Antibody data
- Antibody Data
- Antigen structure
- References [12]
- Comments [0]
- Validations
- Immunocytochemistry [1]
- Other assay [12]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 18-7430 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- ZO-1 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 500 µL
- Storage
- 4° C
Submitted references Methodological study of directed differentiation of pluripotent stem cells into corneal endothelial cells.
Transplantation of GMP-grade human iPSC-derived retinal pigment epithelial cells in rodent model: the first pre-clinical study for safety and efficacy in China.
A C-terminal peptide from type I interferon protects the retina in a mouse model of autoimmune uveitis.
Development of a Scalable Coculture System for Gut Anaerobes and Human Colon Epithelium.
Inhibition of TGF-β1 Signaling by IL-15: A Novel Role for IL-15 in the Control of Renal Epithelial-Mesenchymal Transition: IL-15 Counteracts TGF-β1-Induced EMT in Renal Fibrosis.
Elovanoids are novel cell-specific lipid mediators necessary for neuroprotective signaling for photoreceptor cell integrity.
Directing Differentiation of Pluripotent Stem Cells Toward Retinal Pigment Epithelium Lineage.
Targeting the cAMP and Transforming Growth Factor-β Pathway Increases Proliferation to Promote Re-Epithelialization of Human Stem Cell-Derived Retinal Pigment Epithelium.
A FOXM1 Dependent Mesenchymal-Epithelial Transition in Retinal Pigment Epithelium Cells.
Lung epithelial branching program antagonizes alveolar differentiation.
Integrin Beta 1 suppresses multilayering of a simple epithelium.
Differential expression of claudin tight junction proteins in the human cortical nephron.
Jia L, Diao Y, Fang Y, Yang K, Wang L, Huang Y
Annals of translational medicine 2022 Apr;10(8):482
Annals of translational medicine 2022 Apr;10(8):482
Transplantation of GMP-grade human iPSC-derived retinal pigment epithelial cells in rodent model: the first pre-clinical study for safety and efficacy in China.
Zhang H, Su B, Jiao L, Xu ZH, Zhang CJ, Nie J, Gao ML, Zhang YV, Jin ZB
Annals of translational medicine 2021 Feb;9(3):245
Annals of translational medicine 2021 Feb;9(3):245
A C-terminal peptide from type I interferon protects the retina in a mouse model of autoimmune uveitis.
Ahmed CM, Ildefonso CJ, Johnson HM, Lewin AS
PloS one 2020;15(2):e0227524
PloS one 2020;15(2):e0227524
Development of a Scalable Coculture System for Gut Anaerobes and Human Colon Epithelium.
Sasaki N, Miyamoto K, Maslowski KM, Ohno H, Kanai T, Sato T
Gastroenterology 2020 Jul;159(1):388-390.e5
Gastroenterology 2020 Jul;159(1):388-390.e5
Inhibition of TGF-β1 Signaling by IL-15: A Novel Role for IL-15 in the Control of Renal Epithelial-Mesenchymal Transition: IL-15 Counteracts TGF-β1-Induced EMT in Renal Fibrosis.
Devocelle A, Lecru L, François H, Desterke C, Gallerne C, Eid P, Estelle O, Azzarone B, Giron-Michel J
International journal of cell biology 2019;2019:9151394
International journal of cell biology 2019;2019:9151394
Elovanoids are novel cell-specific lipid mediators necessary for neuroprotective signaling for photoreceptor cell integrity.
Jun B, Mukherjee PK, Asatryan A, Kautzmann MA, Heap J, Gordon WC, Bhattacharjee S, Yang R, Petasis NA, Bazan NG
Scientific reports 2017 Jul 13;7(1):5279
Scientific reports 2017 Jul 13;7(1):5279
Directing Differentiation of Pluripotent Stem Cells Toward Retinal Pigment Epithelium Lineage.
Choudhary P, Booth H, Gutteridge A, Surmacz B, Louca I, Steer J, Kerby J, Whiting PJ
Stem cells translational medicine 2017 Feb;6(2):490-501
Stem cells translational medicine 2017 Feb;6(2):490-501
Targeting the cAMP and Transforming Growth Factor-β Pathway Increases Proliferation to Promote Re-Epithelialization of Human Stem Cell-Derived Retinal Pigment Epithelium.
Choudhary P, Gutteridge A, Impey E, Storer RI, Owen RM, Whiting PJ, Bictash M, Benn CL
Stem cells translational medicine 2016 Jul;5(7):925-37
Stem cells translational medicine 2016 Jul;5(7):925-37
A FOXM1 Dependent Mesenchymal-Epithelial Transition in Retinal Pigment Epithelium Cells.
Choudhary P, Dodsworth BT, Sidders B, Gutteridge A, Michaelides C, Duckworth JK, Whiting PJ, Benn CL
PloS one 2015;10(6):e0130379
PloS one 2015;10(6):e0130379
Lung epithelial branching program antagonizes alveolar differentiation.
Chang DR, Martinez Alanis D, Miller RK, Ji H, Akiyama H, McCrea PD, Chen J
Proceedings of the National Academy of Sciences of the United States of America 2013 Nov 5;110(45):18042-51
Proceedings of the National Academy of Sciences of the United States of America 2013 Nov 5;110(45):18042-51
Integrin Beta 1 suppresses multilayering of a simple epithelium.
Chen J, Krasnow MA
PloS one 2012;7(12):e52886
PloS one 2012;7(12):e52886
Differential expression of claudin tight junction proteins in the human cortical nephron.
Kirk A, Campbell S, Bass P, Mason J, Collins J
Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association 2010 Jul;25(7):2107-19
Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association 2010 Jul;25(7):2107-19
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
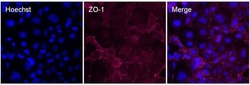
- Experimental details
- Immunofluorescent analysis of ZO-1 (red) in Caco-2 cells. The cells were blocked with 3% BSA in PBS (Product # 37525) for 15 minutes at room temperature, stained with a ZO-1 Rabbit polyclonal antibody (Product # 18-7430) at a concentration of 1:500 in blocking buffer for at least 1 hour at room temperature, and then incubated with a Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 647 conjugate (Product # A27040) at a dilution of 1:1000 for 30 minutes at room temperature (red). Nuclei (blue) were stained with Hoechst 33342 dye (Product # 62249). Images were taken on a Thermo Scientific ToxInsight Instrument at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Primary human RPE cells. ( a ) Confocal images of immunostained primary human RPE cells using specific markers ZO-1 (Zona occludens-1), RPE65 (retinal pigment epithelium-specific 65 kDa protein), MITF (Microphtalmia-associated Transcription Factor), and beta-catenin. ( b ) Light microscopy depicting primary human RPE cell morphology at different passages in culture. Scale bars, 50 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
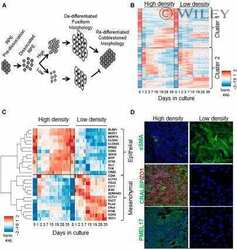
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Itgb1 CKO/- ; Shh Cre/+ mutant lung epithelial cells reorient mitotic spindles and show partial loss of apical-basal polarity. (A) Confocal sections of whole-mount E11 lungs immunostained for markers of mitosis (phospho-Histone 3, pH3), mitotic spindles (acetylated Tubulin, AcTUB) and the epithelium (E-Cadherin, ECAD). Representative images are shown for mitotic cells with no obvious mitotic spindle (prophase) and with the mitotic spindle orientated between 0 to 45 degrees (0deg~45deg) or 45 to 90 degrees (45deg~90deg) relative to the lumenal surface of the epithelium. Mitotic cells in the control lung are exclusively localized within one nuclear diameter of the lumenal surface of the epithelium. The Itgb1 CKO/- ; Shh Cre/+ mutant lung contains mitotic cells localized away from the lumenal surface and sometimes on the basal surface of the epithelium. Grey scale images of the ECAD and AcTUB staining are shown below for the mitotic cells of interest. Note the intense ECAD puncta associated with the basally located Itgb1 mutant nuclei. The upper and lower dashed lines demarcate the lumenal (Lumen) and basal side of the epithelium, respectively. Scale bar, 10 um. (B) Quantification of the percentage of cells in each category in ( A ) out of 110 (control) and 127 (mutant) mitotic cells from 3 E11 lungs. The filled columns and the associated numbers indicate the percentage of mitotic cells that are located more than one nuclear diameter from the lumenal surface of the epit
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 7 Localization of claudin-14: photomicrographs showing serial sections of human renal cortical tissue stained for calbindin ( A ), aquaporin-2 ( B ), claudin-14 ( C ), THP ( D ), L. tetragonolobus ( E ) and ZO-1 ( F ) [claudin-14 stained tubules that were coincident with similar ones that were positive for calbindin (asterisks in A and C) and negative for aquaporin-2, THP and L. tetragonolobus (asterisks in B, D and E), confirming the expression of claudin-14 in the distal tubule; basolateral claudin-14 staining corresponded with tubules showing discrete junctional ZO-1 staining (asterisks in C and F); scale bar 100 um].
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 8 High-magnification images comparing claudin localization with ZO-1 and occludin: photomicrographs showing serial sections of human renal cortical tissue stained for claudin-14 ( A ) and ZO-1 ( B ) with a second sequence of serial sections showing staining of claudin-3 ( C ), -4 ( D ) and -7 ( E ) with occludin ( F ) [claudin-14 was seen in basolateral locations in tubules corresponding to those with discrete tight junction staining for ZO-1 (A and B, square symbol marks corresponding tubule); in serial sections from the same tissue sample, claudin-4 was observed to be mostly in discrete tight junction staining patterns (D), whereas claudin-3 and -7 showed strong basolateral cell membrane staining in addition to tight junction staining (C and E, asterisks mark corresponding tubule); in corresponding tubules, occludin was stained in a discrete pattern consistent with tight junction localization (F); scale bar 50 um].
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 9 Localization of claudin-1: photomicrographs showing serial sections of human renal cortical tissue stained for ZO-1 ( A ), calbindin ( B ), aquaporin-2 ( C ), claudin-1 ( D ), THP ( E ) and L. tetragonolobus ( F ) [claudin-1-, calbindin- and aquaporin-2-stained coincident tubules that were negative for THP and L. tetragonolobus (arrowheads in B-F); there were some calbindin positive tubules that were negative for claudin-1, THP and aquaporin-2 (asterisks in B-E); claudin-1 basolateral membrane location, corresponding with tubules showing discrete junctional ZO-1 staining (arrowheads in A and D); robust ZO-1 staining was seen in glomerular epithelial cells (A); scale bar 100 um].
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 1 A Mesenchymal-Epithelial Transition with temporal FOXM1 expression during RPE culture. A. Immunocytochemistry was performed for CRALBP, ZO1, alphaSMA and Ki67 at Day 0 (D0), Day 3 (D3) and Day 35 (D35). Arrowheads point towards Ki67 positive nuclei. B. Microarray heatmap of the expression profiles of the top 250 genes, ranked by the significance of their expression changes, over time in culture. Raw expression data are mean centred and scaled to unit variance prior to clustering. A schematic of the scaled expression is shown on the right where individual gene profiles are in light grey and the mean expression profile is shown in black. C. Microarray heatmap showing transcript expression for a panel of representative markers over a timecourse of RPE culture. D. Immunocytochemistry for FOXM1 at Day 2 and Day 14 of RPE culture. Arrowheads point towards FOXM1 positive nuclei. E. Quantification of immunocytochemistry showing percentage of nuclei staining positive for FOXM1 over time. Bars represent Mean +- SD (n = 3). F. Expression of FOXM1 transcript measured using qPCR (relative to housekeeping genes ACTB and GAPDH ) in iPSC derived RPE, human foetal RPE and ARPE19 cells over time. Bars represent Mean +- SD (n = 3)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
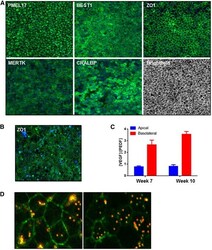
- Experimental details
- Figure 4 Stage 3: Generation of a homogenous and functional retinal pigment epithelium (RPE) population. (A): Two days' postseeding, SHEF1 human embryonic stem cells were treated with Induction Medium 1 for 4 days, followed by Induction Medium 2 for 3 days. At day 9, cells were replated in Induction Medium 3 for a period of 19 days. At day 28, cells were replated in Dulbecco's modified Eagle's medium KSR-XF and cultured for a period of 14 days. Representative images showing immunocytochemistry for indicated RPE markers are shown. Images are captured at x10 magnification. (B): Cells cultured as in panel A were further dissociated and replated on transwells and cultured for a period of 10 weeks. A representative image showing en face immunocytochemistry for ZO-1 is shown. Images are captured at x20 magnification. (C): Spent medium from transwells described in panel B was collected from the top and bottom chambers and quantified for vascular endothelial growth factor (VEGF) and pigment epithelium-derived factor (PEDF) concentration. The ratio of [VEGF]:[PEDF] was quantified in media from the two compartments ( n = 3, +-SD). (D): Representative confocal images showing phagocytosis of fluorescent bead (red) by RPE. ZO-1 immunocytochemistry (green) shows the cell edge, and the presence of bead within the cell boundary indicates internalization by phagocytosis. Images are captured at x63 magnification. Abbreviations: PEDF, pigment epithelium-derived factor; VEGF, vasc
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Primary human RPE cells. ( a ) Confocal images of immunostained primary human RPE cells using specific markers ZO-1 (Zona occludens-1), RPE65 (retinal pigment epithelium-specific 65 kDa protein), MITF (Microphtalmia-associated Transcription Factor), and beta-catenin. ( b ) Light microscopy depicting primary human RPE cell morphology at different passages in culture. Scale bars, 50 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
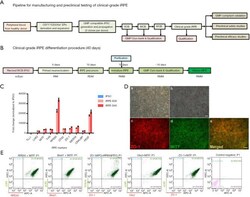
- Experimental details
- Maturation and characterization of human iRPE cells. (A) Scheme of GMP-compatible processes to manufacture and test clinical-grade iPSCs and iRPE cells for preclinical studies. (B) Timeline of manufacturing clinical-grade iRPE cells from iPSC WCB. A GMP-compatible immature iRPE master cell bank was established ~26 days from the start of monolayer iPSC differentiation, and all reagents used were xeno-free and chemically defined. (C) Gene expression analysis of known RPE-specific genes of iRPE cells collected on days 35 and 45 of differentiation. (D) Top panel: Bright field images of the iRPEs in monolayer on days 20 and 40 of differentiation. Bottom panel: Immunostaining of the mature iRPE (day 40) markers ZO-1 (red) and MiTF (green). Scale bars, 100 um. (E) Flow cytometric analysis of mature iRPE cells (day 40) costained for MiTF/RPE65, MiTF/Best1, RPE65/ZO-1, MiTF/Otx2, and MiTF/ZO-1. WCB, working cell bank; RIM, RPE induction medium; RDM, RPE differentiation medium; RMM, RPE maturation medium.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Deformation of hiPSC-induced differentiated NCCs and identification of specific indicators of NCCs. (A) Picture of cells before (D8) and after (D11, D14) differentiation were observed by light microscope. (B) The expression of ZO-1 in induced differentiated cells on day 14, shown by immunofluorescence staining. (C) The expression of corneal endothelial cell-specific indicators ( COL4A1, COL8A1 , COL8A2, ZO-1 , and RPE65 ) in hiPSCs and CECs identified by qRT-PCR. The Y-axis represents normalization to relative fold. ZO-1 , zonula occludens-1; DAPI, 4',6-diamidino-2-phenylindole; COL4A1 , collagen type IV alpha 1; RPE, retinal pigment epithelium; hiPSC, human induced pluripotent stem cell; CEC, corneal endothelial cell; NCC, neural crest cell; qRT-PCR, quantitative reverse transcription polymerase chain reaction.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 rhTGF-beta1-induced EMT in RPTEC and HK-2 cells is inhibited by in vitro rhIL-15 treatment . (a) Analysis of E-cadherin and N-cadherin expressions in 48h-treated HK-2 cells by Western blotting using increasing concentrations of rhIL-15 (0.1-10 ng/mL) +- 3 ng/mL of rhTGF- beta 1. ( * p
 Explore
Explore Validate
Validate Learn
Learn Immunocytochemistry
Immunocytochemistry Immunohistochemistry
Immunohistochemistry