Antibody data
- Antibody Data
- Antigen structure
- References [7]
- Comments [0]
- Validations
- Immunohistochemistry [3]
- Flow cytometry [1]
- Other assay [7]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 14-9457-82 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD45 Monoclonal Antibody (CD45-2B11), eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The CD45-2B11 monoclonal antibody reacts with human CD45, also known as Leukocyte Common Antigen (LCA). CD45 is expressed by all hematopoietic cells excluding circulating erythrocytes and platelets. The cytoplasmic portion of CD45 has tyrosine phosphatase enzymatic activity and plays an important role in lymphocyte proliferation and differentiation. The 2B11 antibody is useful for recognition of normal and neoplastic lymphoid cells. Applications Reported: This CD45-2B11 antibody has been reported for use in flow cytometric analysis, immunoblotting, immunohistochemical staining of formalin-fixed paraffin embedded tissue sections, and immunocytochemistry. Applications Tested: This CD45-2B11 antibody has been tested by immunohistochemisty on formalin-fixed paraffin embedded tissue using high pH antigen retrieval. This can be used at less than or equal to 10 µg/mL. This CD45-2B11 antibody has also been tested by western blot at 5 µg/mL and by flow cytometric analysis on human peripheral blood cells at 0.25 µg/test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest. Purity: Greater than 90%, as determined by SDS-PAGE. Aggregation: Less than 10%, as determined by HPLC. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- CD45-2B11
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- 4° C
Submitted references Exosomes derived from stem cells of human deciduous exfoliated teeth inhibit angiogenesis in vivo and in vitro via the transfer of miR-100-5p and miR-1246.
Macrophage-derived IL-6 trans-signalling as a novel target in the pathogenesis of bronchopulmonary dysplasia.
Ex vivo culture of intact human patient derived pancreatic tumour tissue.
Adoptive transfer of xenoantigen‑stimulated T cell receptor Vβ‑restricted human regulatory T cells prevents porcine islet xenograft rejection in humanized mice.
Glucocorticoid receptor expression in resident and hematopoietic cells in IgG4-related disease.
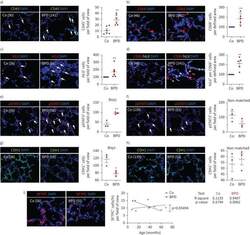
Heterogeneous fibroblasts underlie age-dependent tertiary lymphoid tissues in the kidney.
Inflammatory malignant fibrous histiocytoma: distinction from Hodgkin's disease and non-Hodgkin's lymphoma by a panel of leukocyte markers.
Liu P, Zhang Q, Mi J, Wang S, Xu Q, Zhuang D, Chen W, Liu C, Zhang L, Guo J, Wu X
Stem cell research & therapy 2022 Mar 3;13(1):89
Stem cell research & therapy 2022 Mar 3;13(1):89
Macrophage-derived IL-6 trans-signalling as a novel target in the pathogenesis of bronchopulmonary dysplasia.
Hirani D, Alvira CM, Danopoulos S, Milla C, Donato M, Tian L, Mohr J, Dinger K, Vohlen C, Selle J, V Koningsbruggen-Rietschel S, Barbarino V, Pallasch C, Rose-John S, Odenthal M, Pryhuber GS, Mansouri S, Savai R, Seeger W, Khatri P, Al Alam D, Dötsch J, Alejandre Alcazar MA
The European respiratory journal 2022 Feb;59(2)
The European respiratory journal 2022 Feb;59(2)
Ex vivo culture of intact human patient derived pancreatic tumour tissue.
Kokkinos J, Sharbeen G, Haghighi KS, Ignacio RMC, Kopecky C, Gonzales-Aloy E, Youkhana J, Timpson P, Pereira BA, Ritchie S, Pandzic E, Boyer C, Davis TP, Butler LM, Goldstein D, McCarroll JA, Phillips PA
Scientific reports 2021 Jan 21;11(1):1944
Scientific reports 2021 Jan 21;11(1):1944
Adoptive transfer of xenoantigen‑stimulated T cell receptor Vβ‑restricted human regulatory T cells prevents porcine islet xenograft rejection in humanized mice.
Jin X, Hu M, Gong L, Li H, Wang Y, Ji M, Li H
Molecular medicine reports 2018 Nov;18(5):4457-4467
Molecular medicine reports 2018 Nov;18(5):4457-4467
Glucocorticoid receptor expression in resident and hematopoietic cells in IgG4-related disease.
Iguchi T, Takaori K, Mii A, Sato Y, Suzuki Y, Yoshifuji H, Seno H, Ogawa O, Omori K, Bessho K, Kondo S, Yoshizaki T, Nakashima H, Saito T, Mimori T, Haga H, Kawano M, Yanagita M
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2018 Jun;31(6):890-899
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2018 Jun;31(6):890-899
Heterogeneous fibroblasts underlie age-dependent tertiary lymphoid tissues in the kidney.
Sato Y, Mii A, Hamazaki Y, Fujita H, Nakata H, Masuda K, Nishiyama S, Shibuya S, Haga H, Ogawa O, Shimizu A, Narumiya S, Kaisho T, Arita M, Yanagisawa M, Miyasaka M, Sharma K, Minato N, Kawamoto H, Yanagita M
JCI insight 2016 Jul 21;1(11):e87680
JCI insight 2016 Jul 21;1(11):e87680
Inflammatory malignant fibrous histiocytoma: distinction from Hodgkin's disease and non-Hodgkin's lymphoma by a panel of leukocyte markers.
Khalidi HS, Singleton TP, Weiss SW
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 1997 May;10(5):438-42
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 1997 May;10(5):438-42
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry on formalin-fixed paraffin embedded human pancreas, using 10 µg/mL of Mouse IgG1 K Isotype Control (left) or 10 µg/mL Anti-Human CD45 Purified (right) followed by Anti-Mouse IgG Biotin, and DAB visualization.Nuclei are counterstained with hematoxylin.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
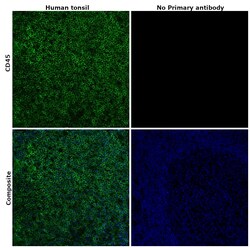
- Experimental details
- Immunohistochemical analysis of CD45 was performed using formalin-fixed paraffin-embedded human tonsil tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - Low pH (10X) (Product # 00-4955-58) diluted to 1X solution in water in a microwave at 100 degree Celsius for 10 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without CD45 Monoclonal Antibody (CD45-2B11), eBioscience™ (Product # 14-9457-82) at 5 µg/mLconcentration in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. Detection was performed using Goat anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32723) at a dilution of 1:2000 in 0.1% normal goat serum for 45 minutes at room temperature. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and deconvoluted externally.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
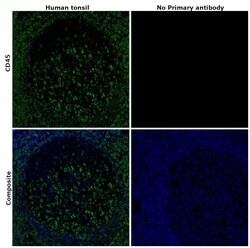
- Experimental details
- Immunohistochemical analysis of CD45 was performed using formalin-fixed paraffin-embedded human tonsil tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - High pH (10X) (Product # 00-4956-58) diluted to 1X solution in water in a microwave at 100 degree Celsius for 10 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without CD45 Monoclonal Antibody (CD45-2B11), eBioscience™ (Product # 14-9457-82) at 5 µg/mLconcentration in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. Detection was performed using Goat anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32723) at a dilution of 1:2000 in 0.1% normal goat serum for 45 minutes at room temperature. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and deconvoluted externally.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
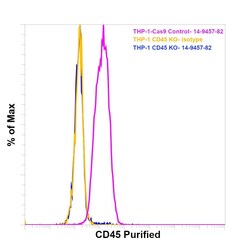
- Experimental details
- Knockout of CD45 was achieved by CRISPR-Cas9 genome editing using LentiArray™ Lentiviral sgRNA (Product # A32042, Assay ID CRISPR664203_LV) and LentiArray Cas9 Lentivirus (Product # A32064). Flow cytometry analysis of CD45 was performed by staining THP-1 CD45 Knock out cells with0.25 µg Mouse IgG1 kappa Isotype Control (P3.6.2.8.1), eBioscience™ (Product # 14-4714-82, yellow histogram) or 0.25 µg CD45 Monoclonal Antibody (CD45-2B11), eBioscience™ (Product # 14-9457-82, blue histogram) followed by Goat anti-Mouse IgG (H+L), Superclonal™ Recombinant Secondary Antibody, Alexa Fluor™ Plus 488 (Product # A55058, 1:1000 dilution). THP-1 Cas9 control cells was also stained with0.25 µg CD45 Monoclonal Antibody (CD45-2B11), eBioscience™ (Product # 14-9457-82, pink histogram) followed by the secondary antibody. Lossof signal was observed in the CD45 KOcells stained with CD45 antibody clone CD45-2B11 but not in the control Cas9cells. Viable cells were used for analysis, as determined by Fixable Viability Dye eFluor™ 780 (Product # 65-0865-18).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
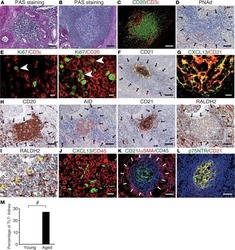
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
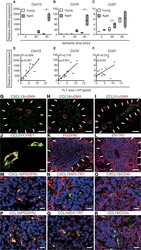
- FIGURE 9 Inflammation and interleukin 6 (IL-6)/signal transducer and activator of transcription 3 (STAT3) signalling in lungs of infants with bronchopulmonary dysplasia (BPD) and non-BPD. a) Representative immunofluorescent staining for immune cells using CD45 as a marker (green) in age-matched BPD and control lungs (Co); the lung identification numbers of the infants are indicated in brackets. Immune cells (CD45 + cells) were counted in 4-12 fields of view per lung. Summary data of the quantification of immune cells (CD45 + cells) per field of view for all infants (n=6 per group). DAPI: 4',6-diamidino-2-phenylindole. b) Representative co-immunofluorescent localisation of CD68 (marker of macrophages, red) in age-matched BPD and Co lungs; white arrows are indicating CD68 + cells. The analysis of CD68 + cells per 6-12 fields of view is shown next to the images; n=6 per group. c) Representative localisation of human IL6 (h IL6 , red) in lungs with BPD and Co; white arrows are indicating h IL6 + in situ hybridisation. The analysis of h IL6 + cells per 6-12 fields of view is shown next to the images; n=6 per group. d) Representative co-immunofluorescent localisation of CD68 (marker of macrophages, red) with h IL6 (white; in situ hybridisation) in age-matched BPD and Co lungs; white arrows are indicating CD68 + and h Il6 + . The analysis of h IL6 mRNA expression per CD68 + cells per 6-12 fields of view is shown next to the images; n=6 per group. e, f) Representative immunofluoresce
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
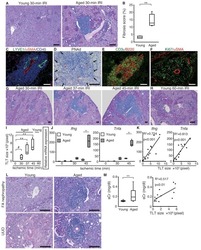
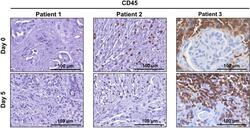
- Figure 6 CD45-positive lymphocytes remain viable for 5 days in human patient derived pancreatic ductal adenocarcinoma tumour explants. Immunohistochemistry was performed for lymphocyte marker CD45 on tumour explants from patients 1-3 at day 0 and day 5.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
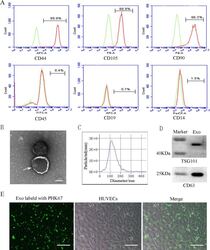
- Characterization and uptake of SHED-Exos. A Surface markers of SHED cells were analyzed by flow cytometry (FACS) and were positive for mesenchymal markers (CD44, CD105 and CD90) and negative for endothelial markers (CD45, CD19 and CD14). B The morphology of exosomes (indicated by arrows) was observed using a transmission electron microscope (TEM). Scale bar = 100 nm. C Particle size distribution of SHED-Exos assessed by nanoparticle tracking analysis (NTA). D Expression of exosome-specific CD63 and TSG101 validated using western blotting. E Efficient uptake of PKH67-labeled exosomes by HUVECs was detected at 24 h. Scale bars = 100 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
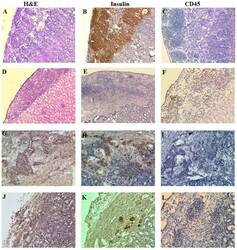
- Figure 9. Histology and immunohistochemical analysis of NICC xenografts. Representative hematoxylin and eosin staining images; and immunohistochemical staining images of porcine insulin and human CD45 in NICC xenograft samples from mice receiving (A-C) no human cells (NICC alone 84 days post-transplantation), (D-F) only human PBMCs (NICC + PBMC 28 days post-PBMC transfer), (G-I) human PBMCs and Xeno-Treg (NICC + PBMC + Xeno-Treg 84 days post-cell transfer) or (J-L) human PBMCs and Poly-Treg (NICC + PBMC + Poly-Treg 63 days post-cell transfer). (A, B, E-I, K and L) Magnification, x200; (C, D and J) magnification, x100. CD, cluster of differentiation; NICC, neonatal porcine islet cell clusters; PBMC, peripheral blood mononuclear cell; Poly-Treg, polyclonal Treg; Tregs, regulatory T cells; Xeno-Treg, Treg with xenoantigen specificity.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunohistochemistry
Immunohistochemistry