Antibody data
- Antibody Data
- Antigen structure
- References [8]
- Comments [0]
- Validations
- Flow cytometry [2]
- Other assay [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 11-9979-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD45RA Monoclonal Antibody (JS-83), FITC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Immunogen sequence: VLPPEAITI LEAEPIRMLE IEGERELPEV SRRELDLLIA EEEEAILLEI PRLPPPAPAE VEGIGEALGP EELRLTGWEP GALLMEVTPP EELRLPAPPS PERRPPVPPP PRRRRRRRLL FWDKETQISP EKFQEQLQTR AHCWECPMVQ PPERTIRGPA ELFRTPTLSG WLPPELLGLW THCAQPPPKA LRRELPEEAA AEEERRKIEV PSEIEVPREA LEPSVPLMVS LEISLEAAEE EKSRISLIPP EERWAWPEVE APEAPALPVV PELPEVPMEM PLVLPPELEL LSLEAVHRAV ALELQANREP DFSSLVSPLS PRRMAARVFY LLLVLSAQQI LHVKQEKPYG RLLIQPGPRF H (188-548 aa encoded by BC010887)
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Green dye
- Isotype
- IgG
- Antibody clone number
- JS-83
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references IL-32γ potentiates tumor immunity in melanoma.
Functionally specialized human CD4(+) T-cell subsets express physicochemically distinct TCRs.
The Changing Landscape of Naive T Cell Receptor Repertoire With Human Aging.
B cells from African American lupus patients exhibit an activated phenotype.
Protective genotypes in HIV infection reflect superior function of KIR3DS1+ over KIR3DL1+ CD8+ T cells.
Age-related decrease in TCR repertoire diversity measured with deep and normalized sequence profiling.
Mother and child T cell receptor repertoires: deep profiling study.
Divergent adaptive and innate immunological responses are observed in humans following blunt trauma.
Gruber T, Kremenovic M, Sadozai H, Rombini N, Baeriswyl L, Maibach F, Modlin RL, Gilliet M, von Werdt D, Hunger RE, Seyed Jafari SM, Parisi G, Abril-Rodriguez G, Ribas A, Schenk M
JCI insight 2020 Sep 17;5(18)
JCI insight 2020 Sep 17;5(18)
Functionally specialized human CD4(+) T-cell subsets express physicochemically distinct TCRs.
Kasatskaya SA, Ladell K, Egorov ES, Miners KL, Davydov AN, Metsger M, Staroverov DB, Matveyshina EK, Shagina IA, Mamedov IZ, Izraelson M, Shelyakin PV, Britanova OV, Price DA, Chudakov DM
eLife 2020 Dec 8;9
eLife 2020 Dec 8;9
The Changing Landscape of Naive T Cell Receptor Repertoire With Human Aging.
Egorov ES, Kasatskaya SA, Zubov VN, Izraelson M, Nakonechnaya TO, Staroverov DB, Angius A, Cucca F, Mamedov IZ, Rosati E, Franke A, Shugay M, Pogorelyy MV, Chudakov DM, Britanova OV
Frontiers in immunology 2018;9:1618
Frontiers in immunology 2018;9:1618
B cells from African American lupus patients exhibit an activated phenotype.
Menard LC, Habte S, Gonsiorek W, Lee D, Banas D, Holloway DA, Manjarrez-Orduno N, Cunningham M, Stetsko D, Casano F, Kansal S, Davis PM, Carman J, Zhang CK, Abidi F, Furie R, Nadler SG, Suchard SJ
JCI insight 2016 Jun 16;1(9):e87310
JCI insight 2016 Jun 16;1(9):e87310
Protective genotypes in HIV infection reflect superior function of KIR3DS1+ over KIR3DL1+ CD8+ T cells.
Zipperlen K, Gallant M, Stapleton S, Heath J, Barrett L, Grant M
Immunology and cell biology 2015 Jan;93(1):67-76
Immunology and cell biology 2015 Jan;93(1):67-76
Age-related decrease in TCR repertoire diversity measured with deep and normalized sequence profiling.
Britanova OV, Putintseva EV, Shugay M, Merzlyak EM, Turchaninova MA, Staroverov DB, Bolotin DA, Lukyanov S, Bogdanova EA, Mamedov IZ, Lebedev YB, Chudakov DM
Journal of immunology (Baltimore, Md. : 1950) 2014 Mar 15;192(6):2689-98
Journal of immunology (Baltimore, Md. : 1950) 2014 Mar 15;192(6):2689-98
Mother and child T cell receptor repertoires: deep profiling study.
Putintseva EV, Britanova OV, Staroverov DB, Merzlyak EM, Turchaninova MA, Shugay M, Bolotin DA, Pogorelyy MV, Mamedov IZ, Bobrynina V, Maschan M, Lebedev YB, Chudakov DM
Frontiers in immunology 2013;4:463
Frontiers in immunology 2013;4:463
Divergent adaptive and innate immunological responses are observed in humans following blunt trauma.
Kasten KR, Goetzman HS, Reid MR, Rasper AM, Adediran SG, Robinson CT, Cave CM, Solomkin JS, Lentsch AB, Johannigman JA, Caldwell CC
BMC immunology 2010 Jan 25;11:4
BMC immunology 2010 Jan 25;11:4
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
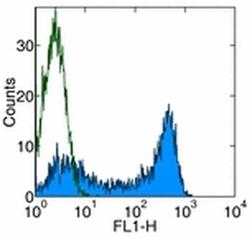
- Experimental details
- Normal human peripheral blood cells were stained with Mouse IgG1 K Isotype Control FITC (Product # 11-4714-42) (open histogram) or Anti-Human CD45RA FITC (filled histogram). Cells in the lymphocyte population were used for analysis.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Normal human peripheral blood cells were stained with Mouse IgG1 kappa Isotype Control, FITC (Product # 11-4714-42) (blue histogram) or CD45RA Monoclonal Antibody, FITC (purple histogram). Cells in the lymphocyte gate were used for analysis.
- Conjugate
- Green dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
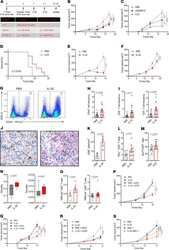
- Experimental details
- Figure 4 IL-32 induces a systemic CD8 + T cell-mediated tumor-specific immune response. ( A ) Experimental setup for in vivo tumor treatments. MC38 and B16F10 were inoculated in C57BL/6J mice, and 4T1 tumors in BALB/c mice ( A - O ). ( B ) Growth curves of IL-32-, cGAMP-, or PBS-treated primary and ( C ) contralateral, nontreated B16F10 melanomas. * P < 0.05, ** P < 0.01, *** P < 0.001. Data are representative of 4 independent experiments, with n = 6 mice per group. ( D ) Kaplan-Meier survival curves of B16F10-bearing mice treated with IL-32 ( n = 6) or PBS ( n = 8). ( E ) Representative growth curves of IL-32-treated and untreated MC38 colon adenocarcinoma ( n = 6) and ( F ) orthotopic 4T1 breast tumors ( n = 10). ( G-O ) On day 14, the primary treated tumors and spleens were harvested for flow cytometric analyses, IHC or TCRbeta chain sequencing. ( G ) Representative flow cytometry plots displaying frequencies of CD45 + immune cells for each treatment group and ( H ) their quantification shown as relative frequencies ( n = 18). ( I ) Relative frequencies of CD8 + and CD4 + T cells as a percentage of live cells ( n = 18). ( J ) Representative immunohistochemical staining and ( K ) morphometric enumeration as cells/mm 2 of CD8 + T cells ( n = 3). Scale bar: 20 mum. ( L ) Relative frequencies of IFN-gamma + cells as percentage of CD8 + T cells ( n = 12). ( M ) Frequencies of Nur77-GFP + cells of CD8 + T cells, as determined by flow cytometry in B16F10-inoculated and PBS- ( n =
- Conjugate
- Green dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry