Antibody data
- Antibody Data
- Antigen structure
- References [181]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [72]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 17-0452-83 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD45R (B220) Monoclonal Antibody (RA3-6B2), APC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The RA3-6B2 monoclonal antibody reacts with exon A-restricted isoform of mouse CD45, a 220 kDa surface molecule. CD45R/B220 epitope is mainly expressed by the B cell lineage from early Pro-B to mature B cells. However, some activated T cells, lymphokine activated killer cells (LAK), NK cell progenitors in the bone marrow, and T cells of the lpr/lpr mutant mouse also express this antigen. Applications Reported: This RA3-6B2 antibody has been reported for use in flow cytometric analysis. Applications Tested: This RA3-6B2 antibody has been tested by flow cytometric analysis of mouse spleen cells. This can be used at less than or equal to 0.25 µg per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest. Excitation: 633-647 nm; Emission: 660 nm; Laser: Red Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human, Mouse
- Host
- Rat
- Isotype
- IgG
- Antibody clone number
- RA3-6B2
- Vial size
- 200 µg
- Concentration
- 0.2 mg/mL
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references RIPK3 and caspase 8 collaborate to limit herpes simplex encephalitis.
B cells imprint adoptively transferred CD8(+) T cells with enhanced tumor immunity.
Lenvatinib for effectively treating antiangiogenic drug-resistant nasopharyngeal carcinoma.
Transient expansion and myofibroblast conversion of adipogenic lineage precursors mediate bone marrow repair after radiation.
A rapid CRISPR competitive assay for in vitro and in vivo discovery of potential drug targets affecting the hematopoietic system.
TNFRSF13B genotypes control immune-mediated pathology by regulating the functions of innate B cells.
Polyvinyl alcohol hydrolysis rate and molecular weight influence human and murine HSC activity ex vivo.
Type 2 diabetic mice enter a state of spontaneous hibernation-like suspended animation following accumulation of uric acid.
Metabolic preconditioning in CD4+ T cells restores inducible immune tolerance in lupus-prone mice.
Targeting Ovarian Carcinoma with TSP-1:CD47 Antagonist TAX2 Activates Anti-Tumor Immunity.
Impaired HA-specific T follicular helper cell and antibody responses to influenza vaccination are linked to inflammation in humans.
The folate cycle enzyme MTHFD2 induces cancer immune evasion through PD-L1 up-regulation.
Cell-permeable transgelin-2 as a potent therapeutic for dendritic cell-based cancer immunotherapy.
Stromal SOX2 Upregulation Promotes Tumorigenesis through the Generation of a SFRP1/2-Expressing Cancer-Associated Fibroblast Population.
Myeloid Cell CK2 Regulates Inflammation and Resistance to Bacterial Infection.
Shp1 Loss Enhances Macrophage Effector Function and Promotes Anti-Tumor Immunity.
TNFAIP3 Plays a Role in Aging of the Hematopoietic System.
G protein-coupled receptor 68 increases the number of B lymphocytes.
The lysophospholipase D enzyme Gdpd3 is required to maintain chronic myelogenous leukaemia stem cells.
Reprogrammed mRNA translation drives resistance to therapeutic targeting of ribosome biogenesis.
Adaptive response to inflammation contributes to sustained myelopoiesis and confers a competitive advantage in myelodysplastic syndrome HSCs.
Retroviral Overexpression of CXCR4 on Murine B-1a Cells and Adoptive Transfer for Targeted B-1a Cell Migration to the Bone Marrow and IgM Production.
Crucial Roles of the RIP Homotypic Interaction Motifs of RIPK3 in RIPK1-Dependent Cell Death and Lymphoproliferative Disease.
High-fat diet induced central adiposity (visceral fat) is associated with increased fibrosis and decreased immune cellularity of the mesenteric lymph node in mice.
Generation and characterization of the Eµ-Irf8 mouse model.
Modest Declines in Proteome Quality Impair Hematopoietic Stem Cell Self-Renewal.
MOSPD2 is a therapeutic target for the treatment of CNS inflammation.
Id1 and Id3 Maintain Steady-State Hematopoiesis by Promoting Sinusoidal Endothelial Cell Survival and Regeneration.
Diverse CD8 T Cell Responses to Viral Infection Revealed by the Collaborative Cross.
Glucocorticoids Promote the Onset of Acute Experimental Colitis and Cancer by Upregulating mTOR Signaling in Intestinal Epithelial Cells.
Tpl2 Protects Against Fulminant Hepatitis Through Mobilization of Myeloid-Derived Suppressor Cells.
Bacteroides fragilis polysaccharide A induces IL-10 secreting B and T cells that prevent viral encephalitis.
Nidogen-1 Contributes to the Interaction Network Involved in Pro-B Cell Retention in the Peri-sinusoidal Hematopoietic Stem Cell Niche.
Cohesin-mediated NF-κB signaling limits hematopoietic stem cell self-renewal in aging and inflammation.
Targeted selection of HIV-specific antibody mutations by engineering B cell maturation.
Modulation of autoimmune pathogenesis by T cell-triggered inflammatory cell death.
Ptpn21 Controls Hematopoietic Stem Cell Homeostasis and Biomechanics.
The Hematopoietic Oxidase NOX2 Regulates Self-Renewal of Leukemic Stem Cells.
Proapoptotic BIM Impacts B Lymphoid Homeostasis by Limiting the Survival of Mature B Cells in a Cell-Autonomous Manner.
Dysregulated TRAF3 and BCL2 Expression Promotes Multiple Classes of Mature Non-hodgkin B Cell Lymphoma in Mice.
ATF3 Sustains IL-22-Induced STAT3 Phosphorylation to Maintain Mucosal Immunity Through Inhibiting Phosphatases.
Orally administered heat-killed Lactobacillus paracasei MCC1849 enhances antigen-specific IgA secretion and induces follicular helper T cells in mice.
EDAG promotes the expansion and survival of human CD34+ cells.
Cbl Ubiquitin Ligases Control B Cell Exit from the Germinal-Center Reaction.
Host Tumor Suppressor p18(INK4c) Functions as a Potent Cell-Intrinsic Inhibitor of Murine Gammaherpesvirus 68 Reactivation and Pathogenesis.
Adrenergic nerve degeneration in bone marrow drives aging of the hematopoietic stem cell niche.
Bone marrow‑derived mesenchymal stem cell‑conditioned medium attenuates tubulointerstitial fibrosis by inhibiting monocyte mobilization in an irreversible model of unilateral ureteral obstruction.
Mitophagy in Intestinal Epithelial Cells Triggers Adaptive Immunity during Tumorigenesis.
miR-143/145 differentially regulate hematopoietic stem and progenitor activity through suppression of canonical TGFβ signaling.
Loss of pleckstrin-2 reverts lethality and vascular occlusions in JAK2V617F-positive myeloproliferative neoplasms.
Tanshinone IIA and Astragaloside IV promote the angiogenesis of mesenchymal stem cell-derived endothelial cell-like cells via upregulation of Cx37, Cx40 and Cx43.
Inactivation of mTORC1 Signaling in Osterix-Expressing Cells Impairs B-cell Differentiation.
IL-2/Anti-IL-2 Complex Attenuates Inflammation and BBB Disruption in Mice Subjected to Traumatic Brain Injury.
The Interleukin (IL)-1R1 pathway is a critical negative regulator of PyMT-mediated mammary tumorigenesis and pulmonary metastasis.
Multiple functional therapeutic effects of TnP: A small stable synthetic peptide derived from fish venom in a mouse model of multiple sclerosis.
ORMDL3 Facilitates the Survival of Splenic B Cells via an ATF6α-Endoplasmic Reticulum Stress-Beclin1 Autophagy Regulatory Pathway.
Diet-induced obesity causes visceral, but not subcutaneous, lymph node hyperplasia via increases in specific immune cell populations.
Inflammasome-driven catecholamine catabolism in macrophages blunts lipolysis during ageing.
DNA-binding of the Tet-transactivator curtails antigen-induced lymphocyte activation in mice.
EZH2 enables germinal centre formation through epigenetic silencing of CDKN1A and an Rb-E2F1 feedback loop.
ADAR1 deletion induces NFκB and interferon signaling dependent liver inflammation and fibrosis.
The creatine kinase pathway is a metabolic vulnerability in EVI1-positive acute myeloid leukemia.
Differential cytokine contributions of perivascular haematopoietic stem cell niches.
Peptidylarginine deiminase 2 is required for tumor necrosis factor alpha-induced citrullination and arthritis, but not neutrophil extracellular trap formation.
Loss of IP(3) Receptor-Mediated Ca(2+) Release in Mouse B Cells Results in Abnormal B Cell Development and Function.
Ubiquitination of hnRNPA1 by TRAF6 links chronic innate immune signaling with myelodysplasia.
Recipient bone marrow assimilates the myeloid/lymphoid reconstitution of distinct fetal hematopoietic stem cells.
Antibody Tumor Targeting Is Enhanced by CD27 Agonists through Myeloid Recruitment.
Irgm1 coordinately regulates autoimmunity and host defense at select mucosal surfaces.
Epithelial Fli1 deficiency drives systemic autoimmunity and fibrosis: Possible roles in scleroderma.
CD4 T Helper Cells Instruct Lymphopenia-Induced Memory-Like CD8 T Cells for Control of Acute LCMV Infection.
Alkylator-Induced and Patient-Derived Xenograft Mouse Models of Therapy-Related Myeloid Neoplasms Model Clinical Disease and Suggest the Presence of Multiple Cell Subpopulations with Leukemia Stem Cell Activity.
Heme Oxygenase-1-Expressing Dendritic Cells Promote Foxp3+ Regulatory T Cell Differentiation and Induce Less Severe Airway Inflammation in Murine Models.
Monocyte Adhesion and Plaque Recruitment During Atherosclerosis Development Is Regulated by the Adapter Protein Chat-H/SHEP1.
Transcriptional regulation of the proto-oncogene Zfp521 by SPI1 (PU.1) and HOXC13.
Reprogramming mouse fibroblasts into engraftable myeloerythroid and lymphoid progenitors.
Epigenetic Memory Underlies Cell-Autonomous Heterogeneous Behavior of Hematopoietic Stem Cells.
Dendritic cell sphingosine-1-phosphate lyase regulates thymic egress.
The 4E-BP-eIF4E axis promotes rapamycin-sensitive growth and proliferation in lymphocytes.
CXCL16-positive dendritic cells enhance invariant natural killer T cell-dependent IFNγ production and tumor control.
Adipose tissue adaptive response to trans-10,cis-12-conjugated linoleic acid engages alternatively activated M2 macrophages.
Caspase-12, but Not Caspase-11, Inhibits Obesity and Insulin Resistance.
Murine iPSC-Derived Macrophages as a Tool for Disease Modeling of Hereditary Pulmonary Alveolar Proteinosis due to Csf2rb Deficiency.
Resident T Cells Are Unable To Control Herpes Simplex Virus-1 Activity in the Brain Ependymal Region during Latency.
miR-22 has a potent anti-tumour role with therapeutic potential in acute myeloid leukaemia.
Type I IFN Does Not Promote Susceptibility to Foodborne Listeria monocytogenes.
Standard sub-thermoneutral caging temperature influences radiosensitivity of hematopoietic stem and progenitor cells.
Tamm-Horsfall Protein Regulates Granulopoiesis and Systemic Neutrophil Homeostasis.
Mitochondrial ATP transporter Ant2 depletion impairs erythropoiesis and B lymphopoiesis.
The Inflammatory Caspases-1 and -11 Mediate the Pathogenesis of Dermatitis in Sharpin-Deficient Mice.
Bcl6 middle domain repressor function is required for T follicular helper cell differentiation and utilizes the corepressor MTA3.
Endothelial Gata5 transcription factor regulates blood pressure.
Core fucosylation of IgG B cell receptor is required for antigen recognition and antibody production.
Disruption of p21-activated kinase 1 gene diminishes atherosclerosis in apolipoprotein E-deficient mice.
BAFF activation of the ERK5 MAP kinase pathway regulates B cell survival.
Apoptotic cells trigger a membrane-initiated pathway to increase ABCA1.
Peroxisomal lipid synthesis regulates inflammation by sustaining neutrophil membrane phospholipid composition and viability.
MHC-mismatched mixed chimerism mediates thymic deletion of cross-reactive autoreactive T cells and prevents insulitis in nonobese diabetic mice.
Natural IgM is produced by CD5- plasma cells that occupy a distinct survival niche in bone marrow.
TGF-β3-expressing CD4+CD25(-)LAG3+ regulatory T cells control humoral immune responses.
Aging-associated inflammation promotes selection for adaptive oncogenic events in B cell progenitors.
B-cell-intrinsic hepatitis C virus expression leads to B-cell-lymphomagenesis and induction of NF-κB signalling.
Self DNA from lymphocytes that have undergone activation-induced cell death enhances murine B cell proliferation and antibody production.
Diet-induced obesity promotes myelopoiesis in hematopoietic stem cells.
α-1-Antitrypsin (AAT)-modified donor cells suppress GVHD but enhance the GVL effect: a role for mitochondrial bioenergetics.
Transcription elongation factor ELL2 drives Ig secretory-specific mRNA production and the unfolded protein response.
CD4+ T cells promote the transition from hypertrophy to heart failure during chronic pressure overload.
TRAF6 is a critical regulator of LMP1 functions in vivo.
A TNF-α-CCL20-CCR6 axis regulates Nod1-induced B cell responses.
Peptidylarginine deiminase 4 contributes to tumor necrosis factor α-induced inflammatory arthritis.
CD41 is a reliable identification and activation marker for murine basophils in the steady state and during helminth and malarial infections.
Rhof promotes murine marginal zone B cell development.
Eosinophils regulate peripheral B cell numbers in both mice and humans.
IκBε is a key regulator of B cell expansion by providing negative feedback on cRel and RelA in a stimulus-specific manner.
Relapsing-remitting central nervous system autoimmunity mediated by GFAP-specific CD8 T cells.
TDP2-dependent non-homologous end-joining protects against topoisomerase II-induced DNA breaks and genome instability in cells and in vivo.
Genome-wide mouse mutagenesis reveals CD45-mediated T cell function as critical in protective immunity to HSV-1.
CD30 is required for activation of a unique subset of interleukin-17A-producing γδ T cells in innate immunity against Mycobacterium bovis Bacillus Calmette-Guerin infection.
VBP15, a novel anti-inflammatory and membrane-stabilizer, improves muscular dystrophy without side effects.
Neuronal and nonneuronal cholinergic structures in the mouse gastrointestinal tract and spleen.
(18)F-FDG-PET/CT imaging in an IL-6- and MYC-driven mouse model of human multiple myeloma affords objective evaluation of plasma cell tumor progression and therapeutic response to the proteasome inhibitor ixazomib.
A type I IFN-Flt3 ligand axis augments plasmacytoid dendritic cell development from common lymphoid progenitors.
AID-expressing germinal center B cells cluster normally within lymph node follicles in the absence of FDC-M1+ CD35+ follicular dendritic cells but dissipate prematurely.
Long-term prevention of chronic allograft rejection by regulatory T-cell immunotherapy involves host Foxp3-expressing T cells.
Antioxidant treatment regulates the humoral immune response during acute viral infection.
Cellular-FLIP, Raji isoform (c-FLIP R) modulates cell death induction upon T-cell activation and infection.
Reciprocal effects of rab7 deletion in activated and neglected T cells.
Thymic damage, impaired negative selection, and development of chronic graft-versus-host disease caused by donor CD4+ and CD8+ T cells.
Lipidoid Nanoparticles Containing PD-L1 siRNA Delivered In Vivo Enter Kupffer Cells and Enhance NK and CD8(+) T Cell-mediated Hepatic Antiviral Immunity.
MHC class II-dependent B cell APC function is required for induction of CNS autoimmunity independent of myelin-specific antibodies.
Myeloid dendritic cells (DCs) of mice susceptible to paracoccidioidomycosis suppress T cell responses whereas myeloid and plasmacytoid DCs from resistant mice induce effector and regulatory T cells.
Th17 effector cells support B cell responses outside of germinal centres.
Induction of IL-10-producing CD1dhighCD5+ regulatory B cells following Babesia microti-infection.
Activation-induced cytidine deaminase-initiated off-target DNA breaks are detected and resolved during S phase.
Biphenotypic B-lymphoid/myeloid cells expressing low levels of Pax5: potential targets of BAL development.
Up-regulation of a HOXA-PBX3 homeobox-gene signature following down-regulation of miR-181 is associated with adverse prognosis in patients with cytogenetically abnormal AML.
Impact of enzymatic tissue disintegration on the level of surface molecule expression and immune cell function.
IKKα-mediated signaling circuitry regulates early B lymphopoiesis during hematopoiesis.
Mechanisms of indirect acute lung injury: a novel role for the coinhibitory receptor, programmed death-1.
Critical role of B cell lymphoma 10 in BAFF-regulated NF-κB activation and survival of anergic B cells.
TRAF binding is required for a distinct subset of in vivo B cell functions of the oncoprotein LMP1.
MyD88 signaling in B cells regulates the production of Th1-dependent antibodies to AAV.
PAD4-mediated neutrophil extracellular trap formation is not required for immunity against influenza infection.
Deletion of genes encoding PU.1 and Spi-B in B cells impairs differentiation and induces pre-B cell acute lymphoblastic leukemia.
Distinct developmental requirements for isolated lymphoid follicle formation in the small and large intestine: RANKL is essential only in the small intestine.
Memory T cells persisting in the brain following MCMV infection induce long-term microglial activation via interferon-γ.
Analysis of the role of IL-21 in development of murine B cell progenitors in the bone marrow.
Role of central leptin signaling in the starvation-induced alteration of B-cell development.
Mn(III) meso-tetrakis-(N-ethylpyridinium-2-yl) porphyrin mitigates total body irradiation-induced long-term bone marrow suppression.
Sensitized phenotypic screening identifies gene dosage sensitive region on chromosome 11 that predisposes to disease in mice.
Microbiota-induced tertiary lymphoid tissues aggravate inflammatory disease in the absence of RORgamma t and LTi cells.
Nuclear export of the NF-κB inhibitor IκBα is required for proper B cell and secondary lymphoid tissue formation.
Enhanced Toll-like receptor (TLR) responses of TNFR-associated factor 3 (TRAF3)-deficient B lymphocytes.
Lung CD103+ dendritic cells efficiently transport influenza virus to the lymph node and load viral antigen onto MHC class I for presentation to CD8 T cells.
Negative selection by IgM superantigen defines a B cell central tolerance compartment and reveals mutations allowing escape.
Sertoli cell-specific deletion of the androgen receptor compromises testicular immune privilege in mice.
Validation of MdmX as a therapeutic target for reactivating p53 in tumors.
Factor VIII delivered by haematopoietic stem cell-derived B cells corrects the phenotype of haemophilia A mice.
The innate immune response to uropathogenic Escherichia coli involves IL-17A in a murine model of urinary tract infection.
Regulation of follicular B cell differentiation by the related E26 transformation-specific transcription factors PU.1, Spi-B, and Spi-C.
The death effector domain protein PEA-15 negatively regulates T-cell receptor signaling.
Peripheral B cell tolerance and function in transgenic mice expressing an IgD superantigen.
c-Myb is required for pro-B cell differentiation.
A critical role of TAK1 in B-cell receptor-mediated nuclear factor kappaB activation.
Endogenous IL-21 restricts CD8+ T cell expansion and is not required for tumor immunity.
Dendritic cells are required for optimal activation of natural killer functions following primary infection with herpes simplex virus type 1.
E47 controls the developmental integrity and cell cycle quiescence of multipotential hematopoietic progenitors.
E47 controls the developmental integrity and cell cycle quiescence of multipotential hematopoietic progenitors.
p27 deficiency cooperates with Bcl-2 but not Bax to promote T-cell lymphoma.
Phospholipase Cgamma2 contributes to light-chain gene activation and receptor editing.
Silent development of memory progenitor B cells.
Silent development of memory progenitor B cells.
B-1 B lymphocytes require Blimp-1 for immunoglobulin secretion.
Negative regulation of activation-induced cytidine deaminase in B cells.
Essential role of phospholipase C gamma 2 in early B-cell development and Myc-mediated lymphomagenesis.
In vivo ablation of CD11c-positive dendritic cells increases susceptibility to herpes simplex virus type 1 infection and diminishes NK and T-cell responses.
A role for brain-derived neurotrophic factor in B cell development.
Impaired immune responses and B-cell proliferation in mice lacking the Id3 gene.
Identification of monoclonal antibodies for immunohistochemical staining of feline B lymphocytes in frozen and formalin-fixed paraffin-embedded tissues.
Surface antigen expression and immunoglobulin gene rearrangement during mouse pre-B cell development.
Abnormalities induced by the mutant gene Ipr: expansion of a unique lymphocyte subset.
Guo H, Koehler HS, Mocarski ES, Dix RD
PLoS pathogens 2022 Sep;18(9):e1010857
PLoS pathogens 2022 Sep;18(9):e1010857
B cells imprint adoptively transferred CD8(+) T cells with enhanced tumor immunity.
Smith AS, Knochelmann HM, Wyatt MM, Rangel Rivera GO, Rivera-Reyes AM, Dwyer CJ, Ware MB, Cole AC, Neskey DM, Rubinstein MP, Liu B, Thaxton JE, Bartee E, Paulos CM
Journal for immunotherapy of cancer 2022 Jan;10(1)
Journal for immunotherapy of cancer 2022 Jan;10(1)
Lenvatinib for effectively treating antiangiogenic drug-resistant nasopharyngeal carcinoma.
Sun Q, Wang Y, Ji H, Sun X, Xie S, Chen L, Li S, Zeng W, Chen R, Tang Q, Zuo J, Hou L, Hosaka K, Lu Y, Liu Y, Ye Y, Yang Y
Cell death & disease 2022 Aug 19;13(8):724
Cell death & disease 2022 Aug 19;13(8):724
Transient expansion and myofibroblast conversion of adipogenic lineage precursors mediate bone marrow repair after radiation.
Zhong L, Yao L, Holdreith N, Yu W, Gui T, Miao Z, Elkaim Y, Li M, Gong Y, Pacifici M, Maity A, Busch TM, Joeng KS, Cengel K, Seale P, Tong W, Qin L
JCI insight 2022 Apr 8;7(7)
JCI insight 2022 Apr 8;7(7)
A rapid CRISPR competitive assay for in vitro and in vivo discovery of potential drug targets affecting the hematopoietic system.
Shen Y, Jiang L, Iyer VS, Raposo B, Dubnovitsky A, Boddul SV, Kasza Z, Wermeling F
Computational and structural biotechnology journal 2021;19:5360-5370
Computational and structural biotechnology journal 2021;19:5360-5370
TNFRSF13B genotypes control immune-mediated pathology by regulating the functions of innate B cells.
de Mattos Barbosa MG, Lefferts AR, Huynh D, Liu H, Zhang Y, Fu B, Barnes J, Samaniego M, Bram RJ, Geha RS, Shikanov A, Prak ETL, Farkash EA, Platt JL, Cascalho M
JCI insight 2021 Sep 8;6(17)
JCI insight 2021 Sep 8;6(17)
Polyvinyl alcohol hydrolysis rate and molecular weight influence human and murine HSC activity ex vivo.
Sudo K, Yamazaki S, Wilkinson AC, Nakauchi H, Nakamura Y
Stem cell research 2021 Oct;56:102531
Stem cell research 2021 Oct;56:102531
Type 2 diabetic mice enter a state of spontaneous hibernation-like suspended animation following accumulation of uric acid.
Zhao Y, Cheng R, Zhao Y, Ge W, Yang Y, Ding Z, Xu X, Wang Z, Wu Z, Zhang J
The Journal of biological chemistry 2021 Oct;297(4):101166
The Journal of biological chemistry 2021 Oct;297(4):101166
Metabolic preconditioning in CD4+ T cells restores inducible immune tolerance in lupus-prone mice.
Wilson CS, Stocks BT, Hoopes EM, Rhoads JP, McNew KL, Major AS, Moore DJ
JCI insight 2021 Oct 8;6(19)
JCI insight 2021 Oct 8;6(19)
Targeting Ovarian Carcinoma with TSP-1:CD47 Antagonist TAX2 Activates Anti-Tumor Immunity.
Jeanne A, Sarazin T, Charlé M, Moali C, Fichel C, Boulagnon-Rombi C, Callewaert M, Andry MC, Diesis E, Delolme F, Rioult D, Dedieu S
Cancers 2021 Oct 7;13(19)
Cancers 2021 Oct 7;13(19)
Impaired HA-specific T follicular helper cell and antibody responses to influenza vaccination are linked to inflammation in humans.
Hill DL, Whyte CE, Innocentin S, Lee JL, Dooley J, Wang J, James EA, Lee JC, Kwok WW, Zand MS, Liston A, Carr EJ, Linterman MA
eLife 2021 Nov 2;10
eLife 2021 Nov 2;10
The folate cycle enzyme MTHFD2 induces cancer immune evasion through PD-L1 up-regulation.
Shang M, Yang H, Yang R, Chen T, Fu Y, Li Y, Fang X, Zhang K, Zhang J, Li H, Cao X, Gu J, Xiao J, Zhang Q, Liu X, Yu Q, Wang T
Nature communications 2021 Mar 29;12(1):1940
Nature communications 2021 Mar 29;12(1):1940
Cell-permeable transgelin-2 as a potent therapeutic for dendritic cell-based cancer immunotherapy.
Kim HR, Park JS, Park JH, Yasmin F, Kim CH, Oh SK, Chung IJ, Jun CD
Journal of hematology & oncology 2021 Mar 17;14(1):43
Journal of hematology & oncology 2021 Mar 17;14(1):43
Stromal SOX2 Upregulation Promotes Tumorigenesis through the Generation of a SFRP1/2-Expressing Cancer-Associated Fibroblast Population.
Kasashima H, Duran A, Martinez-Ordoñez A, Nakanishi Y, Kinoshita H, Linares JF, Reina-Campos M, Kudo Y, L'Hermitte A, Yashiro M, Ohira M, Bao F, Tauriello DVF, Batlle E, Diaz-Meco MT, Moscat J
Developmental cell 2021 Jan 11;56(1):95-110.e10
Developmental cell 2021 Jan 11;56(1):95-110.e10
Myeloid Cell CK2 Regulates Inflammation and Resistance to Bacterial Infection.
Larson SR, Bortell N, Illies A, Crisler WJ, Matsuda JL, Lenz LL
Frontiers in immunology 2020;11:590266
Frontiers in immunology 2020;11:590266
Shp1 Loss Enhances Macrophage Effector Function and Promotes Anti-Tumor Immunity.
Myers DR, Abram CL, Wildes D, Belwafa A, Welsh AMN, Schulze CJ, Choy TJ, Nguyen T, Omaque N, Hu Y, Singh M, Hansen R, Goldsmith MA, Quintana E, Smith JAM, Lowell CA
Frontiers in immunology 2020;11:576310
Frontiers in immunology 2020;11:576310
TNFAIP3 Plays a Role in Aging of the Hematopoietic System.
Smith MA, Culver-Cochran AE, Adelman ER, Rhyasen GW, Ma A, Figueroa ME, Starczynowski DT
Frontiers in immunology 2020;11:536442
Frontiers in immunology 2020;11:536442
G protein-coupled receptor 68 increases the number of B lymphocytes.
He X, Feng S, Hawkins C, Lawley L, Fan W, Xu Y, Zha XM, Fang J
American journal of blood research 2020;10(2):15-21
American journal of blood research 2020;10(2):15-21
The lysophospholipase D enzyme Gdpd3 is required to maintain chronic myelogenous leukaemia stem cells.
Naka K, Ochiai R, Matsubara E, Kondo C, Yang KM, Hoshii T, Araki M, Araki K, Sotomaru Y, Sasaki K, Mitani K, Kim DW, Ooshima A, Kim SJ
Nature communications 2020 Sep 17;11(1):4681
Nature communications 2020 Sep 17;11(1):4681
Reprogrammed mRNA translation drives resistance to therapeutic targeting of ribosome biogenesis.
Kusnadi EP, Trigos AS, Cullinane C, Goode DL, Larsson O, Devlin JR, Chan KT, De Souza DP, McConville MJ, McArthur GA, Thomas G, Sanij E, Poortinga G, Hannan RD, Hannan KM, Kang J, Pearson RB
The EMBO journal 2020 Nov 2;39(21):e105111
The EMBO journal 2020 Nov 2;39(21):e105111
Adaptive response to inflammation contributes to sustained myelopoiesis and confers a competitive advantage in myelodysplastic syndrome HSCs.
Muto T, Walker CS, Choi K, Hueneman K, Smith MA, Gul Z, Garcia-Manero G, Ma A, Zheng Y, Starczynowski DT
Nature immunology 2020 May;21(5):535-545
Nature immunology 2020 May;21(5):535-545
Retroviral Overexpression of CXCR4 on Murine B-1a Cells and Adoptive Transfer for Targeted B-1a Cell Migration to the Bone Marrow and IgM Production.
Upadhye A, Marshall M, Garmey JC, Bender TP, McNamara C
Journal of visualized experiments : JoVE 2020 May 31;(159)
Journal of visualized experiments : JoVE 2020 May 31;(159)
Crucial Roles of the RIP Homotypic Interaction Motifs of RIPK3 in RIPK1-Dependent Cell Death and Lymphoproliferative Disease.
Zhang H, Wu X, Li X, Li M, Li F, Wang L, Zhang X, Zhang Y, Luo Y, Wang H, Jiang Y, Zhang H
Cell reports 2020 May 19;31(7):107650
Cell reports 2020 May 19;31(7):107650
High-fat diet induced central adiposity (visceral fat) is associated with increased fibrosis and decreased immune cellularity of the mesenteric lymph node in mice.
Magnuson AM, Regan DP, Booth AD, Fouts JK, Solt CM, Hill JL, Dow SW, Foster MT
European journal of nutrition 2020 Jun;59(4):1641-1654
European journal of nutrition 2020 Jun;59(4):1641-1654
Generation and characterization of the Eµ-Irf8 mouse model.
Qiu Z, Holder KN, Lin AP, Myers J, Jiang S, Gorena KM, Kinney MC, Aguiar RCT
Cancer genetics 2020 Jul;245:6-16
Cancer genetics 2020 Jul;245:6-16
Modest Declines in Proteome Quality Impair Hematopoietic Stem Cell Self-Renewal.
Hidalgo San Jose L, Sunshine MJ, Dillingham CH, Chua BA, Kruta M, Hong Y, Hatters DM, Signer RAJ
Cell reports 2020 Jan 7;30(1):69-80.e6
Cell reports 2020 Jan 7;30(1):69-80.e6
MOSPD2 is a therapeutic target for the treatment of CNS inflammation.
Yacov N, Kafri P, Salem Y, Propheta-Meiran O, Feldman B, Breitbart E, Mendel I
Clinical and experimental immunology 2020 Aug;201(2):105-120
Clinical and experimental immunology 2020 Aug;201(2):105-120
Id1 and Id3 Maintain Steady-State Hematopoiesis by Promoting Sinusoidal Endothelial Cell Survival and Regeneration.
Gadomski S, Singh SK, Singh S, Sarkar T, Klarmann KD, Berenschot M, Seaman S, Jakubison B, Gudmundsson KO, Lockett S, Keller JR
Cell reports 2020 Apr 28;31(4):107572
Cell reports 2020 Apr 28;31(4):107572
Diverse CD8 T Cell Responses to Viral Infection Revealed by the Collaborative Cross.
Martin MD, Sompallae R, Winborn CS, Harty JT, Badovinac VP
Cell reports 2020 Apr 14;31(2):107508
Cell reports 2020 Apr 14;31(2):107508
Glucocorticoids Promote the Onset of Acute Experimental Colitis and Cancer by Upregulating mTOR Signaling in Intestinal Epithelial Cells.
Zhang Z, Dong L, Jia A, Chen X, Yang Q, Wang Y, Wang Y, Liu R, Cao Y, He Y, Bi Y, Liu G
Cancers 2020 Apr 11;12(4)
Cancers 2020 Apr 11;12(4)
Tpl2 Protects Against Fulminant Hepatitis Through Mobilization of Myeloid-Derived Suppressor Cells.
Xu J, Pei S, Wang Y, Liu J, Qian Y, Huang M, Zhang Y, Xiao Y
Frontiers in immunology 2019;10:1980
Frontiers in immunology 2019;10:1980
Bacteroides fragilis polysaccharide A induces IL-10 secreting B and T cells that prevent viral encephalitis.
Ramakrishna C, Kujawski M, Chu H, Li L, Mazmanian SK, Cantin EM
Nature communications 2019 May 14;10(1):2153
Nature communications 2019 May 14;10(1):2153
Nidogen-1 Contributes to the Interaction Network Involved in Pro-B Cell Retention in the Peri-sinusoidal Hematopoietic Stem Cell Niche.
Balzano M, De Grandis M, Vu Manh TP, Chasson L, Bardin F, Farina A, Sergé A, Bidaut G, Charbord P, Hérault L, Bailly AL, Cartier-Michaud A, Boned A, Dalod M, Duprez E, Genever P, Coles M, Bajenoff M, Xerri L, Aurrand-Lions M, Schiff C, Mancini SJC
Cell reports 2019 Mar 19;26(12):3257-3271.e8
Cell reports 2019 Mar 19;26(12):3257-3271.e8
Cohesin-mediated NF-κB signaling limits hematopoietic stem cell self-renewal in aging and inflammation.
Chen Z, Amro EM, Becker F, Hölzer M, Rasa SMM, Njeru SN, Han B, Di Sanzo S, Chen Y, Tang D, Tao S, Haenold R, Groth M, Romanov VS, Kirkpatrick JM, Kraus JM, Kestler HA, Marz M, Ori A, Neri F, Morita Y, Rudolph KL
The Journal of experimental medicine 2019 Jan 7;216(1):152-175
The Journal of experimental medicine 2019 Jan 7;216(1):152-175
Targeted selection of HIV-specific antibody mutations by engineering B cell maturation.
Saunders KO, Wiehe K, Tian M, Acharya P, Bradley T, Alam SM, Go EP, Scearce R, Sutherland L, Henderson R, Hsu AL, Borgnia MJ, Chen H, Lu X, Wu NR, Watts B, Jiang C, Easterhoff D, Cheng HL, McGovern K, Waddicor P, Chapdelaine-Williams A, Eaton A, Zhang J, Rountree W, Verkoczy L, Tomai M, Lewis MG, Desaire HR, Edwards RJ, Cain DW, Bonsignori M, Montefiori D, Alt FW, Haynes BF
Science (New York, N.Y.) 2019 Dec 6;366(6470)
Science (New York, N.Y.) 2019 Dec 6;366(6470)
Modulation of autoimmune pathogenesis by T cell-triggered inflammatory cell death.
Sasaki K, Himeno A, Nakagawa T, Sasaki Y, Kiyonari H, Iwai K
Nature communications 2019 Aug 28;10(1):3878
Nature communications 2019 Aug 28;10(1):3878
Ptpn21 Controls Hematopoietic Stem Cell Homeostasis and Biomechanics.
Ni F, Yu WM, Wang X, Fay ME, Young KM, Qiu Y, Lam WA, Sulchek TA, Cheng T, Scadden DT, Qu CK
Cell stem cell 2019 Apr 4;24(4):608-620.e6
Cell stem cell 2019 Apr 4;24(4):608-620.e6
The Hematopoietic Oxidase NOX2 Regulates Self-Renewal of Leukemic Stem Cells.
Adane B, Ye H, Khan N, Pei S, Minhajuddin M, Stevens BM, Jones CL, D'Alessandro A, Reisz JA, Zaberezhnyy V, Gasparetto M, Ho TC, Kelly KK, Myers JR, Ashton JM, Siegenthaler J, Kume T, Campbell EL, Pollyea DA, Becker MW, Jordan CT
Cell reports 2019 Apr 2;27(1):238-254.e6
Cell reports 2019 Apr 2;27(1):238-254.e6
Proapoptotic BIM Impacts B Lymphoid Homeostasis by Limiting the Survival of Mature B Cells in a Cell-Autonomous Manner.
Liu R, King A, Bouillet P, Tarlinton DM, Strasser A, Heierhorst J
Frontiers in immunology 2018;9:592
Frontiers in immunology 2018;9:592
Dysregulated TRAF3 and BCL2 Expression Promotes Multiple Classes of Mature Non-hodgkin B Cell Lymphoma in Mice.
Perez-Chacon G, Adrados M, Vallejo-Cremades MT, Lefebvre S, Reed JC, Zapata JM
Frontiers in immunology 2018;9:3114
Frontiers in immunology 2018;9:3114
ATF3 Sustains IL-22-Induced STAT3 Phosphorylation to Maintain Mucosal Immunity Through Inhibiting Phosphatases.
Glal D, Sudhakar JN, Lu HH, Liu MC, Chiang HY, Liu YC, Cheng CF, Shui JW
Frontiers in immunology 2018;9:2522
Frontiers in immunology 2018;9:2522
Orally administered heat-killed Lactobacillus paracasei MCC1849 enhances antigen-specific IgA secretion and induces follicular helper T cells in mice.
Arai S, Iwabuchi N, Takahashi S, Xiao JZ, Abe F, Hachimura S
PloS one 2018;13(6):e0199018
PloS one 2018;13(6):e0199018
EDAG promotes the expansion and survival of human CD34+ cells.
Zhao K, Zheng WW, Dong XM, Yin RH, Gao R, Li X, Liu JF, Zhan YQ, Yu M, Chen H, Ge CH, Ning HM, Yang XM, Li CY
PloS one 2018;13(1):e0190794
PloS one 2018;13(1):e0190794
Cbl Ubiquitin Ligases Control B Cell Exit from the Germinal-Center Reaction.
Li X, Gadzinsky A, Gong L, Tong H, Calderon V, Li Y, Kitamura D, Klein U, Langdon WY, Hou F, Zou YR, Gu H
Immunity 2018 Mar 20;48(3):530-541.e6
Immunity 2018 Mar 20;48(3):530-541.e6
Host Tumor Suppressor p18(INK4c) Functions as a Potent Cell-Intrinsic Inhibitor of Murine Gammaherpesvirus 68 Reactivation and Pathogenesis.
Niemeyer BF, Oko LM, Medina EM, Oldenburg DG, White DW, Cool CD, Clambey ET, van Dyk LF
Journal of virology 2018 Mar 15;92(6)
Journal of virology 2018 Mar 15;92(6)
Adrenergic nerve degeneration in bone marrow drives aging of the hematopoietic stem cell niche.
Maryanovich M, Zahalka AH, Pierce H, Pinho S, Nakahara F, Asada N, Wei Q, Wang X, Ciero P, Xu J, Leftin A, Frenette PS
Nature medicine 2018 Jun;24(6):782-791
Nature medicine 2018 Jun;24(6):782-791
Bone marrow‑derived mesenchymal stem cell‑conditioned medium attenuates tubulointerstitial fibrosis by inhibiting monocyte mobilization in an irreversible model of unilateral ureteral obstruction.
Zheng J, Wang Q, Leng W, Sun X, Peng J
Molecular medicine reports 2018 Jun;17(6):7701-7707
Molecular medicine reports 2018 Jun;17(6):7701-7707
Mitophagy in Intestinal Epithelial Cells Triggers Adaptive Immunity during Tumorigenesis.
Ziegler PK, Bollrath J, Pallangyo CK, Matsutani T, Canli Ö, De Oliveira T, Diamanti MA, Müller N, Gamrekelashvili J, Putoczki T, Horst D, Mankan AK, Öner MG, Müller S, Müller-Höcker J, Kirchner T, Slotta-Huspenina J, Taketo MM, Reinheckel T, Dröse S, Larner AC, Wels WS, Ernst M, Greten TF, Arkan MC, Korn T, Wirth D, Greten FR
Cell 2018 Jun 28;174(1):88-101.e16
Cell 2018 Jun 28;174(1):88-101.e16
miR-143/145 differentially regulate hematopoietic stem and progenitor activity through suppression of canonical TGFβ signaling.
Lam J, van den Bosch M, Wegrzyn J, Parker J, Ibrahim R, Slowski K, Chang L, Martinez-Høyer S, Condorelli G, Boldin M, Deng Y, Umlandt P, Fuller M, Karsan A
Nature communications 2018 Jun 20;9(1):2418
Nature communications 2018 Jun 20;9(1):2418
Loss of pleckstrin-2 reverts lethality and vascular occlusions in JAK2V617F-positive myeloproliferative neoplasms.
Zhao B, Mei Y, Cao L, Zhang J, Sumagin R, Yang J, Gao J, Schipma MJ, Wang Y, Thorsheim C, Zhao L, Stalker T, Stein B, Wen QJ, Crispino JD, Abrams CS, Ji P
The Journal of clinical investigation 2018 Jan 2;128(1):125-140
The Journal of clinical investigation 2018 Jan 2;128(1):125-140
Tanshinone IIA and Astragaloside IV promote the angiogenesis of mesenchymal stem cell-derived endothelial cell-like cells via upregulation of Cx37, Cx40 and Cx43.
Li Z, Zhang S, Cao L, Li W, Ye YC, Shi ZX, Wang ZR, Sun LX, Wang JW, Jia LT, Wang W
Experimental and therapeutic medicine 2018 Feb;15(2):1847-1854
Experimental and therapeutic medicine 2018 Feb;15(2):1847-1854
Inactivation of mTORC1 Signaling in Osterix-Expressing Cells Impairs B-cell Differentiation.
Wang Y, Xiao M, Tao C, Chen J, Wang Z, Yang J, Chen Z, Zou Z, Liu A, Cai D, Jiang Y, Ding C, Li M, Bai X
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research 2018 Apr;33(4):732-742
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research 2018 Apr;33(4):732-742
IL-2/Anti-IL-2 Complex Attenuates Inflammation and BBB Disruption in Mice Subjected to Traumatic Brain Injury.
Gao W, Li F, Zhou Z, Xu X, Wu Y, Zhou S, Yin D, Sun D, Xiong J, Jiang R, Zhang J
Frontiers in neurology 2017;8:281
Frontiers in neurology 2017;8:281
The Interleukin (IL)-1R1 pathway is a critical negative regulator of PyMT-mediated mammary tumorigenesis and pulmonary metastasis.
Dagenais M, Dupaul-Chicoine J, Douglas T, Champagne C, Morizot A, Saleh M
Oncoimmunology 2017;6(3):e1287247
Oncoimmunology 2017;6(3):e1287247
Multiple functional therapeutic effects of TnP: A small stable synthetic peptide derived from fish venom in a mouse model of multiple sclerosis.
Komegae EN, Souza TA, Grund LZ, Lima C, Lopes-Ferreira M
PloS one 2017;12(2):e0171796
PloS one 2017;12(2):e0171796
ORMDL3 Facilitates the Survival of Splenic B Cells via an ATF6α-Endoplasmic Reticulum Stress-Beclin1 Autophagy Regulatory Pathway.
Dang J, Bian X, Ma X, Li J, Long F, Shan S, Yuan Q, Xin Q, Li Y, Gao F, Gong Y, Liu Q
Journal of immunology (Baltimore, Md. : 1950) 2017 Sep 1;199(5):1647-1659
Journal of immunology (Baltimore, Md. : 1950) 2017 Sep 1;199(5):1647-1659
Diet-induced obesity causes visceral, but not subcutaneous, lymph node hyperplasia via increases in specific immune cell populations.
Magnuson AM, Regan DP, Fouts JK, Booth AD, Dow SW, Foster MT
Cell proliferation 2017 Oct;50(5)
Cell proliferation 2017 Oct;50(5)
Inflammasome-driven catecholamine catabolism in macrophages blunts lipolysis during ageing.
Camell CD, Sander J, Spadaro O, Lee A, Nguyen KY, Wing A, Goldberg EL, Youm YH, Brown CW, Elsworth J, Rodeheffer MS, Schultze JL, Dixit VD
Nature 2017 Oct 5;550(7674):119-123
Nature 2017 Oct 5;550(7674):119-123
DNA-binding of the Tet-transactivator curtails antigen-induced lymphocyte activation in mice.
Ottina E, Peperzak V, Schoeler K, Carrington E, Sgonc R, Pellegrini M, Preston S, Herold MJ, Strasser A, Villunger A
Nature communications 2017 Oct 18;8(1):1028
Nature communications 2017 Oct 18;8(1):1028
EZH2 enables germinal centre formation through epigenetic silencing of CDKN1A and an Rb-E2F1 feedback loop.
Béguelin W, Rivas MA, Calvo Fernández MT, Teater M, Purwada A, Redmond D, Shen H, Challman MF, Elemento O, Singh A, Melnick AM
Nature communications 2017 Oct 12;8(1):877
Nature communications 2017 Oct 12;8(1):877
ADAR1 deletion induces NFκB and interferon signaling dependent liver inflammation and fibrosis.
Ben-Shoshan SO, Kagan P, Sultan M, Barabash Z, Dor C, Jacob-Hirsch J, Harmelin A, Pappo O, Marcu-Malina V, Ben-Ari Z, Amariglio N, Rechavi G, Goldstein I, Safran M
RNA biology 2017 May 4;14(5):587-602
RNA biology 2017 May 4;14(5):587-602
The creatine kinase pathway is a metabolic vulnerability in EVI1-positive acute myeloid leukemia.
Fenouille N, Bassil CF, Ben-Sahra I, Benajiba L, Alexe G, Ramos A, Pikman Y, Conway AS, Burgess MR, Li Q, Luciano F, Auberger P, Galinsky I, DeAngelo DJ, Stone RM, Zhang Y, Perkins AS, Shannon K, Hemann MT, Puissant A, Stegmaier K
Nature medicine 2017 Mar;23(3):301-313
Nature medicine 2017 Mar;23(3):301-313
Differential cytokine contributions of perivascular haematopoietic stem cell niches.
Asada N, Kunisaki Y, Pierce H, Wang Z, Fernandez NF, Birbrair A, Ma'ayan A, Frenette PS
Nature cell biology 2017 Mar;19(3):214-223
Nature cell biology 2017 Mar;19(3):214-223
Peptidylarginine deiminase 2 is required for tumor necrosis factor alpha-induced citrullination and arthritis, but not neutrophil extracellular trap formation.
Bawadekar M, Shim D, Johnson CJ, Warner TF, Rebernick R, Damgaard D, Nielsen CH, Pruijn GJM, Nett JE, Shelef MA
Journal of autoimmunity 2017 Jun;80:39-47
Journal of autoimmunity 2017 Jun;80:39-47
Loss of IP(3) Receptor-Mediated Ca(2+) Release in Mouse B Cells Results in Abnormal B Cell Development and Function.
Tang H, Wang H, Lin Q, Fan F, Zhang F, Peng X, Fang X, Liu J, Ouyang K
Journal of immunology (Baltimore, Md. : 1950) 2017 Jul 15;199(2):570-580
Journal of immunology (Baltimore, Md. : 1950) 2017 Jul 15;199(2):570-580
Ubiquitination of hnRNPA1 by TRAF6 links chronic innate immune signaling with myelodysplasia.
Fang J, Bolanos LC, Choi K, Liu X, Christie S, Akunuru S, Kumar R, Wang D, Chen X, Greis KD, Stoilov P, Filippi MD, Maciejewski JP, Garcia-Manero G, Weirauch MT, Salomonis N, Geiger H, Zheng Y, Starczynowski DT
Nature immunology 2017 Feb;18(2):236-245
Nature immunology 2017 Feb;18(2):236-245
Recipient bone marrow assimilates the myeloid/lymphoid reconstitution of distinct fetal hematopoietic stem cells.
Guo XL, Chu L, Ke F, Mu LL, Li Z, Cai JJ, Li HF, Hong DL
Oncotarget 2017 Dec 12;8(65):108981-108988
Oncotarget 2017 Dec 12;8(65):108981-108988
Antibody Tumor Targeting Is Enhanced by CD27 Agonists through Myeloid Recruitment.
Turaj AH, Hussain K, Cox KL, Rose-Zerilli MJJ, Testa J, Dahal LN, Chan HTC, James S, Field VL, Carter MJ, Kim HJ, West JJ, Thomas LJ, He LZ, Keler T, Johnson PWM, Al-Shamkhani A, Thirdborough SM, Beers SA, Cragg MS, Glennie MJ, Lim SH
Cancer cell 2017 Dec 11;32(6):777-791.e6
Cancer cell 2017 Dec 11;32(6):777-791.e6
Irgm1 coordinately regulates autoimmunity and host defense at select mucosal surfaces.
Azzam KM, Madenspacher JH, Cain DW, Lai L, Gowdy KM, Rai P, Janardhan K, Clayton N, Cunningham W, Jensen H, Patel PS, Kearney JF, Taylor GA, Fessler MB
JCI insight 2017 Aug 17;2(16)
JCI insight 2017 Aug 17;2(16)
Epithelial Fli1 deficiency drives systemic autoimmunity and fibrosis: Possible roles in scleroderma.
Takahashi T, Asano Y, Sugawara K, Yamashita T, Nakamura K, Saigusa R, Ichimura Y, Toyama T, Taniguchi T, Akamata K, Noda S, Yoshizaki A, Tsuruta D, Trojanowska M, Sato S
The Journal of experimental medicine 2017 Apr 3;214(4):1129-1151
The Journal of experimental medicine 2017 Apr 3;214(4):1129-1151
CD4 T Helper Cells Instruct Lymphopenia-Induced Memory-Like CD8 T Cells for Control of Acute LCMV Infection.
Schmitt ME, Sitte S, Voehringer D
Frontiers in immunology 2016;7:622
Frontiers in immunology 2016;7:622
Alkylator-Induced and Patient-Derived Xenograft Mouse Models of Therapy-Related Myeloid Neoplasms Model Clinical Disease and Suggest the Presence of Multiple Cell Subpopulations with Leukemia Stem Cell Activity.
Jonas BA, Johnson C, Gratzinger D, Majeti R
PloS one 2016;11(7):e0159189
PloS one 2016;11(7):e0159189
Heme Oxygenase-1-Expressing Dendritic Cells Promote Foxp3+ Regulatory T Cell Differentiation and Induce Less Severe Airway Inflammation in Murine Models.
Wong TH, Chen HA, Gau RJ, Yen JH, Suen JL
PloS one 2016;11(12):e0168919
PloS one 2016;11(12):e0168919
Monocyte Adhesion and Plaque Recruitment During Atherosclerosis Development Is Regulated by the Adapter Protein Chat-H/SHEP1.
Herbin O, Regelmann AG, Ramkhelawon B, Weinstein EG, Moore KJ, Alexandropoulos K
Arteriosclerosis, thrombosis, and vascular biology 2016 Sep;36(9):1791-801
Arteriosclerosis, thrombosis, and vascular biology 2016 Sep;36(9):1791-801
Transcriptional regulation of the proto-oncogene Zfp521 by SPI1 (PU.1) and HOXC13.
Yu M, Al-Dallal S, Al-Haj L, Panjwani S, McCartney AS, Edwards SM, Manjunath P, Walker C, Awgulewitsch A, Hentges KE
Genesis (New York, N.Y. : 2000) 2016 Oct;54(10):519-533
Genesis (New York, N.Y. : 2000) 2016 Oct;54(10):519-533
Reprogramming mouse fibroblasts into engraftable myeloerythroid and lymphoid progenitors.
Cheng H, Ang HY, A El Farran C, Li P, Fang HT, Liu TM, Kong SL, Chin ML, Ling WY, Lim EK, Li H, Huber T, Loh KM, Loh YH, Lim B
Nature communications 2016 Nov 21;7:13396
Nature communications 2016 Nov 21;7:13396
Epigenetic Memory Underlies Cell-Autonomous Heterogeneous Behavior of Hematopoietic Stem Cells.
Yu VWC, Yusuf RZ, Oki T, Wu J, Saez B, Wang X, Cook C, Baryawno N, Ziller MJ, Lee E, Gu H, Meissner A, Lin CP, Kharchenko PV, Scadden DT
Cell 2016 Nov 17;167(5):1310-1322.e17
Cell 2016 Nov 17;167(5):1310-1322.e17
Dendritic cell sphingosine-1-phosphate lyase regulates thymic egress.
Zamora-Pineda J, Kumar A, Suh JH, Zhang M, Saba JD
The Journal of experimental medicine 2016 Nov 14;213(12):2773-2791
The Journal of experimental medicine 2016 Nov 14;213(12):2773-2791
The 4E-BP-eIF4E axis promotes rapamycin-sensitive growth and proliferation in lymphocytes.
So L, Lee J, Palafox M, Mallya S, Woxland CG, Arguello M, Truitt ML, Sonenberg N, Ruggero D, Fruman DA
Science signaling 2016 May 31;9(430):ra57
Science signaling 2016 May 31;9(430):ra57
CXCL16-positive dendritic cells enhance invariant natural killer T cell-dependent IFNγ production and tumor control.
Veinotte L, Gebremeskel S, Johnston B
Oncoimmunology 2016 Jun;5(6):e1160979
Oncoimmunology 2016 Jun;5(6):e1160979
Adipose tissue adaptive response to trans-10,cis-12-conjugated linoleic acid engages alternatively activated M2 macrophages.
Pini M, Touch S, Poirier H, Dalmas E, Niot I, Rouault C, Druart C, Delzenne N, Clément K, André S, Guerre-Millo M
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2016 Jan;30(1):241-51
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2016 Jan;30(1):241-51
Caspase-12, but Not Caspase-11, Inhibits Obesity and Insulin Resistance.
Skeldon AM, Morizot A, Douglas T, Santoro N, Kursawe R, Kozlitina J, Caprio S, Mehal WZ, Saleh M
Journal of immunology (Baltimore, Md. : 1950) 2016 Jan 1;196(1):437-47
Journal of immunology (Baltimore, Md. : 1950) 2016 Jan 1;196(1):437-47
Murine iPSC-Derived Macrophages as a Tool for Disease Modeling of Hereditary Pulmonary Alveolar Proteinosis due to Csf2rb Deficiency.
Mucci A, Kunkiel J, Suzuki T, Brennig S, Glage S, Kühnel MP, Ackermann M, Happle C, Kuhn A, Schambach A, Trapnell BC, Hansen G, Moritz T, Lachmann N
Stem cell reports 2016 Aug 9;7(2):292-305
Stem cell reports 2016 Aug 9;7(2):292-305
Resident T Cells Are Unable To Control Herpes Simplex Virus-1 Activity in the Brain Ependymal Region during Latency.
Menendez CM, Jinkins JK, Carr DJ
Journal of immunology (Baltimore, Md. : 1950) 2016 Aug 15;197(4):1262-75
Journal of immunology (Baltimore, Md. : 1950) 2016 Aug 15;197(4):1262-75
miR-22 has a potent anti-tumour role with therapeutic potential in acute myeloid leukaemia.
Jiang X, Hu C, Arnovitz S, Bugno J, Yu M, Zuo Z, Chen P, Huang H, Ulrich B, Gurbuxani S, Weng H, Strong J, Wang Y, Li Y, Salat J, Li S, Elkahloun AG, Yang Y, Neilly MB, Larson RA, Le Beau MM, Herold T, Bohlander SK, Liu PP, Zhang J, Li Z, He C, Jin J, Hong S, Chen J
Nature communications 2016 Apr 26;7:11452
Nature communications 2016 Apr 26;7:11452
Type I IFN Does Not Promote Susceptibility to Foodborne Listeria monocytogenes.
Pitts MG, Myers-Morales T, D'Orazio SE
Journal of immunology (Baltimore, Md. : 1950) 2016 Apr 1;196(7):3109-16
Journal of immunology (Baltimore, Md. : 1950) 2016 Apr 1;196(7):3109-16
Standard sub-thermoneutral caging temperature influences radiosensitivity of hematopoietic stem and progenitor cells.
Povinelli BJ, Kokolus KM, Eng JW, Dougher CW, Curtin L, Capitano ML, Sailsbury-Ruf CT, Repasky EA, Nemeth MJ
PloS one 2015;10(3):e0120078
PloS one 2015;10(3):e0120078
Tamm-Horsfall Protein Regulates Granulopoiesis and Systemic Neutrophil Homeostasis.
Micanovic R, Chitteti BR, Dagher PC, Srour EF, Khan S, Hato T, Lyle A, Tong Y, Wu XR, El-Achkar TM
Journal of the American Society of Nephrology : JASN 2015 Sep;26(9):2172-82
Journal of the American Society of Nephrology : JASN 2015 Sep;26(9):2172-82
Mitochondrial ATP transporter Ant2 depletion impairs erythropoiesis and B lymphopoiesis.
Cho J, Seo J, Lim CH, Yang L, Shiratsuchi T, Lee MH, Chowdhury RR, Kasahara H, Kim JS, Oh SP, Lee YJ, Terada N
Cell death and differentiation 2015 Sep;22(9):1437-50
Cell death and differentiation 2015 Sep;22(9):1437-50
The Inflammatory Caspases-1 and -11 Mediate the Pathogenesis of Dermatitis in Sharpin-Deficient Mice.
Douglas T, Champagne C, Morizot A, Lapointe JM, Saleh M
Journal of immunology (Baltimore, Md. : 1950) 2015 Sep 1;195(5):2365-73
Journal of immunology (Baltimore, Md. : 1950) 2015 Sep 1;195(5):2365-73
Bcl6 middle domain repressor function is required for T follicular helper cell differentiation and utilizes the corepressor MTA3.
Nance JP, Bélanger S, Johnston RJ, Hu JK, Takemori T, Crotty S
Proceedings of the National Academy of Sciences of the United States of America 2015 Oct 27;112(43):13324-9
Proceedings of the National Academy of Sciences of the United States of America 2015 Oct 27;112(43):13324-9
Endothelial Gata5 transcription factor regulates blood pressure.
Messaoudi S, He Y, Gutsol A, Wight A, Hébert RL, Vilmundarson RO, Makrigiannis AP, Chalmers J, Hamet P, Tremblay J, McPherson R, Stewart AFR, Touyz RM, Nemer M
Nature communications 2015 Nov 30;6:8835
Nature communications 2015 Nov 30;6:8835
Core fucosylation of IgG B cell receptor is required for antigen recognition and antibody production.
Li W, Yu R, Ma B, Yang Y, Jiao X, Liu Y, Cao H, Dong W, Liu L, Ma K, Fukuda T, Liu Q, Ma T, Wang Z, Gu J, Zhang J, Taniguchi N
Journal of immunology (Baltimore, Md. : 1950) 2015 Mar 15;194(6):2596-606
Journal of immunology (Baltimore, Md. : 1950) 2015 Mar 15;194(6):2596-606
Disruption of p21-activated kinase 1 gene diminishes atherosclerosis in apolipoprotein E-deficient mice.
Singh NK, Kotla S, Dyukova E, Traylor JG Jr, Orr AW, Chernoff J, Marion TN, Rao GN
Nature communications 2015 Jun 24;6:7450
Nature communications 2015 Jun 24;6:7450
BAFF activation of the ERK5 MAP kinase pathway regulates B cell survival.
Jacque E, Schweighoffer E, Tybulewicz VL, Ley SC
The Journal of experimental medicine 2015 Jun 1;212(6):883-92
The Journal of experimental medicine 2015 Jun 1;212(6):883-92
Apoptotic cells trigger a membrane-initiated pathway to increase ABCA1.
Fond AM, Lee CS, Schulman IG, Kiss RS, Ravichandran KS
The Journal of clinical investigation 2015 Jul 1;125(7):2748-58
The Journal of clinical investigation 2015 Jul 1;125(7):2748-58
Peroxisomal lipid synthesis regulates inflammation by sustaining neutrophil membrane phospholipid composition and viability.
Lodhi IJ, Wei X, Yin L, Feng C, Adak S, Abou-Ezzi G, Hsu FF, Link DC, Semenkovich CF
Cell metabolism 2015 Jan 6;21(1):51-64
Cell metabolism 2015 Jan 6;21(1):51-64
MHC-mismatched mixed chimerism mediates thymic deletion of cross-reactive autoreactive T cells and prevents insulitis in nonobese diabetic mice.
Racine JJ, Zhang M, Wang M, Morales W, Shen C, Zeng D
Journal of immunology (Baltimore, Md. : 1950) 2015 Jan 1;194(1):407-17
Journal of immunology (Baltimore, Md. : 1950) 2015 Jan 1;194(1):407-17
Natural IgM is produced by CD5- plasma cells that occupy a distinct survival niche in bone marrow.
Reynolds AE, Kuraoka M, Kelsoe G
Journal of immunology (Baltimore, Md. : 1950) 2015 Jan 1;194(1):231-42
Journal of immunology (Baltimore, Md. : 1950) 2015 Jan 1;194(1):231-42
TGF-β3-expressing CD4+CD25(-)LAG3+ regulatory T cells control humoral immune responses.
Okamura T, Sumitomo S, Morita K, Iwasaki Y, Inoue M, Nakachi S, Komai T, Shoda H, Miyazaki J, Fujio K, Yamamoto K
Nature communications 2015 Feb 19;6:6329
Nature communications 2015 Feb 19;6:6329
Aging-associated inflammation promotes selection for adaptive oncogenic events in B cell progenitors.
Henry CJ, Casás-Selves M, Kim J, Zaberezhnyy V, Aghili L, Daniel AE, Jimenez L, Azam T, McNamee EN, Clambey ET, Klawitter J, Serkova NJ, Tan AC, Dinarello CA, DeGregori J
The Journal of clinical investigation 2015 Dec;125(12):4666-80
The Journal of clinical investigation 2015 Dec;125(12):4666-80
B-cell-intrinsic hepatitis C virus expression leads to B-cell-lymphomagenesis and induction of NF-κB signalling.
Kasama Y, Mizukami T, Kusunoki H, Peveling-Oberhag J, Nishito Y, Ozawa M, Kohara M, Mizuochi T, Tsukiyama-Kohara K
PloS one 2014;9(3):e91373
PloS one 2014;9(3):e91373
Self DNA from lymphocytes that have undergone activation-induced cell death enhances murine B cell proliferation and antibody production.
Lu Q, Wang JY, Wang L, Jiang X, Chu Y
PloS one 2014;9(10):e109095
PloS one 2014;9(10):e109095
Diet-induced obesity promotes myelopoiesis in hematopoietic stem cells.
Singer K, DelProposto J, Morris DL, Zamarron B, Mergian T, Maley N, Cho KW, Geletka L, Subbaiah P, Muir L, Martinez-Santibanez G, Lumeng CN
Molecular metabolism 2014 Sep;3(6):664-75
Molecular metabolism 2014 Sep;3(6):664-75
α-1-Antitrypsin (AAT)-modified donor cells suppress GVHD but enhance the GVL effect: a role for mitochondrial bioenergetics.
Marcondes AM, Karoopongse E, Lesnikova M, Margineantu D, Welte T, Dinarello CA, Hockenbery D, Janciauskiene S, Deeg HJ
Blood 2014 Oct 30;124(18):2881-91
Blood 2014 Oct 30;124(18):2881-91
Transcription elongation factor ELL2 drives Ig secretory-specific mRNA production and the unfolded protein response.
Park KS, Bayles I, Szlachta-McGinn A, Paul J, Boiko J, Santos P, Liu J, Wang Z, Borghesi L, Milcarek C
Journal of immunology (Baltimore, Md. : 1950) 2014 Nov 1;193(9):4663-74
Journal of immunology (Baltimore, Md. : 1950) 2014 Nov 1;193(9):4663-74
CD4+ T cells promote the transition from hypertrophy to heart failure during chronic pressure overload.
Laroumanie F, Douin-Echinard V, Pozzo J, Lairez O, Tortosa F, Vinel C, Delage C, Calise D, Dutaur M, Parini A, Pizzinat N
Circulation 2014 May 27;129(21):2111-24
Circulation 2014 May 27;129(21):2111-24
TRAF6 is a critical regulator of LMP1 functions in vivo.
Arcipowski KM, Stunz LL, Bishop GA
International immunology 2014 Mar;26(3):149-58
International immunology 2014 Mar;26(3):149-58
A TNF-α-CCL20-CCR6 axis regulates Nod1-induced B cell responses.
Paradis M, Mindt BC, Duerr CU, Rojas OL, Ng D, Boulianne B, McCarthy DD, Yu MD, Summers deLuca LE, Ward LA, Waldron JB, Philpott DJ, Gommerman JL, Fritz JH
Journal of immunology (Baltimore, Md. : 1950) 2014 Mar 15;192(6):2787-99
Journal of immunology (Baltimore, Md. : 1950) 2014 Mar 15;192(6):2787-99
Peptidylarginine deiminase 4 contributes to tumor necrosis factor α-induced inflammatory arthritis.
Shelef MA, Sokolove J, Lahey LJ, Wagner CA, Sackmann EK, Warner TF, Wang Y, Beebe DJ, Robinson WH, Huttenlocher A
Arthritis & rheumatology (Hoboken, N.J.) 2014 Jun;66(6):1482-91
Arthritis & rheumatology (Hoboken, N.J.) 2014 Jun;66(6):1482-91
CD41 is a reliable identification and activation marker for murine basophils in the steady state and during helminth and malarial infections.
Bakocevic N, Claser C, Yoshikawa S, Jones LA, Chew S, Goh CC, Malleret B, Larbi A, Ginhoux F, de Lafaille MC, Karasuyama H, Renia L, Ng LG
European journal of immunology 2014 Jun;44(6):1823-34
European journal of immunology 2014 Jun;44(6):1823-34
Rhof promotes murine marginal zone B cell development.
Kishimoto M, Matsuda T, Yanase S, Katsumi A, Suzuki N, Ikejiri M, Takagi A, Ikawa M, Kojima T, Kunishima S, Kiyoi H, Naoe T, Matsushita T, Maruyama M
Nagoya journal of medical science 2014 Aug;76(3-4):293-305
Nagoya journal of medical science 2014 Aug;76(3-4):293-305
Eosinophils regulate peripheral B cell numbers in both mice and humans.
Wong TW, Doyle AD, Lee JJ, Jelinek DF
Journal of immunology (Baltimore, Md. : 1950) 2014 Apr 15;192(8):3548-58
Journal of immunology (Baltimore, Md. : 1950) 2014 Apr 15;192(8):3548-58
IκBε is a key regulator of B cell expansion by providing negative feedback on cRel and RelA in a stimulus-specific manner.
Alves BN, Tsui R, Almaden J, Shokhirev MN, Davis-Turak J, Fujimoto J, Birnbaum H, Ponomarenko J, Hoffmann A
Journal of immunology (Baltimore, Md. : 1950) 2014 Apr 1;192(7):3121-32
Journal of immunology (Baltimore, Md. : 1950) 2014 Apr 1;192(7):3121-32
Relapsing-remitting central nervous system autoimmunity mediated by GFAP-specific CD8 T cells.
Sasaki K, Bean A, Shah S, Schutten E, Huseby PG, Peters B, Shen ZT, Vanguri V, Liggitt D, Huseby ES
Journal of immunology (Baltimore, Md. : 1950) 2014 Apr 1;192(7):3029-42
Journal of immunology (Baltimore, Md. : 1950) 2014 Apr 1;192(7):3029-42
TDP2-dependent non-homologous end-joining protects against topoisomerase II-induced DNA breaks and genome instability in cells and in vivo.
Gómez-Herreros F, Romero-Granados R, Zeng Z, Alvarez-Quilón A, Quintero C, Ju L, Umans L, Vermeire L, Huylebroeck D, Caldecott KW, Cortés-Ledesma F
PLoS genetics 2013;9(3):e1003226
PLoS genetics 2013;9(3):e1003226
Genome-wide mouse mutagenesis reveals CD45-mediated T cell function as critical in protective immunity to HSV-1.
Caignard G, Leiva-Torres GA, Leney-Greene M, Charbonneau B, Dumaine A, Fodil-Cornu N, Pyzik M, Cingolani P, Schwartzentruber J, Dupaul-Chicoine J, Guo H, Saleh M, Veillette A, Lathrop M, Blanchette M, Majewski J, Pearson A, Vidal SM
PLoS pathogens 2013 Sep;9(9):e1003637
PLoS pathogens 2013 Sep;9(9):e1003637
CD30 is required for activation of a unique subset of interleukin-17A-producing γδ T cells in innate immunity against Mycobacterium bovis Bacillus Calmette-Guerin infection.
Guo Y, Sun X, Shibata K, Yamada H, Muta H, Podack ER, Yoshikai Y
Infection and immunity 2013 Oct;81(10):3923-34
Infection and immunity 2013 Oct;81(10):3923-34
VBP15, a novel anti-inflammatory and membrane-stabilizer, improves muscular dystrophy without side effects.
Heier CR, Damsker JM, Yu Q, Dillingham BC, Huynh T, Van der Meulen JH, Sali A, Miller BK, Phadke A, Scheffer L, Quinn J, Tatem K, Jordan S, Dadgar S, Rodriguez OC, Albanese C, Calhoun M, Gordish-Dressman H, Jaiswal JK, Connor EM, McCall JM, Hoffman EP, Reeves EK, Nagaraju K
EMBO molecular medicine 2013 Oct;5(10):1569-85
EMBO molecular medicine 2013 Oct;5(10):1569-85
Neuronal and nonneuronal cholinergic structures in the mouse gastrointestinal tract and spleen.
Gautron L, Rutkowski JM, Burton MD, Wei W, Wan Y, Elmquist JK
The Journal of comparative neurology 2013 Nov;521(16):3741-67
The Journal of comparative neurology 2013 Nov;521(16):3741-67
(18)F-FDG-PET/CT imaging in an IL-6- and MYC-driven mouse model of human multiple myeloma affords objective evaluation of plasma cell tumor progression and therapeutic response to the proteasome inhibitor ixazomib.
Duncan K, Rosean TR, Tompkins VS, Olivier A, Sompallae R, Zhan F, Tricot G, Acevedo MR, Ponto LL, Walsh SA, Tygrett LT, Berger AJ, Waldschmidt T, Morse HC 3rd, Sunderland JJ, Janz S
Blood cancer journal 2013 Nov 29;3(11):e165
Blood cancer journal 2013 Nov 29;3(11):e165
A type I IFN-Flt3 ligand axis augments plasmacytoid dendritic cell development from common lymphoid progenitors.
Chen YL, Chen TT, Pai LM, Wesoly J, Bluyssen HA, Lee CK
The Journal of experimental medicine 2013 Nov 18;210(12):2515-22
The Journal of experimental medicine 2013 Nov 18;210(12):2515-22
AID-expressing germinal center B cells cluster normally within lymph node follicles in the absence of FDC-M1+ CD35+ follicular dendritic cells but dissipate prematurely.
Boulianne B, Le MX, Ward LA, Meng L, Haddad D, Li C, Martin A, Gommerman JL
Journal of immunology (Baltimore, Md. : 1950) 2013 Nov 1;191(9):4521-30
Journal of immunology (Baltimore, Md. : 1950) 2013 Nov 1;191(9):4521-30
Long-term prevention of chronic allograft rejection by regulatory T-cell immunotherapy involves host Foxp3-expressing T cells.
Pasquet L, Douet JY, Sparwasser T, Romagnoli P, van Meerwijk JP
Blood 2013 May 23;121(21):4303-10
Blood 2013 May 23;121(21):4303-10
Antioxidant treatment regulates the humoral immune response during acute viral infection.
Crump KE, Langston PK, Rajkarnikar S, Grayson JM
Journal of virology 2013 Mar;87(5):2577-86
Journal of virology 2013 Mar;87(5):2577-86
Cellular-FLIP, Raji isoform (c-FLIP R) modulates cell death induction upon T-cell activation and infection.
Telieps T, Ewald F, Gereke M, Annemann M, Rauter Y, Schuster M, Ueffing N, von Smolinski D, Gruber AD, Bruder D, Schmitz I
European journal of immunology 2013 Jun;43(6):1499-510
European journal of immunology 2013 Jun;43(6):1499-510
Reciprocal effects of rab7 deletion in activated and neglected T cells.
Roy SG, Stevens MW, So L, Edinger AL
Autophagy 2013 Jul;9(7):1009-23
Autophagy 2013 Jul;9(7):1009-23
Thymic damage, impaired negative selection, and development of chronic graft-versus-host disease caused by donor CD4+ and CD8+ T cells.
Wu T, Young JS, Johnston H, Ni X, Deng R, Racine J, Wang M, Wang A, Todorov I, Wang J, Zeng D
Journal of immunology (Baltimore, Md. : 1950) 2013 Jul 1;191(1):488-99
Journal of immunology (Baltimore, Md. : 1950) 2013 Jul 1;191(1):488-99
Lipidoid Nanoparticles Containing PD-L1 siRNA Delivered In Vivo Enter Kupffer Cells and Enhance NK and CD8(+) T Cell-mediated Hepatic Antiviral Immunity.
Dolina JS, Sung SS, Novobrantseva TI, Nguyen TM, Hahn YS
Molecular therapy. Nucleic acids 2013 Feb 19;2(2):e72
Molecular therapy. Nucleic acids 2013 Feb 19;2(2):e72
MHC class II-dependent B cell APC function is required for induction of CNS autoimmunity independent of myelin-specific antibodies.
Molnarfi N, Schulze-Topphoff U, Weber MS, Patarroyo JC, Prod'homme T, Varrin-Doyer M, Shetty A, Linington C, Slavin AJ, Hidalgo J, Jenne DE, Wekerle H, Sobel RA, Bernard CC, Shlomchik MJ, Zamvil SS
The Journal of experimental medicine 2013 Dec 16;210(13):2921-37
The Journal of experimental medicine 2013 Dec 16;210(13):2921-37
Myeloid dendritic cells (DCs) of mice susceptible to paracoccidioidomycosis suppress T cell responses whereas myeloid and plasmacytoid DCs from resistant mice induce effector and regulatory T cells.
Pina A, de Araujo EF, Felonato M, Loures FV, Feriotti C, Bernardino S, Barbuto JA, Calich VL
Infection and immunity 2013 Apr;81(4):1064-77
Infection and immunity 2013 Apr;81(4):1064-77
Th17 effector cells support B cell responses outside of germinal centres.
Patakas A, Benson RA, Withers DR, Conigliaro P, McInnes IB, Brewer JM, Garside P
PloS one 2012;7(11):e49715
PloS one 2012;7(11):e49715
Induction of IL-10-producing CD1dhighCD5+ regulatory B cells following Babesia microti-infection.
Jeong YI, Hong SH, Cho SH, Lee WJ, Lee SE
PloS one 2012;7(10):e46553
PloS one 2012;7(10):e46553
Activation-induced cytidine deaminase-initiated off-target DNA breaks are detected and resolved during S phase.
Hasham MG, Snow KJ, Donghia NM, Branca JA, Lessard MD, Stavnezer J, Shopland LS, Mills KD
Journal of immunology (Baltimore, Md. : 1950) 2012 Sep 1;189(5):2374-82
Journal of immunology (Baltimore, Md. : 1950) 2012 Sep 1;189(5):2374-82
Biphenotypic B-lymphoid/myeloid cells expressing low levels of Pax5: potential targets of BAL development.
Simmons S, Knoll M, Drewell C, Wolf I, Mollenkopf HJ, Bouquet C, Melchers F
Blood 2012 Nov 1;120(18):3688-98
Blood 2012 Nov 1;120(18):3688-98
Up-regulation of a HOXA-PBX3 homeobox-gene signature following down-regulation of miR-181 is associated with adverse prognosis in patients with cytogenetically abnormal AML.
Li Z, Huang H, Li Y, Jiang X, Chen P, Arnovitz S, Radmacher MD, Maharry K, Elkahloun A, Yang X, He C, He M, Zhang Z, Dohner K, Neilly MB, Price C, Lussier YA, Zhang Y, Larson RA, Le Beau MM, Caligiuri MA, Bullinger L, Valk PJ, Delwel R, Lowenberg B, Liu PP, Marcucci G, Bloomfield CD, Rowley JD, Chen J
Blood 2012 Mar 8;119(10):2314-24
Blood 2012 Mar 8;119(10):2314-24
Impact of enzymatic tissue disintegration on the level of surface molecule expression and immune cell function.
Autengruber A, Gereke M, Hansen G, Hennig C, Bruder D
European journal of microbiology & immunology 2012 Jun;2(2):112-20
European journal of microbiology & immunology 2012 Jun;2(2):112-20
IKKα-mediated signaling circuitry regulates early B lymphopoiesis during hematopoiesis.
Balkhi MY, Willette-Brown J, Zhu F, Chen Z, Liu S, Guttridge DC, Karin M, Hu Y
Blood 2012 Jun 7;119(23):5467-77
Blood 2012 Jun 7;119(23):5467-77
Mechanisms of indirect acute lung injury: a novel role for the coinhibitory receptor, programmed death-1.
Monaghan SF, Thakkar RK, Heffernan DS, Huang X, Chung CS, Lomas-Neira J, Cioffi WG, Ayala A
Annals of surgery 2012 Jan;255(1):158-64
Annals of surgery 2012 Jan;255(1):158-64
Critical role of B cell lymphoma 10 in BAFF-regulated NF-κB activation and survival of anergic B cells.
Yu M, Chen Y, He Y, Podd A, Fu G, Wright JA, Kleiman E, Khan WN, Wen R, Wang D
Journal of immunology (Baltimore, Md. : 1950) 2012 Dec 1;189(11):5185-93
Journal of immunology (Baltimore, Md. : 1950) 2012 Dec 1;189(11):5185-93
TRAF binding is required for a distinct subset of in vivo B cell functions of the oncoprotein LMP1.
Arcipowski KM, Bishop GA
Journal of immunology (Baltimore, Md. : 1950) 2012 Dec 1;189(11):5165-70
Journal of immunology (Baltimore, Md. : 1950) 2012 Dec 1;189(11):5165-70
MyD88 signaling in B cells regulates the production of Th1-dependent antibodies to AAV.
Sudres M, Ciré S, Vasseur V, Brault L, Da Rocha S, Boisgérault F, Le Bec C, Gross DA, Blouin V, Ryffel B, Galy A
Molecular therapy : the journal of the American Society of Gene Therapy 2012 Aug;20(8):1571-81
Molecular therapy : the journal of the American Society of Gene Therapy 2012 Aug;20(8):1571-81
PAD4-mediated neutrophil extracellular trap formation is not required for immunity against influenza infection.
Hemmers S, Teijaro JR, Arandjelovic S, Mowen KA
PloS one 2011;6(7):e22043
PloS one 2011;6(7):e22043
Deletion of genes encoding PU.1 and Spi-B in B cells impairs differentiation and induces pre-B cell acute lymphoblastic leukemia.
Sokalski KM, Li SK, Welch I, Cadieux-Pitre HA, Gruca MR, DeKoter RP
Blood 2011 Sep 8;118(10):2801-8
Blood 2011 Sep 8;118(10):2801-8
Distinct developmental requirements for isolated lymphoid follicle formation in the small and large intestine: RANKL is essential only in the small intestine.
Knoop KA, Butler BR, Kumar N, Newberry RD, Williams IR
The American journal of pathology 2011 Oct;179(4):1861-71
The American journal of pathology 2011 Oct;179(4):1861-71
Memory T cells persisting in the brain following MCMV infection induce long-term microglial activation via interferon-γ.
Mutnal MB, Hu S, Little MR, Lokensgard JR
Journal of neurovirology 2011 Oct;17(5):424-37
Journal of neurovirology 2011 Oct;17(5):424-37
Analysis of the role of IL-21 in development of murine B cell progenitors in the bone marrow.
Simard N, Konforte D, Tran AH, Esufali J, Leonard WJ, Paige CJ
Journal of immunology (Baltimore, Md. : 1950) 2011 May 1;186(9):5244-53
Journal of immunology (Baltimore, Md. : 1950) 2011 May 1;186(9):5244-53
Role of central leptin signaling in the starvation-induced alteration of B-cell development.
Tanaka M, Suganami T, Kim-Saijo M, Toda C, Tsuiji M, Ochi K, Kamei Y, Minokoshi Y, Ogawa Y
The Journal of neuroscience : the official journal of the Society for Neuroscience 2011 Jun 8;31(23):8373-80
The Journal of neuroscience : the official journal of the Society for Neuroscience 2011 Jun 8;31(23):8373-80
Mn(III) meso-tetrakis-(N-ethylpyridinium-2-yl) porphyrin mitigates total body irradiation-induced long-term bone marrow suppression.
Li H, Wang Y, Pazhanisamy SK, Shao L, Batinic-Haberle I, Meng A, Zhou D
Free radical biology & medicine 2011 Jul 1;51(1):30-7
Free radical biology & medicine 2011 Jul 1;51(1):30-7
Sensitized phenotypic screening identifies gene dosage sensitive region on chromosome 11 that predisposes to disease in mice.
Ermakova O, Piszczek L, Luciani L, Cavalli FM, Ferreira T, Farley D, Rizzo S, Paolicelli RC, Al-Banchaabouchi M, Nerlov C, Moriggl R, Luscombe NM, Gross C
EMBO molecular medicine 2011 Jan;3(1):50-66
EMBO molecular medicine 2011 Jan;3(1):50-66
Microbiota-induced tertiary lymphoid tissues aggravate inflammatory disease in the absence of RORgamma t and LTi cells.
Lochner M, Ohnmacht C, Presley L, Bruhns P, Si-Tahar M, Sawa S, Eberl G
The Journal of experimental medicine 2011 Jan 17;208(1):125-34
The Journal of experimental medicine 2011 Jan 17;208(1):125-34
Nuclear export of the NF-κB inhibitor IκBα is required for proper B cell and secondary lymphoid tissue formation.
Wuerzberger-Davis SM, Chen Y, Yang DT, Kearns JD, Bates PW, Lynch C, Ladell NC, Yu M, Podd A, Zeng H, Huang TT, Wen R, Hoffmann A, Wang D, Miyamoto S
Immunity 2011 Feb 25;34(2):188-200
Immunity 2011 Feb 25;34(2):188-200
Enhanced Toll-like receptor (TLR) responses of TNFR-associated factor 3 (TRAF3)-deficient B lymphocytes.
Xie P, Poovassery J, Stunz LL, Smith SM, Schultz ML, Carlin LE, Bishop GA
Journal of leukocyte biology 2011 Dec;90(6):1149-57
Journal of leukocyte biology 2011 Dec;90(6):1149-57
Lung CD103+ dendritic cells efficiently transport influenza virus to the lymph node and load viral antigen onto MHC class I for presentation to CD8 T cells.
Ho AW, Prabhu N, Betts RJ, Ge MQ, Dai X, Hutchinson PE, Lew FC, Wong KL, Hanson BJ, Macary PA, Kemeny DM
Journal of immunology (Baltimore, Md. : 1950) 2011 Dec 1;187(11):6011-21
Journal of immunology (Baltimore, Md. : 1950) 2011 Dec 1;187(11):6011-21
Negative selection by IgM superantigen defines a B cell central tolerance compartment and reveals mutations allowing escape.
Duong BH, Ota T, Aoki-Ota M, Cooper AB, Ait-Azzouzene D, Vela JL, Gavin AL, Nemazee D
Journal of immunology (Baltimore, Md. : 1950) 2011 Dec 1;187(11):5596-605
Journal of immunology (Baltimore, Md. : 1950) 2011 Dec 1;187(11):5596-605
Sertoli cell-specific deletion of the androgen receptor compromises testicular immune privilege in mice.
Meng J, Greenlee AR, Taub CJ, Braun RE
Biology of reproduction 2011 Aug;85(2):254-60
Biology of reproduction 2011 Aug;85(2):254-60
Validation of MdmX as a therapeutic target for reactivating p53 in tumors.
Garcia D, Warr MR, Martins CP, Brown Swigart L, Passegué E, Evan GI
Genes & development 2011 Aug 15;25(16):1746-57
Genes & development 2011 Aug 15;25(16):1746-57
Factor VIII delivered by haematopoietic stem cell-derived B cells corrects the phenotype of haemophilia A mice.
Ramezani A, Zweier-Renn LA, Hawley RG
Thrombosis and haemostasis 2011 Apr;105(4):676-87
Thrombosis and haemostasis 2011 Apr;105(4):676-87
The innate immune response to uropathogenic Escherichia coli involves IL-17A in a murine model of urinary tract infection.
Sivick KE, Schaller MA, Smith SN, Mobley HL
Journal of immunology (Baltimore, Md. : 1950) 2010 Feb 15;184(4):2065-75
Journal of immunology (Baltimore, Md. : 1950) 2010 Feb 15;184(4):2065-75
Regulation of follicular B cell differentiation by the related E26 transformation-specific transcription factors PU.1, Spi-B, and Spi-C.
DeKoter RP, Geadah M, Khoosal S, Xu LS, Thillainadesan G, Torchia J, Chin SS, Garrett-Sinha LA
Journal of immunology (Baltimore, Md. : 1950) 2010 Dec 15;185(12):7374-84
Journal of immunology (Baltimore, Md. : 1950) 2010 Dec 15;185(12):7374-84
The death effector domain protein PEA-15 negatively regulates T-cell receptor signaling.
Pastorino S, Renganathan H, Caliva MJ, Filbert EL, Opoku-Ansah J, Sulzmaier FJ, Gawecka JE, Werlen G, Shaw AS, Ramos JW
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2010 Aug;24(8):2818-28
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2010 Aug;24(8):2818-28
Peripheral B cell tolerance and function in transgenic mice expressing an IgD superantigen.
Duong BH, Ota T, Aït-Azzouzene D, Aoki-Ota M, Vela JL, Huber C, Walsh K, Gavin AL, Nemazee D
Journal of immunology (Baltimore, Md. : 1950) 2010 Apr 15;184(8):4143-58
Journal of immunology (Baltimore, Md. : 1950) 2010 Apr 15;184(8):4143-58
c-Myb is required for pro-B cell differentiation.
Fahl SP, Crittenden RB, Allman D, Bender TP
Journal of immunology (Baltimore, Md. : 1950) 2009 Nov 1;183(9):5582-92
Journal of immunology (Baltimore, Md. : 1950) 2009 Nov 1;183(9):5582-92
A critical role of TAK1 in B-cell receptor-mediated nuclear factor kappaB activation.
Schuman J, Chen Y, Podd A, Yu M, Liu HH, Wen R, Chen ZJ, Wang D
Blood 2009 May 7;113(19):4566-74
Blood 2009 May 7;113(19):4566-74
Endogenous IL-21 restricts CD8+ T cell expansion and is not required for tumor immunity.
Søndergaard H, Coquet JM, Uldrich AP, McLaughlin N, Godfrey DI, Sivakumar PV, Skak K, Smyth MJ
Journal of immunology (Baltimore, Md. : 1950) 2009 Dec 1;183(11):7326-36
Journal of immunology (Baltimore, Md. : 1950) 2009 Dec 1;183(11):7326-36
Dendritic cells are required for optimal activation of natural killer functions following primary infection with herpes simplex virus type 1.
Kassim SH, Rajasagi NK, Ritz BW, Pruett SB, Gardner EM, Chervenak R, Jennings SR
Journal of virology 2009 Apr;83(7):3175-86
Journal of virology 2009 Apr;83(7):3175-86
E47 controls the developmental integrity and cell cycle quiescence of multipotential hematopoietic progenitors.
Yang Q, Kardava L, St Leger A, Martincic K, Varnum-Finney B, Bernstein ID, Milcarek C, Borghesi L
Journal of immunology (Baltimore, Md. : 1950) 2008 Nov 1;181(9):5885-94
Journal of immunology (Baltimore, Md. : 1950) 2008 Nov 1;181(9):5885-94
E47 controls the developmental integrity and cell cycle quiescence of multipotential hematopoietic progenitors.
Yang Q, Kardava L, St Leger A, Martincic K, Varnum-Finney B, Bernstein ID, Milcarek C, Borghesi L
Journal of immunology (Baltimore, Md. : 1950) 2008 Nov 1;181(9):5885-94
Journal of immunology (Baltimore, Md. : 1950) 2008 Nov 1;181(9):5885-94
p27 deficiency cooperates with Bcl-2 but not Bax to promote T-cell lymphoma.
Cheng N, van de Wetering CI, Knudson CM
PloS one 2008 Apr 2;3(4):e1911
PloS one 2008 Apr 2;3(4):e1911
Phospholipase Cgamma2 contributes to light-chain gene activation and receptor editing.
Bai L, Chen Y, He Y, Dai X, Lin X, Wen R, Wang D
Molecular and cellular biology 2007 Sep;27(17):5957-67
Molecular and cellular biology 2007 Sep;27(17):5957-67
Silent development of memory progenitor B cells.
Aviszus K, Zhang X, Wysocki LJ
Journal of immunology (Baltimore, Md. : 1950) 2007 Oct 15;179(8):5181-90
Journal of immunology (Baltimore, Md. : 1950) 2007 Oct 15;179(8):5181-90
Silent development of memory progenitor B cells.
Aviszus K, Zhang X, Wysocki LJ
Journal of immunology (Baltimore, Md. : 1950) 2007 Oct 15;179(8):5181-90
Journal of immunology (Baltimore, Md. : 1950) 2007 Oct 15;179(8):5181-90
B-1 B lymphocytes require Blimp-1 for immunoglobulin secretion.
Savitsky D, Calame K
The Journal of experimental medicine 2006 Oct 2;203(10):2305-14
The Journal of experimental medicine 2006 Oct 2;203(10):2305-14
Negative regulation of activation-induced cytidine deaminase in B cells.
Muto T, Okazaki IM, Yamada S, Tanaka Y, Kinoshita K, Muramatsu M, Nagaoka H, Honjo T
Proceedings of the National Academy of Sciences of the United States of America 2006 Feb 21;103(8):2752-7
Proceedings of the National Academy of Sciences of the United States of America 2006 Feb 21;103(8):2752-7
Essential role of phospholipase C gamma 2 in early B-cell development and Myc-mediated lymphomagenesis.
Wen R, Chen Y, Bai L, Fu G, Schuman J, Dai X, Zeng H, Yang C, Stephan RP, Cleveland JL, Wang D
Molecular and cellular biology 2006 Dec;26(24):9364-76
Molecular and cellular biology 2006 Dec;26(24):9364-76
In vivo ablation of CD11c-positive dendritic cells increases susceptibility to herpes simplex virus type 1 infection and diminishes NK and T-cell responses.
Kassim SH, Rajasagi NK, Zhao X, Chervenak R, Jennings SR
Journal of virology 2006 Apr;80(8):3985-93
Journal of virology 2006 Apr;80(8):3985-93
A role for brain-derived neurotrophic factor in B cell development.
Schuhmann B, Dietrich A, Sel S, Hahn C, Klingenspor M, Lommatzsch M, Gudermann T, Braun A, Renz H, Nockher WA
Journal of neuroimmunology 2005 Jun;163(1-2):15-23
Journal of neuroimmunology 2005 Jun;163(1-2):15-23
Impaired immune responses and B-cell proliferation in mice lacking the Id3 gene.
Pan L, Sato S, Frederick JP, Sun XH, Zhuang Y
Molecular and cellular biology 1999 Sep;19(9):5969-80
Molecular and cellular biology 1999 Sep;19(9):5969-80
Identification of monoclonal antibodies for immunohistochemical staining of feline B lymphocytes in frozen and formalin-fixed paraffin-embedded tissues.
Monteith CE, Chelack BJ, Davis WC, Haines DM
Canadian journal of veterinary research = Revue canadienne de recherche veterinaire 1996 Jul;60(3):193-8
Canadian journal of veterinary research = Revue canadienne de recherche veterinaire 1996 Jul;60(3):193-8
Surface antigen expression and immunoglobulin gene rearrangement during mouse pre-B cell development.
Coffman RL
Immunological reviews 1982;69:5-23
Immunological reviews 1982;69:5-23
Abnormalities induced by the mutant gene Ipr: expansion of a unique lymphocyte subset.
Morse HC 3rd, Davidson WF, Yetter RA, Murphy ED, Roths JB, Coffman RL
Journal of immunology (Baltimore, Md. : 1950) 1982 Dec;129(6):2612-5
Journal of immunology (Baltimore, Md. : 1950) 1982 Dec;129(6):2612-5
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Staining of C57Bl/6 splenocytes with Anti-Mouse CD3 PerCP-eFluor® 710 (Product # 46-0032-82) and 0.125 µg of Rat IgG2a K Isotype Control APC (Product # 17-4321-81) (left) or 0.125 µg of Anti-Human/Mouse CD45R (B220) APC (right). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
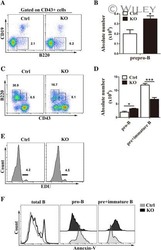
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
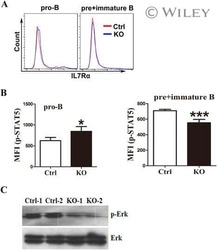
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
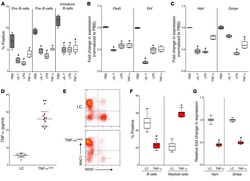
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
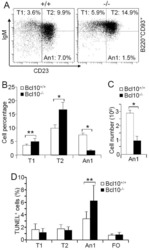
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
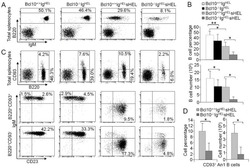
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
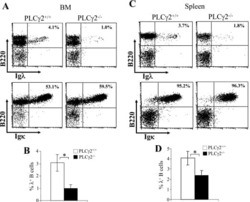
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
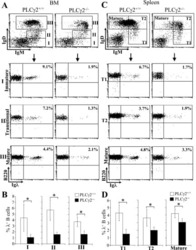
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
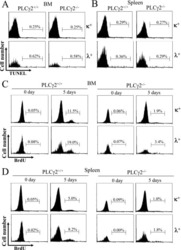
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
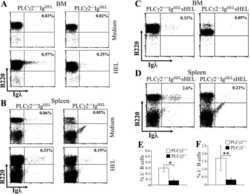
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
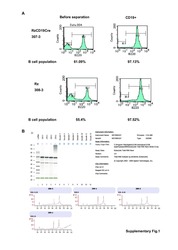
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
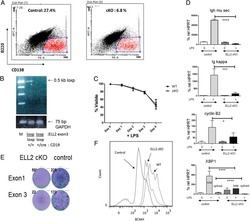
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
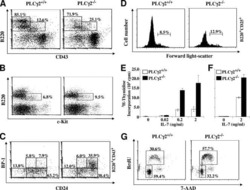
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
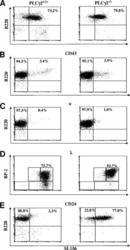
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
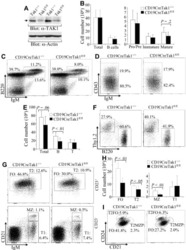
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
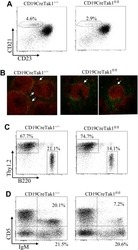
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
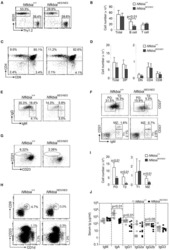
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
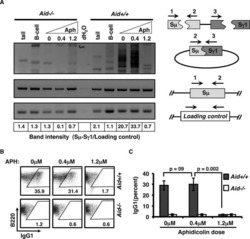
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
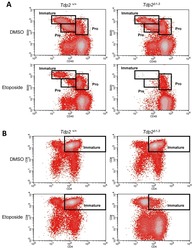
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
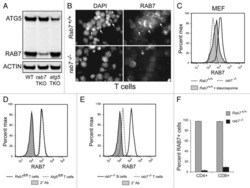
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
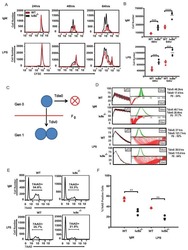
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
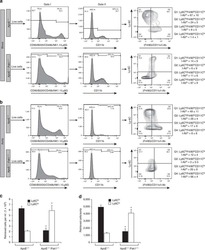
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
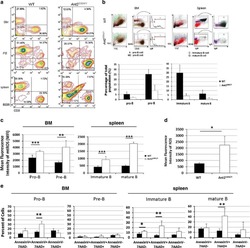
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
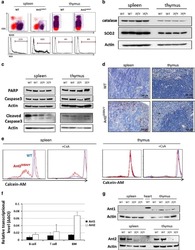
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
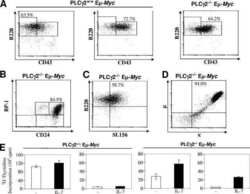
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
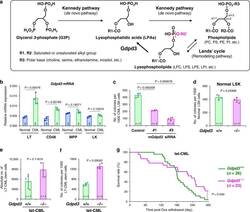
- Experimental details
- Fig. 1 Gdpd3 is implicated in CML disease initiation in vivo. a Diagram of pathways of lysophospholipid biosynthesis. G3P is converted into LPAs, and LPAs are then converted into phospholipids by the addition of polar bases via the Kennedy (de novo) pathway. The Lands'' cycle (remodelling pathway) generates lysophospholipids of distinct composition by substituting fatty acid ester and polar base groups of phospholipids. Lysophospholipase D Gdpd3 converts lysophospholipids back into LPAs by catalysing hydrolysis (magenta dotted line). (PC Phosphatidylcholine, PS Phosphatidylserine, PE Phosphatidylethanolamine, PI Phosphatidylinositol, LPC Lysophosphatidylcholine, LPS Lysophosphatidylserine, LPE Lysophosphatidylethanolamine, LPI Lysophosphatidylinositol). b qRT-PCR determination of Gdpd3 mRNA expression in LT-stem (LT), CD48, MPP, and LK cells (see Supplementary Fig.2) isolated from Gdpd3 +/+ tet-CML-affected (SCL-tTA + TRE-BCR-ABL1 + ) mice (one male, six females) or normal littermate (SCL-tTA + ) mice (four males, four females). Data are the mean ratio +- s.d. of transcript levels normalised to Actb ( n = 3 biologically independent samples) ( P -value, unpaired two-sided Student''s t -test). c Quantitation of the colony-forming capacity of Gdpd3 +/+ CML-LSK cells that were transduced with/without Cy3-labelled siRNA targetting mouse Gdpd3 mRNA (mGdpd3 siRNA #1 or #3). Cy3 + and Cy3 - CML-LSK cells were purified at 3 days post-transduction and plated in semi-solid methylcellulo
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
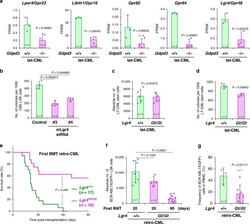
- Experimental details
- Fig. 7 Lgr4/Gpr48 is involved in CML stem cell self-renewal in vivo. a RNA-Seq determinations of mRNA levels of the indicated GPCR family genes in LT-CML stem cells isolated from Gdpd3 +/+ tet-CML-affected mice (22 males, 12 females) and Gdpd3 -/- tet-CML-affected mice (five males, five females). Results are expressed as FPKM (see Methods). Data are the mean FPKM +- s.d. ( n = 3 biologically independent samples) ( P -value, unpaired two-sided Student''s t -test). Results of the MA-plot and GO term enrichment analyses for these RNA-Seq data are shown in Supplementary Fig. 11a, b . b Quantitation of the colony-forming capacity of Gdpd3 +/+ CML-LSK cells that were transduced with/without Cy3-labelled siRNA targetting mouse Lgr4/Gpr48 mRNA (mLgr4 #3 or Lgr4 #4). Cy3 + and Cy3 - CML-LSK cells were purified at 3 days post-transduction and plated in a semi-solid methylcellulose medium. Data are the mean colony number +- s.d. ( n = 3) and are representative of three biologically independent experiments. ( P -value compared with control, unpaired two-sided Student''s t -test). The relevant FACS data are shown in Supplementary Fig. 12 . c Absolute numbers of LT-CML stem cells isolated from BM of the two hind limbs of Lgr4 +/+ tet-CML-affected mice (four males, one female) and Lgr4 Gt/Gt tet-CML-affected mice (five females) ( n = 5 biologically independent samples). Data are the mean absolute numbers +- s.d. of LT-CML stem cells ( P -value, unpaired two-sided Student''s t -test). (See S
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
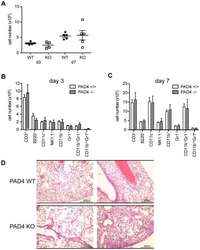
- Experimental details
- Figure 4 Lung leukocyte infiltration during influenza infection is comparable between PAD4 WT and KO mice. Experimental setup as described in Figure 2 . (A-C) Five mice per group were analyzed at d3 or d7 post infection. Leukocytes were isolated from infected lungs. (A) Total cell numbers of lung-infiltrating leukocytes at d3 and d7 p.i. Each symbol represents an individual mouse, filled squares represent PAD4 WT, open squares depict PAD4 KO mice. (B+C) The subsets of infiltrating leukocytes were enumerated by flow cytometry both at d3 p.i. (B) and d7 p.i. (C). Gated populations are indicated at the bottom of the graph. Total numbers of infiltrating cells are shown. WT mice are depicted in white bars, KO mice as grey bars. Bars represent mean + SEM. (D) Lungs for histological examination were harvested at d8 p.i. (5 mice/group). Leukocyte infiltration was assessed on H&E stained paraffin sections. Sections from two representative mice per group are shown. The scale bar indicates 200 um. Data is representative of two independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
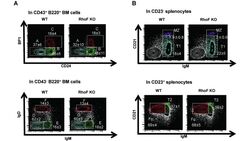
- Experimental details
- Fig. 4 Analysis of B cell development in bone marrow and spleen. A. FACS analysis of BM B cells. BM cells from WT and RhoF KO mice (6-7 weeks old, n = 5, male: 40%) were stained with antibodies against CD43, B220, BP1, and CD24 and analyzed by flow cytometry. The number shows the percentage (mean +- standard deviation) of the indicated subpopulation within the parent population; Fraction A (germline pro-B cells), fraction B (DJ-rearranged pro-B cells), fraction C (Early pre-B cells), fraction D (Late pre-B cells), fraction E (newly formed B cells), and fraction F (follicular-type recirculating B cells). The figures are representatives of three independent experiments. B. FACS analysis of B cells from the spleen. Splenocytes from WT and RhoF KO mice (6-7 weeks old, n = 6, male: 33%) were stained with antibodies against CD23, CD21, and IgM and analyzed by flow cytometry. The number shows the percentage (mean +- standard deviation) of the indicated subpopulation within the parent population; T1 (Transitional 1), T2 (Transitional 2), Fo (mature follicular B cells), and MZ (MZ B cells). C. The number of B cell subsets in BM was calculated by multiplying the total number of viable (trypan blue negative) BM cells by the fraction of the target population in viable (7AAD negative) cells. The data are shown as mean +- standard deviation (6-7 weeks old, n = 5, male: 40%). White bars: WT. Black bars: RhoF KO. D. The number of B cell subsets in the spleen was also calculated by multiplyin
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
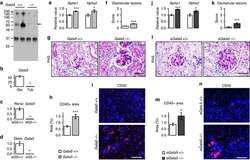
- Experimental details
- Figure 2 Loss of Gata5 from renal endothelial cells leads to renal alterations. ( a ) GATA5 is expressed in the kidney as assessed by western blot performed on total kidney extracts. ( b ) Gata5 is essentially expressed in the glomeruli as assessed by qPCR on isolated glomeruli (Glo) and microdissected tubules (Tub) from Wt mice kidneys. ( n =3-5 per group). The results are reported as mean+-s.e.m. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
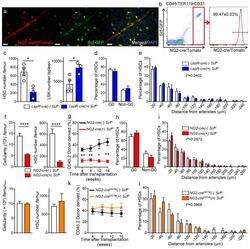
- Experimental details
- Figure 5 NG2-cre, but not NG2-cre ERTM , targeted cells are the source of Scf in the bone marrow (a) Whole-mount sternum from NG2-cre/ iTdTomato/ Scf-GFP mice, anti-VE-cadherin. Representative images from 3 mice. Scale bars, 20 mum. (b) Representative FACS plot showing percentage of NG2-cre/ iTdTomato + cells within CD45 - TER119 - CD31 - Scf-GFP + cells. n=3 mice. (c-e) Analyses of LepR-cre/ Scf fl/- mice. (c) Numbers of HSCs (left) in BM and LSK cells in spleen (right). n=4 mice for cre (-), n=3 mice for cre (+). (d) FACS analyses of HSC (CD150 + CD48 - LSK) cell cycle with Ki-67 and Hoechst 33342 staining. n=5 mice for cre (-), n=6 mice for cre (+). (e) HSC localization relative to arterioles. Error bars: n=3 mice. P value has been calculated using n=272 HSCs for cre (-), 293 HSCs for cre (+) pooled from 3 mice per group. P =0.3402. (f-i) Analyses of NG2-cre/ Scf fl/- mice. (f) Numbers of total BM cells (left) and CD150 + CD48 - LSK HSCs (right) in BM. n=5 mice for cre (-), n=7 mice for cre (+). (g) Percentages of donor-derived cells after competitive reconstitution. n=5 mice for cre (-), n=7 mice for cre (+). (h) FACS analyses of HSC cell cycle with Ki-67 and Hoechst 33342 staining. n=6 mice for cre (-), n=7 mice for cre (+). (i) HSC localization relative to arterioles. Error bars: n=3 mice. P value has been calculated using n=224 HSCs for cre (-), 274 HSCs for cre (+) pooled from 3 mice per group. P =0.2872. (j-l) Analyses of NG2-cre ERTM / Scf fl/- mice. (j) Absolute nu
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
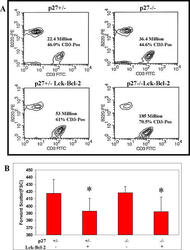
- Experimental details
- Figure 2 Splenic T cell hyperplasia and T cell size in p27 -/- Lck-Bcl-2 mice. A) Total splenic cells from mice of the indicated genotypes were isolated and stained with anti-B220-PE and anti-CD3-FITC antibodies as described in the Materials and Methods . The percentage of CD3-positive cells and the total number of splenic T cells (total # of splenic cells X %CD3) is shown. The data are representative of at least three mice from each genotype. B) The Mean Forward Scatter of CD3 positive splenic T cells was determined by staining with an anti-CD3 antibody as described in the Materials and Methods . The Mean+-SD of at least 5 mice is shown for the genotypes indicated. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 7. NOX2 and FOXC1 Are Important for the Growth of Primary Human Myeloid Leukemia Cells (A) The Cancer Genome Atlas (TCGA) database for AML was used to analyze the expression levels of several NADPH-dependent oxidases (NOX1-NOX5), accessory subunits (NCF1, NCF2, NCF4, NOXA1, NOXO1, p22Phox, RAC1, and RAC2) as well as related oxido-reductase enzymes (dual-oxidases 1,2, A2). The reads per kilobase of transcript per million mapped reads (RPKM) values for each gene in a total of 188 AMLs are shown. (B) RNA-seq analysis was performed on functionally validated leukemic stem cells isolated from human primary AMLs. The RPKM value for each gene is shown. Unpublished data are used with permission. Additional supporting data are shown in Figure S7A . (C) Equal numbers of control or shNOX2-transduced primary AML cells were cultured in vitro in the presence of 10 ng/mL of IL-3, SCF, and FL3, and the relative percent expansion is reported for each specimen. n = 3, mean +- SD. *p < 0.05; **p < 0.01. (D) Control and shNOX2 primary AML cells were purified and cultured in vitro for 12 days. Annexin V, DAPI staining was performed to evaluate the degree of apoptotic cell death. A representative flow plot (left) and quantitation of 3 technical triplicates (right) are shown. Additional data are shown in Figure S7B . (E) The relative expression level of NOX2 , CEBP epsilon, Elane , and CTSG is shown in AML specimens in which NOX2 was knocked down using shRNAs. Additional supporting data are p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 6 Assessments of organ damage and mouse metabolites during HLSA or recovery from HLSA. A , upper left panel , MMP-1, IL-1beta, and CRP levels in plasma had no significant change between the control group, HLSA group, and R-HLSA group. Upper right panel , CK-MB as a marker of heart injury in the plasma had no significant change between the control group, HLSA group, and R-HLSA group. Lower left panel , creatinine and BUN as markers of kidney injury in the plasma slightly increased in the HLSA group and returned to normal level after recovery from HLSA. Lower right panel , AST and ALT as markers of liver injury in the plasma showed no change between the control group, HLSA group, and R-HLSA group. Data were presented as mean +- SD (N = 6; ANOVA: ** p < 0.01). B , upper panel , food consumption and water consumption 3 days before and 3 days after a single ATP-induced HLSA depicted no change. Data were presented as mean +- SD (N = 6; ANOVA: p > 0.05). Lower panel , metabolomics analysis of scores plot from PCA analysis based on 1 H NMR data from the plasma, brain, liver, and kidney of the control group, HLSA group, and R-HLSA group. The PCA score showcased clusters correspond to metabolic patterns in different groups, with each point representing one sample. Circles represent 95% confidence interval for each score in each group (see also Figs. S5-S8 ). C , draining lymph nodes of mice that underwent HLSA once a day for 10 consecutive days were analyzed with FACS. CD44 and
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 5 CD4 surface expression of CD45RB and global glycosylation are altered in SLE123 mice. ( A and B ) Cell surface expression of CD45RB and total CD45 were measured by flow cytometry on CD4 + T cells. SLE123 demonstrated a downregulation of CD45RB on the cell surface as compared with B6 CD4 + T cells. There was no difference in pan-CD45 expression. Quantified in B . ( C ) CD45 is composed of an intracellular region that controls cytoskeletal binding and its phosphatase activity. The extracellular domain is composed of a region with fibronectin repeats that is heavily N-glycosylated. An alternatively spliced region is heavily O-glycosylated, and this region imparts unique functions to each CD45 isoform. Additionally, a sialic acid residue on the B portion of this region is essential for the binding of therapeutic aCD45RB. ( D and E ) We utilized lectins to detect the level of alpha-2,3-linked sialylation (MALII), O-linked glycosylation (Jacalin), and N-linked glycosylation (PHA-L) in CD4 + T cells from B6 and SLE123 mice. We determined SLE123 CD4 + T cells had reduced levels of alpha-2,3-linked sialic acids and O-glycosylation and an increase in N-glycosylation. Quantified in E . ( F ) Utilizing an antibody that detects a desialylated form of anti-CD45RB, we determined SLE123 CD4 + T cells possessed increased binding of this antibody compared with B6. ( G and H ) To determine the binding of therapeutic aCD45RB to B6 and SLE123 CD4 + T cells, we incubated splenocytes from
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
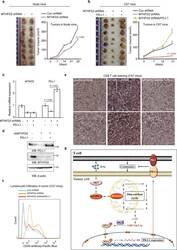
- Experimental details
- Fig. 7 Upregulation of PD-L1 by MTHFD2 is required for tumorigenesis. a - b A total of 2 x 10 6 ( a ) or 5 x 10 6 ( b ) Pan02 cells stably infected with lentivirus carrying indicated shRNAs or exogenous expressed PD-L1, were subcutaneously injected into athymic nude mice ( a ) or C57 mice ( b ), tumor volume was calculated every 7 days. Tumor xenografts at the 28th day in nude mice ( a ) or the 21th day in C57 mice ( b ) were shown. Data represent the means +- s.e.m ( n = 8 mice per group); p value (Student's t test, two-sided) with control is presented. c , PD-L1 mRNA levels in tumor tissues in C57 mice were analyzed by real-time PCR. The values are presented as mean +- s.e.m ( n = 3); p values (Student's t test, two-sided) with control or the indicated groups are presented. d The lysates of 8 pooled tumor tissues in C57 mice were subjected to immunoblotting analyses using the indicated antibodies. e Immunohistochemical staining was performed on tumor sections in C57 mice with anti-CD8 antibody. Representative images are shown. Scale bars, 50 mum. Histological semi-quantification was performed. f Cells digested from indicate tumor tissues in C57 mice were stained with anti-CD45 antibody and subjected to flow cytometric analyses (also see Supplementary Fig. 7b ). Representative images (1 out of 3 experiments) are shown. g A schematic model showing the role of MTHFD2 in tumor immune evasion. MTHFD2 promotes PD-L1 mediated tumor immune evasion through the folate-cycle-UTP-UDP-G
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
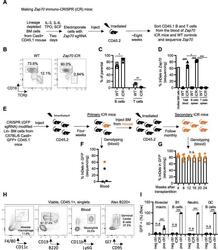
- Experimental details
- Fig. 2 Generating immuno-CRISPR (iCR) mice and evaluating the CRISPR-mediated modifications by sequencing. ( A ) Model describing the experimental setup where CD45.1 + Lin- BM cells were modified by CRISPR targeting Zap70 and grafted into irradiated CD45.2 + recipients. ( B ) Flow cytometry analysis of cells in the blood of Zap70 iCR mice and WT control mice eight weeks post transplantation. Cells gated on viable, CD45.1+, single lymphocytes. ( C ) Quantification of B and T cells in the blood of WT and Zap70 iCR mice in (B). ( D ) Analysis of the level of mutations in the sgRNA targeted Zap70 region in the BM cells used for transplantation, total cells from the blood, as well as in B and T cells sorted from the spleen of Zap70 iCR mice and WT control mice 8 weeks after engraftment. ( E ) Model describing the experimental setup where a secondary transplantation was used to amplify the population of successfully modified mice. ( F ) Analysis of the level of mutations in the sgRNA targeted GFP region in blood cells of the GFP iCR mice four weeks after transplantation, in an experiment with low efficiency. One mouse showed good knockout efficiency (labeled in orange) and was used as BM donor for secondary transplantation. ( G ) Kinetics of the level of mutations of GFP in the secondary iCR mice. ( H ) As examples, representative flow cytometry plots and ( I ) GFP + cell population percentage of alveolar macrophages, B1 cells, neutrophils and germinal center B cells from secondary
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
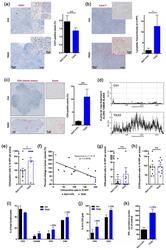
- Experimental details
- Figure 4 Proof-of-mechanism study for TAX2 use as an anti-tumor immunomodulatory drug. The effects of TAX2 peptide treatment on the activation of an anti-tumor immune response were investigated in two syngeneic models of epithelial ovarian carcinoma. We inoculated 5 x 10 6 ID8 cells either subcutaneously or intraperitoneally to C57BL/6JRj mice in order to mimic primary tumor growth ( a - h ) and metastatic dissemination (peritoneal carcinosis, i - l ), respectively. TAX2 treatments (30 mg * kg -1 BW) vs. vehicle injections (0.9% NaCl) were performed thrice weekly for 4 and 8 weeks for the s.c. and i.p. ID8 models, respectively ( n = 11 per group for both models). ( a - h ) IHC staining of s.c. ovarian carcinoma allografts for the analysis of the vascular/lymphovascular features, as well as immune-cell-infiltration profile. ( a ) Representative microphotograph for CD31 immunostaining of intra-tumor vascular structures. Histogram displays results of automated quantification for the percent of CD31-positive pixels across the whole tumor section (mean +- SEM, Mann-Whitney U test; n.s., not significant). ( b ) Lymphatic vessel density analysis. Microscopic views are shown, while histogram displays the number of Lyve-1-positive functional lymphatic vessels in 5 high power fields (HPF), as determined by a pathologist who was blinded to the treatment (mean +- SEM, Mann-Whitney U test, * p < 0.05). ( c - f ) Macroscopic views (x20 magnification) of s.c. tumor allograft sections after
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
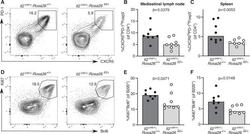
- Experimental details
- Figure 8. Increased IL-2 production impairs T follicular helper (Tfh) cell formation and the germinal centre response. Assessment of the Tfh cell and germinal centre response in Il2 cre/+ ; Rosa26 stop-flox-Il2/+ transgenic mice that do not switch off IL-2 production, and Il2 cre/+ ; Rosa26 +/+ control mice 12 days after influenza A infection. Flow cytometric contour plots ( A ) and quantification of the percentage of CXCR5 high PD-1 high Foxp3 - CD4 + Tfh cells in the mediastinal lymph node ( B ) and spleen ( C ). Flow cytometric contour plots ( D ) and quantification of the percentage of Bcl6 + Ki67 + B220 + germinal centre B cells in the mediastinal lymph node ( E ) and spleen ( F ). The height of the bars indicates the median, each symbol represents one mouse, data are pooled from two independent experiments. p-Values calculated between genotype groups by Mann-Whitney U test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
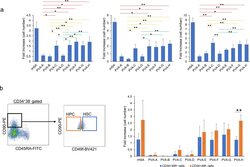
- Experimental details
- Fig. 2. Effect of different PVA hydrolysis rates and molecular weights on ex vivo cultured human cord blood CD34 + hematopoietic stem and progenitor cells. a , Human cord blood CD34 + cells were cultured in PVA media and cell numbers were counted at day 14. Three independent experiments were performed with cord blood CD34 + cells derived from different donors. Cell cultures were started from 7 x 10 3 (Exp.1) or 5 x 10 3 (Exp. 2 and 3) CD34 + cells. Mean +- S.D is shown. Statistical significance was calculated using ANOVA followed by Tukey-Kramer; *p < 0.05, **p < 0.01. b , Proliferation of the two different CD34 + cell fractions after 7-day culture in PVA-containing media. Fifty cells were sorted into four wells in a 96-well plate and cell numbers in each well were counted at 7-day. Statistical significance was calculated using ANOVA and Tukey-Kramer. **p < 0.01.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
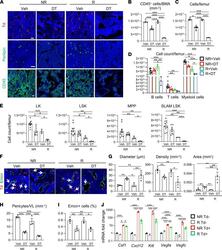
- Experimental details
- Figure 6 MALP ablation blocks bone marrow recovery after radiation. ( A ) Representative fluorescence images of Td + cells, Perilipin + LiLAs, and CD45 + hematopoietic cells in femoral bone marrow of Adipoq/Td/DTR mice after receiving 2 weeks of vehicle (Veh) or DT injections with or without prior radiation. Scale bar: 20 mum (top), 100 mum (middle), and 20 mum (bottom). ( B ) Quantification of CD45 + cells per bone marrow area. n = 3-6 mice/group. ( C ) Bone marrow cells were flushed from femurs and counted. n = 3-8 mice/group. ( D ) Cell counts of hematopoietic lineage cells in the bone marrow. n = 3-11 mice/group. B cells = B220 + , T cells = CD3 + , myeloid cells = Gr1 + and/or Mac1 + . ( E ) Cell counts of HSPCs. n = 3-11 mice/group. LK, Lineage - cKit + , LSK, Lineage - Sca1 + cKit + , SLAM LSK, Lineage - Sca1 + cKit + CD48 - CD150 + , MPP, Lineage - Sca1 + cKit + CD48 + CD150 - . ( F ) Representative fluorescence images of Adipoq/Td/DTR femoral bone marrow with Emcn staining (vessels). Arrows point to Td + pericytes. Scale bar: 20 mum. ( G ) Quantification of bone marrow vessel diameter, density, and area. ( H ) The number of pericytes per vessel length (VL) was measured. n = 3-4 mice/group. ( I ) The percentage of Emcn + endothelial cells in bone marrow was measured by flow cytometry. n = 3-4 mice/group. ( J ) qRT-PCR analysis of hematopoietic and angiogenic factors in sorted Td - and Td + cells from bone marrow before and after radiation. n = 4 mice/group. Statistica
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
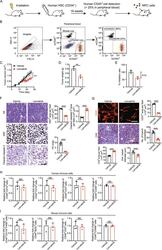
- Experimental details
- Lenvatinib insignificantly affects the immune microenvironment in NPC in humanized NSG mice. A Schematic diagram of the establishment of humanized NSG mice. B Representative FACS analysis of human CD45 + cells in mouse peripheral blood. Human CD45 + cell percentage greater than 25% was considered successful in modeling. C - E Tumor growth ( C ), tumor weights ( D ) were measured in vehicle-, anti-VEGF-, and lenvatinib-treated NPC tumors. The tumor inhibition ratio were calculated ( E ) ( n = 3 samples per group). F Representative micrographs of Ki67 + proliferative cells and cleaved caspase-3 + apoptotic cells in vehicle-, anti-VEGF-, and lenvatinib-treated NPC tumors. Scale bar = 50 mum. Quantification of Ki67 + , cleaved caspase-3 + signals, and PA index in vehicle-, anti-VEGF-, and lenvatinib-treated NPC tumors. ( n = 8 random fields per group) G Representative micrographs of CD31 + microvessels and CA9 + hypoxic areas in vehicle-, anti-VEGF-, and lenvatinib-treated NPC tumors. Scale bar in upper panel = 100 mum, scale bar in lower panel = 50 mum. Quantification of CD31 + tumor vessel parameters and CA9 + signals in vehicle-, anti-VEGF-, and lenvatinib-treated NPC tumors ( n = 8 random fields per group). H Quantification of hCD45 + hCD14 + population, hCD45 + hCD19 + population, hCD45 + hCD3 + population, and hCD45 + hCD56 + population in the NPC TME ( n = 3 samples per group). I Quantification of mCD45 + mCD11b + mF4/80 + population, mCD45 + mB220 + population, mCD45 + mC
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
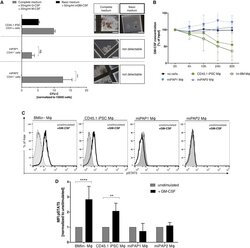
- Experimental details
- Figure 6 Disease Modeling of herPAP Using miPAP iPSCs (A) Number of CD41 + progenitor-derived colonies in methylcellulose-based clonogenic assays employing complete (IL-6, erythropoietin, SCF, IL-3, and supplemented with 20 ng/ml human G-CSF + 20 ng/ml murine GM-CSF) or basic medium (50 ng/ml murine GM-CSF only; independent experiments, n = 3 miPAP1 and 2, n = 2 CD45.1 iPSC, mean +- SD) and representative pictures of colonies. Scale bars, 500 mum. (B) GM-CSF clearance assay comparing miPAP-Mphi with CD45.1(10.3) iPSC-Mphi, BMlin - -Mphi, and no cells incubated with 2 ng/ml GM-CSF: at indicated time points (0, 4, 10, 24, and 30 hr) GM-CSF concentrations in supernatants were analyzed by ELISA, normalized to 0 hr (independent experiments, n = 3 BMlin - and CD45.1 iPSC, n = 2 miPAP1 and 2, mean +- SD). (C and D) Flow cytometry plots of STAT5 phosphorylation levels upon stimulation with mGM-CSF (C) and (D) summary of mean fluorescence intensity (MFI) data (independent experiments, n = 3 BMlin - and CD45.1 iPSC, n = 2 miPAP1 and 2, mean +- SD). ns, not significant; ** p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
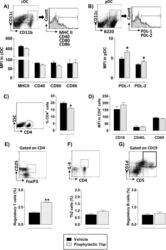
- Experimental details
- Fig 6 Tn P acts systemically during the induction phase modulating DCs and induces regulatory cells. Splenocytes from EAE mice treated with vehicle or Tn P (n = 5/group) were isolated at day 7, and analyzed. A ) The MFI of MHC class II, CD40, CD80, CD86 in cDC (CD11c+CD11b+) and B ) MFI of PDL-1 and PDL-2 in pDC (CD11c+B220 low ) were analyzed by flow cytometry (50,000 events). C ) The percentage of CD4+ cells, D ) and the MFI of CD18, CD40L, and CD69 in the CD4+ gate were analyzed by flow cytometry. E , F , G ) The percentages of FOXP3-positive CD4+CD25+ Treg, IL-4-positive CD4 Th2 cells and CD5-positive CD19+CD1d+ Breg cells were analyzed by flow cytometry. Values in the bar graphs are the mean +- SEM. * p < 0.05 and ** p < 0.01 compared with vehicle-treated EAE-mice.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
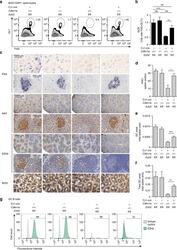
- Experimental details
- Fig. 1 In vivo depletion of Cdkn1a rescues GC formation in Ezh2 -/- mice. Ezh2 fl/fl , Cdkn1a -/- , Ezh2 fl/fl ;Cgamma1-cre and Ezh2 fl/fl ;Cgamma1-cre; Cdkn1a -/- littermate mice were immunized with SRBC to induce germinal center (GC) formation and were killed 10 days later. a Flow cytometry plot of one representative mouse spleen per group. The gated area shows the percentage of GC B cells (GL7 + FAS + ) within live B cells (B220 + DAPI-, see Supplementary Fig. 2A for gating strategy). b Average of GC B populations of each group of mice quantified by flow cytometry as in a ( n = 7 mice per group). c Formalin fixed paraffin embedded splenic tissue was stained for PNA, Ki67, EZH2, and B220. One representative picture of three spleens analyzed per group is shown. d - f Quantification of PNA staining from c ( n = 3 spleens per group). d ""#GC/spleen section"" is the count of all GC per spleen section. e ""GC area/total spleen area"" is the quantified area of each individual GC divided by the total area of the spleen section. f ""Total GC area/total spleen area"" is the sum of all GC quantified areas in a certain section divided by the total area of that spleen section. g Splenocytes were permeabilized and stained for GC B and EZH2 using a fluorochrome-conjugated anti EZH2 antibody. The gated area shows the percentage of GC B cells (GL7 + FAS + ) within live B cells (B220 + DAPI-, see Supplementary Fig. 2A for gating strategy) that are EZH2 positive. The flow plot shown is repre
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
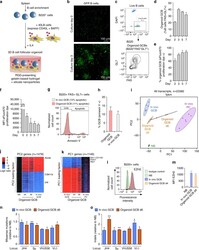
- Experimental details
- Fig. 3 Characterization of a 3D B cell follicular organoid to model the GC reaction. a Scheme of 3D B cell follicular organoid fabrication. Splenic B cells are co-encapsulated with 40LB stromal cells and IL4 into an RGD-presenting nanocomposite hydrogel that contain gelatin ionically cross-linked with synthetic silicate nanoparticles. b Representative fluorescence pictures of splenic GFP B cells in 3D organoid culture. c Flow cytometry plot of a 3D B cell follicular organoid. The gated area on the top shows the live B cells (B220 + DAPI-) and on the bottom plot, the organoid GC B cells (GL7 + FAS + ) within live B cells. d Average of GC B populations of organoids quantified as in c . e Average of percentage of proliferating organoid GC B populations, quantified as indicated in Supplementary Fig. 4B . f MFI of proliferation dye from e . g Flow cytometry plot of GC B cells stained with annexinV. h Average of percentage of apoptotic GC B cells quantified as in g . i RNA-seq profiles of organoid GC B cells after 4 and 6 days in culture (organoid GCB d4, n = 4 spleens, and d6, n = 3 spleens) were projected into the principal component space defined by GC B cells sorted from immunized mice (in vivo GCB, n = 6 mice), CD138 + plasma cells (in vivo PC, n = 6 mice) and FAS-GL7-IgD + B220 + naive B cells (NB, n = 3 mice). j , k Heat maps of gene expression level of GC B cells showed in i , represented as log2 ratio relative to mean naive B cells j and to mean plasma cells k . PC1/2 = pr
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
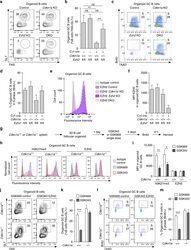
- Experimental details
- Fig. 4 CDKN1A repression by EZH2 is required for GC B cell cycle progression. a - f Organoids were generated using B cells isolated from Ezh2 fl/fl ;Cgamma1-cre, Cdkn1a -/- , Ezh2 fl/fl ;Cgamma1-cre; Cdkn1a -/- and Ezh2 fl/fl control mice ( n = 3 mice per group) and were harvested for flow cytometry analysis after 4 days in culture. a Flow cytometry plots of representative organoids from each genotype. The gated area shows the percentage of organoid GC B cells (GL7 + FAS + ) within live B cells (B220 + DAPI-). b Average of percentage of organoid GC B populations quantified by flow cytometry as in a ( n = 3 biological replicates per group). c Organoids received a BrdU pulse of 2 h before harvest. Cell cycle was analyzed by BrdU staining and 7AAD to measure DNA content. The representative gated area shows the percentage of organoid GC B cells (GL7 + FAS + B220 + DAPI-) that are in S phase (BrdU + ). d Average of percentage of organoid GC B populations in S phase of each group of genotype quantified by flow cytometry as in c ( n = 3 biological replicates per group). e Organoid cells were permeabilized and stained for EZH2 and GC markers GL7, FAS, and B220 to identify organoid GC B cells. The flow cytometry plot shows one representative sample per genotype group. f MFI of EZH2 in organoid GC B cells ( n = 3) quantified by flow cytometry as in e . g - m Organoids were generated using B cells isolated from 3 Cdkn1a +/+ and 3 Cdkn1a -/- mice. g Scheme of EZH2 inhibitor GSK343 and Br
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 1. VEGF and bFGF promote the differentiation of MSCs into EC-like cells. (A) Expression of MSC surface markers was confirmed using flow cytometry. (B) Morphological features of isolated MSCs and MSC-derived EC-like cells following induction as observed under an inverted microscope. The red and white arrows indicate spindle-shaped and polygon-shaped cells, respectively. Magnification, x20. (C) Factor VIII expression was detected in MSC-derived EC-like cells via immunohistochemistry and light microscopy with brown staining indicating the positive signal. VEGF, vascular endothelial growth factor; bFGF, basic fibroblast growth factor; MSCs, mesenchymal stem cells; EC, endothelial cell; Ctrl, control; CD, cluster of differentiation.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 1 Loss of miR-143/145 results in reduced LT-HSC frequency. a Frequency of long-term-HSC (LT-HSC, CD45 + EPCR + CD48 - CD150 + ), LSK/short-term HSC (Lin - Sca1 + c-Kit + ), common myeloid progenitors (CMP, Lin - Sca1 - c-Kit + CD34 + CD16/32 lo ), granulocyte-macrophage progenitors (GMP, Lin - Sca1 - c-Kit + CD34 + CD16/32 hi ), and megakaryocyte-erythrocyte progenitors (MEPs, Lin - Sca1 - c-Kit + CD34 - CD16/32 lo ) in the marrow of 8-12-week- old wild-type (WT), miR-143/145 +/- , and miR-143/145 -/- mice, as analyzed by flow cytometry (median +- 1.5 IQR, WT n = 11, 143/145 +/- n = 9, 143/145 -/- n = 8). b Marrow cellularity (2 femurs and 2 tibias) (WT n = 3, 143/145 +/- n = 4, 143/145 -/- n = 8). c Colony-forming unit (CFU) assay of marrow cells from WT and miR-143/145 -/- mice (mean +- SEM, WT n = 11, 143/145 -/- n = 10). d Primary CFU cells were replated in equal proportions per condition, normalized to the number of input cells, to generate secondary CFUs (mean +- SEM, WT n = 9, 143/145 -/- n = 7). e Estimate of HSC frequency in WT and miR-143/145 -/- mice by limiting dilution assay. Shown is a log-fraction plot of the limiting dilution model. The slope of the line is the log-active cell fraction. The dotted lines give the 95% CI
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 4 Surgical denervation of young bone marrow induces premature HSC and niche aging (a) Schematic illustration of surgical denervation experiment. The sciatic and femoral nerves were transected in young (2 months old) C57BL/6 mice and the mice were analyzed 4 months post surgery, at 6 months of age. (b) Absolute numbers of HSCs (lineage - Sca-1 + c-Kit + CD48 - CD150 + ) isolated from Sham and Denervated (Den) femurs (n=7 mice). (c, d) Total peripheral blood (CD45.2 + ), blood myeloid (Mac-1 + CD45.2 + ) and blood B cell (B220 + CD45.2 + ) donor chimerism at the indicated time points post transplantation (c) and bone marrow donor chimerism (Total CD45.2 + (BM); Myeloid (My), Mac-1 + CD45.2 + ; B cell (B), B220 + CD45.2 +; and T cell (T), CD4 + /CD8 + CD45.2 + ) 5 months after primary transplantation of 200 HSCs derived from either sham or denervated (Den) femurs and transplanted in competition with young BM competitor cells (n=4 sham, 6 denervated mice) (left) and 5 months after secondary transplantation of 3 x 10 6 bone marrow from primary recipients (right) (d). (e) Left, representative confocal z -stack projections of HSCs sorted from sham or denervated femurs and stained with Cdc42, Tubulin and DAPI. Scale bar, 10 mum. Right, quantification of the percentage of Cdc42 and Tubulin polarized HSCs out of total HSCs scored (total of 356 sham and 353 denervated HSCs isolated from 4 mice per group). (f) Left, representative confocal z -stack pro
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 1 ATF3 maintains intestinal homeostasis. (A) Comparison of colon length between naive mice as indicated. (B) Colon crypts from mice were isolated by shaking colon fragments in EDTA and counted under light microscopy. (C) Flow cytometry analysis of Ki67 and CD24 expression in ileum crypts, gated on the CD45 - EpCAM + populations, from the indicated naive mice. (D) Representative micrographs showing intestinal organoids derived from naive mice. (E) Quantitative real-time PCR analysis of cell cycle genes in naive ileum organoids at day 6 of culture (""n"" indicates organoids derived from 4 mice each group). (F) Representative confocal images of whole mount tissues with co-immunofluorescence staining of UEA-1 and WGA in naive ileum villi. Results were from at least two independent experiments and ""n"" refers to the number of mice unless indicated otherwise. All mice were at the age of 2~3 months old when analyzed. Statistical analysis was done using Multiple T -test on Prism software. * P < 0.05, ** P < 0.005, *** P < 0.0005.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
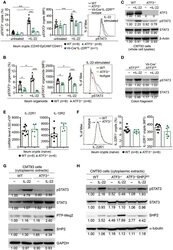
- Experimental details
- Figure 7 ATF3 promotes IL-22-induced STAT3 phosphorylation by suppressing phosphatases. (A) Freshly isolated ileum crypts, or (B) ileum organoids at day 6 of culture, were stimulated with IL-22, followed by fixation and intracellular staining of phospho-STAT3, and analyzed by flow cytometry. Western blot analysis of (C) IL-22-stimulated CMT93 cells, or (D) IL-22-stimuated colon fragments isolated from the indicated mice, for the expression of the indicated proteins. (E) Quantitative real-time PCR analysis of IL-22R1 and IL-10R2 mRNA levels in freshly isolated ileum crypts from mice. (F) Flow cytometry analysis of IL-22R1 in freshly isolated ileum crypt cells gated on the CD45 - EpCAM + population. (G,H) Western blot analysis of unstimulated or IL-22-stimulated CMT93 cells for the indicated proteins. ATF3 -/- CMT93 cells with SHP2 knockdown (ATF3 -/- SHP2 KD ) were indicated. Images were representative of four independent experiments (G-H) . Results were from two independent experiments (A-F) . ""n"" refers to the number of mice analyzed (A,B,E,F) . Statistical analysis was done by multiple comparison in Two-way ANOVA test using Prism software. * P < 0.05, ** P < 0.005, *** P < 0.0005.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
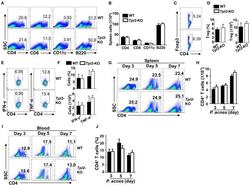
- Experimental details
- Figure 2 Tpl2 didn't affect peripheral immune activation during FH pathogenesis. The splenic cells or peripheral blood immune cells were isolated from P. acnes -primed WT and Tpl2 -KO mice at day 3, 5, and 7 as described in Materials and methods ( n = 4 mice/group), and subjected for flow cytometry analysis. (A-F) Flow cytometry analysis of the frequencies and absolute numbers of CD4 + T cells, CD8 + T cells, B220 + B cells, CD11c + dendritic cells (A,B) , CD4 + Foxp3 + Treg cells (C,D) , and IFN-gamma- and TNF-alpha-producing pathogenic Th1 cells (E,F) in the spleens of WT and Tpl2 -KO mice at day 7 after P. acnes priming. Data are presented as representative plots of the frequencies of immune cell subpopulations (A,C,E) and a summary graph of the cell frequencies or absolute cell numbers (B,D,F) . (G-J) Flow cytometry analysis of the frequencies and absolute numbers of CD4 + T cells in the spleens (G,H) or peripheral blood (I,J) of WT and Tpl2 -KO mice at day 3, 5, and 7 after P. acnes priming. Data are presented as representative plots of the frequencies of immune cell subpopulations (G,I) and a summary graph of the cell frequencies or absolute cell numbers (H,J) . Results are mean +- SD from three independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
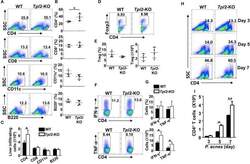
- Experimental details
- Figure 3 Tpl2 reduced liver infiltration of pathogenic CD4 + T cells. Liver-infiltrating immune cells were isolated from P. acnes -primed WT and Tpl2 -KO mice at day 3, 5, and 7 as described in Materials and methods ( n = 4 mice/group). (A-G) Flow cytometry analysis of the frequencies and absolute numbers of CD4 + and CD8 + T cells, B220 + B cell, CD11c + dendritic cells (A-C) , CD4 + Foxp3 + Treg cells (D,E) , and IFN-gamma- and TNF-alpha-producing pathogenic Th1 cells (F,G) in the livers of WT and Tpl2 -KO mice at day 7 after P. acnes priming. Data are presented as representative plots of the frequencies of immune cell subpopulations (A,D,F) and a summary graph of the frequencies and absolute cell numbers (B,C,E,G) . (H,I) Flow cytometry analysis of the frequencies and absolute numbers of CD4 + T cells in the livers of WT and Tpl2 -KO mice at day 3, 5, and 7 after P. acnes priming. Data are presented as representative plots of the frequencies of immune cell subpopulations (H) and a summary graph of the absolute cell numbers (I) . Results are mean +- SD from three independent experiments. Two-tailed Student's t -tests were performed. * P < 0.05; ** P < 0.01.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
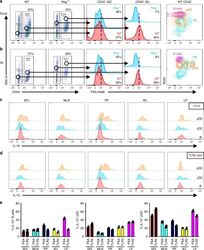
- Experimental details
- Fig. 6 TLR2 + macrophages, pDCs, and PB in the small intestine bind PSA and induce IL-10 secretion. Gating strategy for mononuclear cells isolated from a duodenum (Duod) and b ileum (Ile) of WT and Rag mice analyzed for binding of fluorescent A488-conjugated PSA (left two plots). CD45 - intra-epithelial cells (CD45 - IEC: middle histogram) and CD45 + gated intra-epithelial leukocytes (CD45 + IEL: second right histogram) isolated from the a Duod and b Ile of WT (red) and Rag (blue) mice were analyzed for reactivity to PSA-A488. CD45 + CD11c - B220 + B cells, PDCA1 + B220 + CD11c + pDCs and CD138 + B220 + PB and B220 low PC isolated from a Duod and b Ile of WT mice were analyzed for PSA reactivity (right histogram). Flow cytometry plots show CD11c + PDCA1 - cDC, PDCA1 + B220 + pDC, and B220 + CD19 + B cells isolated from spleen, MLN, PP, IEL, or LP of WT mice were stimulated with c PSA or d LTA-SA (TLR2 agonist) and analyzed for IL-10 expression. e Plots summarize data from c and d and show % IL-10 + B cells (left), cDCs (middle), and pDCs (right) from spleen, MLN, PP, IEL, and LP stimulated with PSA or TLR2 (LTA-SA) ( n = 3 mice). All data show mean +- SEM
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
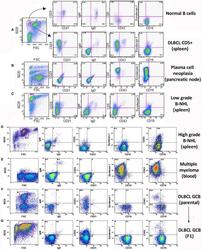
- Experimental details
- Figure 2 Analysis of the B lymphocyte populations expanded in TRAF3/BCL2 double-tg mice with lymphoid dyscrasias. Three-color flow-cytometry analysis was performed to determine the phenotype of expanded B lymphocyte populations. Gating of the expanded population was based on the CD45R/B220 and FSC plot of each sample analyzed and is indicated in the figure. The surface molecules analyzed are indicated in the plots, as well as the percentage of cells found in each quadrant. The quadrants settings were selected based on the staining of isotype-controls (not shown). The tissue source where the analyzed lymphocytes were extracted from and the type of B cell malignancy developed by the TRAF3/BCL2 double-tg mice, according to the flow-cytometry and immunohistochemical analysis, is indicated in the figure.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
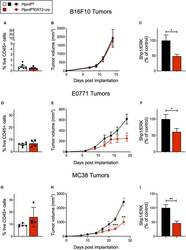
- Experimental details
- Figure 4 Ptpn6 deletion drives robust anti-tumor immunity in two immune-rich syngeneic tumor lines. (A) Flow cytometric analysis of live CD45 + cells from B16F10 melanoma tumors isolated from tamoxifen-treated Ptpn6 fl/fl and Ptpn6 fl/fl ERT2-cre mice at day 14 post tumor implantation. (B) B16F10 tumor volume measurements in tamoxifen-treated Ptpn6 fl/fl ERT2-cre and Ptpn6 fl/fl mice. Each data point represents the average tumor volume of all mice in a given group. Data is representative of three independent experiments with 5-7 mice/group. (C) Shp1 protein relative to total Erk2 protein in peripheral blood cells from indicated mice 14 days after initial tamoxifen dose (200 mg/kg bid for 4 days) and 7 days after B16F10 tumor cells were implanted. Data is representative of at least three independent experiments with 5-7 mice per group. (D) Flow cytometric analysis of live CD45 + cells from E0771 tumors isolated from tamoxifen-treated Ptpn6 fl/fl and Ptpn6 fl/fl ERT2-cre mice at day 19 post tumor implantation. (E) E0771 tumor volume measurements in tamoxifen-treated Ptpn6 fl/fl ERT2-cre and Ptpn6 fl/fl mice. Each data point represents the average tumor volume of all mice in a given group. Data is representative of three independent experiments with 4-5 mice per group. Statistical significance was calculated at each time point using an unpaired t -test. (F) Shp1 protein relative to total Erk2 protein in peripheral blood cells from indicated mice 14 days after initial tamoxifen d
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
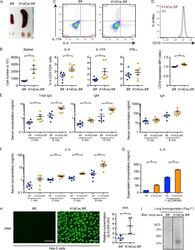
- Experimental details
- Figure 5. K14Cre;fl/fl mice exhibit prominent autoimmunity with Th2/Th17 cell polarization and abnormal B cell activation leading to autoantibody production. (A) A representative image of the gross appearance of spleens and inguinal lymph nodes from 3-mo-old fl/fl and K14Cre;fl/fl mice. (B) The number of splenocytes in 3-mo-old fl/fl and K14Cre;fl/fl mice was counted. n = 4 per each group. (C) Lymphocytes isolated from inguinal lymph nodes were stained for CD3 and CD4 and intracellularly stained for IL-4, IL-17A, and IFN-gamma. (Top) Representative two-dimensional plots for IL-4 and IL-17A in CD3 + CD4 + cells. (Bottom) The percentages of cells positive for IL-4, IL-17A, and IFN-gamma are summarized. n = 7 mice per each group. (D) Splenocytes from 3-mo-old fl/fl and K14Cre;fl/fl mice were stained for B220 and CD19. B220 + CD19 + cells were analyzed for CD19 expression. A representative histogram of mean fluorescence intensity (MFI; top) and the comparison of mean fluorescence intensities (bottom) are shown. n = 4 mice per genotype. (E) Sera from 3- and 8-mo-old mice were analyzed for the concentrations of total IgG, IgM, and IgA. n = 5-7 mice per genotype. Note that the results are shown with log scale. (F) Sera from 1-, 2-, 3-, and 8-mo-old mice were analyzed for the concentration of IL-6. n = 6-7 mice per genotype. (G) B cells were isolated from splenocytes of 3-mo-old mice and cultured for 48 h, either unstimulated or stimulated with anti-CD40 antibody (alphaCD40Ab). The c
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
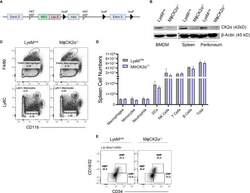
- Experimental details
- Figure 1 Development of myeloid cell populations is not detectably altered in MphCK2alpha -/- mice. (A) Schematic of Csnk2a targeting vector. (B) Immunoblots of lysates from BMDMs, splenocytes, or peritoneal cavity cells of LysM cre or LysM cre CK2alpha fl/fl (MphCK2alpha - / - ) mice were probed for CK2alpha or beta-actin as a loading control. n = 2. (C) Splenic myeloid cell populations were stained and analyzed by flow cytometry. Plots depict gating for representative F4/80+ macrophages, Ly6C+ monocytes, and neutrophils (also Ly6G+). (D) Cells from spleen were stained for flow cytometry to enumerate immune cell populations. Data are from pooled experiments, n = 4-12 mice/group. (E) Cells from bone marrow were stained for flow cytometry to evaluate bone marrow precursor populations including common myeloid progenitors (CMPs), granulocyte-monocyte progenitors (GMPs), and megakaryocyte-erythrocyte progenitor (MEP). Cells were pre-gated on Lin-(B220, CD3, CD11b, GR-1 and TER-119), Sca-1-, cKit-. Density plots are representative from two experiments.
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry