Antibody data
- Antibody Data
- Antigen structure
- References [9]
- Comments [0]
- Validations
- Other assay [4]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MHCD45RA01 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD45RA Monoclonal Antibody (MEM-56), FITC
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Commonly used, FITC conjugates provide relatively high absorptivity, excellent fluorescence quantum yield, and good water solubility.
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Green dye
- Isotype
- IgG
- Antibody clone number
- MEM-56
- Vial size
- 500 µL
- Storage
- 4° C, store in dark
Submitted references In vitro differentiation of human pluripotent stem cells into the B lineage using OP9-MS5 co-culture.
Inflammation-driven deaminase deregulation fuels human pre-leukemia stem cell evolution.
Unravelling Intratumoral Heterogeneity through High-Sensitivity Single-Cell Mutational Analysis and Parallel RNA Sequencing.
FUNDAMANT: an interventional 72-week phase 1 follow-up study of AADvac1, an active immunotherapy against tau protein pathology in Alzheimer's disease.
DOT1L inhibition attenuates graft-versus-host disease by allogeneic T cells in adoptive immunotherapy models.
A Human IPS Model Implicates Embryonic B-Myeloid Fate Restriction as Developmental Susceptibility to B Acute Lymphoblastic Leukemia-Associated ETV6-RUNX1.
Transient stimulation expands superior antitumor T cells for adoptive therapy.
Prospective isolation of human clonogenic common myeloid progenitors.
CD3 hyporesponsiveness and in vitro apoptosis are features of T cells from both malignant and nonmalignant secondary lymphoid organs.
Richardson SE, Ghazanfari R, Chhetri J, Enver T, Böiers C
STAR protocols 2021 Jun 18;2(2):100420
STAR protocols 2021 Jun 18;2(2):100420
Inflammation-driven deaminase deregulation fuels human pre-leukemia stem cell evolution.
Jiang Q, Isquith J, Ladel L, Mark A, Holm F, Mason C, He Y, Mondala P, Oliver I, Pham J, Ma W, Reynoso E, Ali S, Morris IJ, Diep R, Nasamran C, Xu G, Sasik R, Rosenthal SB, Birmingham A, Coso S, Pineda G, Crews L, Donohoe ME, Venter JC, Whisenant T, Mesa RA, Alexandrov LB, Fisch KM, Jamieson C
Cell reports 2021 Jan 26;34(4):108670
Cell reports 2021 Jan 26;34(4):108670
Unravelling Intratumoral Heterogeneity through High-Sensitivity Single-Cell Mutational Analysis and Parallel RNA Sequencing.
Rodriguez-Meira A, Buck G, Clark SA, Povinelli BJ, Alcolea V, Louka E, McGowan S, Hamblin A, Sousos N, Barkas N, Giustacchini A, Psaila B, Jacobsen SEW, Thongjuea S, Mead AJ
Molecular cell 2019 Mar 21;73(6):1292-1305.e8
Molecular cell 2019 Mar 21;73(6):1292-1305.e8
FUNDAMANT: an interventional 72-week phase 1 follow-up study of AADvac1, an active immunotherapy against tau protein pathology in Alzheimer's disease.
Novak P, Schmidt R, Kontsekova E, Kovacech B, Smolek T, Katina S, Fialova L, Prcina M, Parrak V, Dal-Bianco P, Brunner M, Staffen W, Rainer M, Ondrus M, Ropele S, Smisek M, Sivak R, Zilka N, Winblad B, Novak M
Alzheimer's research & therapy 2018 Oct 24;10(1):108
Alzheimer's research & therapy 2018 Oct 24;10(1):108
DOT1L inhibition attenuates graft-versus-host disease by allogeneic T cells in adoptive immunotherapy models.
Kagoya Y, Nakatsugawa M, Saso K, Guo T, Anczurowski M, Wang CH, Butler MO, Arrowsmith CH, Hirano N
Nature communications 2018 May 15;9(1):1915
Nature communications 2018 May 15;9(1):1915
A Human IPS Model Implicates Embryonic B-Myeloid Fate Restriction as Developmental Susceptibility to B Acute Lymphoblastic Leukemia-Associated ETV6-RUNX1.
Böiers C, Richardson SE, Laycock E, Zriwil A, Turati VA, Brown J, Wray JP, Wang D, James C, Herrero J, Sitnicka E, Karlsson S, Smith AJH, Jacobsen SEW, Enver T
Developmental cell 2018 Feb 5;44(3):362-377.e7
Developmental cell 2018 Feb 5;44(3):362-377.e7
Transient stimulation expands superior antitumor T cells for adoptive therapy.
Kagoya Y, Nakatsugawa M, Ochi T, Cen Y, Guo T, Anczurowski M, Saso K, Butler MO, Hirano N
JCI insight 2017 Jan 26;2(2):e89580
JCI insight 2017 Jan 26;2(2):e89580
Prospective isolation of human clonogenic common myeloid progenitors.
Manz MG, Miyamoto T, Akashi K, Weissman IL
Proceedings of the National Academy of Sciences of the United States of America 2002 Sep 3;99(18):11872-7
Proceedings of the National Academy of Sciences of the United States of America 2002 Sep 3;99(18):11872-7
CD3 hyporesponsiveness and in vitro apoptosis are features of T cells from both malignant and nonmalignant secondary lymphoid organs.
Agrawal S, Marquet J, Delfau-Larue MH, Copie-Bergman C, Jouault H, Reyes F, Bensussan A, Farcet JP
The Journal of clinical investigation 1998 Nov 1;102(9):1715-23
The Journal of clinical investigation 1998 Nov 1;102(9):1715-23
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
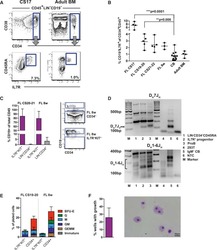
- Fig. 3 DOT1L inhibition delays the incidence of GVHD in a xenogeneic model. a CD3 + T cells were stimulated with aAPC/mOKT3, expanded for 12 days in the presence of 0.5 muM SGC0946 or DMSO, and subsequently infused into irradiated NSG mice (10 million T cells per mouse). b , c Relative body weight of the mouse compared to the weight prior to T-cell infusion. Sequential data at the indicated time points ( b ) and the body weight at day 14 ( c ) are shown ( n = 8 mice, unpaired two-sided t -test). d , e Kaplan-Meier analysis for more than 10% weight-loss-free survival ( d ) and overall survival ( e ) following T-cell transplantation ( n = 8 mice, log-rank test). The data shown are representative of three independent experiments. f Histological assessment of the indicated organs showing lymphocyte infiltration (hematoxylin and eosin staining) on day 21 following T-cell transplantation. Scale bar, 100 mum. g Human CD4 + or CD8 + T-cell chimerism in the peripheral blood 14 days following T-cell transfer ( n = 8 mice, unpaired two-sided t -test). h - j Surface expression of CD45RA, CD62L and CCR7 was analyzed in the persistent T cells within the peripheral blood on day 14. Representative FACS plots ( h ) and frequency of CD45RA + CD62L + CCR7 + cells within the CD4 + or CD8 + T-cell population ( i ) or within the total peripheral blood mononuclear cell population ( j ) are shown ( n = 8 mice, unpaired two-sided t -test). NS, not significant. Horizontal lines represent the means +-
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
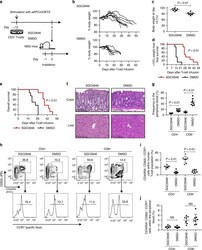
- Figure 8 Flow cytometry analysis from approximately day 31 of co-culture Cells were harvested after 3 weeks of MS5 co-culture and stained for surface markers. (A) Cells were gated for singlets and scatter and then for viability (7AAD) and CD45 expression as indicated in the figure (top panel). Expression of CD19 and CD34 is shown within the 7AAD - CD45 + fraction (second panel, left). The CD19 positive fraction is further gated for expression of CD10 and CD20 (right). Counting beads were used in this experiment and can be seen in the scatter plot in the top middle panel. (B) Cells were gated for singlets and scatter (as in Figure 8 A, plots not shown). Contour plot of viability (7AAD) and CD45 expression as shown in the figure (left). Further gating on 7AAD - CD45 + cells for B lymphoid (CD19) and, within the CD19 - gate, myeloid (CD11b and CD14) markers are shown to the right.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
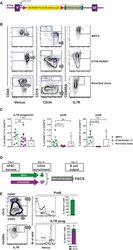
- Figure 5 An hIPSC Model of ETV6-RUNX1 Shows a Block in B Lineage Commitment (A) Genome engineering strategy. A constitutive knockin cassette, encoding a splice acceptor (SA), cDNA for RUNX1 exons II-VIII V5 tagged at the C terminus, and linked to the mVenus fluorescent reporter by a self-cleaving furin/T2A peptide is inserted by CRISPR-directed homologous recombination toward the 5' end of ETV6 intron V. The cassette is flanked by LoxP sites and includes a 3' triple stop/poly(A) tail followed by an FLP floxable NeoR + selection cassette. (B) Control MIFF3 (top panel), ETV6-RUNX1 hPSCs (middle panel) and reverted ETV6-RUNX1 hPSCs (bottom panel) were cultured according to Figure 4 A and analyzed by flow cytometry for Venus reporter (left), proB (CD34 + CD19 + ) and preB (CD34 - CD19 + ) cells (middle), and IL-7R + progenitor (right). Viable cells were gated as indicated. (C) Frequencies of IL-7R + progenitor, proB, and preB cells in MIFF3 cells compared with ETV6-RUNX1 iPSCs and reverted ETV6-RUNX1 hPSCs analyzed at D27-31. Percentage of Venus - (MIFF3/reverted clone) or Venus + (ETV6-RUNX1 hIPSC) CD45 + blood cells. Each dot represents one replicate. Only samples with 8,000 cells or more were considered. Mean +- SD. Statistics were performed using MIFF3 as control group, ns, not significant. (D) Schematic drawing of in vitro competitive assay. Equal numbers of ETV6-RUNX1 and wild-type MIFF3-derived CD34 + cells from D10 of OP9 co-culture were seeded onto MS5 in B differentiati
- Conjugate
- Green dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry Other assay
Other assay