix functionalized with collagen type I. ( a ) SDS-PAGE of RAD or RAD/COL cell-free gels; ( b ) phase-contrast microscopy images of fibroblast 24 h after cell embedding within 0.15% RAD or RAD/COL gels. Scale bars represent 50 um; ( c ) Western blot bands of FAK and Erk1/2 and its phosphorylated forms in PANC-1 cells cultured in 0.15% (soft) and 0.5% (stiff) RAD16-I gels mixed with increasing amounts of collagen I; ( d ) densitometry of pFAK Y397 /FAK shown in ( c ). GAPDH was used as loading control. Statistical differences are indicated as ** for p value < 0.01, *** for p value < 0.001 and **** for p value < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
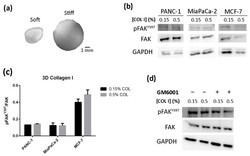
- Cell culture in 3D collagen type I gels. ( a ) Macroscopic view of collagen 3D constructs containing PANC-1 cells; ( b ) Western blot bands of pFAK Y397 and total FAK in PANC-1, MiaPaCa-2 and MCF-7 cells cultured in 0.15% (soft) and 0.5% (stiff) collagen gels; ( c ) densitometry of bands shown in ( b ) represented as pFAK Y397 /FAK; ( d ) Western blot bands of pFAK Y397 and total FAK in PANC-1 cells cultured in 0.15% (soft) and 0.5% (stiff) collagen gels in the presence of GM6001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Images of REF52 cells labeled for four sets of five focal-adhesion-associated components. REF52 cells, stably expressing beta 3 -integrin-GFP, were fixed 24 hours after plating and labeled for actin and paxillin, as well as for: (A) vinculin and alpha-actinin, (B) zyxin and alpha-actinin, (C) FAK and alpha-actinin, (D) vinculin and phosphotyrosine (PY). (A') REF52 cells treated for 3 hours with 100 uM of the Rho-kinase inhibitor Y-27632 and labeled as in (A). The fluorophores used here include: Cy5 (for the IR1 channel), Alexa-750 (IR2), Cy3 or Alexa-555 (red), GFP (green) and CPITC (blue). Images were acquired using selective excitation and emission filter sets for five fluorescent channels. Scale bar, 10 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
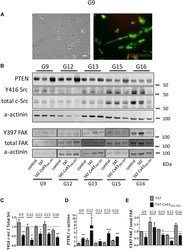
- Figure 1 TAT-Cx43 266-283 Targets c-Src, PTEN, and FAK in Primary GSCs (A) Phase-contrast images (left) and SOX-2 (red) and Nestin (green) immunostaining (right) of the same field showing that primary G9 GSCs express these GSC markers. Scale bar, 50 mum. (B) Primary G9, G12, G13, G15, and G16 GSCs were incubated with 50 muM TAT or 50 muM TAT-Cx43 266-283 . After 24 hr, the PTEN, total c-Src, Y416 c-Src, total FAK, and Y397 FAK levels were analyzed by western blotting. (C-E) Quantification of the Y416 c-Src/total c-Src (C), PTEN/alpha-actinin (D), and Y379 FAK/total FAK (E) ratios. The results were normalized with their corresponding controls (assigned a value of 1; dotted line) and are the means +- SEM of at least three independent experiments (ANOVA; *** p < 0.001, ** p < 0.01, * p < 0.05; TAT-Cx43 266-283 versus TAT). See also Figure S1 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
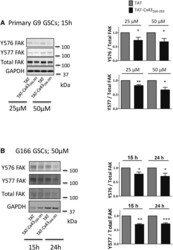
- Figure 2 TAT-Cx43 266-283 Reduces Y576 and Y577 FAK Phosphorylation in GSCs Primary G9 (A) or G166 (B) GSCs were incubated with TAT or TAT-Cx43 266-283 . Western blot analysis for FAK, Y576 FAK, and Y577 FAK. The results were normalized with their corresponding TAT (assigned a value of 1) and are the means +- SEM of at least four independent experiments ( *** p < 0.001, ** p < 0.01, * p < 0.05; TAT-Cx43 266-283 versus TAT, t test). See also Figure S2 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
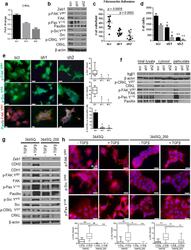
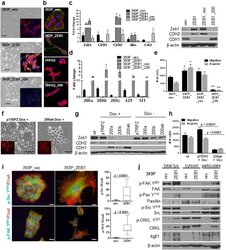
- Figure 1 Zeb1 expression induces EMT/miR-200 repression and FAK pathway activation in previously non-invasive cells. ( a ) Zeb1 expression causes a morphology change (middle panel) and is reversed upon re-expression of miR-200 (lower panel). Cells were stained with Integrin alpha6 (red), ZO-1 (green) and DAPI. ( b ) 393P cells with constitutive Zeb1 expression and 344SQ cells with constitutive miR-200 family expression grown on agarose and stained with phalloidin (red), DAPI and cortactin (green, lower panels). ( c ) Quantitative RT-PCR (left) and Western blot (right) analysis of EMT markers in the indicated cell lines. ( d ) Taqman RT-PCR for the miR-200 family members in the indicated cell lines. ( e ) In vitro Transwell migration and invasion assay for the 393P cell line panel. *p < 0.004, **p < 0.001 ( f ) Induction of miR-200a and b in H157 causes a morphology change. ( g ) Western Blot analysis of EMT markers in H157 + /- miR-200a and/or b. ( h ) In vitro Transwell migration and invasion assay for the inducible H157 cells. ( i ) Immunofluorescence of 393P_vec and 393P_ZEB1 cells stained for p-Src Y 418 , p-FAK Y 861 , and phalloidin (left); an average of 15-30 cells was counted for focal adhesions/cell with individual puncta considered a focal adhesion. Scale bar is 200 mum. (J) Western blot analysis of total cell lysates, cytosolic or membrane particulate fractions from 393P_vec and 393P_ZEB1 cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
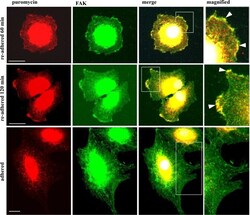
- Experimental details
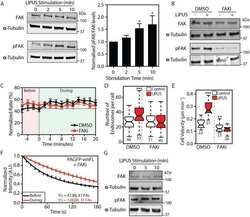
- Fig 9 Puromycin-terminated proteins colocalize with focal adhesion kinase (FAK). Confocal microscopy of synovial fibroblasts that were re-adhered for 60 and 120 min, fed with puromycin, double stained using anti-puromycin and FAK antibodies, followed by visualization with Alexa 488 and 594 secondary antibodies, showing colocalization of puromycylated proteins with FAK at the cell membrane during adhesion only (white arrows in magnified area). Objective 40x, size bar = 20 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Effect of DNDA on the expression of phospho and total aPKCs, and FAK in metastatic (H1299, A549) lung cancer cells. ( A ) Western blot analysis for the effect of DNDA in A549 and H1299 lung cancer cells following 3 days of treatment with DNDA 10muM on the expression of phospho and total PKC-iota, PKC-zeta, and FAK. Equal amounts of protein (30 mug) were loaded in SDS-PAGE as normalized by loading control beta-actin. ( B ) The bar graph depicts the densitometry of phospho PKC-iota, PKC-zeta, and FAK levels upon DNDA 10muM treatment in H1299 lung cancer cells and ( C ) A549 lung cancer cells. The data represents three independent experiments, Mean +- S.D. * represents p value where * < 0.05, ** < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
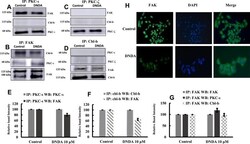
- Experimental details
- Figure 7 ( A-D ) Immunoprecipitation results showing the association of PKC-iota with FAK and FAK with Cbl-b. Initially, 3 mug of PKC-iota, PKC-zeta, FAK and Cbl-b were IP separately from whole cell lysates of control and DNDA treated A549 NSCLC cells and probed with various antibodies to determine the association between PKC-iota, PKC-zeta, FAK and Cbl-b. Immunoblots showed the association of PKC-iota with FAK, Cbl-b with FAK. In FAK IP samples, cleaved fragments of FAK were observed with the treatment of DNDA. There was no association found between PKC-zeta with FAK or Cbl-b. ( E-G ) Bar graphs show the densitometry of the associated proteins between control and DNDA treated cells ( H ) Immunofluorescence of FAK also complements FAK cleavage with treatment of DNDA when compared to the cells treated with vehicle (DMSO).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
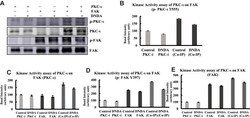
- Experimental details
- Figure 8 Kinase activity assay of PKC-iota on FAK phosphorylation. ( A ) A549 cells were treated with either DMSO or DNDA 10muM for 3 days. PKC-iota and FAK were IP (3 mug) separately and co-IP from whole cell extracts using specific antibodies. The proteins were then separated by SDS-PAGE, followed by immunoblotting to determine the p- PKC-iota at T555, PKC-i, p-FAK (Y-397) and FAK. The inhibition of PKC-iota activity by DNDA, inhibits the phosphorylation of FAK. ( B-E ) Densitometry was used to quantify the intensity of respective bands. The data represents three independent experiments; Mean +- S.D.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
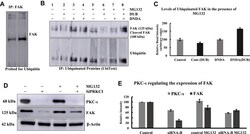
- Experimental details
- Figure 9 Determination of ubiquitination of FAK in A549 lung cancer cells by the treatment with DNDA 10muM for 3 days. ( A ) The IP of FAK (3 mug) was immunoblotted for ubiquitin and found that there was an increase in the FAK (125 kDa) band in the DNDA treated. ( B, C ) UbiTest was employed to determine the ubiquitination of FAK. The results showed the FAK (125 kDa) and cleaved FAK (100 kDa) were ubiquitinated with the DNDA treatment. ( D ) To explore the PKC-iota involvement in the ubiquitination of FAK, after transient transfection, cells were treated with 20muM proteasomal inhibitor MG-132 or DMSO for 4 hrs before collecting the lysates. The FAK protein expression was concurrently decreased when PKC-iota was repressed in the absence of MG132. However, FAK expression was increased when PKC-iota was repressed in the presence of MG132. ( E ) The intensity of respective bands were quantified by using densitometry. This result indicate that the ubiquitinated FAK undergoes proteasomal degradation and PKC-iota regulates this process.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunoprecipitation
Immunoprecipitation