Antibody data
- Antibody Data
- Antigen structure
- References [22]
- Comments [0]
- Validations
- Immunocytochemistry [2]
- Other assay [10]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 339194 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- ZO-1 Monoclonal Antibody (ZO1-1A12), Alexa Fluor™ 594
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Reactivity
- Human, Canine
- Host
- Mouse
- Conjugate
- Red dye
- Isotype
- IgG
- Antibody clone number
- ZO1-1A12
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- 4° C, store in dark
Submitted references A Novel Method of Mouse RPE Explant Culture and Effective Introduction of Transgenes Using Adenoviral Transduction for In Vitro Studies in AMD.
An integrative proteomics method identifies a regulator of translation during stem cell maintenance and differentiation.
Human colorectal cancer-on-chip model to study the microenvironmental influence on early metastatic spread.
Disparate temperature-dependent virus-host dynamics for SARS-CoV-2 and SARS-CoV in the human respiratory epithelium.
The Na+, K+-ATPase β1 subunit regulates epithelial tight junctions via MRCKα.
High-purity production and precise editing of DNA base editing ribonucleoproteins.
A human-airway-on-a-chip for the rapid identification of candidate antiviral therapeutics and prophylactics.
Anti-Inflammatory and Anti-Oxidative Synergistic Effect of Vitamin D and Nutritional Complex on Retinal Pigment Epithelial and Endothelial Cell Lines against Age-Related Macular Degeneration.
Caco-2 Cells for Measuring Intestinal Cholesterol Transport - Possibilities and Limitations.
Vitamin D Protects against Oxidative Stress and Inflammation in Human Retinal Cells.
3D brain angiogenesis model to reconstitute functional human blood-brain barrier in vitro.
In Vitro High-Content Imaging-Based Phenotypic Analysis of Bronchial 3D Organotypic Air-Liquid Interface Cultures.
A role for mast cells in geographic atrophy.
Optimization of Normal Human Bronchial Epithelial (NHBE) Cell 3D Cultures for in vitro Lung Model Studies.
Evidence for a regulated Ca(2+) entry in proximal tubular cells and its implication in calcium stone formation.
Repeat-Associated Non-ATG (RAN) Translation in Fuchs' Endothelial Corneal Dystrophy.
Lymphatic deletion of calcitonin receptor-like receptor exacerbates intestinal inflammation.
Paracellular epithelial sodium transport maximizes energy efficiency in the kidney.
Restoration of mesenchymal retinal pigmented epithelial cells by TGFβ pathway inhibitors: implications for age-related macular degeneration.
Real-time estimation of paracellular permeability of cerebral endothelial cells by capacitance sensor array.
PARD3 Inactivation in Lung Squamous Cell Carcinomas Impairs STAT3 and Promotes Malignant Invasion.
Hox proteins drive cell segregation and non-autonomous apical remodelling during hindbrain segmentation.
Shang P, Stepicheva NA, Liu H, Chowdhury O, Franks J, Sun M, Hose S, Ghosh S, Yazdankhah M, Strizhakova A, Stolz DB, Zigler JS Jr, Sinha D
International journal of molecular sciences 2021 Nov 5;22(21)
International journal of molecular sciences 2021 Nov 5;22(21)
An integrative proteomics method identifies a regulator of translation during stem cell maintenance and differentiation.
Sabatier P, Beusch CM, Saei AA, Aoun M, Moruzzi N, Coelho A, Leijten N, Nordenskjöld M, Micke P, Maltseva D, Tonevitsky AG, Millischer V, Carlos Villaescusa J, Kadekar S, Gaetani M, Altynbekova K, Kel A, Berggren PO, Simonson O, Grinnemo KH, Holmdahl R, Rodin S, Zubarev RA
Nature communications 2021 Nov 12;12(1):6558
Nature communications 2021 Nov 12;12(1):6558
Human colorectal cancer-on-chip model to study the microenvironmental influence on early metastatic spread.
Strelez C, Chilakala S, Ghaffarian K, Lau R, Spiller E, Ung N, Hixon D, Yoon AY, Sun RX, Lenz HJ, Katz JE, Mumenthaler SM
iScience 2021 May 21;24(5):102509
iScience 2021 May 21;24(5):102509
Disparate temperature-dependent virus-host dynamics for SARS-CoV-2 and SARS-CoV in the human respiratory epithelium.
V'kovski P, Gultom M, Kelly JN, Steiner S, Russeil J, Mangeat B, Cora E, Pezoldt J, Holwerda M, Kratzel A, Laloli L, Wider M, Portmann J, Tran T, Ebert N, Stalder H, Hartmann R, Gardeux V, Alpern D, Deplancke B, Thiel V, Dijkman R
PLoS biology 2021 Mar;19(3):e3001158
PLoS biology 2021 Mar;19(3):e3001158
The Na+, K+-ATPase β1 subunit regulates epithelial tight junctions via MRCKα.
Bai H, Zhou R, Barravecchia M, Norman R, Friedman A, Yu D, Lin X, Young JL, Dean DA
JCI insight 2021 Feb 22;6(4)
JCI insight 2021 Feb 22;6(4)
High-purity production and precise editing of DNA base editing ribonucleoproteins.
Jang HK, Jo DH, Lee SN, Cho CS, Jeong YK, Jung Y, Yu J, Kim JH, Woo JS, Bae S
Science advances 2021 Aug;7(35)
Science advances 2021 Aug;7(35)
A human-airway-on-a-chip for the rapid identification of candidate antiviral therapeutics and prophylactics.
Si L, Bai H, Rodas M, Cao W, Oh CY, Jiang A, Moller R, Hoagland D, Oishi K, Horiuchi S, Uhl S, Blanco-Melo D, Albrecht RA, Liu WC, Jordan T, Nilsson-Payant BE, Golynker I, Frere J, Logue J, Haupt R, McGrath M, Weston S, Zhang T, Plebani R, Soong M, Nurani A, Kim SM, Zhu DY, Benam KH, Goyal G, Gilpin SE, Prantil-Baun R, Gygi SP, Powers RK, Carlson KE, Frieman M, tenOever BR, Ingber DE
Nature biomedical engineering 2021 Aug;5(8):815-829
Nature biomedical engineering 2021 Aug;5(8):815-829
Anti-Inflammatory and Anti-Oxidative Synergistic Effect of Vitamin D and Nutritional Complex on Retinal Pigment Epithelial and Endothelial Cell Lines against Age-Related Macular Degeneration.
Hernandez M, Recalde S, González-Zamora J, Bilbao-Malavé V, Sáenz de Viteri M, Bezunartea J, Moreno-Orduña M, Belza I, Barrio-Barrio J, Fernandez-Robredo P, García-Layana A
Nutrients 2021 Apr 23;13(5)
Nutrients 2021 Apr 23;13(5)
Caco-2 Cells for Measuring Intestinal Cholesterol Transport - Possibilities and Limitations.
Hiebl V, Schachner D, Ladurner A, Heiss EH, Stangl H, Dirsch VM
Biological procedures online 2020;22:7
Biological procedures online 2020;22:7
Vitamin D Protects against Oxidative Stress and Inflammation in Human Retinal Cells.
Fernandez-Robredo P, González-Zamora J, Recalde S, Bilbao-Malavé V, Bezunartea J, Hernandez M, Garcia-Layana A
Antioxidants (Basel, Switzerland) 2020 Sep 8;9(9)
Antioxidants (Basel, Switzerland) 2020 Sep 8;9(9)
3D brain angiogenesis model to reconstitute functional human blood-brain barrier in vitro.
Lee S, Chung M, Lee SR, Jeon NL
Biotechnology and bioengineering 2020 Mar;117(3):748-762
Biotechnology and bioengineering 2020 Mar;117(3):748-762
In Vitro High-Content Imaging-Based Phenotypic Analysis of Bronchial 3D Organotypic Air-Liquid Interface Cultures.
Marescotti D, Bovard D, Morelli M, Sandoz A, Luettich K, Frentzel S, Peitsch M, Hoeng J
SLAS technology 2020 Jun;25(3):247-252
SLAS technology 2020 Jun;25(3):247-252
A role for mast cells in geographic atrophy.
Ogura S, Baldeosingh R, Bhutto IA, Kambhampati SP, Scott McLeod D, Edwards MM, Rais R, Schubert W, Lutty GA
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2020 Aug;34(8):10117-10131
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2020 Aug;34(8):10117-10131
Optimization of Normal Human Bronchial Epithelial (NHBE) Cell 3D Cultures for in vitro Lung Model Studies.
Rayner RE, Makena P, Prasad GL, Cormet-Boyaka E
Scientific reports 2019 Jan 24;9(1):500
Scientific reports 2019 Jan 24;9(1):500
Evidence for a regulated Ca(2+) entry in proximal tubular cells and its implication in calcium stone formation.
Ibeh CL, Yiu AJ, Kanaras YL, Paal E, Birnbaumer L, Jose PA, Bandyopadhyay BC
Journal of cell science 2019 Apr 30;132(9)
Journal of cell science 2019 Apr 30;132(9)
Repeat-Associated Non-ATG (RAN) Translation in Fuchs' Endothelial Corneal Dystrophy.
Soragni E, Petrosyan L, Rinkoski TA, Wieben ED, Baratz KH, Fautsch MP, Gottesfeld JM
Investigative ophthalmology & visual science 2018 Apr 1;59(5):1888-1896
Investigative ophthalmology & visual science 2018 Apr 1;59(5):1888-1896
Lymphatic deletion of calcitonin receptor-like receptor exacerbates intestinal inflammation.
Davis RB, Kechele DO, Blakeney ES, Pawlak JB, Caron KM
JCI insight 2017 Mar 23;2(6):e92465
JCI insight 2017 Mar 23;2(6):e92465
Paracellular epithelial sodium transport maximizes energy efficiency in the kidney.
Pei L, Solis G, Nguyen MT, Kamat N, Magenheimer L, Zhuo M, Li J, Curry J, McDonough AA, Fields TA, Welch WJ, Yu AS
The Journal of clinical investigation 2016 Jul 1;126(7):2509-18
The Journal of clinical investigation 2016 Jul 1;126(7):2509-18
Restoration of mesenchymal retinal pigmented epithelial cells by TGFβ pathway inhibitors: implications for age-related macular degeneration.
Radeke MJ, Radeke CM, Shih YH, Hu J, Bok D, Johnson LV, Coffey PJ
Genome medicine 2015;7(1):58
Genome medicine 2015;7(1):58
Real-time estimation of paracellular permeability of cerebral endothelial cells by capacitance sensor array.
Hyun Jo D, Lee R, Hyoung Kim J, Oh Jun H, Geol Lee T, Hun Kim J
Scientific reports 2015 Jun 5;5:11014
Scientific reports 2015 Jun 5;5:11014
PARD3 Inactivation in Lung Squamous Cell Carcinomas Impairs STAT3 and Promotes Malignant Invasion.
Bonastre E, Verdura S, Zondervan I, Facchinetti F, Lantuejoul S, Chiara MD, Rodrigo JP, Carretero J, Condom E, Vidal A, Sidransky D, Villanueva A, Roz L, Brambilla E, Savola S, Sanchez-Cespedes M
Cancer research 2015 Apr 1;75(7):1287-97
Cancer research 2015 Apr 1;75(7):1287-97
Hox proteins drive cell segregation and non-autonomous apical remodelling during hindbrain segmentation.
Prin F, Serpente P, Itasaki N, Gould AP
Development (Cambridge, England) 2014 Apr;141(7):1492-502
Development (Cambridge, England) 2014 Apr;141(7):1492-502
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
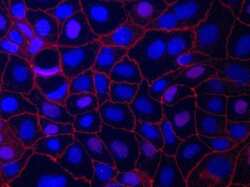
- Experimental details
- Immunofluorescent staining of ZO-1 in Human Caco-2 cells using mouse anti ZO-1 monoclonal antibody - Alexa Fluor® 594 (Product # 339194). DNA is counter stained with blue Hoechst 33258 (Product # H3569).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
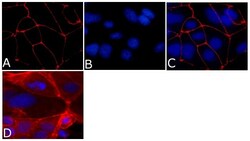
- Experimental details
- Immunofluorescence analysis of ZO-1/TJP1 Antibody, Alexa Fluor® 594 conjugate (ZO1-1A12) was done on 90% confluent log phase CaCo2 cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton™ X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with ZO-1/TJP1 Antibody, Alexa Fluor® 594 conjugate (ZO1-1A12) (Product # 339194) at 1µg/mL in 1% BSA and incubated for 3 hours at room temperature (Panel a: red). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). Panel c: red). Panel c is a merged image showing cell junctional localization. Panel d is a no primary antibody control. The images were captured at 40X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
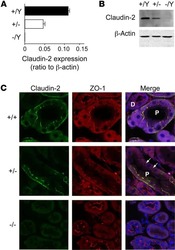
- Fig. 6. Hox4 proteins are necessary and sufficient to induce non-autonomous apical cell enlargement. (A-H) Confocal immunostaining for Hoxb4 (green) and ZO-1 (red) on E10.5 flat-mounted mouse hindbrains of wild-type ( Hoxb4 +/+ ; Hoxd4 +/+ ) (A,B), Hoxb4 +/+ ; Hoxd4 -/- (D), Hoxb4 +/- ; Hoxd4 -/- (F) and Hox4 double-mutant (H,I) embryos at a lateral level of the r6/r7 boundary region. The r6/r7 boundary or its presumptive position (x) are indicated in A and H. (B,I) Confocal z-projections of similar magnifications of the r6/r7 boundary region as in D (corresponding to dotted boxes in A and H) showing the apical cell outlines (yellow) used for quantitation. (C,E,G,J) Apical areas (mum 2 ) with respect to the anteroposterior position of the cells in B,D,F,I, respectively. Hoxb4 + (green) and Hoxb4 - (black) cells are indicated. (K,L) Confocal z -projections and ZO-1 apical outlines (yellow) of the chick r5 neuroepithelium electroporated with mouse Hoxb4-mGFP . (K) Mouse Hoxb4/GFP + cells (green) form a cluster with a well-defined interface (dotted line). (L) Corresponding apical cell areas (mum 2 ) with respect to anteroposterior position for mouse Hoxb4 + (green) and mouse Hoxb4 - (black) cells. Anterior is towards the left.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
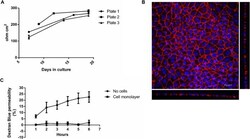
- Fig. 1 a-c Quality control steps for the cultivation of Caco-2 cells on filter inserts. Increase in TEER over the cultivation period ( a ). Caco-2 cells were seeded onto filter inserts applying Protocol #2 and TEER was measured as detailed in Supplementary Protocol #3 (Additional file 1 ). The data points represent mean +- SD from three inserts from the respective plate. Representative image of a Caco-2 cell monolayer grown on a filter insert for 21 days ( b ). Depicted is a maximum projection of the top view and the optical cross sections obtained by CLSM. Cell nuclei are shown in blue (DAPI) and the tight junction protein ZO-1 in red (Alexa Fluor 594 conjugated anti ZO-1 antibody). Staining was carried out as described in Protocol #6. Dextran Blue permeability of a filter insert without cells and a filter insert with a fully differentiated cell monolayer ( c ). Filter inserts were incubated with Dextran Blue as described in Supplementary Protocol #4 (Additional file 1 ) over a period of 6 h and sampling every hour. The data points represent mean +- SD from three to four independent experiments
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
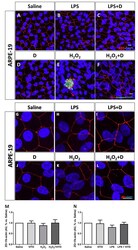
- Figure 4 Integrity of ARPE-19 cells evaluated by zonula occludens-1 (ZO-1) (red) and caspase-3 (green) immunofluorescence. VITD (1 nM, 1 h; ( D )) did not affect ZO-1 structure compared to saline ( A , G ). Lipopolysaccharide (LPS) ( B , H ) and H 2 O 2 ( E , K ) addition damaged tight junctions and concomitant incubation with VITD (1 nM, 1 h; ( C , I ) and ( F , L )) restored the altered structure. ( G-L ) show the apical junction in higher magnification. Caspase-3 was highly observed in the H 2 O 2 group ( E ) compared to saline ( A ) and VITD ( D ). VITD addition showed restoration, and caspase-3 activation was absent ( F ). Nuclei were labeled with DAPI (blue). Scale bar: 20 um. Densitometry of ZO-1 expression in ARPE-19 cells under oxidative stress ( M ) and inflammatory ( N ) conditions. Although a tendency to reduce the ZO-1 expression was observed, no statistical differences were found. VITD restored values similar to saline group. n = 3.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
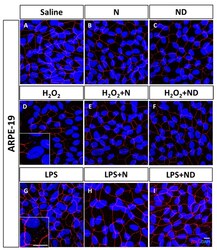
- Figure 1 Junctional integrity of ARPE-19 cells evaluated by ZO-1 (red) fluorescence under a confocal imaging system. Application of N and ND treatments (62.34 mug/mL each) did not affect tight junctions, cell integrity, and structure ( B , C ) compared to saline ( A ). Addition of H 2 O 2 (2h; 1600 muM) and LPS (24 h; 20 mug/mL) to induce oxidative stress and inflammatory-like conditions, respectively, damaged tight junctions ( D , G ), while incubation with N and ND treatments during the last hour of the induction recovered the altered structure ( E , F , H , I ). Nuclei were labelled with DAPI (blue). Scale bar: 20 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
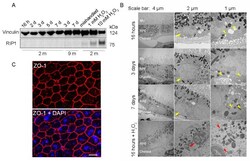
- Figure 2 RPE flatmounts are viable for at least one week after culture in vitro . ( A ) RPE flatmounts were prepared from 2- and 9-month-old wild-type C57BL/6J mice and cultured in complete medium with or without H 2 O 2 for up to one week. Protein lysates were prepared from the RPE flatmounts and subjected to Western blotting analysis. The cell death marker RIP1 was used to evaluate cell viability; vinculin was used as an internal control. As a positive control: RPE flatmounts treated with either 1 mM or 10 mM H 2 O 2 had significant expression of RIP1. Further, the RPE flatmount that was cultured for 7 days without changing the medium (mishandled sample) showed a slight amount of RIP1, whereas the RPE flatmounts cultured with frequent medium changes did not express RIP1, suggesting that the RPE cells obtained even from older mice (9 months) were viable after at least 7 days of culture. ( B ) RPE flatmounts were obtained from 3-month-old wild-type C57BL/6J mice and cultured in complete medium with or without H 2 O 2 treatment for up to one week. RPE flatmounts were then fixed in 2.5% glutaraldehyde and processed for transmission electron microscopy (TEM). The TEM imaging of the RPE flatmounts cultured for 16 h, 3 days, and 7 days showed that these RPE cells have normal nuclei (yellow arrows) and organelles, such as mitochondria (yellow arrowheads), and abundant microvilli. H 2 O 2 -treated RPE flatmounts showed cell death symptoms, including chromatin condensation and fragme
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
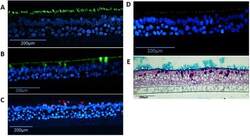
- Figure 6 Detection of differentiated cells in NHBE pseudostratified epithelial layer generated from passage 1 cultures. Immunofluorescence for ( A ) Ciliated cells (alpha-tubulin, green), ( B ) goblet cells (MUC5B, green), ( C ) goblet cells (MUC5AC, red), ( D ) ZO-1 tight junctions (red), nuclear stain (DAPI - blue); and staining for ( E ) periodic acid-Schiff/alcian blue for mucus producing goblet cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
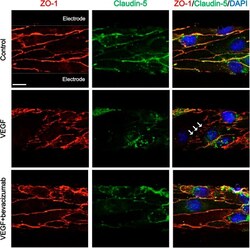
- Figure 5 Immunocytochemical staining of tight junction proteins in cerebral endothelial cells with different paracellular permeability. White arrows indicate disrupted alignment of tight junction proteins. Scale bars, 10 mum.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry