Antibody data
- Antibody Data
- Antigen structure
- References [194]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [75]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 12-0452-83 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD45R (B220) Monoclonal Antibody (RA3-6B2), PE, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The RA3-6B2 monoclonal antibody reacts with exon A-restricted isoform of mouse CD45, a 220 kDa surface molecule. CD45R/B220 epitope is mainly expressed by the B cell lineage from early Pro-B to mature B cells. However, some activated T cells, lymphokine activated killer cells (LAK), NK cell progenitors in the bone marrow, and T cells of the lpr/lpr mutant mouse also express this antigen. Applications Reported: The RA3-6B2 antibody has been reported for use in flow cytometric analysis. Applications Tested: The RA3-6B2 antibody has been tested by flow cytometric analysis of mouse splenocytes. This can be used at less than or equal to 0.5 µg per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest. Excitation: 488-561 nm; Emission: 578 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human, Mouse
- Host
- Rat
- Conjugate
- Yellow dye
- Isotype
- IgG
- Antibody clone number
- RA3-6B2
- Vial size
- 200 µg
- Concentration
- 0.2 mg/mL
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references XIAP promotes the expansion and limits the contraction of CD8 T cell response through cell extrinsic and intrinsic mechanisms respectively.
IL6Myc mouse is an immunocompetent model for the development of aggressive multiple myeloma.
Loss of phosphatidylinositol-4-phosphate 5-kinase type-1 gamma (Pip5k1c) in mesenchymal stem cells leads to osteopenia by impairing bone remodeling.
Long-term culture-expanded alveolar macrophages restore their full epigenetic identity after transfer in vivo.
Sphk2 deletion is involved in structural abnormalities and Th17 response but does not aggravate colon inflammation induced by sub-chronic stress.
Tet-mediated DNA demethylation regulates specification of hematopoietic stem and progenitor cells during mammalian embryogenesis.
Lenvatinib for effectively treating antiangiogenic drug-resistant nasopharyngeal carcinoma.
Krüppel-like factor 2 controls IgA plasma cell compartmentalization and IgA responses.
Transient expansion and myofibroblast conversion of adipogenic lineage precursors mediate bone marrow repair after radiation.
A rapid CRISPR competitive assay for in vitro and in vivo discovery of potential drug targets affecting the hematopoietic system.
Polyvinyl alcohol hydrolysis rate and molecular weight influence human and murine HSC activity ex vivo.
Type 2 diabetic mice enter a state of spontaneous hibernation-like suspended animation following accumulation of uric acid.
Metabolic preconditioning in CD4+ T cells restores inducible immune tolerance in lupus-prone mice.
Impaired HA-specific T follicular helper cell and antibody responses to influenza vaccination are linked to inflammation in humans.
The folate cycle enzyme MTHFD2 induces cancer immune evasion through PD-L1 up-regulation.
Aged hematopoietic stem cells are refractory to bloodborne systemic rejuvenation interventions.
Defining the Mechanistic Correlates of Protection Conferred by Whole-Cell Vaccination against Pseudomonas aeruginosa Acute Murine Pneumonia.
Induction of Foxp3 and activation of Tregs by HSP gp96 for treatment of autoimmune diseases.
Fascin inhibitor increases intratumoral dendritic cell activation and anti-cancer immunity.
Loss of FGFR3 Delays Acute Myeloid Leukemogenesis by Programming Weakly Pathogenic CD117-Positive Leukemia Stem-Like Cells.
Myeloid Cell CK2 Regulates Inflammation and Resistance to Bacterial Infection.
Shp1 Loss Enhances Macrophage Effector Function and Promotes Anti-Tumor Immunity.
The Innate Immune Response to Infection Induces Erythropoietin-Dependent Replenishment of the Dendritic Cell Compartment.
The lysophospholipase D enzyme Gdpd3 is required to maintain chronic myelogenous leukaemia stem cells.
Restraining Lysosomal Activity Preserves Hematopoietic Stem Cell Quiescence and Potency.
Deregulated Notch and Wnt signaling activates early-stage myeloid regeneration pathways in leukemia.
Analysis of Myeloid Cells in Mouse Tissues with Flow Cytometry.
Doxycycline Reduces Scar Thickness and Improves Collagen Architecture.
Protective role of neuronal and lymphoid cannabinoid CB(2) receptors in neuropathic pain.
RNA Demethylase ALKBH5 Selectively Promotes Tumorigenesis and Cancer Stem Cell Self-Renewal in Acute Myeloid Leukemia.
mDia1/3-dependent actin polymerization spatiotemporally controls LAT phosphorylation by Zap70 at the immune synapse.
Modest Declines in Proteome Quality Impair Hematopoietic Stem Cell Self-Renewal.
Prodrug-Loaded Zirconium Carbide Nanosheets as a Novel Biophotonic Nanoplatform for Effective Treatment of Cancer.
BAD inactivation exacerbates rheumatoid arthritis pathology by promoting survival of sublining macrophages.
SARS-CoV-2 mRNA Vaccines Foster Potent Antigen-Specific Germinal Center Responses Associated with Neutralizing Antibody Generation.
Inhibition of EZH2 ameliorates bacteria-induced liver injury by repressing RUNX1 in dendritic cells.
Conditional antibody expression to avoid central B cell deletion in humanized HIV-1 vaccine mouse models.
Tpl2 Protects Against Fulminant Hepatitis Through Mobilization of Myeloid-Derived Suppressor Cells.
Ubiquitination of RIPK1 suppresses programmed cell death by regulating RIPK1 kinase activation during embryogenesis.
Incorporating B cell activating factor (BAFF) into the membrane of rabies virus (RABV) particles improves the speed and magnitude of vaccine-induced antibody responses.
Bacteroides fragilis polysaccharide A induces IL-10 secreting B and T cells that prevent viral encephalitis.
A Regulatory Circuit Controlling the Dynamics of NFκB cRel Transitions B Cells from Proliferation to Plasma Cell Differentiation.
p53-TP53-Induced Glycolysis Regulator Mediated Glycolytic Suppression Attenuates DNA Damage and Genomic Instability in Fanconi Anemia Hematopoietic Stem Cells.
N-Cadherin-Expressing Bone and Marrow Stromal Progenitor Cells Maintain Reserve Hematopoietic Stem Cells.
Aging Induces an Nlrp3 Inflammasome-Dependent Expansion of Adipose B Cells That Impairs Metabolic Homeostasis.
Cutaneous TRPV1(+) Neurons Trigger Protective Innate Type 17 Anticipatory Immunity.
Ptpn21 Controls Hematopoietic Stem Cell Homeostasis and Biomechanics.
Metabolic Imaging Reveals a Unique Preference of Symmetric Cell Division and Homing of Leukemia-Initiating Cells in an Endosteal Niche.
The Hematopoietic Oxidase NOX2 Regulates Self-Renewal of Leukemic Stem Cells.
Proapoptotic BIM Impacts B Lymphoid Homeostasis by Limiting the Survival of Mature B Cells in a Cell-Autonomous Manner.
De Novo Fatty Acid Synthesis During Mycobacterial Infection Is a Prerequisite for the Function of Highly Proliferative T Cells, But Not for Dendritic Cells or Macrophages.
Role of TFH Cells in Promoting T Helper 17-Induced Neuroinflammation.
Dysregulated TRAF3 and BCL2 Expression Promotes Multiple Classes of Mature Non-hodgkin B Cell Lymphoma in Mice.
ATF3 Sustains IL-22-Induced STAT3 Phosphorylation to Maintain Mucosal Immunity Through Inhibiting Phosphatases.
The RNA-binding protein ROD1/PTBP3 cotranscriptionally defines AID-loading sites to mediate antibody class switch in mammalian genomes.
Live imaging and tracking of genome regions in CRISPR/dCas9 knock-in mice.
Retinoid-Sensitive Epigenetic Regulation of the Hoxb Cluster Maintains Normal Hematopoiesis and Inhibits Leukemogenesis.
Host Tumor Suppressor p18(INK4c) Functions as a Potent Cell-Intrinsic Inhibitor of Murine Gammaherpesvirus 68 Reactivation and Pathogenesis.
Adrenergic nerve degeneration in bone marrow drives aging of the hematopoietic stem cell niche.
Expression of mutant Asxl1 perturbs hematopoiesis and promotes susceptibility to leukemic transformation.
Mitophagy in Intestinal Epithelial Cells Triggers Adaptive Immunity during Tumorigenesis.
miR-143/145 differentially regulate hematopoietic stem and progenitor activity through suppression of canonical TGFβ signaling.
Tanshinone IIA and Astragaloside IV promote the angiogenesis of mesenchymal stem cell-derived endothelial cell-like cells via upregulation of Cx37, Cx40 and Cx43.
Betaine Ameliorates Experimental Autoimmune Encephalomyelitis by Inhibiting Dendritic Cell-Derived IL-6 Production and Th17 Differentiation.
Clonal expansion and myeloid leukemia progression modeled by multiplex gene editing of murine hematopoietic progenitor cells.
Inactivation of mTORC1 Signaling in Osterix-Expressing Cells Impairs B-cell Differentiation.
Investigation of tumor-tumor interactions in a double human cervical carcinoma xenograft model in nude mice.
A Circular RNA Protects Dormant Hematopoietic Stem Cells from DNA Sensor cGAS-Mediated Exhaustion.
The Interleukin (IL)-1R1 pathway is a critical negative regulator of PyMT-mediated mammary tumorigenesis and pulmonary metastasis.
Multiple functional therapeutic effects of TnP: A small stable synthetic peptide derived from fish venom in a mouse model of multiple sclerosis.
Mononuclear Phagocytes Are Dispensable for Cardiac Remodeling in Established Pressure-Overload Heart Failure.
DNA-binding of the Tet-transactivator curtails antigen-induced lymphocyte activation in mice.
EZH2 enables germinal centre formation through epigenetic silencing of CDKN1A and an Rb-E2F1 feedback loop.
Interleukin-33-Activated Islet-Resident Innate Lymphoid Cells Promote Insulin Secretion through Myeloid Cell Retinoic Acid Production.
Ceramide synthesis regulates T cell activity and GVHD development.
A high-yield isolation and enrichment strategy for human lung microvascular endothelial cells.
Differential cytokine contributions of perivascular haematopoietic stem cell niches.
Decoupling the Functional Pleiotropy of Stem Cell Factor by Tuning c-Kit Signaling.
Rad52 competes with Ku70/Ku86 for binding to S-region DSB ends to modulate antibody class-switch DNA recombination.
Paeoniflorin Ameliorates Experimental Autoimmune Encephalomyelitis via Inhibition of Dendritic Cell Function and Th17 Cell Differentiation.
Irgm1 coordinately regulates autoimmunity and host defense at select mucosal surfaces.
Epithelial Fli1 deficiency drives systemic autoimmunity and fibrosis: Possible roles in scleroderma.
Eradication of Tumors through Simultaneous Ablation of CD276/B7-H3-Positive Tumor Cells and Tumor Vasculature.
Distinctive Expression of Bcl-2 Factors in Regulatory T Cells Determines a Pharmacological Target to Induce Immunological Tolerance.
Enhancement of Oral Tolerance Induction in DO11.10 Mice by Lactobacillus gasseri OLL2809 via Increase of Effector Regulatory T Cells.
Opposing Effects of Fasting Metabolism on Tissue Tolerance in Bacterial and Viral Inflammation.
Flow cytometric analysis of inflammatory and resident myeloid populations in mouse ocular inflammatory models.
Mast cell depletion in the preclinical phase of collagen-induced arthritis reduces clinical outcome by lowering the inflammatory cytokine profile.
NLRP2 controls age-associated maternal fertility.
Murine iPSC-Derived Macrophages as a Tool for Disease Modeling of Hereditary Pulmonary Alveolar Proteinosis due to Csf2rb Deficiency.
Resident T Cells Are Unable To Control Herpes Simplex Virus-1 Activity in the Brain Ependymal Region during Latency.
Sphingosine 1-Phosphate Receptor 3-Deficient Dendritic Cells Modulate Splenic Responses to Ischemia-Reperfusion Injury.
Kappa opioid receptor activation alleviates experimental autoimmune encephalomyelitis and promotes oligodendrocyte-mediated remyelination.
Metabolic reprogramming of alloantigen-activated T cells after hematopoietic cell transplantation.
Immunostimulatory Effects Triggered by Enterococcus faecalis CECT7121 Probiotic Strain Involve Activation of Dendritic Cells and Interferon-Gamma Production.
The proximal J kappa germline-transcript promoter facilitates receptor editing through control of ordered recombination.
Mitochondrial ATP transporter Ant2 depletion impairs erythropoiesis and B lymphopoiesis.
Acute DNA damage activates the tumour suppressor p53 to promote radiation-induced lymphoma.
CD93 Marks a Non-Quiescent Human Leukemia Stem Cell Population and Is Required for Development of MLL-Rearranged Acute Myeloid Leukemia.
Endothelial Gata5 transcription factor regulates blood pressure.
Disruption of p21-activated kinase 1 gene diminishes atherosclerosis in apolipoprotein E-deficient mice.
BAFF activation of the ERK5 MAP kinase pathway regulates B cell survival.
The development of mixed cryoglobulinemia in Capillaria hepatica-infected mice is associated with the capillaria antigen-induced selective proliferation of splenic B-1a cells in response to interleukin-5 stimulation.
B cells modulate systemic responses to Pneumocystis murina lung infection and protect on-demand hematopoiesis via T cell-independent innate mechanisms when type I interferon signaling is absent.
Aging-associated inflammation promotes selection for adaptive oncogenic events in B cell progenitors.
Lactobacillus gasseri SBT2055 induces TGF-β expression in dendritic cells and activates TLR2 signal to produce IgA in the small intestine.
B-cell-intrinsic hepatitis C virus expression leads to B-cell-lymphomagenesis and induction of NF-κB signalling.
Acquired initiating mutations in early hematopoietic cells of CLL patients.
Age-dependent enterocyte invasion and microcolony formation by Salmonella.
Targeting multiple types of tumors using NKG2D-coated iron oxide nanoparticles.
Transcription elongation factor ELL2 drives Ig secretory-specific mRNA production and the unfolded protein response.
Immunosuppression by N-methyl-D-aspartate receptor antagonists is mediated through inhibition of Kv1.3 and KCa3.1 channels in T cells.
TRAF6 is a critical regulator of LMP1 functions in vivo.
TLR7 ligand augments GM-CSF-initiated antitumor immunity through activation of plasmacytoid dendritic cells.
Antigen capture and archiving by lymphatic endothelial cells following vaccination or viral infection.
Oral combined therapy with probiotics and alloantigen induces B cell-dependent long-lasting specific tolerance.
Histone deacetylase inhibitors upregulate B cell microRNAs that silence AID and Blimp-1 expression for epigenetic modulation of antibody and autoantibody responses.
Rhof promotes murine marginal zone B cell development.
The Vi capsular polysaccharide enables Salmonella enterica serovar typhi to evade microbe-guided neutrophil chemotaxis.
IκBε is a key regulator of B cell expansion by providing negative feedback on cRel and RelA in a stimulus-specific manner.
TDP2-dependent non-homologous end-joining protects against topoisomerase II-induced DNA breaks and genome instability in cells and in vivo.
Properties of immature myeloid progenitors with nitric-oxide-dependent immunosuppressive activity isolated from bone marrow of tumor-free mice.
Prolactin levels correlate with abnormal B cell maturation in MRL and MRL/lpr mouse models of systemic lupus erythematosus-like disease.
c-Rel is an essential transcription factor for the development of acute graft-versus-host disease in mice.
Usp16 contributes to somatic stem-cell defects in Down's syndrome.
IL-33 signaling regulates innate and adaptive immunity to Cryptococcus neoformans.
B lymphocytes trigger monocyte mobilization and impair heart function after acute myocardial infarction.
Homologous recombination into the eosinophil peroxidase locus generates a strain of mice expressing Cre recombinase exclusively in eosinophils.
Reciprocal effects of rab7 deletion in activated and neglected T cells.
Candida albicans stimulates in vivo differentiation of haematopoietic stem and progenitor cells towards macrophages by a TLR2-dependent signalling.
Streptomycin-induced inflammation enhances Escherichia coli gut colonization through nitrate respiration.
Novel apidaecin 1b analogs with superior serum stabilities for treatment of infections by gram-negative pathogens.
Interferon gamma and T cells inhibit osteogenesis induced by allogeneic mesenchymal stromal cells.
HDL and Glut1 inhibition reverse a hypermetabolic state in mouse models of myeloproliferative disorders.
MHC class II-dependent B cell APC function is required for induction of CNS autoimmunity independent of myelin-specific antibodies.
Interaction of CD99 with its paralog CD99L2 positively regulates CD99L2 trafficking to cell surfaces.
Minicircle DNA is superior to plasmid DNA in eliciting antigen-specific CD8+ T-cell responses.
11β-hydroxysteroid dehydrogenase type 1 deficiency in bone marrow-derived cells reduces atherosclerosis.
Role of suppressor of cytokine signaling-1 in murine atherosclerosis.
Retention of anergy and inhibition of antibody responses during acute γ herpesvirus 68 infection.
Activation-induced cytidine deaminase-initiated off-target DNA breaks are detected and resolved during S phase.
Myeloid-derived suppressor cells from tumor-bearing mice impair TGF-β-induced differentiation of CD4+CD25+FoxP3+ Tregs from CD4+CD25-FoxP3- T cells.
Trogocytosis of CD80 and CD86 by induced regulatory T cells.
Up-regulation of a HOXA-PBX3 homeobox-gene signature following down-regulation of miR-181 is associated with adverse prognosis in patients with cytogenetically abnormal AML.
IKKα-mediated signaling circuitry regulates early B lymphopoiesis during hematopoiesis.
Transcription factor RORα is critical for nuocyte development.
Metalloproteinase-mediated Shedding of Integrin β2 promotes macrophage efflux from inflammatory sites.
Critical role of B cell lymphoma 10 in BAFF-regulated NF-κB activation and survival of anergic B cells.
Candida albicans induces selective development of macrophages and monocyte derived dendritic cells by a TLR2 dependent signalling.
PAD4-mediated neutrophil extracellular trap formation is not required for immunity against influenza infection.
Study of camelpox virus pathogenesis in athymic nude mice.
Antiasthmatic drugs targeting the cysteinyl leukotriene receptor 1 alleviate central nervous system inflammatory cell infiltration and pathogenesis of experimental autoimmune encephalomyelitis.
Online nanoflow multidimensional fractionation for high efficiency phosphopeptide analysis.
Prevention of GVHD while sparing GVL effect by targeting Th1 and Th17 transcription factor T-bet and RORγt in mice.
CONTINUOUS REPOPULATION OF LYMPHOCYTE SUBSETS IN TRANSPLANTED MYCOBACTERIAL GRANULOMAS.
Induction of Noxa-mediated apoptosis by modified vaccinia virus Ankara depends on viral recognition by cytosolic helicases, leading to IRF-3/IFN-β-dependent induction of pro-apoptotic Noxa.
Indoleamine 2,3-dioxygenase and regulatory dendritic cells contribute to the allograft protection induced by infusion of donor-specific splenic stromal cells.
Uracil residues dependent on the deaminase AID in immunoglobulin gene variable and switch regions.
Protein kinase D2 is an essential regulator of murine myoblast differentiation.
Auto-antibody production and glomerulonephritis in congenic Slamf1-/- and Slamf2-/- [B6.129] but not in Slamf1-/- and Slamf2-/- [BALB/c.129] mice.
Nuclear export of the NF-κB inhibitor IκBα is required for proper B cell and secondary lymphoid tissue formation.
mGluR6 transcripts in non-neuronal tissues.
AID dysregulation in lupus-prone MRL/Fas(lpr/lpr) mice increases class switch DNA recombination and promotes interchromosomal c-Myc/IgH loci translocations: modulation by HoxC4.
Th-1 lymphocytes induce dendritic cell tumor killing activity by an IFN-γ-dependent mechanism.
TLR8 deficiency leads to autoimmunity in mice.
Estrogen receptors bind to and activate the HOXC4/HoxC4 promoter to potentiate HoxC4-mediated activation-induced cytosine deaminase induction, immunoglobulin class switch DNA recombination, and somatic hypermutation.
Impaired apoptotic cell clearance in the germinal center by Mer-deficient tingible body macrophages leads to enhanced antibody-forming cell and germinal center responses.
HVCN1 modulates BCR signal strength via regulation of BCR-dependent generation of reactive oxygen species.
Increased hematopoietic cells in the mertk-/- mouse peritoneal cavity: a result of augmented migration.
CD11c+ dendritic cells and B cells contribute to the tumoricidal activity of anti-DR5 antibody therapy in established tumors.
CD4 T-cell suppression by cells from Toxoplasma gondii-infected retinas is mediated by surface protein PD-L1.
B cell depletion reduces the development of atherosclerosis in mice.
Gene expression profiling of leukemia stem cells.
Critical role of the interleukin-17/interleukin-17 receptor axis in regulating host susceptibility to respiratory infection with Chlamydia species.
HoxC4 binds to the promoter of the cytidine deaminase AID gene to induce AID expression, class-switch DNA recombination and somatic hypermutation.
A critical role of TAK1 in B-cell receptor-mediated nuclear factor kappaB activation.
STAT6-mediated suppression of erythropoiesis in an experimental model of malarial anemia.
Lupus-prone MRL/faslpr/lpr mice display increased AID expression and extensive DNA lesions, comprising deletions and insertions, in the immunoglobulin locus: concurrent upregulation of somatic hypermutation and class switch DNA recombination.
The cellular niche of Listeria monocytogenes infection changes rapidly in the spleen.
Differential roles of migratory and resident DCs in T cell priming after mucosal or skin HSV-1 infection.
BLNK suppresses pre-B-cell leukemogenesis through inhibition of JAK3.
4-1BB functions as a survival factor in dendritic cells.
Optimization of methods to study pulmonary dendritic cell migration reveals distinct capacities of DC subsets to acquire soluble versus particulate antigen.
IL-7 specifies B cell fate at the common lymphoid progenitor to pre-proB transition stage by maintaining early B cell factor expression.
IL-7 specifies B cell fate at the common lymphoid progenitor to pre-proB transition stage by maintaining early B cell factor expression.
p27 deficiency cooperates with Bcl-2 but not Bax to promote T-cell lymphoma.
Phospholipase Cgamma2 contributes to light-chain gene activation and receptor editing.
Immature hematopoietic cells display selective requirements for adhesion- and degranulation-promoting adaptor protein in development and homeostatsis.
STAT3 positively regulates an early step in B-cell development.
Distinct roles of phosphoinositide-3 kinase and phospholipase Cgamma2 in B-cell receptor-mediated signal transduction.
Essential role of phospholipase C gamma 2 in early B-cell development and Myc-mediated lymphomagenesis.
A role for brain-derived neurotrophic factor in B cell development.
Impaired immune responses and B-cell proliferation in mice lacking the Id3 gene.
Identification of monoclonal antibodies for immunohistochemical staining of feline B lymphocytes in frozen and formalin-fixed paraffin-embedded tissues.
Thakker P, Ariana A, Hajjar S, Cai D, Rijal D, Sad S
PLoS pathogens 2023 Jun;19(6):e1011455
PLoS pathogens 2023 Jun;19(6):e1011455
IL6Myc mouse is an immunocompetent model for the development of aggressive multiple myeloma.
Pisano MD, Sun F, Cheng Y, Parashar D, Zhou V, Jing X, Sompallae R, Abrudan J, Zimmermann MT, Mathison A, Janz S, Pufall MA
Haematologica 2023 Dec 1;108(12):3372-3383
Haematologica 2023 Dec 1;108(12):3372-3383
Loss of phosphatidylinositol-4-phosphate 5-kinase type-1 gamma (Pip5k1c) in mesenchymal stem cells leads to osteopenia by impairing bone remodeling.
Yan Q, Gao H, Yao Q, Ling K, Xiao G
The Journal of biological chemistry 2022 Mar;298(3):101639
The Journal of biological chemistry 2022 Mar;298(3):101639
Long-term culture-expanded alveolar macrophages restore their full epigenetic identity after transfer in vivo.
Subramanian S, Busch CJ, Molawi K, Geirsdottir L, Maurizio J, Vargas Aguilar S, Belahbib H, Gimenez G, Yuda RAA, Burkon M, Favret J, Gholamhosseinian Najjar S, de Laval B, Kandalla PK, Sarrazin S, Alexopoulou L, Sieweke MH
Nature immunology 2022 Mar;23(3):458-468
Nature immunology 2022 Mar;23(3):458-468
Sphk2 deletion is involved in structural abnormalities and Th17 response but does not aggravate colon inflammation induced by sub-chronic stress.
Martín-Hernández D, Gutiérrez IL, González-Prieto M, MacDowell KS, Robledo-Montaña J, Tendilla-Beltrán H, Calleja-Rodríguez N, Bris ÁG, Ulecia-Morón C, Moreno B, Caso JR, García-Bueno B, Rodrigues-Mascarenhas S, Marín-Jiménez I, Leza JC, Menchén L
Scientific reports 2022 Mar 8;12(1):4073
Scientific reports 2022 Mar 8;12(1):4073
Tet-mediated DNA demethylation regulates specification of hematopoietic stem and progenitor cells during mammalian embryogenesis.
Ma L, Tang Q, Gao X, Lee J, Lei R, Suzuki M, Zheng D, Ito K, Frenette PS, Dawlaty MM
Science advances 2022 Mar 4;8(9):eabm3470
Science advances 2022 Mar 4;8(9):eabm3470
Lenvatinib for effectively treating antiangiogenic drug-resistant nasopharyngeal carcinoma.
Sun Q, Wang Y, Ji H, Sun X, Xie S, Chen L, Li S, Zeng W, Chen R, Tang Q, Zuo J, Hou L, Hosaka K, Lu Y, Liu Y, Ye Y, Yang Y
Cell death & disease 2022 Aug 19;13(8):724
Cell death & disease 2022 Aug 19;13(8):724
Krüppel-like factor 2 controls IgA plasma cell compartmentalization and IgA responses.
Wittner J, Schulz SR, Steinmetz TD, Berges J, Hauke M, Channell WM, Cunningham AF, Hauser AE, Hutloff A, Mielenz D, Jäck HM, Schuh W
Mucosal immunology 2022 Apr;15(4):668-682
Mucosal immunology 2022 Apr;15(4):668-682
Transient expansion and myofibroblast conversion of adipogenic lineage precursors mediate bone marrow repair after radiation.
Zhong L, Yao L, Holdreith N, Yu W, Gui T, Miao Z, Elkaim Y, Li M, Gong Y, Pacifici M, Maity A, Busch TM, Joeng KS, Cengel K, Seale P, Tong W, Qin L
JCI insight 2022 Apr 8;7(7)
JCI insight 2022 Apr 8;7(7)
A rapid CRISPR competitive assay for in vitro and in vivo discovery of potential drug targets affecting the hematopoietic system.
Shen Y, Jiang L, Iyer VS, Raposo B, Dubnovitsky A, Boddul SV, Kasza Z, Wermeling F
Computational and structural biotechnology journal 2021;19:5360-5370
Computational and structural biotechnology journal 2021;19:5360-5370
Polyvinyl alcohol hydrolysis rate and molecular weight influence human and murine HSC activity ex vivo.
Sudo K, Yamazaki S, Wilkinson AC, Nakauchi H, Nakamura Y
Stem cell research 2021 Oct;56:102531
Stem cell research 2021 Oct;56:102531
Type 2 diabetic mice enter a state of spontaneous hibernation-like suspended animation following accumulation of uric acid.
Zhao Y, Cheng R, Zhao Y, Ge W, Yang Y, Ding Z, Xu X, Wang Z, Wu Z, Zhang J
The Journal of biological chemistry 2021 Oct;297(4):101166
The Journal of biological chemistry 2021 Oct;297(4):101166
Metabolic preconditioning in CD4+ T cells restores inducible immune tolerance in lupus-prone mice.
Wilson CS, Stocks BT, Hoopes EM, Rhoads JP, McNew KL, Major AS, Moore DJ
JCI insight 2021 Oct 8;6(19)
JCI insight 2021 Oct 8;6(19)
Impaired HA-specific T follicular helper cell and antibody responses to influenza vaccination are linked to inflammation in humans.
Hill DL, Whyte CE, Innocentin S, Lee JL, Dooley J, Wang J, James EA, Lee JC, Kwok WW, Zand MS, Liston A, Carr EJ, Linterman MA
eLife 2021 Nov 2;10
eLife 2021 Nov 2;10
The folate cycle enzyme MTHFD2 induces cancer immune evasion through PD-L1 up-regulation.
Shang M, Yang H, Yang R, Chen T, Fu Y, Li Y, Fang X, Zhang K, Zhang J, Li H, Cao X, Gu J, Xiao J, Zhang Q, Liu X, Yu Q, Wang T
Nature communications 2021 Mar 29;12(1):1940
Nature communications 2021 Mar 29;12(1):1940
Aged hematopoietic stem cells are refractory to bloodborne systemic rejuvenation interventions.
Ho TT, Dellorusso PV, Verovskaya EV, Bakker ST, Flach J, Smith LK, Ventura PB, Lansinger OM, Hérault A, Zhang SY, Kang YA, Mitchell CA, Villeda SA, Passegué E
The Journal of experimental medicine 2021 Jul 5;218(7)
The Journal of experimental medicine 2021 Jul 5;218(7)
Defining the Mechanistic Correlates of Protection Conferred by Whole-Cell Vaccination against Pseudomonas aeruginosa Acute Murine Pneumonia.
Sen-Kilic E, Blackwood CB, Huckaby AB, Horspool AM, Weaver KL, Malkowski AC, Witt WT, Bevere JR, Damron FH, Barbier M
Infection and immunity 2021 Jan 19;89(2)
Infection and immunity 2021 Jan 19;89(2)
Induction of Foxp3 and activation of Tregs by HSP gp96 for treatment of autoimmune diseases.
Xu Y, Liu E, Xie X, Wang J, Zheng H, Ju Y, Chen L, Li C, Zhou X, Li Z, Li X, Meng S
iScience 2021 Dec 17;24(12):103445
iScience 2021 Dec 17;24(12):103445
Fascin inhibitor increases intratumoral dendritic cell activation and anti-cancer immunity.
Wang Y, Song M, Liu M, Zhang G, Zhang X, Li MO, Ma X, Zhang JJ, Huang XY
Cell reports 2021 Apr 6;35(1):108948
Cell reports 2021 Apr 6;35(1):108948
Loss of FGFR3 Delays Acute Myeloid Leukemogenesis by Programming Weakly Pathogenic CD117-Positive Leukemia Stem-Like Cells.
Guo C, Ran Q, Sun C, Zhou T, Yang X, Zhang J, Pang S, Xiao Y
Frontiers in pharmacology 2020;11:632809
Frontiers in pharmacology 2020;11:632809
Myeloid Cell CK2 Regulates Inflammation and Resistance to Bacterial Infection.
Larson SR, Bortell N, Illies A, Crisler WJ, Matsuda JL, Lenz LL
Frontiers in immunology 2020;11:590266
Frontiers in immunology 2020;11:590266
Shp1 Loss Enhances Macrophage Effector Function and Promotes Anti-Tumor Immunity.
Myers DR, Abram CL, Wildes D, Belwafa A, Welsh AMN, Schulze CJ, Choy TJ, Nguyen T, Omaque N, Hu Y, Singh M, Hansen R, Goldsmith MA, Quintana E, Smith JAM, Lowell CA
Frontiers in immunology 2020;11:576310
Frontiers in immunology 2020;11:576310
The Innate Immune Response to Infection Induces Erythropoietin-Dependent Replenishment of the Dendritic Cell Compartment.
Einwächter H, Heiseke A, Schlitzer A, Gasteiger G, Adler H, Voehringer D, Manz MG, Ruzsics Z, Dölken L, Koszinowski UH, Sparwasser T, Reindl W, Jordan S
Frontiers in immunology 2020;11:1627
Frontiers in immunology 2020;11:1627
The lysophospholipase D enzyme Gdpd3 is required to maintain chronic myelogenous leukaemia stem cells.
Naka K, Ochiai R, Matsubara E, Kondo C, Yang KM, Hoshii T, Araki M, Araki K, Sotomaru Y, Sasaki K, Mitani K, Kim DW, Ooshima A, Kim SJ
Nature communications 2020 Sep 17;11(1):4681
Nature communications 2020 Sep 17;11(1):4681
Restraining Lysosomal Activity Preserves Hematopoietic Stem Cell Quiescence and Potency.
Liang R, Arif T, Kalmykova S, Kasianov A, Lin M, Menon V, Qiu J, Bernitz JM, Moore K, Lin F, Benson DL, Tzavaras N, Mahajan M, Papatsenko D, Ghaffari S
Cell stem cell 2020 Mar 5;26(3):359-376.e7
Cell stem cell 2020 Mar 5;26(3):359-376.e7
Deregulated Notch and Wnt signaling activates early-stage myeloid regeneration pathways in leukemia.
Kang YA, Pietras EM, Passegué E
The Journal of experimental medicine 2020 Mar 2;217(3)
The Journal of experimental medicine 2020 Mar 2;217(3)
Analysis of Myeloid Cells in Mouse Tissues with Flow Cytometry.
Liu Z, Gu Y, Shin A, Zhang S, Ginhoux F
STAR protocols 2020 Jun 19;1(1):100029
STAR protocols 2020 Jun 19;1(1):100029
Doxycycline Reduces Scar Thickness and Improves Collagen Architecture.
Moore AL, desJardins-Park HE, Duoto BA, Mascharak S, Murphy MP, Irizarry DM, Foster DS, Jones RE, Barnes LA, Marshall CD, Ransom RC, Wernig G, Longaker MT
Annals of surgery 2020 Jul;272(1):183-193
Annals of surgery 2020 Jul;272(1):183-193
Protective role of neuronal and lymphoid cannabinoid CB(2) receptors in neuropathic pain.
Cabañero D, Ramírez-López A, Drews E, Schmöle A, Otte DM, Wawrzczak-Bargiela A, Huerga Encabo H, Kummer S, Ferrer-Montiel A, Przewlocki R, Zimmer A, Maldonado R
eLife 2020 Jul 20;9
eLife 2020 Jul 20;9
RNA Demethylase ALKBH5 Selectively Promotes Tumorigenesis and Cancer Stem Cell Self-Renewal in Acute Myeloid Leukemia.
Shen C, Sheng Y, Zhu AC, Robinson S, Jiang X, Dong L, Chen H, Su R, Yin Z, Li W, Deng X, Chen Y, Hu YC, Weng H, Huang H, Prince E, Cogle CR, Sun M, Zhang B, Chen CW, Marcucci G, He C, Qian Z, Chen J
Cell stem cell 2020 Jul 2;27(1):64-80.e9
Cell stem cell 2020 Jul 2;27(1):64-80.e9
mDia1/3-dependent actin polymerization spatiotemporally controls LAT phosphorylation by Zap70 at the immune synapse.
Thumkeo D, Katsura Y, Nishimura Y, Kanchanawong P, Tohyama K, Ishizaki T, Kitajima S, Takahashi C, Hirata T, Watanabe N, Krummel MF, Narumiya S
Science advances 2020 Jan;6(1):eaay2432
Science advances 2020 Jan;6(1):eaay2432
Modest Declines in Proteome Quality Impair Hematopoietic Stem Cell Self-Renewal.
Hidalgo San Jose L, Sunshine MJ, Dillingham CH, Chua BA, Kruta M, Hong Y, Hatters DM, Signer RAJ
Cell reports 2020 Jan 7;30(1):69-80.e6
Cell reports 2020 Jan 7;30(1):69-80.e6
Prodrug-Loaded Zirconium Carbide Nanosheets as a Novel Biophotonic Nanoplatform for Effective Treatment of Cancer.
Liu Q, Xie Z, Qiu M, Shim I, Yang Y, Xie S, Yang Q, Wang D, Chen S, Fan T, Ding B, Guo Z, Adah D, Yao X, Zhang Y, Wu H, Wu Z, Wei C, Wang H, Kim HS, Zou Q, Yan Q, Cai Z, Kim JS, Liu LP, Zhang H, Cao Y
Advanced science (Weinheim, Baden-Wurttemberg, Germany) 2020 Dec;7(24):2001191
Advanced science (Weinheim, Baden-Wurttemberg, Germany) 2020 Dec;7(24):2001191
BAD inactivation exacerbates rheumatoid arthritis pathology by promoting survival of sublining macrophages.
Li J, Zhang L, Zheng Y, Shao R, Liang Q, Yu W, Wang H, Zou W, Wang D, Xiang J, Lin A
eLife 2020 Dec 3;9
eLife 2020 Dec 3;9
SARS-CoV-2 mRNA Vaccines Foster Potent Antigen-Specific Germinal Center Responses Associated with Neutralizing Antibody Generation.
Lederer K, Castaño D, Gómez Atria D, Oguin TH 3rd, Wang S, Manzoni TB, Muramatsu H, Hogan MJ, Amanat F, Cherubin P, Lundgreen KA, Tam YK, Fan SHY, Eisenlohr LC, Maillard I, Weissman D, Bates P, Krammer F, Sempowski GD, Pardi N, Locci M
Immunity 2020 Dec 15;53(6):1281-1295.e5
Immunity 2020 Dec 15;53(6):1281-1295.e5
Inhibition of EZH2 ameliorates bacteria-induced liver injury by repressing RUNX1 in dendritic cells.
Wang Y, Wang Q, Wang B, Gu Y, Yu H, Yang W, Ren X, Qian F, Zhao X, Xiao Y, Zhang Y, Jin M, Zhu M
Cell death & disease 2020 Dec 1;11(11):1024
Cell death & disease 2020 Dec 1;11(11):1024
Conditional antibody expression to avoid central B cell deletion in humanized HIV-1 vaccine mouse models.
Tian M, McGovern K, Cheng HL, Waddicor P, Rieble L, Dao M, Chen Y, Kimble MT, Cantor E, Manfredonia N, Judson R, Chapdelaine-Williams A, Cain DW, Haynes BF, Alt FW
Proceedings of the National Academy of Sciences of the United States of America 2020 Apr 7;117(14):7929-7940
Proceedings of the National Academy of Sciences of the United States of America 2020 Apr 7;117(14):7929-7940
Tpl2 Protects Against Fulminant Hepatitis Through Mobilization of Myeloid-Derived Suppressor Cells.
Xu J, Pei S, Wang Y, Liu J, Qian Y, Huang M, Zhang Y, Xiao Y
Frontiers in immunology 2019;10:1980
Frontiers in immunology 2019;10:1980
Ubiquitination of RIPK1 suppresses programmed cell death by regulating RIPK1 kinase activation during embryogenesis.
Zhang X, Zhang H, Xu C, Li X, Li M, Wu X, Pu W, Zhou B, Wang H, Li D, Ding Q, Ying H, Wang H, Zhang H
Nature communications 2019 Sep 13;10(1):4158
Nature communications 2019 Sep 13;10(1):4158
Incorporating B cell activating factor (BAFF) into the membrane of rabies virus (RABV) particles improves the speed and magnitude of vaccine-induced antibody responses.
Plummer JR, McGettigan JP
PLoS neglected tropical diseases 2019 Nov;13(11):e0007800
PLoS neglected tropical diseases 2019 Nov;13(11):e0007800
Bacteroides fragilis polysaccharide A induces IL-10 secreting B and T cells that prevent viral encephalitis.
Ramakrishna C, Kujawski M, Chu H, Li L, Mazmanian SK, Cantin EM
Nature communications 2019 May 14;10(1):2153
Nature communications 2019 May 14;10(1):2153
A Regulatory Circuit Controlling the Dynamics of NFκB cRel Transitions B Cells from Proliferation to Plasma Cell Differentiation.
Roy K, Mitchell S, Liu Y, Ohta S, Lin YS, Metzig MO, Nutt SL, Hoffmann A
Immunity 2019 Mar 19;50(3):616-628.e6
Immunity 2019 Mar 19;50(3):616-628.e6
p53-TP53-Induced Glycolysis Regulator Mediated Glycolytic Suppression Attenuates DNA Damage and Genomic Instability in Fanconi Anemia Hematopoietic Stem Cells.
Li X, Wu L, Zopp M, Kopelov S, Du W
Stem cells (Dayton, Ohio) 2019 Jul;37(7):937-947
Stem cells (Dayton, Ohio) 2019 Jul;37(7):937-947
N-Cadherin-Expressing Bone and Marrow Stromal Progenitor Cells Maintain Reserve Hematopoietic Stem Cells.
Zhao M, Tao F, Venkatraman A, Li Z, Smith SE, Unruh J, Chen S, Ward C, Qian P, Perry JM, Marshall H, Wang J, He XC, Li L
Cell reports 2019 Jan 15;26(3):652-669.e6
Cell reports 2019 Jan 15;26(3):652-669.e6
Aging Induces an Nlrp3 Inflammasome-Dependent Expansion of Adipose B Cells That Impairs Metabolic Homeostasis.
Camell CD, Günther P, Lee A, Goldberg EL, Spadaro O, Youm YH, Bartke A, Hubbard GB, Ikeno Y, Ruddle NH, Schultze J, Dixit VD
Cell metabolism 2019 Dec 3;30(6):1024-1039.e6
Cell metabolism 2019 Dec 3;30(6):1024-1039.e6
Cutaneous TRPV1(+) Neurons Trigger Protective Innate Type 17 Anticipatory Immunity.
Cohen JA, Edwards TN, Liu AW, Hirai T, Jones MR, Wu J, Li Y, Zhang S, Ho J, Davis BM, Albers KM, Kaplan DH
Cell 2019 Aug 8;178(4):919-932.e14
Cell 2019 Aug 8;178(4):919-932.e14
Ptpn21 Controls Hematopoietic Stem Cell Homeostasis and Biomechanics.
Ni F, Yu WM, Wang X, Fay ME, Young KM, Qiu Y, Lam WA, Sulchek TA, Cheng T, Scadden DT, Qu CK
Cell stem cell 2019 Apr 4;24(4):608-620.e6
Cell stem cell 2019 Apr 4;24(4):608-620.e6
Metabolic Imaging Reveals a Unique Preference of Symmetric Cell Division and Homing of Leukemia-Initiating Cells in an Endosteal Niche.
Hao X, Gu H, Chen C, Huang D, Zhao Y, Xie L, Zou Y, Shu HS, Zhang Y, He X, Lai X, Zhang X, Zhou BO, Zhang CC, Chen GQ, Yu Z, Yang Y, Zheng J
Cell metabolism 2019 Apr 2;29(4):950-965.e6
Cell metabolism 2019 Apr 2;29(4):950-965.e6
The Hematopoietic Oxidase NOX2 Regulates Self-Renewal of Leukemic Stem Cells.
Adane B, Ye H, Khan N, Pei S, Minhajuddin M, Stevens BM, Jones CL, D'Alessandro A, Reisz JA, Zaberezhnyy V, Gasparetto M, Ho TC, Kelly KK, Myers JR, Ashton JM, Siegenthaler J, Kume T, Campbell EL, Pollyea DA, Becker MW, Jordan CT
Cell reports 2019 Apr 2;27(1):238-254.e6
Cell reports 2019 Apr 2;27(1):238-254.e6
Proapoptotic BIM Impacts B Lymphoid Homeostasis by Limiting the Survival of Mature B Cells in a Cell-Autonomous Manner.
Liu R, King A, Bouillet P, Tarlinton DM, Strasser A, Heierhorst J
Frontiers in immunology 2018;9:592
Frontiers in immunology 2018;9:592
De Novo Fatty Acid Synthesis During Mycobacterial Infection Is a Prerequisite for the Function of Highly Proliferative T Cells, But Not for Dendritic Cells or Macrophages.
Stüve P, Minarrieta L, Erdmann H, Arnold-Schrauf C, Swallow M, Guderian M, Krull F, Hölscher A, Ghorbani P, Behrends J, Abraham WR, Hölscher C, Sparwasser TD, Berod L
Frontiers in immunology 2018;9:495
Frontiers in immunology 2018;9:495
Role of TFH Cells in Promoting T Helper 17-Induced Neuroinflammation.
Quinn JL, Kumar G, Agasing A, Ko RM, Axtell RC
Frontiers in immunology 2018;9:382
Frontiers in immunology 2018;9:382
Dysregulated TRAF3 and BCL2 Expression Promotes Multiple Classes of Mature Non-hodgkin B Cell Lymphoma in Mice.
Perez-Chacon G, Adrados M, Vallejo-Cremades MT, Lefebvre S, Reed JC, Zapata JM
Frontiers in immunology 2018;9:3114
Frontiers in immunology 2018;9:3114
ATF3 Sustains IL-22-Induced STAT3 Phosphorylation to Maintain Mucosal Immunity Through Inhibiting Phosphatases.
Glal D, Sudhakar JN, Lu HH, Liu MC, Chiang HY, Liu YC, Cheng CF, Shui JW
Frontiers in immunology 2018;9:2522
Frontiers in immunology 2018;9:2522
The RNA-binding protein ROD1/PTBP3 cotranscriptionally defines AID-loading sites to mediate antibody class switch in mammalian genomes.
Chen J, Cai Z, Bai M, Yu X, Zhang C, Cao C, Hu X, Wang L, Su R, Wang D, Wang L, Yao Y, Ye R, Hou B, Yu Y, Yu S, Li J, Xue Y
Cell research 2018 Oct;28(10):981-995
Cell research 2018 Oct;28(10):981-995
Live imaging and tracking of genome regions in CRISPR/dCas9 knock-in mice.
Duan J, Lu G, Hong Y, Hu Q, Mai X, Guo J, Si X, Wang F, Zhang Y
Genome biology 2018 Nov 8;19(1):192
Genome biology 2018 Nov 8;19(1):192
Retinoid-Sensitive Epigenetic Regulation of the Hoxb Cluster Maintains Normal Hematopoiesis and Inhibits Leukemogenesis.
Qian P, De Kumar B, He XC, Nolte C, Gogol M, Ahn Y, Chen S, Li Z, Xu H, Perry JM, Hu D, Tao F, Zhao M, Han Y, Hall K, Peak A, Paulson A, Zhao C, Venkatraman A, Box A, Perera A, Haug JS, Parmely T, Li H, Krumlauf R, Li L
Cell stem cell 2018 May 3;22(5):740-754.e7
Cell stem cell 2018 May 3;22(5):740-754.e7
Host Tumor Suppressor p18(INK4c) Functions as a Potent Cell-Intrinsic Inhibitor of Murine Gammaherpesvirus 68 Reactivation and Pathogenesis.
Niemeyer BF, Oko LM, Medina EM, Oldenburg DG, White DW, Cool CD, Clambey ET, van Dyk LF
Journal of virology 2018 Mar 15;92(6)
Journal of virology 2018 Mar 15;92(6)
Adrenergic nerve degeneration in bone marrow drives aging of the hematopoietic stem cell niche.
Maryanovich M, Zahalka AH, Pierce H, Pinho S, Nakahara F, Asada N, Wei Q, Wang X, Ciero P, Xu J, Leftin A, Frenette PS
Nature medicine 2018 Jun;24(6):782-791
Nature medicine 2018 Jun;24(6):782-791
Expression of mutant Asxl1 perturbs hematopoiesis and promotes susceptibility to leukemic transformation.
Nagase R, Inoue D, Pastore A, Fujino T, Hou HA, Yamasaki N, Goyama S, Saika M, Kanai A, Sera Y, Horikawa S, Ota Y, Asada S, Hayashi Y, Kawabata KC, Takeda R, Tien HF, Honda H, Abdel-Wahab O, Kitamura T
The Journal of experimental medicine 2018 Jun 4;215(6):1729-1747
The Journal of experimental medicine 2018 Jun 4;215(6):1729-1747
Mitophagy in Intestinal Epithelial Cells Triggers Adaptive Immunity during Tumorigenesis.
Ziegler PK, Bollrath J, Pallangyo CK, Matsutani T, Canli Ö, De Oliveira T, Diamanti MA, Müller N, Gamrekelashvili J, Putoczki T, Horst D, Mankan AK, Öner MG, Müller S, Müller-Höcker J, Kirchner T, Slotta-Huspenina J, Taketo MM, Reinheckel T, Dröse S, Larner AC, Wels WS, Ernst M, Greten TF, Arkan MC, Korn T, Wirth D, Greten FR
Cell 2018 Jun 28;174(1):88-101.e16
Cell 2018 Jun 28;174(1):88-101.e16
miR-143/145 differentially regulate hematopoietic stem and progenitor activity through suppression of canonical TGFβ signaling.
Lam J, van den Bosch M, Wegrzyn J, Parker J, Ibrahim R, Slowski K, Chang L, Martinez-Høyer S, Condorelli G, Boldin M, Deng Y, Umlandt P, Fuller M, Karsan A
Nature communications 2018 Jun 20;9(1):2418
Nature communications 2018 Jun 20;9(1):2418
Tanshinone IIA and Astragaloside IV promote the angiogenesis of mesenchymal stem cell-derived endothelial cell-like cells via upregulation of Cx37, Cx40 and Cx43.
Li Z, Zhang S, Cao L, Li W, Ye YC, Shi ZX, Wang ZR, Sun LX, Wang JW, Jia LT, Wang W
Experimental and therapeutic medicine 2018 Feb;15(2):1847-1854
Experimental and therapeutic medicine 2018 Feb;15(2):1847-1854
Betaine Ameliorates Experimental Autoimmune Encephalomyelitis by Inhibiting Dendritic Cell-Derived IL-6 Production and Th17 Differentiation.
Yang C, Lai W, Zhou J, Zheng X, Cai Y, Yang W, Xie S, Gao Y, Du C
Journal of immunology (Baltimore, Md. : 1950) 2018 Feb 15;200(4):1316-1324
Journal of immunology (Baltimore, Md. : 1950) 2018 Feb 15;200(4):1316-1324
Clonal expansion and myeloid leukemia progression modeled by multiplex gene editing of murine hematopoietic progenitor cells.
Shi X, Kitano A, Jiang Y, Luu V, Hoegenauer KA, Nakada D
Experimental hematology 2018 Aug;64:33-44.e5
Experimental hematology 2018 Aug;64:33-44.e5
Inactivation of mTORC1 Signaling in Osterix-Expressing Cells Impairs B-cell Differentiation.
Wang Y, Xiao M, Tao C, Chen J, Wang Z, Yang J, Chen Z, Zou Z, Liu A, Cai D, Jiang Y, Ding C, Li M, Bai X
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research 2018 Apr;33(4):732-742
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research 2018 Apr;33(4):732-742
Investigation of tumor-tumor interactions in a double human cervical carcinoma xenograft model in nude mice.
Mertens B, Cristina de Araujo Nogueira T, Topalis D, Stranska R, Snoeck R, Andrei G
Oncotarget 2018 Apr 24;9(31):21978-22000
Oncotarget 2018 Apr 24;9(31):21978-22000
A Circular RNA Protects Dormant Hematopoietic Stem Cells from DNA Sensor cGAS-Mediated Exhaustion.
Xia P, Wang S, Ye B, Du Y, Li C, Xiong Z, Qu Y, Fan Z
Immunity 2018 Apr 17;48(4):688-701.e7
Immunity 2018 Apr 17;48(4):688-701.e7
The Interleukin (IL)-1R1 pathway is a critical negative regulator of PyMT-mediated mammary tumorigenesis and pulmonary metastasis.
Dagenais M, Dupaul-Chicoine J, Douglas T, Champagne C, Morizot A, Saleh M
Oncoimmunology 2017;6(3):e1287247
Oncoimmunology 2017;6(3):e1287247
Multiple functional therapeutic effects of TnP: A small stable synthetic peptide derived from fish venom in a mouse model of multiple sclerosis.
Komegae EN, Souza TA, Grund LZ, Lima C, Lopes-Ferreira M
PloS one 2017;12(2):e0171796
PloS one 2017;12(2):e0171796
Mononuclear Phagocytes Are Dispensable for Cardiac Remodeling in Established Pressure-Overload Heart Failure.
Patel B, Ismahil MA, Hamid T, Bansal SS, Prabhu SD
PloS one 2017;12(1):e0170781
PloS one 2017;12(1):e0170781
DNA-binding of the Tet-transactivator curtails antigen-induced lymphocyte activation in mice.
Ottina E, Peperzak V, Schoeler K, Carrington E, Sgonc R, Pellegrini M, Preston S, Herold MJ, Strasser A, Villunger A
Nature communications 2017 Oct 18;8(1):1028
Nature communications 2017 Oct 18;8(1):1028
EZH2 enables germinal centre formation through epigenetic silencing of CDKN1A and an Rb-E2F1 feedback loop.
Béguelin W, Rivas MA, Calvo Fernández MT, Teater M, Purwada A, Redmond D, Shen H, Challman MF, Elemento O, Singh A, Melnick AM
Nature communications 2017 Oct 12;8(1):877
Nature communications 2017 Oct 12;8(1):877
Interleukin-33-Activated Islet-Resident Innate Lymphoid Cells Promote Insulin Secretion through Myeloid Cell Retinoic Acid Production.
Dalmas E, Lehmann FM, Dror E, Wueest S, Thienel C, Borsigova M, Stawiski M, Traunecker E, Lucchini FC, Dapito DH, Kallert SM, Guigas B, Pattou F, Kerr-Conte J, Maechler P, Girard JP, Konrad D, Wolfrum C, Böni-Schnetzler M, Finke D, Donath MY
Immunity 2017 Nov 21;47(5):928-942.e7
Immunity 2017 Nov 21;47(5):928-942.e7
Ceramide synthesis regulates T cell activity and GVHD development.
Sofi MH, Heinrichs J, Dany M, Nguyen H, Dai M, Bastian D, Schutt S, Wu Y, Daenthanasanmak A, Gencer S, Zivkovic A, Szulc Z, Stark H, Liu C, Chang YJ, Ogretmen B, Yu XZ
JCI insight 2017 May 18;2(10)
JCI insight 2017 May 18;2(10)
A high-yield isolation and enrichment strategy for human lung microvascular endothelial cells.
Gaskill C, Majka SM
Pulmonary circulation 2017 Mar;7(1):108-116
Pulmonary circulation 2017 Mar;7(1):108-116
Differential cytokine contributions of perivascular haematopoietic stem cell niches.
Asada N, Kunisaki Y, Pierce H, Wang Z, Fernandez NF, Birbrair A, Ma'ayan A, Frenette PS
Nature cell biology 2017 Mar;19(3):214-223
Nature cell biology 2017 Mar;19(3):214-223
Decoupling the Functional Pleiotropy of Stem Cell Factor by Tuning c-Kit Signaling.
Ho CCM, Chhabra A, Starkl P, Schnorr PJ, Wilmes S, Moraga I, Kwon HS, Gaudenzio N, Sibilano R, Wehrman TS, Gakovic M, Sockolosky JT, Tiffany MR, Ring AM, Piehler J, Weissman IL, Galli SJ, Shizuru JA, Garcia KC
Cell 2017 Mar 9;168(6):1041-1052.e18
Cell 2017 Mar 9;168(6):1041-1052.e18
Rad52 competes with Ku70/Ku86 for binding to S-region DSB ends to modulate antibody class-switch DNA recombination.
Zan H, Tat C, Qiu Z, Taylor JR, Guerrero JA, Shen T, Casali P
Nature communications 2017 Feb 8;8:14244
Nature communications 2017 Feb 8;8:14244
Paeoniflorin Ameliorates Experimental Autoimmune Encephalomyelitis via Inhibition of Dendritic Cell Function and Th17 Cell Differentiation.
Zhang H, Qi Y, Yuan Y, Cai L, Xu H, Zhang L, Su B, Nie H
Scientific reports 2017 Feb 6;7:41887
Scientific reports 2017 Feb 6;7:41887
Irgm1 coordinately regulates autoimmunity and host defense at select mucosal surfaces.
Azzam KM, Madenspacher JH, Cain DW, Lai L, Gowdy KM, Rai P, Janardhan K, Clayton N, Cunningham W, Jensen H, Patel PS, Kearney JF, Taylor GA, Fessler MB
JCI insight 2017 Aug 17;2(16)
JCI insight 2017 Aug 17;2(16)
Epithelial Fli1 deficiency drives systemic autoimmunity and fibrosis: Possible roles in scleroderma.
Takahashi T, Asano Y, Sugawara K, Yamashita T, Nakamura K, Saigusa R, Ichimura Y, Toyama T, Taniguchi T, Akamata K, Noda S, Yoshizaki A, Tsuruta D, Trojanowska M, Sato S
The Journal of experimental medicine 2017 Apr 3;214(4):1129-1151
The Journal of experimental medicine 2017 Apr 3;214(4):1129-1151
Eradication of Tumors through Simultaneous Ablation of CD276/B7-H3-Positive Tumor Cells and Tumor Vasculature.
Seaman S, Zhu Z, Saha S, Zhang XM, Yang MY, Hilton MB, Morris K, Szot C, Morris H, Swing DA, Tessarollo L, Smith SW, Degrado S, Borkin D, Jain N, Scheiermann J, Feng Y, Wang Y, Li J, Welsch D, DeCrescenzo G, Chaudhary A, Zudaire E, Klarmann KD, Keller JR, Dimitrov DS, St Croix B
Cancer cell 2017 Apr 10;31(4):501-515.e8
Cancer cell 2017 Apr 10;31(4):501-515.e8
Distinctive Expression of Bcl-2 Factors in Regulatory T Cells Determines a Pharmacological Target to Induce Immunological Tolerance.
Gabriel SS, Bon N, Chen J, Wekerle T, Bushell A, Fehr T, Cippà PE
Frontiers in immunology 2016;7:73
Frontiers in immunology 2016;7:73
Enhancement of Oral Tolerance Induction in DO11.10 Mice by Lactobacillus gasseri OLL2809 via Increase of Effector Regulatory T Cells.
Aoki-Yoshida A, Yamada K, Hachimura S, Sashihara T, Ikegami S, Shimizu M, Totsuka M
PloS one 2016;11(7):e0158643
PloS one 2016;11(7):e0158643
Opposing Effects of Fasting Metabolism on Tissue Tolerance in Bacterial and Viral Inflammation.
Wang A, Huen SC, Luan HH, Yu S, Zhang C, Gallezot JD, Booth CJ, Medzhitov R
Cell 2016 Sep 8;166(6):1512-1525.e12
Cell 2016 Sep 8;166(6):1512-1525.e12
Flow cytometric analysis of inflammatory and resident myeloid populations in mouse ocular inflammatory models.
Liyanage SE, Gardner PJ, Ribeiro J, Cristante E, Sampson RD, Luhmann UF, Ali RR, Bainbridge JW
Experimental eye research 2016 Oct;151:160-70
Experimental eye research 2016 Oct;151:160-70
Mast cell depletion in the preclinical phase of collagen-induced arthritis reduces clinical outcome by lowering the inflammatory cytokine profile.
van der Velden D, Lagraauw HM, Wezel A, Launay P, Kuiper J, Huizinga TW, Toes RE, Bot I, Stoop JN
Arthritis research & therapy 2016 Jun 13;18(1):138
Arthritis research & therapy 2016 Jun 13;18(1):138
NLRP2 controls age-associated maternal fertility.
Kuchmiy AA, D'Hont J, Hochepied T, Lamkanfi M
The Journal of experimental medicine 2016 Dec 12;213(13):2851-2860
The Journal of experimental medicine 2016 Dec 12;213(13):2851-2860
Murine iPSC-Derived Macrophages as a Tool for Disease Modeling of Hereditary Pulmonary Alveolar Proteinosis due to Csf2rb Deficiency.
Mucci A, Kunkiel J, Suzuki T, Brennig S, Glage S, Kühnel MP, Ackermann M, Happle C, Kuhn A, Schambach A, Trapnell BC, Hansen G, Moritz T, Lachmann N
Stem cell reports 2016 Aug 9;7(2):292-305
Stem cell reports 2016 Aug 9;7(2):292-305
Resident T Cells Are Unable To Control Herpes Simplex Virus-1 Activity in the Brain Ependymal Region during Latency.
Menendez CM, Jinkins JK, Carr DJ
Journal of immunology (Baltimore, Md. : 1950) 2016 Aug 15;197(4):1262-75
Journal of immunology (Baltimore, Md. : 1950) 2016 Aug 15;197(4):1262-75
Sphingosine 1-Phosphate Receptor 3-Deficient Dendritic Cells Modulate Splenic Responses to Ischemia-Reperfusion Injury.
Bajwa A, Huang L, Kurmaeva E, Gigliotti JC, Ye H, Miller J, Rosin DL, Lobo PI, Okusa MD
Journal of the American Society of Nephrology : JASN 2016 Apr;27(4):1076-90
Journal of the American Society of Nephrology : JASN 2016 Apr;27(4):1076-90
Kappa opioid receptor activation alleviates experimental autoimmune encephalomyelitis and promotes oligodendrocyte-mediated remyelination.
Du C, Duan Y, Wei W, Cai Y, Chai H, Lv J, Du X, Zhu J, Xie X
Nature communications 2016 Apr 4;7:11120
Nature communications 2016 Apr 4;7:11120
Metabolic reprogramming of alloantigen-activated T cells after hematopoietic cell transplantation.
Nguyen HD, Chatterjee S, Haarberg KM, Wu Y, Bastian D, Heinrichs J, Fu J, Daenthanasanmak A, Schutt S, Shrestha S, Liu C, Wang H, Chi H, Mehrotra S, Yu XZ
The Journal of clinical investigation 2016 Apr 1;126(4):1337-52
The Journal of clinical investigation 2016 Apr 1;126(4):1337-52
Immunostimulatory Effects Triggered by Enterococcus faecalis CECT7121 Probiotic Strain Involve Activation of Dendritic Cells and Interferon-Gamma Production.
Molina MA, Díaz AM, Hesse C, Ginter W, Gentilini MV, Nuñez GG, Canellada AM, Sparwasser T, Berod L, Castro MS, Manghi MA
PloS one 2015;10(5):e0127262
PloS one 2015;10(5):e0127262
The proximal J kappa germline-transcript promoter facilitates receptor editing through control of ordered recombination.
Vettermann C, Timblin GA, Lim V, Lai EC, Schlissel MS
PloS one 2015;10(1):e0113824
PloS one 2015;10(1):e0113824
Mitochondrial ATP transporter Ant2 depletion impairs erythropoiesis and B lymphopoiesis.
Cho J, Seo J, Lim CH, Yang L, Shiratsuchi T, Lee MH, Chowdhury RR, Kasahara H, Kim JS, Oh SP, Lee YJ, Terada N
Cell death and differentiation 2015 Sep;22(9):1437-50
Cell death and differentiation 2015 Sep;22(9):1437-50
Acute DNA damage activates the tumour suppressor p53 to promote radiation-induced lymphoma.
Lee CL, Castle KD, Moding EJ, Blum JM, Williams N, Luo L, Ma Y, Borst LB, Kim Y, Kirsch DG
Nature communications 2015 Sep 24;6:8477
Nature communications 2015 Sep 24;6:8477
CD93 Marks a Non-Quiescent Human Leukemia Stem Cell Population and Is Required for Development of MLL-Rearranged Acute Myeloid Leukemia.
Iwasaki M, Liedtke M, Gentles AJ, Cleary ML
Cell stem cell 2015 Oct 1;17(4):412-21
Cell stem cell 2015 Oct 1;17(4):412-21
Endothelial Gata5 transcription factor regulates blood pressure.
Messaoudi S, He Y, Gutsol A, Wight A, Hébert RL, Vilmundarson RO, Makrigiannis AP, Chalmers J, Hamet P, Tremblay J, McPherson R, Stewart AFR, Touyz RM, Nemer M
Nature communications 2015 Nov 30;6:8835
Nature communications 2015 Nov 30;6:8835
Disruption of p21-activated kinase 1 gene diminishes atherosclerosis in apolipoprotein E-deficient mice.
Singh NK, Kotla S, Dyukova E, Traylor JG Jr, Orr AW, Chernoff J, Marion TN, Rao GN
Nature communications 2015 Jun 24;6:7450
Nature communications 2015 Jun 24;6:7450
BAFF activation of the ERK5 MAP kinase pathway regulates B cell survival.
Jacque E, Schweighoffer E, Tybulewicz VL, Ley SC
The Journal of experimental medicine 2015 Jun 1;212(6):883-92
The Journal of experimental medicine 2015 Jun 1;212(6):883-92
The development of mixed cryoglobulinemia in Capillaria hepatica-infected mice is associated with the capillaria antigen-induced selective proliferation of splenic B-1a cells in response to interleukin-5 stimulation.
Aihara N, Kamiie J, Yamada M, Shirota K
The American journal of pathology 2015 Jan;185(1):172-84
The American journal of pathology 2015 Jan;185(1):172-84
B cells modulate systemic responses to Pneumocystis murina lung infection and protect on-demand hematopoiesis via T cell-independent innate mechanisms when type I interferon signaling is absent.
Hoyt TR, Dobrinen E, Kochetkova I, Meissner N
Infection and immunity 2015 Feb;83(2):743-58
Infection and immunity 2015 Feb;83(2):743-58
Aging-associated inflammation promotes selection for adaptive oncogenic events in B cell progenitors.
Henry CJ, Casás-Selves M, Kim J, Zaberezhnyy V, Aghili L, Daniel AE, Jimenez L, Azam T, McNamee EN, Clambey ET, Klawitter J, Serkova NJ, Tan AC, Dinarello CA, DeGregori J
The Journal of clinical investigation 2015 Dec;125(12):4666-80
The Journal of clinical investigation 2015 Dec;125(12):4666-80
Lactobacillus gasseri SBT2055 induces TGF-β expression in dendritic cells and activates TLR2 signal to produce IgA in the small intestine.
Sakai F, Hosoya T, Ono-Ohmachi A, Ukibe K, Ogawa A, Moriya T, Kadooka Y, Shiozaki T, Nakagawa H, Nakayama Y, Miyazaki T
PloS one 2014;9(8):e105370
PloS one 2014;9(8):e105370
B-cell-intrinsic hepatitis C virus expression leads to B-cell-lymphomagenesis and induction of NF-κB signalling.
Kasama Y, Mizukami T, Kusunoki H, Peveling-Oberhag J, Nishito Y, Ozawa M, Kohara M, Mizuochi T, Tsukiyama-Kohara K
PloS one 2014;9(3):e91373
PloS one 2014;9(3):e91373
Acquired initiating mutations in early hematopoietic cells of CLL patients.
Damm F, Mylonas E, Cosson A, Yoshida K, Della Valle V, Mouly E, Diop M, Scourzic L, Shiraishi Y, Chiba K, Tanaka H, Miyano S, Kikushige Y, Davi F, Lambert J, Gautheret D, Merle-Béral H, Sutton L, Dessen P, Solary E, Akashi K, Vainchenker W, Mercher T, Droin N, Ogawa S, Nguyen-Khac F, Bernard OA
Cancer discovery 2014 Sep;4(9):1088-101
Cancer discovery 2014 Sep;4(9):1088-101
Age-dependent enterocyte invasion and microcolony formation by Salmonella.
Zhang K, Dupont A, Torow N, Gohde F, Leschner S, Lienenklaus S, Weiss S, Brinkmann MM, Kühnel M, Hensel M, Fulde M, Hornef MW
PLoS pathogens 2014 Sep;10(9):e1004385
PLoS pathogens 2014 Sep;10(9):e1004385
Targeting multiple types of tumors using NKG2D-coated iron oxide nanoparticles.
Wu MR, Cook WJ, Zhang T, Sentman CL
Nanotechnology 2014 Nov 28;25(47):475101
Nanotechnology 2014 Nov 28;25(47):475101
Transcription elongation factor ELL2 drives Ig secretory-specific mRNA production and the unfolded protein response.
Park KS, Bayles I, Szlachta-McGinn A, Paul J, Boiko J, Santos P, Liu J, Wang Z, Borghesi L, Milcarek C
Journal of immunology (Baltimore, Md. : 1950) 2014 Nov 1;193(9):4663-74
Journal of immunology (Baltimore, Md. : 1950) 2014 Nov 1;193(9):4663-74
Immunosuppression by N-methyl-D-aspartate receptor antagonists is mediated through inhibition of Kv1.3 and KCa3.1 channels in T cells.
Kahlfuß S, Simma N, Mankiewicz J, Bose T, Lowinus T, Klein-Hessling S, Sprengel R, Schraven B, Heine M, Bommhardt U
Molecular and cellular biology 2014 Mar;34(5):820-31
Molecular and cellular biology 2014 Mar;34(5):820-31
TRAF6 is a critical regulator of LMP1 functions in vivo.
Arcipowski KM, Stunz LL, Bishop GA
International immunology 2014 Mar;26(3):149-58
International immunology 2014 Mar;26(3):149-58
TLR7 ligand augments GM-CSF-initiated antitumor immunity through activation of plasmacytoid dendritic cells.
Narusawa M, Inoue H, Sakamoto C, Matsumura Y, Takahashi A, Inoue T, Watanabe A, Miyamoto S, Miura Y, Hijikata Y, Tanaka Y, Inoue M, Takayama K, Okazaki T, Hasegawa M, Nakanishi Y, Tani K
Cancer immunology research 2014 Jun;2(6):568-80
Cancer immunology research 2014 Jun;2(6):568-80
Antigen capture and archiving by lymphatic endothelial cells following vaccination or viral infection.
Tamburini BA, Burchill MA, Kedl RM
Nature communications 2014 Jun 6;5:3989
Nature communications 2014 Jun 6;5:3989
Oral combined therapy with probiotics and alloantigen induces B cell-dependent long-lasting specific tolerance.
Mercadante AC, Perobelli SM, Alves AP, Gonçalves-Silva T, Mello W, Gomes-Santos AC, Miyoshi A, Azevedo V, Faria AM, Bonomo A
Journal of immunology (Baltimore, Md. : 1950) 2014 Feb 15;192(4):1928-37
Journal of immunology (Baltimore, Md. : 1950) 2014 Feb 15;192(4):1928-37
Histone deacetylase inhibitors upregulate B cell microRNAs that silence AID and Blimp-1 expression for epigenetic modulation of antibody and autoantibody responses.
White CA, Pone EJ, Lam T, Tat C, Hayama KL, Li G, Zan H, Casali P
Journal of immunology (Baltimore, Md. : 1950) 2014 Dec 15;193(12):5933-50
Journal of immunology (Baltimore, Md. : 1950) 2014 Dec 15;193(12):5933-50
Rhof promotes murine marginal zone B cell development.
Kishimoto M, Matsuda T, Yanase S, Katsumi A, Suzuki N, Ikejiri M, Takagi A, Ikawa M, Kojima T, Kunishima S, Kiyoi H, Naoe T, Matsushita T, Maruyama M
Nagoya journal of medical science 2014 Aug;76(3-4):293-305
Nagoya journal of medical science 2014 Aug;76(3-4):293-305
The Vi capsular polysaccharide enables Salmonella enterica serovar typhi to evade microbe-guided neutrophil chemotaxis.
Wangdi T, Lee CY, Spees AM, Yu C, Kingsbury DD, Winter SE, Hastey CJ, Wilson RP, Heinrich V, Bäumler AJ
PLoS pathogens 2014 Aug;10(8):e1004306
PLoS pathogens 2014 Aug;10(8):e1004306
IκBε is a key regulator of B cell expansion by providing negative feedback on cRel and RelA in a stimulus-specific manner.
Alves BN, Tsui R, Almaden J, Shokhirev MN, Davis-Turak J, Fujimoto J, Birnbaum H, Ponomarenko J, Hoffmann A
Journal of immunology (Baltimore, Md. : 1950) 2014 Apr 1;192(7):3121-32
Journal of immunology (Baltimore, Md. : 1950) 2014 Apr 1;192(7):3121-32
TDP2-dependent non-homologous end-joining protects against topoisomerase II-induced DNA breaks and genome instability in cells and in vivo.
Gómez-Herreros F, Romero-Granados R, Zeng Z, Alvarez-Quilón A, Quintero C, Ju L, Umans L, Vermeire L, Huylebroeck D, Caldecott KW, Cortés-Ledesma F
PLoS genetics 2013;9(3):e1003226
PLoS genetics 2013;9(3):e1003226
Properties of immature myeloid progenitors with nitric-oxide-dependent immunosuppressive activity isolated from bone marrow of tumor-free mice.
Forghani P, Harris W, Giver CR, Mirshafiey A, Galipeau J, Waller EK
PloS one 2013;8(7):e64837
PloS one 2013;8(7):e64837
Prolactin levels correlate with abnormal B cell maturation in MRL and MRL/lpr mouse models of systemic lupus erythematosus-like disease.
Legorreta-Haquet MV, Flores-Fernández R, Blanco-Favela F, Fuentes-Pananá EM, Chávez-Sánchez L, Hernández-González R, Tesoro-Cruz E, Arriaga-Pizano L, Chávez-Rueda AK
Clinical & developmental immunology 2013;2013:287469
Clinical & developmental immunology 2013;2013:287469
c-Rel is an essential transcription factor for the development of acute graft-versus-host disease in mice.
Yu Y, Wang D, Kaosaard K, Liu C, Fu J, Haarberg K, Anasetti C, Beg AA, Yu XZ
European journal of immunology 2013 Sep;43(9):2327-37
European journal of immunology 2013 Sep;43(9):2327-37
Usp16 contributes to somatic stem-cell defects in Down's syndrome.
Adorno M, Sikandar S, Mitra SS, Kuo A, Nicolis Di Robilant B, Haro-Acosta V, Ouadah Y, Quarta M, Rodriguez J, Qian D, Reddy VM, Cheshier S, Garner CC, Clarke MF
Nature 2013 Sep 19;501(7467):380-4
Nature 2013 Sep 19;501(7467):380-4
IL-33 signaling regulates innate and adaptive immunity to Cryptococcus neoformans.
Flaczyk A, Duerr CU, Shourian M, Lafferty EI, Fritz JH, Qureshi ST
Journal of immunology (Baltimore, Md. : 1950) 2013 Sep 1;191(5):2503-13
Journal of immunology (Baltimore, Md. : 1950) 2013 Sep 1;191(5):2503-13
B lymphocytes trigger monocyte mobilization and impair heart function after acute myocardial infarction.
Zouggari Y, Ait-Oufella H, Bonnin P, Simon T, Sage AP, Guérin C, Vilar J, Caligiuri G, Tsiantoulas D, Laurans L, Dumeau E, Kotti S, Bruneval P, Charo IF, Binder CJ, Danchin N, Tedgui A, Tedder TF, Silvestre JS, Mallat Z
Nature medicine 2013 Oct;19(10):1273-80
Nature medicine 2013 Oct;19(10):1273-80
Homologous recombination into the eosinophil peroxidase locus generates a strain of mice expressing Cre recombinase exclusively in eosinophils.
Doyle AD, Jacobsen EA, Ochkur SI, Willetts L, Shim K, Neely J, Kloeber J, Lesuer WE, Pero RS, Lacy P, Moqbel R, Lee NA, Lee JJ
Journal of leukocyte biology 2013 Jul;94(1):17-24
Journal of leukocyte biology 2013 Jul;94(1):17-24
Reciprocal effects of rab7 deletion in activated and neglected T cells.
Roy SG, Stevens MW, So L, Edinger AL
Autophagy 2013 Jul;9(7):1009-23
Autophagy 2013 Jul;9(7):1009-23
Candida albicans stimulates in vivo differentiation of haematopoietic stem and progenitor cells towards macrophages by a TLR2-dependent signalling.
Megías J, Maneu V, Salvador P, Gozalbo D, Gil ML
Cellular microbiology 2013 Jul;15(7):1143-53
Cellular microbiology 2013 Jul;15(7):1143-53
Streptomycin-induced inflammation enhances Escherichia coli gut colonization through nitrate respiration.
Spees AM, Wangdi T, Lopez CA, Kingsbury DD, Xavier MN, Winter SE, Tsolis RM, Bäumler AJ
mBio 2013 Jul 2;4(4)
mBio 2013 Jul 2;4(4)
Novel apidaecin 1b analogs with superior serum stabilities for treatment of infections by gram-negative pathogens.
Berthold N, Czihal P, Fritsche S, Sauer U, Schiffer G, Knappe D, Alber G, Hoffmann R
Antimicrobial agents and chemotherapy 2013 Jan;57(1):402-9
Antimicrobial agents and chemotherapy 2013 Jan;57(1):402-9
Interferon gamma and T cells inhibit osteogenesis induced by allogeneic mesenchymal stromal cells.
Dighe AS, Yang S, Madhu V, Balian G, Cui Q
Journal of orthopaedic research : official publication of the Orthopaedic Research Society 2013 Feb;31(2):227-34
Journal of orthopaedic research : official publication of the Orthopaedic Research Society 2013 Feb;31(2):227-34
HDL and Glut1 inhibition reverse a hypermetabolic state in mouse models of myeloproliferative disorders.
Gautier EL, Westerterp M, Bhagwat N, Cremers S, Shih A, Abdel-Wahab O, Lütjohann D, Randolph GJ, Levine RL, Tall AR, Yvan-Charvet L
The Journal of experimental medicine 2013 Feb 11;210(2):339-53
The Journal of experimental medicine 2013 Feb 11;210(2):339-53
MHC class II-dependent B cell APC function is required for induction of CNS autoimmunity independent of myelin-specific antibodies.
Molnarfi N, Schulze-Topphoff U, Weber MS, Patarroyo JC, Prod'homme T, Varrin-Doyer M, Shetty A, Linington C, Slavin AJ, Hidalgo J, Jenne DE, Wekerle H, Sobel RA, Bernard CC, Shlomchik MJ, Zamvil SS
The Journal of experimental medicine 2013 Dec 16;210(13):2921-37
The Journal of experimental medicine 2013 Dec 16;210(13):2921-37
Interaction of CD99 with its paralog CD99L2 positively regulates CD99L2 trafficking to cell surfaces.
Nam G, Lee YK, Lee HY, Ma MJ, Araki M, Araki K, Lee S, Lee IS, Choi EY
Journal of immunology (Baltimore, Md. : 1950) 2013 Dec 1;191(11):5730-42
Journal of immunology (Baltimore, Md. : 1950) 2013 Dec 1;191(11):5730-42
Minicircle DNA is superior to plasmid DNA in eliciting antigen-specific CD8+ T-cell responses.
Dietz WM, Skinner NE, Hamilton SE, Jund MD, Heitfeld SM, Litterman AJ, Hwu P, Chen ZY, Salazar AM, Ohlfest JR, Blazar BR, Pennell CA, Osborn MJ
Molecular therapy : the journal of the American Society of Gene Therapy 2013 Aug;21(8):1526-35
Molecular therapy : the journal of the American Society of Gene Therapy 2013 Aug;21(8):1526-35
11β-hydroxysteroid dehydrogenase type 1 deficiency in bone marrow-derived cells reduces atherosclerosis.
Kipari T, Hadoke PW, Iqbal J, Man TY, Miller E, Coutinho AE, Zhang Z, Sullivan KM, Mitic T, Livingstone DE, Schrecker C, Samuel K, White CI, Bouhlel MA, Chinetti-Gbaguidi G, Staels B, Andrew R, Walker BR, Savill JS, Chapman KE, Seckl JR
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2013 Apr;27(4):1519-31
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2013 Apr;27(4):1519-31
Role of suppressor of cytokine signaling-1 in murine atherosclerosis.
Grothusen C, Schuett H, Hillmer A, Lumpe S, Grote K, Ballmaier M, Bleich A, Glage S, Tietge UJ, Luchtefeld M, Schieffer B
PloS one 2012;7(12):e51608
PloS one 2012;7(12):e51608
Retention of anergy and inhibition of antibody responses during acute γ herpesvirus 68 infection.
Getahun A, Smith MJ, Kogut I, van Dyk LF, Cambier JC
Journal of immunology (Baltimore, Md. : 1950) 2012 Sep 15;189(6):2965-74
Journal of immunology (Baltimore, Md. : 1950) 2012 Sep 15;189(6):2965-74
Activation-induced cytidine deaminase-initiated off-target DNA breaks are detected and resolved during S phase.
Hasham MG, Snow KJ, Donghia NM, Branca JA, Lessard MD, Stavnezer J, Shopland LS, Mills KD
Journal of immunology (Baltimore, Md. : 1950) 2012 Sep 1;189(5):2374-82
Journal of immunology (Baltimore, Md. : 1950) 2012 Sep 1;189(5):2374-82
Myeloid-derived suppressor cells from tumor-bearing mice impair TGF-β-induced differentiation of CD4+CD25+FoxP3+ Tregs from CD4+CD25-FoxP3- T cells.
Centuori SM, Trad M, LaCasse CJ, Alizadeh D, Larmonier CB, Hanke NT, Kartchner J, Janikashvili N, Bonnotte B, Larmonier N, Katsanis E
Journal of leukocyte biology 2012 Nov;92(5):987-97
Journal of leukocyte biology 2012 Nov;92(5):987-97
Trogocytosis of CD80 and CD86 by induced regulatory T cells.
Gu P, Gao JF, D'Souza CA, Kowalczyk A, Chou KY, Zhang L
Cellular & molecular immunology 2012 Mar;9(2):136-46
Cellular & molecular immunology 2012 Mar;9(2):136-46
Up-regulation of a HOXA-PBX3 homeobox-gene signature following down-regulation of miR-181 is associated with adverse prognosis in patients with cytogenetically abnormal AML.
Li Z, Huang H, Li Y, Jiang X, Chen P, Arnovitz S, Radmacher MD, Maharry K, Elkahloun A, Yang X, He C, He M, Zhang Z, Dohner K, Neilly MB, Price C, Lussier YA, Zhang Y, Larson RA, Le Beau MM, Caligiuri MA, Bullinger L, Valk PJ, Delwel R, Lowenberg B, Liu PP, Marcucci G, Bloomfield CD, Rowley JD, Chen J
Blood 2012 Mar 8;119(10):2314-24
Blood 2012 Mar 8;119(10):2314-24
IKKα-mediated signaling circuitry regulates early B lymphopoiesis during hematopoiesis.
Balkhi MY, Willette-Brown J, Zhu F, Chen Z, Liu S, Guttridge DC, Karin M, Hu Y
Blood 2012 Jun 7;119(23):5467-77
Blood 2012 Jun 7;119(23):5467-77
Transcription factor RORα is critical for nuocyte development.
Wong SH, Walker JA, Jolin HE, Drynan LF, Hams E, Camelo A, Barlow JL, Neill DR, Panova V, Koch U, Radtke F, Hardman CS, Hwang YY, Fallon PG, McKenzie AN
Nature immunology 2012 Jan 22;13(3):229-36
Nature immunology 2012 Jan 22;13(3):229-36
Metalloproteinase-mediated Shedding of Integrin β2 promotes macrophage efflux from inflammatory sites.
Gomez IG, Tang J, Wilson CL, Yan W, Heinecke JW, Harlan JM, Raines EW
The Journal of biological chemistry 2012 Feb 10;287(7):4581-9
The Journal of biological chemistry 2012 Feb 10;287(7):4581-9
Critical role of B cell lymphoma 10 in BAFF-regulated NF-κB activation and survival of anergic B cells.
Yu M, Chen Y, He Y, Podd A, Fu G, Wright JA, Kleiman E, Khan WN, Wen R, Wang D
Journal of immunology (Baltimore, Md. : 1950) 2012 Dec 1;189(11):5185-93
Journal of immunology (Baltimore, Md. : 1950) 2012 Dec 1;189(11):5185-93
Candida albicans induces selective development of macrophages and monocyte derived dendritic cells by a TLR2 dependent signalling.
Yáñez A, Megías J, O'Connor JE, Gozalbo D, Gil ML
PloS one 2011;6(9):e24761
PloS one 2011;6(9):e24761
PAD4-mediated neutrophil extracellular trap formation is not required for immunity against influenza infection.
Hemmers S, Teijaro JR, Arandjelovic S, Mowen KA
PloS one 2011;6(7):e22043
PloS one 2011;6(7):e22043
Study of camelpox virus pathogenesis in athymic nude mice.
Duraffour S, Matthys P, van den Oord JJ, De Schutter T, Mitera T, Snoeck R, Andrei G
PloS one 2011;6(6):e21561
PloS one 2011;6(6):e21561
Antiasthmatic drugs targeting the cysteinyl leukotriene receptor 1 alleviate central nervous system inflammatory cell infiltration and pathogenesis of experimental autoimmune encephalomyelitis.
Wang L, Du C, Lv J, Wei W, Cui Y, Xie X
Journal of immunology (Baltimore, Md. : 1950) 2011 Sep 1;187(5):2336-45
Journal of immunology (Baltimore, Md. : 1950) 2011 Sep 1;187(5):2336-45
Online nanoflow multidimensional fractionation for high efficiency phosphopeptide analysis.
Ficarro SB, Zhang Y, Carrasco-Alfonso MJ, Garg B, Adelmant G, Webber JT, Luckey CJ, Marto JA
Molecular & cellular proteomics : MCP 2011 Nov;10(11):O111.011064
Molecular & cellular proteomics : MCP 2011 Nov;10(11):O111.011064
Prevention of GVHD while sparing GVL effect by targeting Th1 and Th17 transcription factor T-bet and RORγt in mice.
Yu Y, Wang D, Liu C, Kaosaard K, Semple K, Anasetti C, Yu XZ
Blood 2011 Nov 3;118(18):5011-20
Blood 2011 Nov 3;118(18):5011-20
CONTINUOUS REPOPULATION OF LYMPHOCYTE SUBSETS IN TRANSPLANTED MYCOBACTERIAL GRANULOMAS.
Schreiber HA, Harding JS, Altamirano CJ, Hunt O, Hulseberg PD, Fabry Z, Sandor M
European journal of microbiology & immunology 2011 Mar;1(1):59-69
European journal of microbiology & immunology 2011 Mar;1(1):59-69
Induction of Noxa-mediated apoptosis by modified vaccinia virus Ankara depends on viral recognition by cytosolic helicases, leading to IRF-3/IFN-β-dependent induction of pro-apoptotic Noxa.
Eitz Ferrer P, Potthoff S, Kirschnek S, Gasteiger G, Kastenmüller W, Ludwig H, Paschen SA, Villunger A, Sutter G, Drexler I, Häcker G
PLoS pathogens 2011 Jun;7(6):e1002083
PLoS pathogens 2011 Jun;7(6):e1002083
Indoleamine 2,3-dioxygenase and regulatory dendritic cells contribute to the allograft protection induced by infusion of donor-specific splenic stromal cells.
Liu L, Duan L, Gong M, Dai H, Gong Q, Zheng F, Tan Z, Wang C, Gong F, Fang M
Cellular & molecular immunology 2011 Jan;8(1):31-40
Cellular & molecular immunology 2011 Jan;8(1):31-40
Uracil residues dependent on the deaminase AID in immunoglobulin gene variable and switch regions.
Maul RW, Saribasak H, Martomo SA, McClure RL, Yang W, Vaisman A, Gramlich HS, Schatz DG, Woodgate R, Wilson DM 3rd, Gearhart PJ
Nature immunology 2011 Jan;12(1):70-6
Nature immunology 2011 Jan;12(1):70-6
Protein kinase D2 is an essential regulator of murine myoblast differentiation.
Kleger A, Loebnitz C, Pusapati GV, Armacki M, Müller M, Tümpel S, Illing A, Hartmann D, Brunner C, Liebau S, Rudolph KL, Adler G, Seufferlein T
PloS one 2011 Jan 27;6(1):e14599
PloS one 2011 Jan 27;6(1):e14599
Auto-antibody production and glomerulonephritis in congenic Slamf1-/- and Slamf2-/- [B6.129] but not in Slamf1-/- and Slamf2-/- [BALB/c.129] mice.
Keszei M, Latchman YE, Vanguri VK, Brown DR, Detre C, Morra M, Arancibia-Carcamo CV, Paul E, Calpe S, Castro W, Wang N, Terhorst C, Sharpe AH
International immunology 2011 Feb;23(2):149-58
International immunology 2011 Feb;23(2):149-58
Nuclear export of the NF-κB inhibitor IκBα is required for proper B cell and secondary lymphoid tissue formation.
Wuerzberger-Davis SM, Chen Y, Yang DT, Kearns JD, Bates PW, Lynch C, Ladell NC, Yu M, Podd A, Zeng H, Huang TT, Wen R, Hoffmann A, Wang D, Miyamoto S
Immunity 2011 Feb 25;34(2):188-200
Immunity 2011 Feb 25;34(2):188-200
mGluR6 transcripts in non-neuronal tissues.
Vardi T, Fina M, Zhang L, Dhingra A, Vardi N
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2011 Dec;59(12):1076-86
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2011 Dec;59(12):1076-86
AID dysregulation in lupus-prone MRL/Fas(lpr/lpr) mice increases class switch DNA recombination and promotes interchromosomal c-Myc/IgH loci translocations: modulation by HoxC4.
White CA, Seth Hawkins J, Pone EJ, Yu ES, Al-Qahtani A, Mai T, Zan H, Casali P
Autoimmunity 2011 Dec;44(8):585-98
Autoimmunity 2011 Dec;44(8):585-98
Th-1 lymphocytes induce dendritic cell tumor killing activity by an IFN-γ-dependent mechanism.
LaCasse CJ, Janikashvili N, Larmonier CB, Alizadeh D, Hanke N, Kartchner J, Situ E, Centuori S, Har-Noy M, Bonnotte B, Katsanis E, Larmonier N
Journal of immunology (Baltimore, Md. : 1950) 2011 Dec 15;187(12):6310-7
Journal of immunology (Baltimore, Md. : 1950) 2011 Dec 15;187(12):6310-7
TLR8 deficiency leads to autoimmunity in mice.
Demaria O, Pagni PP, Traub S, de Gassart A, Branzk N, Murphy AJ, Valenzuela DM, Yancopoulos GD, Flavell RA, Alexopoulou L
The Journal of clinical investigation 2010 Oct;120(10):3651-62
The Journal of clinical investigation 2010 Oct;120(10):3651-62
Estrogen receptors bind to and activate the HOXC4/HoxC4 promoter to potentiate HoxC4-mediated activation-induced cytosine deaminase induction, immunoglobulin class switch DNA recombination, and somatic hypermutation.
Mai T, Zan H, Zhang J, Hawkins JS, Xu Z, Casali P
The Journal of biological chemistry 2010 Nov 26;285(48):37797-810
The Journal of biological chemistry 2010 Nov 26;285(48):37797-810
Impaired apoptotic cell clearance in the germinal center by Mer-deficient tingible body macrophages leads to enhanced antibody-forming cell and germinal center responses.
Rahman ZS, Shao WH, Khan TN, Zhen Y, Cohen PL
Journal of immunology (Baltimore, Md. : 1950) 2010 Nov 15;185(10):5859-68
Journal of immunology (Baltimore, Md. : 1950) 2010 Nov 15;185(10):5859-68
HVCN1 modulates BCR signal strength via regulation of BCR-dependent generation of reactive oxygen species.
Capasso M, Bhamrah MK, Henley T, Boyd RS, Langlais C, Cain K, Dinsdale D, Pulford K, Khan M, Musset B, Cherny VV, Morgan D, Gascoyne RD, Vigorito E, DeCoursey TE, MacLennan IC, Dyer MJ
Nature immunology 2010 Mar;11(3):265-72
Nature immunology 2010 Mar;11(3):265-72
Increased hematopoietic cells in the mertk-/- mouse peritoneal cavity: a result of augmented migration.
Williams JC, Wagner NJ, Earp HS, Vilen BJ, Matsushima GK
Journal of immunology (Baltimore, Md. : 1950) 2010 Jun 15;184(12):6637-48
Journal of immunology (Baltimore, Md. : 1950) 2010 Jun 15;184(12):6637-48
CD11c+ dendritic cells and B cells contribute to the tumoricidal activity of anti-DR5 antibody therapy in established tumors.
Haynes NM, Hawkins ED, Li M, McLaughlin NM, Hämmerling GJ, Schwendener R, Winoto A, Wensky A, Yagita H, Takeda K, Kershaw MH, Darcy PK, Smyth MJ
Journal of immunology (Baltimore, Md. : 1950) 2010 Jul 1;185(1):532-41
Journal of immunology (Baltimore, Md. : 1950) 2010 Jul 1;185(1):532-41
CD4 T-cell suppression by cells from Toxoplasma gondii-infected retinas is mediated by surface protein PD-L1.
Charles E, Joshi S, Ash JD, Fox BA, Farris AD, Bzik DJ, Lang ML, Blader IJ
Infection and immunity 2010 Aug;78(8):3484-92
Infection and immunity 2010 Aug;78(8):3484-92
B cell depletion reduces the development of atherosclerosis in mice.
Ait-Oufella H, Herbin O, Bouaziz JD, Binder CJ, Uyttenhove C, Laurans L, Taleb S, Van Vré E, Esposito B, Vilar J, Sirvent J, Van Snick J, Tedgui A, Tedder TF, Mallat Z
The Journal of experimental medicine 2010 Aug 2;207(8):1579-87
The Journal of experimental medicine 2010 Aug 2;207(8):1579-87
Gene expression profiling of leukemia stem cells.
Krivtsov AV, Wang Y, Feng Z, Armstrong SA
Methods in molecular biology (Clifton, N.J.) 2009;538:231-46
Methods in molecular biology (Clifton, N.J.) 2009;538:231-46
Critical role of the interleukin-17/interleukin-17 receptor axis in regulating host susceptibility to respiratory infection with Chlamydia species.
Zhou X, Chen Q, Moore J, Kolls JK, Halperin S, Wang J
Infection and immunity 2009 Nov;77(11):5059-70
Infection and immunity 2009 Nov;77(11):5059-70
HoxC4 binds to the promoter of the cytidine deaminase AID gene to induce AID expression, class-switch DNA recombination and somatic hypermutation.
Park SR, Zan H, Pal Z, Zhang J, Al-Qahtani A, Pone EJ, Xu Z, Mai T, Casali P
Nature immunology 2009 May;10(5):540-50
Nature immunology 2009 May;10(5):540-50
A critical role of TAK1 in B-cell receptor-mediated nuclear factor kappaB activation.
Schuman J, Chen Y, Podd A, Yu M, Liu HH, Wen R, Chen ZJ, Wang D
Blood 2009 May 7;113(19):4566-74
Blood 2009 May 7;113(19):4566-74
STAT6-mediated suppression of erythropoiesis in an experimental model of malarial anemia.
Thawani N, Tam M, Stevenson MM
Haematologica 2009 Feb;94(2):195-204
Haematologica 2009 Feb;94(2):195-204
Lupus-prone MRL/faslpr/lpr mice display increased AID expression and extensive DNA lesions, comprising deletions and insertions, in the immunoglobulin locus: concurrent upregulation of somatic hypermutation and class switch DNA recombination.
Zan H, Zhang J, Ardeshna S, Xu Z, Park SR, Casali P
Autoimmunity 2009 Feb;42(2):89-103
Autoimmunity 2009 Feb;42(2):89-103
The cellular niche of Listeria monocytogenes infection changes rapidly in the spleen.
Aoshi T, Carrero JA, Konjufca V, Koide Y, Unanue ER, Miller MJ
European journal of immunology 2009 Feb;39(2):417-25
European journal of immunology 2009 Feb;39(2):417-25
Differential roles of migratory and resident DCs in T cell priming after mucosal or skin HSV-1 infection.
Lee HK, Zamora M, Linehan MM, Iijima N, Gonzalez D, Haberman A, Iwasaki A
The Journal of experimental medicine 2009 Feb 16;206(2):359-70
The Journal of experimental medicine 2009 Feb 16;206(2):359-70
BLNK suppresses pre-B-cell leukemogenesis through inhibition of JAK3.
Nakayama J, Yamamoto M, Hayashi K, Satoh H, Bundo K, Kubo M, Goitsuka R, Farrar MA, Kitamura D
Blood 2009 Feb 12;113(7):1483-92
Blood 2009 Feb 12;113(7):1483-92
4-1BB functions as a survival factor in dendritic cells.
Choi BK, Kim YH, Kwon PM, Lee SC, Kang SW, Kim MS, Lee MJ, Kwon BS
Journal of immunology (Baltimore, Md. : 1950) 2009 Apr 1;182(7):4107-15
Journal of immunology (Baltimore, Md. : 1950) 2009 Apr 1;182(7):4107-15
Optimization of methods to study pulmonary dendritic cell migration reveals distinct capacities of DC subsets to acquire soluble versus particulate antigen.
Jakubzick C, Helft J, Kaplan TJ, Randolph GJ
Journal of immunological methods 2008 Sep 15;337(2):121-31
Journal of immunological methods 2008 Sep 15;337(2):121-31
IL-7 specifies B cell fate at the common lymphoid progenitor to pre-proB transition stage by maintaining early B cell factor expression.
Kikuchi K, Kasai H, Watanabe A, Lai AY, Kondo M
Journal of immunology (Baltimore, Md. : 1950) 2008 Jul 1;181(1):383-92
Journal of immunology (Baltimore, Md. : 1950) 2008 Jul 1;181(1):383-92
IL-7 specifies B cell fate at the common lymphoid progenitor to pre-proB transition stage by maintaining early B cell factor expression.
Kikuchi K, Kasai H, Watanabe A, Lai AY, Kondo M
Journal of immunology (Baltimore, Md. : 1950) 2008 Jul 1;181(1):383-92
Journal of immunology (Baltimore, Md. : 1950) 2008 Jul 1;181(1):383-92
p27 deficiency cooperates with Bcl-2 but not Bax to promote T-cell lymphoma.
Cheng N, van de Wetering CI, Knudson CM
PloS one 2008 Apr 2;3(4):e1911
PloS one 2008 Apr 2;3(4):e1911
Phospholipase Cgamma2 contributes to light-chain gene activation and receptor editing.
Bai L, Chen Y, He Y, Dai X, Lin X, Wen R, Wang D
Molecular and cellular biology 2007 Sep;27(17):5957-67
Molecular and cellular biology 2007 Sep;27(17):5957-67
Immature hematopoietic cells display selective requirements for adhesion- and degranulation-promoting adaptor protein in development and homeostatsis.
Dluzniewska J, Zou L, Harmon IR, Ellingson MT, Peterson EJ
European journal of immunology 2007 Nov;37(11):3208-19
European journal of immunology 2007 Nov;37(11):3208-19
STAT3 positively regulates an early step in B-cell development.
Chou WC, Levy DE, Lee CK
Blood 2006 Nov 1;108(9):3005-11
Blood 2006 Nov 1;108(9):3005-11
Distinct roles of phosphoinositide-3 kinase and phospholipase Cgamma2 in B-cell receptor-mediated signal transduction.
Dai X, Chen Y, Schuman J, Hua Z, Adamson JW, Wen R, Wang D
Molecular and cellular biology 2006 Jan;26(1):88-99
Molecular and cellular biology 2006 Jan;26(1):88-99
Essential role of phospholipase C gamma 2 in early B-cell development and Myc-mediated lymphomagenesis.
Wen R, Chen Y, Bai L, Fu G, Schuman J, Dai X, Zeng H, Yang C, Stephan RP, Cleveland JL, Wang D
Molecular and cellular biology 2006 Dec;26(24):9364-76
Molecular and cellular biology 2006 Dec;26(24):9364-76
A role for brain-derived neurotrophic factor in B cell development.
Schuhmann B, Dietrich A, Sel S, Hahn C, Klingenspor M, Lommatzsch M, Gudermann T, Braun A, Renz H, Nockher WA
Journal of neuroimmunology 2005 Jun;163(1-2):15-23
Journal of neuroimmunology 2005 Jun;163(1-2):15-23
Impaired immune responses and B-cell proliferation in mice lacking the Id3 gene.
Pan L, Sato S, Frederick JP, Sun XH, Zhuang Y
Molecular and cellular biology 1999 Sep;19(9):5969-80
Molecular and cellular biology 1999 Sep;19(9):5969-80
Identification of monoclonal antibodies for immunohistochemical staining of feline B lymphocytes in frozen and formalin-fixed paraffin-embedded tissues.
Monteith CE, Chelack BJ, Davis WC, Haines DM
Canadian journal of veterinary research = Revue canadienne de recherche veterinaire 1996 Jul;60(3):193-8
Canadian journal of veterinary research = Revue canadienne de recherche veterinaire 1996 Jul;60(3):193-8
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
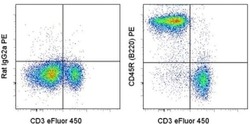
- Experimental details
- Staining of C57Bl/6 splenocytes with Anti-Mouse CD3 eFluor® 450 (Product # 48-0032-82) and 0.25 µg of Rat IgG2a K Isotype Control PE (Product # 12-4321-80) (left) or 0.25 µg of Anti-Human/Mouse CD45R (B220) PE (right). Total viable cells were used for analysis.
- Conjugate
- Yellow dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
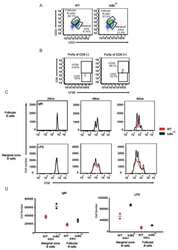
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
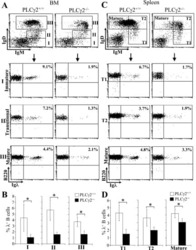
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
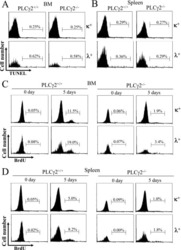
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
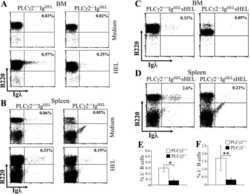
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
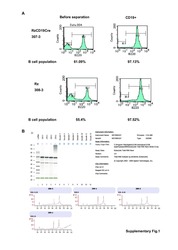
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
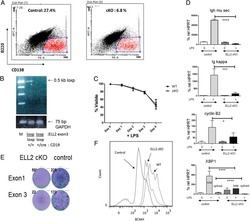
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
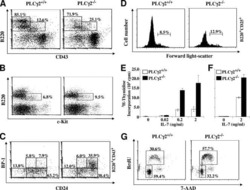
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
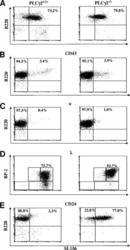
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
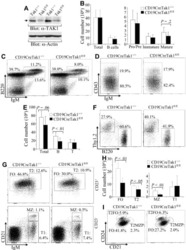
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
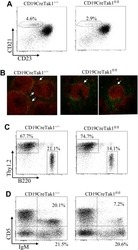
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
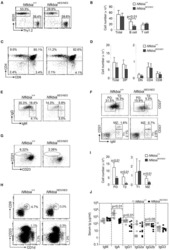
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
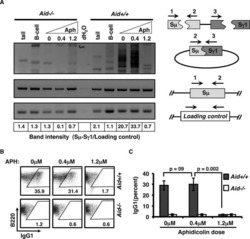
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
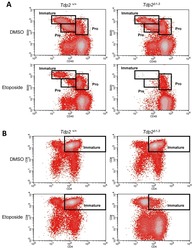
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
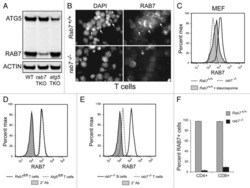
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
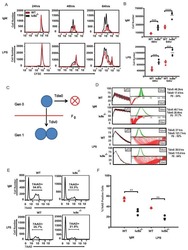
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
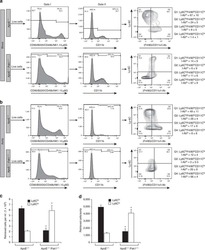
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
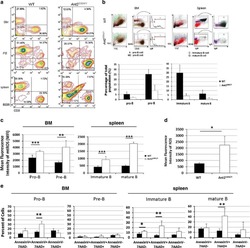
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
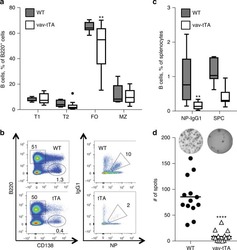
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
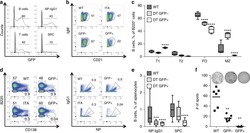
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
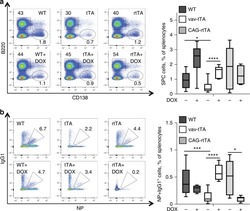
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
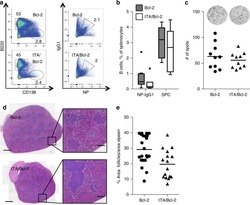
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
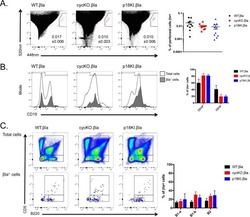
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
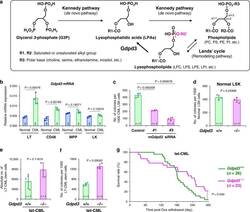
- Experimental details
- Fig. 1 Gdpd3 is implicated in CML disease initiation in vivo. a Diagram of pathways of lysophospholipid biosynthesis. G3P is converted into LPAs, and LPAs are then converted into phospholipids by the addition of polar bases via the Kennedy (de novo) pathway. The Lands'' cycle (remodelling pathway) generates lysophospholipids of distinct composition by substituting fatty acid ester and polar base groups of phospholipids. Lysophospholipase D Gdpd3 converts lysophospholipids back into LPAs by catalysing hydrolysis (magenta dotted line). (PC Phosphatidylcholine, PS Phosphatidylserine, PE Phosphatidylethanolamine, PI Phosphatidylinositol, LPC Lysophosphatidylcholine, LPS Lysophosphatidylserine, LPE Lysophosphatidylethanolamine, LPI Lysophosphatidylinositol). b qRT-PCR determination of Gdpd3 mRNA expression in LT-stem (LT), CD48, MPP, and LK cells (see Supplementary Fig.2) isolated from Gdpd3 +/+ tet-CML-affected (SCL-tTA + TRE-BCR-ABL1 + ) mice (one male, six females) or normal littermate (SCL-tTA + ) mice (four males, four females). Data are the mean ratio +- s.d. of transcript levels normalised to Actb ( n = 3 biologically independent samples) ( P -value, unpaired two-sided Student''s t -test). c Quantitation of the colony-forming capacity of Gdpd3 +/+ CML-LSK cells that were transduced with/without Cy3-labelled siRNA targetting mouse Gdpd3 mRNA (mGdpd3 siRNA #1 or #3). Cy3 + and Cy3 - CML-LSK cells were purified at 3 days post-transduction and plated in semi-solid methylcellulo
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
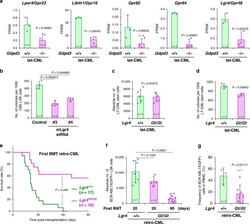
- Experimental details
- Fig. 7 Lgr4/Gpr48 is involved in CML stem cell self-renewal in vivo. a RNA-Seq determinations of mRNA levels of the indicated GPCR family genes in LT-CML stem cells isolated from Gdpd3 +/+ tet-CML-affected mice (22 males, 12 females) and Gdpd3 -/- tet-CML-affected mice (five males, five females). Results are expressed as FPKM (see Methods). Data are the mean FPKM +- s.d. ( n = 3 biologically independent samples) ( P -value, unpaired two-sided Student''s t -test). Results of the MA-plot and GO term enrichment analyses for these RNA-Seq data are shown in Supplementary Fig. 11a, b . b Quantitation of the colony-forming capacity of Gdpd3 +/+ CML-LSK cells that were transduced with/without Cy3-labelled siRNA targetting mouse Lgr4/Gpr48 mRNA (mLgr4 #3 or Lgr4 #4). Cy3 + and Cy3 - CML-LSK cells were purified at 3 days post-transduction and plated in a semi-solid methylcellulose medium. Data are the mean colony number +- s.d. ( n = 3) and are representative of three biologically independent experiments. ( P -value compared with control, unpaired two-sided Student''s t -test). The relevant FACS data are shown in Supplementary Fig. 12 . c Absolute numbers of LT-CML stem cells isolated from BM of the two hind limbs of Lgr4 +/+ tet-CML-affected mice (four males, one female) and Lgr4 Gt/Gt tet-CML-affected mice (five females) ( n = 5 biologically independent samples). Data are the mean absolute numbers +- s.d. of LT-CML stem cells ( P -value, unpaired two-sided Student''s t -test). (See S
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
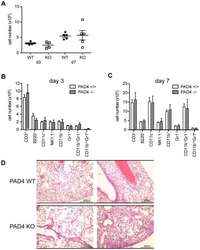
- Experimental details
- Figure 4 Lung leukocyte infiltration during influenza infection is comparable between PAD4 WT and KO mice. Experimental setup as described in Figure 2 . (A-C) Five mice per group were analyzed at d3 or d7 post infection. Leukocytes were isolated from infected lungs. (A) Total cell numbers of lung-infiltrating leukocytes at d3 and d7 p.i. Each symbol represents an individual mouse, filled squares represent PAD4 WT, open squares depict PAD4 KO mice. (B+C) The subsets of infiltrating leukocytes were enumerated by flow cytometry both at d3 p.i. (B) and d7 p.i. (C). Gated populations are indicated at the bottom of the graph. Total numbers of infiltrating cells are shown. WT mice are depicted in white bars, KO mice as grey bars. Bars represent mean + SEM. (D) Lungs for histological examination were harvested at d8 p.i. (5 mice/group). Leukocyte infiltration was assessed on H&E stained paraffin sections. Sections from two representative mice per group are shown. The scale bar indicates 200 um. Data is representative of two independent experiments.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
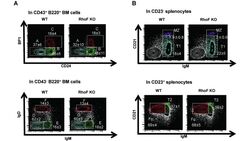
- Experimental details
- Fig. 4 Analysis of B cell development in bone marrow and spleen. A. FACS analysis of BM B cells. BM cells from WT and RhoF KO mice (6-7 weeks old, n = 5, male: 40%) were stained with antibodies against CD43, B220, BP1, and CD24 and analyzed by flow cytometry. The number shows the percentage (mean +- standard deviation) of the indicated subpopulation within the parent population; Fraction A (germline pro-B cells), fraction B (DJ-rearranged pro-B cells), fraction C (Early pre-B cells), fraction D (Late pre-B cells), fraction E (newly formed B cells), and fraction F (follicular-type recirculating B cells). The figures are representatives of three independent experiments. B. FACS analysis of B cells from the spleen. Splenocytes from WT and RhoF KO mice (6-7 weeks old, n = 6, male: 33%) were stained with antibodies against CD23, CD21, and IgM and analyzed by flow cytometry. The number shows the percentage (mean +- standard deviation) of the indicated subpopulation within the parent population; T1 (Transitional 1), T2 (Transitional 2), Fo (mature follicular B cells), and MZ (MZ B cells). C. The number of B cell subsets in BM was calculated by multiplying the total number of viable (trypan blue negative) BM cells by the fraction of the target population in viable (7AAD negative) cells. The data are shown as mean +- standard deviation (6-7 weeks old, n = 5, male: 40%). White bars: WT. Black bars: RhoF KO. D. The number of B cell subsets in the spleen was also calculated by multiplyin
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 2 Loss of Gata5 from renal endothelial cells leads to renal alterations. ( a ) GATA5 is expressed in the kidney as assessed by western blot performed on total kidney extracts. ( b ) Gata5 is essentially expressed in the glomeruli as assessed by qPCR on isolated glomeruli (Glo) and microdissected tubules (Tub) from Wt mice kidneys. ( n =3-5 per group). The results are reported as mean+-s.e.m. * P
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 5 NG2-cre, but not NG2-cre ERTM , targeted cells are the source of Scf in the bone marrow (a) Whole-mount sternum from NG2-cre/ iTdTomato/ Scf-GFP mice, anti-VE-cadherin. Representative images from 3 mice. Scale bars, 20 mum. (b) Representative FACS plot showing percentage of NG2-cre/ iTdTomato + cells within CD45 - TER119 - CD31 - Scf-GFP + cells. n=3 mice. (c-e) Analyses of LepR-cre/ Scf fl/- mice. (c) Numbers of HSCs (left) in BM and LSK cells in spleen (right). n=4 mice for cre (-), n=3 mice for cre (+). (d) FACS analyses of HSC (CD150 + CD48 - LSK) cell cycle with Ki-67 and Hoechst 33342 staining. n=5 mice for cre (-), n=6 mice for cre (+). (e) HSC localization relative to arterioles. Error bars: n=3 mice. P value has been calculated using n=272 HSCs for cre (-), 293 HSCs for cre (+) pooled from 3 mice per group. P =0.3402. (f-i) Analyses of NG2-cre/ Scf fl/- mice. (f) Numbers of total BM cells (left) and CD150 + CD48 - LSK HSCs (right) in BM. n=5 mice for cre (-), n=7 mice for cre (+). (g) Percentages of donor-derived cells after competitive reconstitution. n=5 mice for cre (-), n=7 mice for cre (+). (h) FACS analyses of HSC cell cycle with Ki-67 and Hoechst 33342 staining. n=6 mice for cre (-), n=7 mice for cre (+). (i) HSC localization relative to arterioles. Error bars: n=3 mice. P value has been calculated using n=224 HSCs for cre (-), 274 HSCs for cre (+) pooled from 3 mice per group. P =0.2872. (j-l) Analyses of NG2-cre ERTM / Scf fl/- mice. (j) Absolute nu
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
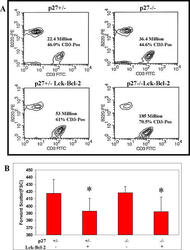
- Experimental details
- Figure 2 Splenic T cell hyperplasia and T cell size in p27 -/- Lck-Bcl-2 mice. A) Total splenic cells from mice of the indicated genotypes were isolated and stained with anti-B220-PE and anti-CD3-FITC antibodies as described in the Materials and Methods . The percentage of CD3-positive cells and the total number of splenic T cells (total # of splenic cells X %CD3) is shown. The data are representative of at least three mice from each genotype. B) The Mean Forward Scatter of CD3 positive splenic T cells was determined by staining with an anti-CD3 antibody as described in the Materials and Methods . The Mean+-SD of at least 5 mice is shown for the genotypes indicated. * P
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 7. NOX2 and FOXC1 Are Important for the Growth of Primary Human Myeloid Leukemia Cells (A) The Cancer Genome Atlas (TCGA) database for AML was used to analyze the expression levels of several NADPH-dependent oxidases (NOX1-NOX5), accessory subunits (NCF1, NCF2, NCF4, NOXA1, NOXO1, p22Phox, RAC1, and RAC2) as well as related oxido-reductase enzymes (dual-oxidases 1,2, A2). The reads per kilobase of transcript per million mapped reads (RPKM) values for each gene in a total of 188 AMLs are shown. (B) RNA-seq analysis was performed on functionally validated leukemic stem cells isolated from human primary AMLs. The RPKM value for each gene is shown. Unpublished data are used with permission. Additional supporting data are shown in Figure S7A . (C) Equal numbers of control or shNOX2-transduced primary AML cells were cultured in vitro in the presence of 10 ng/mL of IL-3, SCF, and FL3, and the relative percent expansion is reported for each specimen. n = 3, mean +- SD. *p < 0.05; **p < 0.01. (D) Control and shNOX2 primary AML cells were purified and cultured in vitro for 12 days. Annexin V, DAPI staining was performed to evaluate the degree of apoptotic cell death. A representative flow plot (left) and quantitation of 3 technical triplicates (right) are shown. Additional data are shown in Figure S7B . (E) The relative expression level of NOX2 , CEBP epsilon, Elane , and CTSG is shown in AML specimens in which NOX2 was knocked down using shRNAs. Additional supporting data are p
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 6 Assessments of organ damage and mouse metabolites during HLSA or recovery from HLSA. A , upper left panel , MMP-1, IL-1beta, and CRP levels in plasma had no significant change between the control group, HLSA group, and R-HLSA group. Upper right panel , CK-MB as a marker of heart injury in the plasma had no significant change between the control group, HLSA group, and R-HLSA group. Lower left panel , creatinine and BUN as markers of kidney injury in the plasma slightly increased in the HLSA group and returned to normal level after recovery from HLSA. Lower right panel , AST and ALT as markers of liver injury in the plasma showed no change between the control group, HLSA group, and R-HLSA group. Data were presented as mean +- SD (N = 6; ANOVA: ** p < 0.01). B , upper panel , food consumption and water consumption 3 days before and 3 days after a single ATP-induced HLSA depicted no change. Data were presented as mean +- SD (N = 6; ANOVA: p > 0.05). Lower panel , metabolomics analysis of scores plot from PCA analysis based on 1 H NMR data from the plasma, brain, liver, and kidney of the control group, HLSA group, and R-HLSA group. The PCA score showcased clusters correspond to metabolic patterns in different groups, with each point representing one sample. Circles represent 95% confidence interval for each score in each group (see also Figs. S5-S8 ). C , draining lymph nodes of mice that underwent HLSA once a day for 10 consecutive days were analyzed with FACS. CD44 and
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 5 CD4 surface expression of CD45RB and global glycosylation are altered in SLE123 mice. ( A and B ) Cell surface expression of CD45RB and total CD45 were measured by flow cytometry on CD4 + T cells. SLE123 demonstrated a downregulation of CD45RB on the cell surface as compared with B6 CD4 + T cells. There was no difference in pan-CD45 expression. Quantified in B . ( C ) CD45 is composed of an intracellular region that controls cytoskeletal binding and its phosphatase activity. The extracellular domain is composed of a region with fibronectin repeats that is heavily N-glycosylated. An alternatively spliced region is heavily O-glycosylated, and this region imparts unique functions to each CD45 isoform. Additionally, a sialic acid residue on the B portion of this region is essential for the binding of therapeutic aCD45RB. ( D and E ) We utilized lectins to detect the level of alpha-2,3-linked sialylation (MALII), O-linked glycosylation (Jacalin), and N-linked glycosylation (PHA-L) in CD4 + T cells from B6 and SLE123 mice. We determined SLE123 CD4 + T cells had reduced levels of alpha-2,3-linked sialic acids and O-glycosylation and an increase in N-glycosylation. Quantified in E . ( F ) Utilizing an antibody that detects a desialylated form of anti-CD45RB, we determined SLE123 CD4 + T cells possessed increased binding of this antibody compared with B6. ( G and H ) To determine the binding of therapeutic aCD45RB to B6 and SLE123 CD4 + T cells, we incubated splenocytes from
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
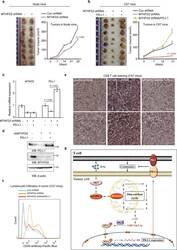
- Experimental details
- Fig. 7 Upregulation of PD-L1 by MTHFD2 is required for tumorigenesis. a - b A total of 2 x 10 6 ( a ) or 5 x 10 6 ( b ) Pan02 cells stably infected with lentivirus carrying indicated shRNAs or exogenous expressed PD-L1, were subcutaneously injected into athymic nude mice ( a ) or C57 mice ( b ), tumor volume was calculated every 7 days. Tumor xenografts at the 28th day in nude mice ( a ) or the 21th day in C57 mice ( b ) were shown. Data represent the means +- s.e.m ( n = 8 mice per group); p value (Student's t test, two-sided) with control is presented. c , PD-L1 mRNA levels in tumor tissues in C57 mice were analyzed by real-time PCR. The values are presented as mean +- s.e.m ( n = 3); p values (Student's t test, two-sided) with control or the indicated groups are presented. d The lysates of 8 pooled tumor tissues in C57 mice were subjected to immunoblotting analyses using the indicated antibodies. e Immunohistochemical staining was performed on tumor sections in C57 mice with anti-CD8 antibody. Representative images are shown. Scale bars, 50 mum. Histological semi-quantification was performed. f Cells digested from indicate tumor tissues in C57 mice were stained with anti-CD45 antibody and subjected to flow cytometric analyses (also see Supplementary Fig. 7b ). Representative images (1 out of 3 experiments) are shown. g A schematic model showing the role of MTHFD2 in tumor immune evasion. MTHFD2 promotes PD-L1 mediated tumor immune evasion through the folate-cycle-UTP-UDP-G
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
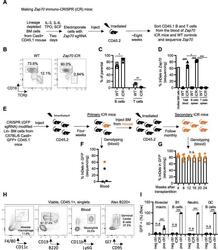
- Experimental details
- Fig. 2 Generating immuno-CRISPR (iCR) mice and evaluating the CRISPR-mediated modifications by sequencing. ( A ) Model describing the experimental setup where CD45.1 + Lin- BM cells were modified by CRISPR targeting Zap70 and grafted into irradiated CD45.2 + recipients. ( B ) Flow cytometry analysis of cells in the blood of Zap70 iCR mice and WT control mice eight weeks post transplantation. Cells gated on viable, CD45.1+, single lymphocytes. ( C ) Quantification of B and T cells in the blood of WT and Zap70 iCR mice in (B). ( D ) Analysis of the level of mutations in the sgRNA targeted Zap70 region in the BM cells used for transplantation, total cells from the blood, as well as in B and T cells sorted from the spleen of Zap70 iCR mice and WT control mice 8 weeks after engraftment. ( E ) Model describing the experimental setup where a secondary transplantation was used to amplify the population of successfully modified mice. ( F ) Analysis of the level of mutations in the sgRNA targeted GFP region in blood cells of the GFP iCR mice four weeks after transplantation, in an experiment with low efficiency. One mouse showed good knockout efficiency (labeled in orange) and was used as BM donor for secondary transplantation. ( G ) Kinetics of the level of mutations of GFP in the secondary iCR mice. ( H ) As examples, representative flow cytometry plots and ( I ) GFP + cell population percentage of alveolar macrophages, B1 cells, neutrophils and germinal center B cells from secondary
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
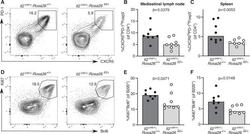
- Experimental details
- Figure 8. Increased IL-2 production impairs T follicular helper (Tfh) cell formation and the germinal centre response. Assessment of the Tfh cell and germinal centre response in Il2 cre/+ ; Rosa26 stop-flox-Il2/+ transgenic mice that do not switch off IL-2 production, and Il2 cre/+ ; Rosa26 +/+ control mice 12 days after influenza A infection. Flow cytometric contour plots ( A ) and quantification of the percentage of CXCR5 high PD-1 high Foxp3 - CD4 + Tfh cells in the mediastinal lymph node ( B ) and spleen ( C ). Flow cytometric contour plots ( D ) and quantification of the percentage of Bcl6 + Ki67 + B220 + germinal centre B cells in the mediastinal lymph node ( E ) and spleen ( F ). The height of the bars indicates the median, each symbol represents one mouse, data are pooled from two independent experiments. p-Values calculated between genotype groups by Mann-Whitney U test.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
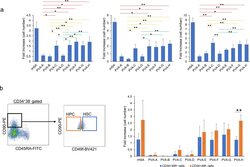
- Experimental details
- Fig. 2. Effect of different PVA hydrolysis rates and molecular weights on ex vivo cultured human cord blood CD34 + hematopoietic stem and progenitor cells. a , Human cord blood CD34 + cells were cultured in PVA media and cell numbers were counted at day 14. Three independent experiments were performed with cord blood CD34 + cells derived from different donors. Cell cultures were started from 7 x 10 3 (Exp.1) or 5 x 10 3 (Exp. 2 and 3) CD34 + cells. Mean +- S.D is shown. Statistical significance was calculated using ANOVA followed by Tukey-Kramer; *p < 0.05, **p < 0.01. b , Proliferation of the two different CD34 + cell fractions after 7-day culture in PVA-containing media. Fifty cells were sorted into four wells in a 96-well plate and cell numbers in each well were counted at 7-day. Statistical significance was calculated using ANOVA and Tukey-Kramer. **p < 0.01.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
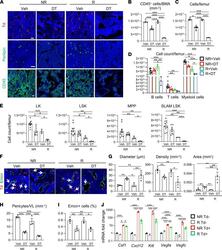
- Experimental details
- Figure 6 MALP ablation blocks bone marrow recovery after radiation. ( A ) Representative fluorescence images of Td + cells, Perilipin + LiLAs, and CD45 + hematopoietic cells in femoral bone marrow of Adipoq/Td/DTR mice after receiving 2 weeks of vehicle (Veh) or DT injections with or without prior radiation. Scale bar: 20 mum (top), 100 mum (middle), and 20 mum (bottom). ( B ) Quantification of CD45 + cells per bone marrow area. n = 3-6 mice/group. ( C ) Bone marrow cells were flushed from femurs and counted. n = 3-8 mice/group. ( D ) Cell counts of hematopoietic lineage cells in the bone marrow. n = 3-11 mice/group. B cells = B220 + , T cells = CD3 + , myeloid cells = Gr1 + and/or Mac1 + . ( E ) Cell counts of HSPCs. n = 3-11 mice/group. LK, Lineage - cKit + , LSK, Lineage - Sca1 + cKit + , SLAM LSK, Lineage - Sca1 + cKit + CD48 - CD150 + , MPP, Lineage - Sca1 + cKit + CD48 + CD150 - . ( F ) Representative fluorescence images of Adipoq/Td/DTR femoral bone marrow with Emcn staining (vessels). Arrows point to Td + pericytes. Scale bar: 20 mum. ( G ) Quantification of bone marrow vessel diameter, density, and area. ( H ) The number of pericytes per vessel length (VL) was measured. n = 3-4 mice/group. ( I ) The percentage of Emcn + endothelial cells in bone marrow was measured by flow cytometry. n = 3-4 mice/group. ( J ) qRT-PCR analysis of hematopoietic and angiogenic factors in sorted Td - and Td + cells from bone marrow before and after radiation. n = 4 mice/group. Statistica
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
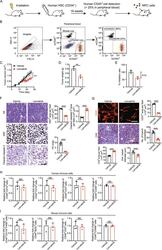
- Experimental details
- Lenvatinib insignificantly affects the immune microenvironment in NPC in humanized NSG mice. A Schematic diagram of the establishment of humanized NSG mice. B Representative FACS analysis of human CD45 + cells in mouse peripheral blood. Human CD45 + cell percentage greater than 25% was considered successful in modeling. C - E Tumor growth ( C ), tumor weights ( D ) were measured in vehicle-, anti-VEGF-, and lenvatinib-treated NPC tumors. The tumor inhibition ratio were calculated ( E ) ( n = 3 samples per group). F Representative micrographs of Ki67 + proliferative cells and cleaved caspase-3 + apoptotic cells in vehicle-, anti-VEGF-, and lenvatinib-treated NPC tumors. Scale bar = 50 mum. Quantification of Ki67 + , cleaved caspase-3 + signals, and PA index in vehicle-, anti-VEGF-, and lenvatinib-treated NPC tumors. ( n = 8 random fields per group) G Representative micrographs of CD31 + microvessels and CA9 + hypoxic areas in vehicle-, anti-VEGF-, and lenvatinib-treated NPC tumors. Scale bar in upper panel = 100 mum, scale bar in lower panel = 50 mum. Quantification of CD31 + tumor vessel parameters and CA9 + signals in vehicle-, anti-VEGF-, and lenvatinib-treated NPC tumors ( n = 8 random fields per group). H Quantification of hCD45 + hCD14 + population, hCD45 + hCD19 + population, hCD45 + hCD3 + population, and hCD45 + hCD56 + population in the NPC TME ( n = 3 samples per group). I Quantification of mCD45 + mCD11b + mF4/80 + population, mCD45 + mB220 + population, mCD45 + mC
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
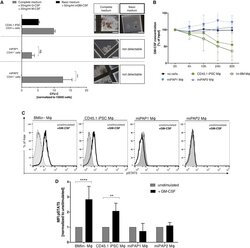
- Experimental details
- Figure 6 Disease Modeling of herPAP Using miPAP iPSCs (A) Number of CD41 + progenitor-derived colonies in methylcellulose-based clonogenic assays employing complete (IL-6, erythropoietin, SCF, IL-3, and supplemented with 20 ng/ml human G-CSF + 20 ng/ml murine GM-CSF) or basic medium (50 ng/ml murine GM-CSF only; independent experiments, n = 3 miPAP1 and 2, n = 2 CD45.1 iPSC, mean +- SD) and representative pictures of colonies. Scale bars, 500 mum. (B) GM-CSF clearance assay comparing miPAP-Mphi with CD45.1(10.3) iPSC-Mphi, BMlin - -Mphi, and no cells incubated with 2 ng/ml GM-CSF: at indicated time points (0, 4, 10, 24, and 30 hr) GM-CSF concentrations in supernatants were analyzed by ELISA, normalized to 0 hr (independent experiments, n = 3 BMlin - and CD45.1 iPSC, n = 2 miPAP1 and 2, mean +- SD). (C and D) Flow cytometry plots of STAT5 phosphorylation levels upon stimulation with mGM-CSF (C) and (D) summary of mean fluorescence intensity (MFI) data (independent experiments, n = 3 BMlin - and CD45.1 iPSC, n = 2 miPAP1 and 2, mean +- SD). ns, not significant; ** p
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
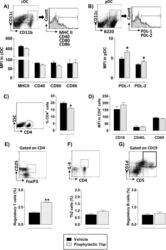
- Experimental details
- Fig 6 Tn P acts systemically during the induction phase modulating DCs and induces regulatory cells. Splenocytes from EAE mice treated with vehicle or Tn P (n = 5/group) were isolated at day 7, and analyzed. A ) The MFI of MHC class II, CD40, CD80, CD86 in cDC (CD11c+CD11b+) and B ) MFI of PDL-1 and PDL-2 in pDC (CD11c+B220 low ) were analyzed by flow cytometry (50,000 events). C ) The percentage of CD4+ cells, D ) and the MFI of CD18, CD40L, and CD69 in the CD4+ gate were analyzed by flow cytometry. E , F , G ) The percentages of FOXP3-positive CD4+CD25+ Treg, IL-4-positive CD4 Th2 cells and CD5-positive CD19+CD1d+ Breg cells were analyzed by flow cytometry. Values in the bar graphs are the mean +- SEM. * p < 0.05 and ** p < 0.01 compared with vehicle-treated EAE-mice.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 1 In vivo depletion of Cdkn1a rescues GC formation in Ezh2 -/- mice. Ezh2 fl/fl , Cdkn1a -/- , Ezh2 fl/fl ;Cgamma1-cre and Ezh2 fl/fl ;Cgamma1-cre; Cdkn1a -/- littermate mice were immunized with SRBC to induce germinal center (GC) formation and were killed 10 days later. a Flow cytometry plot of one representative mouse spleen per group. The gated area shows the percentage of GC B cells (GL7 + FAS + ) within live B cells (B220 + DAPI-, see Supplementary Fig. 2A for gating strategy). b Average of GC B populations of each group of mice quantified by flow cytometry as in a ( n = 7 mice per group). c Formalin fixed paraffin embedded splenic tissue was stained for PNA, Ki67, EZH2, and B220. One representative picture of three spleens analyzed per group is shown. d - f Quantification of PNA staining from c ( n = 3 spleens per group). d ""#GC/spleen section"" is the count of all GC per spleen section. e ""GC area/total spleen area"" is the quantified area of each individual GC divided by the total area of the spleen section. f ""Total GC area/total spleen area"" is the sum of all GC quantified areas in a certain section divided by the total area of that spleen section. g Splenocytes were permeabilized and stained for GC B and EZH2 using a fluorochrome-conjugated anti EZH2 antibody. The gated area shows the percentage of GC B cells (GL7 + FAS + ) within live B cells (B220 + DAPI-, see Supplementary Fig. 2A for gating strategy) that are EZH2 positive. The flow plot shown is repre
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 3 Characterization of a 3D B cell follicular organoid to model the GC reaction. a Scheme of 3D B cell follicular organoid fabrication. Splenic B cells are co-encapsulated with 40LB stromal cells and IL4 into an RGD-presenting nanocomposite hydrogel that contain gelatin ionically cross-linked with synthetic silicate nanoparticles. b Representative fluorescence pictures of splenic GFP B cells in 3D organoid culture. c Flow cytometry plot of a 3D B cell follicular organoid. The gated area on the top shows the live B cells (B220 + DAPI-) and on the bottom plot, the organoid GC B cells (GL7 + FAS + ) within live B cells. d Average of GC B populations of organoids quantified as in c . e Average of percentage of proliferating organoid GC B populations, quantified as indicated in Supplementary Fig. 4B . f MFI of proliferation dye from e . g Flow cytometry plot of GC B cells stained with annexinV. h Average of percentage of apoptotic GC B cells quantified as in g . i RNA-seq profiles of organoid GC B cells after 4 and 6 days in culture (organoid GCB d4, n = 4 spleens, and d6, n = 3 spleens) were projected into the principal component space defined by GC B cells sorted from immunized mice (in vivo GCB, n = 6 mice), CD138 + plasma cells (in vivo PC, n = 6 mice) and FAS-GL7-IgD + B220 + naive B cells (NB, n = 3 mice). j , k Heat maps of gene expression level of GC B cells showed in i , represented as log2 ratio relative to mean naive B cells j and to mean plasma cells k . PC1/2 = pr
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 4 CDKN1A repression by EZH2 is required for GC B cell cycle progression. a - f Organoids were generated using B cells isolated from Ezh2 fl/fl ;Cgamma1-cre, Cdkn1a -/- , Ezh2 fl/fl ;Cgamma1-cre; Cdkn1a -/- and Ezh2 fl/fl control mice ( n = 3 mice per group) and were harvested for flow cytometry analysis after 4 days in culture. a Flow cytometry plots of representative organoids from each genotype. The gated area shows the percentage of organoid GC B cells (GL7 + FAS + ) within live B cells (B220 + DAPI-). b Average of percentage of organoid GC B populations quantified by flow cytometry as in a ( n = 3 biological replicates per group). c Organoids received a BrdU pulse of 2 h before harvest. Cell cycle was analyzed by BrdU staining and 7AAD to measure DNA content. The representative gated area shows the percentage of organoid GC B cells (GL7 + FAS + B220 + DAPI-) that are in S phase (BrdU + ). d Average of percentage of organoid GC B populations in S phase of each group of genotype quantified by flow cytometry as in c ( n = 3 biological replicates per group). e Organoid cells were permeabilized and stained for EZH2 and GC markers GL7, FAS, and B220 to identify organoid GC B cells. The flow cytometry plot shows one representative sample per genotype group. f MFI of EZH2 in organoid GC B cells ( n = 3) quantified by flow cytometry as in e . g - m Organoids were generated using B cells isolated from 3 Cdkn1a +/+ and 3 Cdkn1a -/- mice. g Scheme of EZH2 inhibitor GSK343 and Br
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 1. VEGF and bFGF promote the differentiation of MSCs into EC-like cells. (A) Expression of MSC surface markers was confirmed using flow cytometry. (B) Morphological features of isolated MSCs and MSC-derived EC-like cells following induction as observed under an inverted microscope. The red and white arrows indicate spindle-shaped and polygon-shaped cells, respectively. Magnification, x20. (C) Factor VIII expression was detected in MSC-derived EC-like cells via immunohistochemistry and light microscopy with brown staining indicating the positive signal. VEGF, vascular endothelial growth factor; bFGF, basic fibroblast growth factor; MSCs, mesenchymal stem cells; EC, endothelial cell; Ctrl, control; CD, cluster of differentiation.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 1 Loss of miR-143/145 results in reduced LT-HSC frequency. a Frequency of long-term-HSC (LT-HSC, CD45 + EPCR + CD48 - CD150 + ), LSK/short-term HSC (Lin - Sca1 + c-Kit + ), common myeloid progenitors (CMP, Lin - Sca1 - c-Kit + CD34 + CD16/32 lo ), granulocyte-macrophage progenitors (GMP, Lin - Sca1 - c-Kit + CD34 + CD16/32 hi ), and megakaryocyte-erythrocyte progenitors (MEPs, Lin - Sca1 - c-Kit + CD34 - CD16/32 lo ) in the marrow of 8-12-week- old wild-type (WT), miR-143/145 +/- , and miR-143/145 -/- mice, as analyzed by flow cytometry (median +- 1.5 IQR, WT n = 11, 143/145 +/- n = 9, 143/145 -/- n = 8). b Marrow cellularity (2 femurs and 2 tibias) (WT n = 3, 143/145 +/- n = 4, 143/145 -/- n = 8). c Colony-forming unit (CFU) assay of marrow cells from WT and miR-143/145 -/- mice (mean +- SEM, WT n = 11, 143/145 -/- n = 10). d Primary CFU cells were replated in equal proportions per condition, normalized to the number of input cells, to generate secondary CFUs (mean +- SEM, WT n = 9, 143/145 -/- n = 7). e Estimate of HSC frequency in WT and miR-143/145 -/- mice by limiting dilution assay. Shown is a log-fraction plot of the limiting dilution model. The slope of the line is the log-active cell fraction. The dotted lines give the 95% CI
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 4 Surgical denervation of young bone marrow induces premature HSC and niche aging (a) Schematic illustration of surgical denervation experiment. The sciatic and femoral nerves were transected in young (2 months old) C57BL/6 mice and the mice were analyzed 4 months post surgery, at 6 months of age. (b) Absolute numbers of HSCs (lineage - Sca-1 + c-Kit + CD48 - CD150 + ) isolated from Sham and Denervated (Den) femurs (n=7 mice). (c, d) Total peripheral blood (CD45.2 + ), blood myeloid (Mac-1 + CD45.2 + ) and blood B cell (B220 + CD45.2 + ) donor chimerism at the indicated time points post transplantation (c) and bone marrow donor chimerism (Total CD45.2 + (BM); Myeloid (My), Mac-1 + CD45.2 + ; B cell (B), B220 + CD45.2 +; and T cell (T), CD4 + /CD8 + CD45.2 + ) 5 months after primary transplantation of 200 HSCs derived from either sham or denervated (Den) femurs and transplanted in competition with young BM competitor cells (n=4 sham, 6 denervated mice) (left) and 5 months after secondary transplantation of 3 x 10 6 bone marrow from primary recipients (right) (d). (e) Left, representative confocal z -stack projections of HSCs sorted from sham or denervated femurs and stained with Cdc42, Tubulin and DAPI. Scale bar, 10 mum. Right, quantification of the percentage of Cdc42 and Tubulin polarized HSCs out of total HSCs scored (total of 356 sham and 353 denervated HSCs isolated from 4 mice per group). (f) Left, representative confocal z -stack pro
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 1 ATF3 maintains intestinal homeostasis. (A) Comparison of colon length between naive mice as indicated. (B) Colon crypts from mice were isolated by shaking colon fragments in EDTA and counted under light microscopy. (C) Flow cytometry analysis of Ki67 and CD24 expression in ileum crypts, gated on the CD45 - EpCAM + populations, from the indicated naive mice. (D) Representative micrographs showing intestinal organoids derived from naive mice. (E) Quantitative real-time PCR analysis of cell cycle genes in naive ileum organoids at day 6 of culture (""n"" indicates organoids derived from 4 mice each group). (F) Representative confocal images of whole mount tissues with co-immunofluorescence staining of UEA-1 and WGA in naive ileum villi. Results were from at least two independent experiments and ""n"" refers to the number of mice unless indicated otherwise. All mice were at the age of 2~3 months old when analyzed. Statistical analysis was done using Multiple T -test on Prism software. * P < 0.05, ** P < 0.005, *** P < 0.0005.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 7 ATF3 promotes IL-22-induced STAT3 phosphorylation by suppressing phosphatases. (A) Freshly isolated ileum crypts, or (B) ileum organoids at day 6 of culture, were stimulated with IL-22, followed by fixation and intracellular staining of phospho-STAT3, and analyzed by flow cytometry. Western blot analysis of (C) IL-22-stimulated CMT93 cells, or (D) IL-22-stimuated colon fragments isolated from the indicated mice, for the expression of the indicated proteins. (E) Quantitative real-time PCR analysis of IL-22R1 and IL-10R2 mRNA levels in freshly isolated ileum crypts from mice. (F) Flow cytometry analysis of IL-22R1 in freshly isolated ileum crypt cells gated on the CD45 - EpCAM + population. (G,H) Western blot analysis of unstimulated or IL-22-stimulated CMT93 cells for the indicated proteins. ATF3 -/- CMT93 cells with SHP2 knockdown (ATF3 -/- SHP2 KD ) were indicated. Images were representative of four independent experiments (G-H) . Results were from two independent experiments (A-F) . ""n"" refers to the number of mice analyzed (A,B,E,F) . Statistical analysis was done by multiple comparison in Two-way ANOVA test using Prism software. * P < 0.05, ** P < 0.005, *** P < 0.0005.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
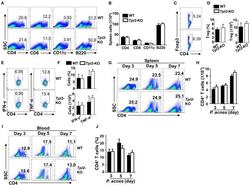
- Experimental details
- Figure 2 Tpl2 didn't affect peripheral immune activation during FH pathogenesis. The splenic cells or peripheral blood immune cells were isolated from P. acnes -primed WT and Tpl2 -KO mice at day 3, 5, and 7 as described in Materials and methods ( n = 4 mice/group), and subjected for flow cytometry analysis. (A-F) Flow cytometry analysis of the frequencies and absolute numbers of CD4 + T cells, CD8 + T cells, B220 + B cells, CD11c + dendritic cells (A,B) , CD4 + Foxp3 + Treg cells (C,D) , and IFN-gamma- and TNF-alpha-producing pathogenic Th1 cells (E,F) in the spleens of WT and Tpl2 -KO mice at day 7 after P. acnes priming. Data are presented as representative plots of the frequencies of immune cell subpopulations (A,C,E) and a summary graph of the cell frequencies or absolute cell numbers (B,D,F) . (G-J) Flow cytometry analysis of the frequencies and absolute numbers of CD4 + T cells in the spleens (G,H) or peripheral blood (I,J) of WT and Tpl2 -KO mice at day 3, 5, and 7 after P. acnes priming. Data are presented as representative plots of the frequencies of immune cell subpopulations (G,I) and a summary graph of the cell frequencies or absolute cell numbers (H,J) . Results are mean +- SD from three independent experiments.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
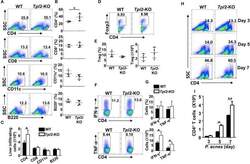
- Experimental details
- Figure 3 Tpl2 reduced liver infiltration of pathogenic CD4 + T cells. Liver-infiltrating immune cells were isolated from P. acnes -primed WT and Tpl2 -KO mice at day 3, 5, and 7 as described in Materials and methods ( n = 4 mice/group). (A-G) Flow cytometry analysis of the frequencies and absolute numbers of CD4 + and CD8 + T cells, B220 + B cell, CD11c + dendritic cells (A-C) , CD4 + Foxp3 + Treg cells (D,E) , and IFN-gamma- and TNF-alpha-producing pathogenic Th1 cells (F,G) in the livers of WT and Tpl2 -KO mice at day 7 after P. acnes priming. Data are presented as representative plots of the frequencies of immune cell subpopulations (A,D,F) and a summary graph of the frequencies and absolute cell numbers (B,C,E,G) . (H,I) Flow cytometry analysis of the frequencies and absolute numbers of CD4 + T cells in the livers of WT and Tpl2 -KO mice at day 3, 5, and 7 after P. acnes priming. Data are presented as representative plots of the frequencies of immune cell subpopulations (H) and a summary graph of the absolute cell numbers (I) . Results are mean +- SD from three independent experiments. Two-tailed Student's t -tests were performed. * P < 0.05; ** P < 0.01.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
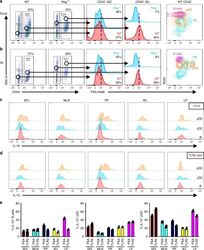
- Experimental details
- Fig. 6 TLR2 + macrophages, pDCs, and PB in the small intestine bind PSA and induce IL-10 secretion. Gating strategy for mononuclear cells isolated from a duodenum (Duod) and b ileum (Ile) of WT and Rag mice analyzed for binding of fluorescent A488-conjugated PSA (left two plots). CD45 - intra-epithelial cells (CD45 - IEC: middle histogram) and CD45 + gated intra-epithelial leukocytes (CD45 + IEL: second right histogram) isolated from the a Duod and b Ile of WT (red) and Rag (blue) mice were analyzed for reactivity to PSA-A488. CD45 + CD11c - B220 + B cells, PDCA1 + B220 + CD11c + pDCs and CD138 + B220 + PB and B220 low PC isolated from a Duod and b Ile of WT mice were analyzed for PSA reactivity (right histogram). Flow cytometry plots show CD11c + PDCA1 - cDC, PDCA1 + B220 + pDC, and B220 + CD19 + B cells isolated from spleen, MLN, PP, IEL, or LP of WT mice were stimulated with c PSA or d LTA-SA (TLR2 agonist) and analyzed for IL-10 expression. e Plots summarize data from c and d and show % IL-10 + B cells (left), cDCs (middle), and pDCs (right) from spleen, MLN, PP, IEL, and LP stimulated with PSA or TLR2 (LTA-SA) ( n = 3 mice). All data show mean +- SEM
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
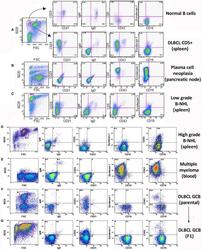
- Experimental details
- Figure 2 Analysis of the B lymphocyte populations expanded in TRAF3/BCL2 double-tg mice with lymphoid dyscrasias. Three-color flow-cytometry analysis was performed to determine the phenotype of expanded B lymphocyte populations. Gating of the expanded population was based on the CD45R/B220 and FSC plot of each sample analyzed and is indicated in the figure. The surface molecules analyzed are indicated in the plots, as well as the percentage of cells found in each quadrant. The quadrants settings were selected based on the staining of isotype-controls (not shown). The tissue source where the analyzed lymphocytes were extracted from and the type of B cell malignancy developed by the TRAF3/BCL2 double-tg mice, according to the flow-cytometry and immunohistochemical analysis, is indicated in the figure.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 4 Ptpn6 deletion drives robust anti-tumor immunity in two immune-rich syngeneic tumor lines. (A) Flow cytometric analysis of live CD45 + cells from B16F10 melanoma tumors isolated from tamoxifen-treated Ptpn6 fl/fl and Ptpn6 fl/fl ERT2-cre mice at day 14 post tumor implantation. (B) B16F10 tumor volume measurements in tamoxifen-treated Ptpn6 fl/fl ERT2-cre and Ptpn6 fl/fl mice. Each data point represents the average tumor volume of all mice in a given group. Data is representative of three independent experiments with 5-7 mice/group. (C) Shp1 protein relative to total Erk2 protein in peripheral blood cells from indicated mice 14 days after initial tamoxifen dose (200 mg/kg bid for 4 days) and 7 days after B16F10 tumor cells were implanted. Data is representative of at least three independent experiments with 5-7 mice per group. (D) Flow cytometric analysis of live CD45 + cells from E0771 tumors isolated from tamoxifen-treated Ptpn6 fl/fl and Ptpn6 fl/fl ERT2-cre mice at day 19 post tumor implantation. (E) E0771 tumor volume measurements in tamoxifen-treated Ptpn6 fl/fl ERT2-cre and Ptpn6 fl/fl mice. Each data point represents the average tumor volume of all mice in a given group. Data is representative of three independent experiments with 4-5 mice per group. Statistical significance was calculated at each time point using an unpaired t -test. (F) Shp1 protein relative to total Erk2 protein in peripheral blood cells from indicated mice 14 days after initial tamoxifen d
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 5. K14Cre;fl/fl mice exhibit prominent autoimmunity with Th2/Th17 cell polarization and abnormal B cell activation leading to autoantibody production. (A) A representative image of the gross appearance of spleens and inguinal lymph nodes from 3-mo-old fl/fl and K14Cre;fl/fl mice. (B) The number of splenocytes in 3-mo-old fl/fl and K14Cre;fl/fl mice was counted. n = 4 per each group. (C) Lymphocytes isolated from inguinal lymph nodes were stained for CD3 and CD4 and intracellularly stained for IL-4, IL-17A, and IFN-gamma. (Top) Representative two-dimensional plots for IL-4 and IL-17A in CD3 + CD4 + cells. (Bottom) The percentages of cells positive for IL-4, IL-17A, and IFN-gamma are summarized. n = 7 mice per each group. (D) Splenocytes from 3-mo-old fl/fl and K14Cre;fl/fl mice were stained for B220 and CD19. B220 + CD19 + cells were analyzed for CD19 expression. A representative histogram of mean fluorescence intensity (MFI; top) and the comparison of mean fluorescence intensities (bottom) are shown. n = 4 mice per genotype. (E) Sera from 3- and 8-mo-old mice were analyzed for the concentrations of total IgG, IgM, and IgA. n = 5-7 mice per genotype. Note that the results are shown with log scale. (F) Sera from 1-, 2-, 3-, and 8-mo-old mice were analyzed for the concentration of IL-6. n = 6-7 mice per genotype. (G) B cells were isolated from splenocytes of 3-mo-old mice and cultured for 48 h, either unstimulated or stimulated with anti-CD40 antibody (alphaCD40Ab). The c
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 1 Development of myeloid cell populations is not detectably altered in MphCK2alpha -/- mice. (A) Schematic of Csnk2a targeting vector. (B) Immunoblots of lysates from BMDMs, splenocytes, or peritoneal cavity cells of LysM cre or LysM cre CK2alpha fl/fl (MphCK2alpha - / - ) mice were probed for CK2alpha or beta-actin as a loading control. n = 2. (C) Splenic myeloid cell populations were stained and analyzed by flow cytometry. Plots depict gating for representative F4/80+ macrophages, Ly6C+ monocytes, and neutrophils (also Ly6G+). (D) Cells from spleen were stained for flow cytometry to enumerate immune cell populations. Data are from pooled experiments, n = 4-12 mice/group. (E) Cells from bone marrow were stained for flow cytometry to evaluate bone marrow precursor populations including common myeloid progenitors (CMPs), granulocyte-monocyte progenitors (GMPs), and megakaryocyte-erythrocyte progenitor (MEP). Cells were pre-gated on Lin-(B220, CD3, CD11b, GR-1 and TER-119), Sca-1-, cKit-. Density plots are representative from two experiments.
- Conjugate
- Yellow dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry